Abstract
The Crk family of adaptors is implicated in regulating various biological and pathological processes such as cell proliferation, adhesion, migration, invasion, phagocytosis, and survival. A large number of studies have shown that Crk plays an important role in aggressive and malignant behaviors of human cancers. In immunohistochemical analyses and gene-expression profiles, enhanced expression of Crk has been identified in adenocarcinomas of lung, breast, and stomach and in sarcomas and glioma. Overexpression of Crk in tumor cells induces the prominent tyrosine phosphorylations of scaffolding molecules such as p130Cas and paxillin through Src family tyrosine kinases and stimulates the activation loop of intracellular signalling, ultimately contributing to the increased motility and aggressive potential of cancer cells. Crk proteins thus are not simply conduits for intracellular signal transduction but also can control the amplitude of signalling. This review summarizes the significance of Crk and its mediated signaling assemblies, particularly in regulating tumor metastasis and invasion, and discusses the possibilities that they are potential cancer therapeutic targets.
Crk Adaptor Proteins and the Role in Human Malignancies
CT10 regulator of kinase (Crk) was originally identified as an oncogene product of v-Crk encoded in a chicken tumor retrovirus, CT10, that comprised Src homology 2 (SH2) and SH3 domains. 1 The cellular homologues CrkI and CrkII, and the related Crk-like (CrkL) are ubiquitously expressed and conserved across eukaryotic organisms.2-4 CrkI and CrkII are splice variants, where CrkI shares a structure analogous to v-Crk, and CrkII possesses an extended C-terminus containing a proline-rich linker and an atypical SH3 domain (SH3C).5,6 More recently, the third splice variant of crk, named CrkIII, has been identified and is predicted to encode a protein with a truncated SH3C; however, the biological significance of this molecule has remained obscure. 7
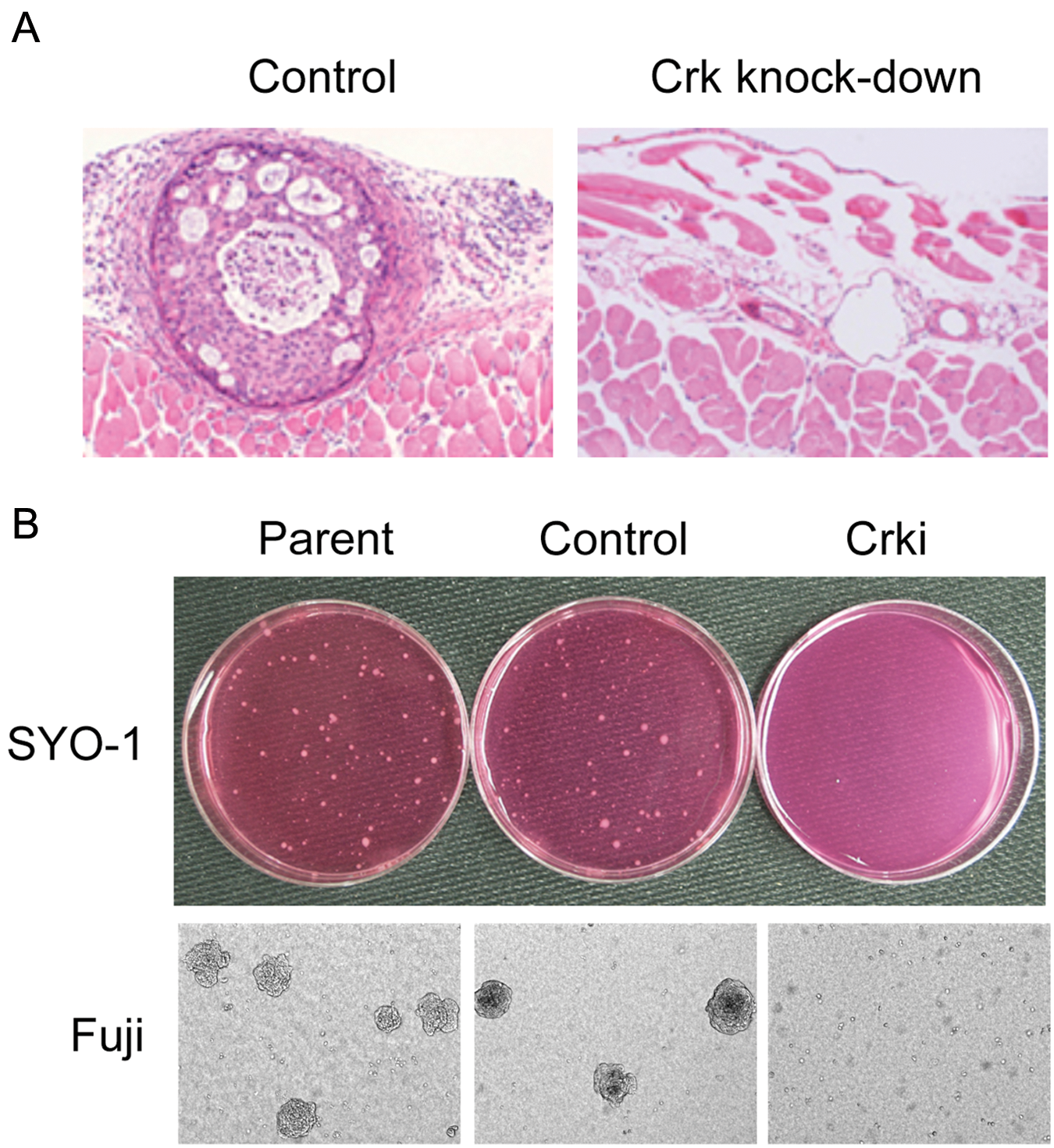
Crk proteins are dysregulated in several human malignancies; indeed, overexpression of Crk proteins has been reported in various human cancers.8,9 Immunohistochemical analysis has illustrated enhanced expression of Crk proteins in adenocarcinomas of lung, breast, and stomach and also in sarcomas (Fig. 1). In an analysis of gene-expression profiles of 86 primary lung adenocarcinomas, increased Crk expression has shown to be a predictive marker for poor prognosis and shorter survival. Simultaneous down-regulation of both CrkI and CrkII by siRNA approach demonstrated the essential role of Crk in the malignant features of human ovarian cancer cells (Fig. 2), 10 synovial sarcoma cells (Fig. 2), 11 and brain tumors, such as glioblastoma cells. 12
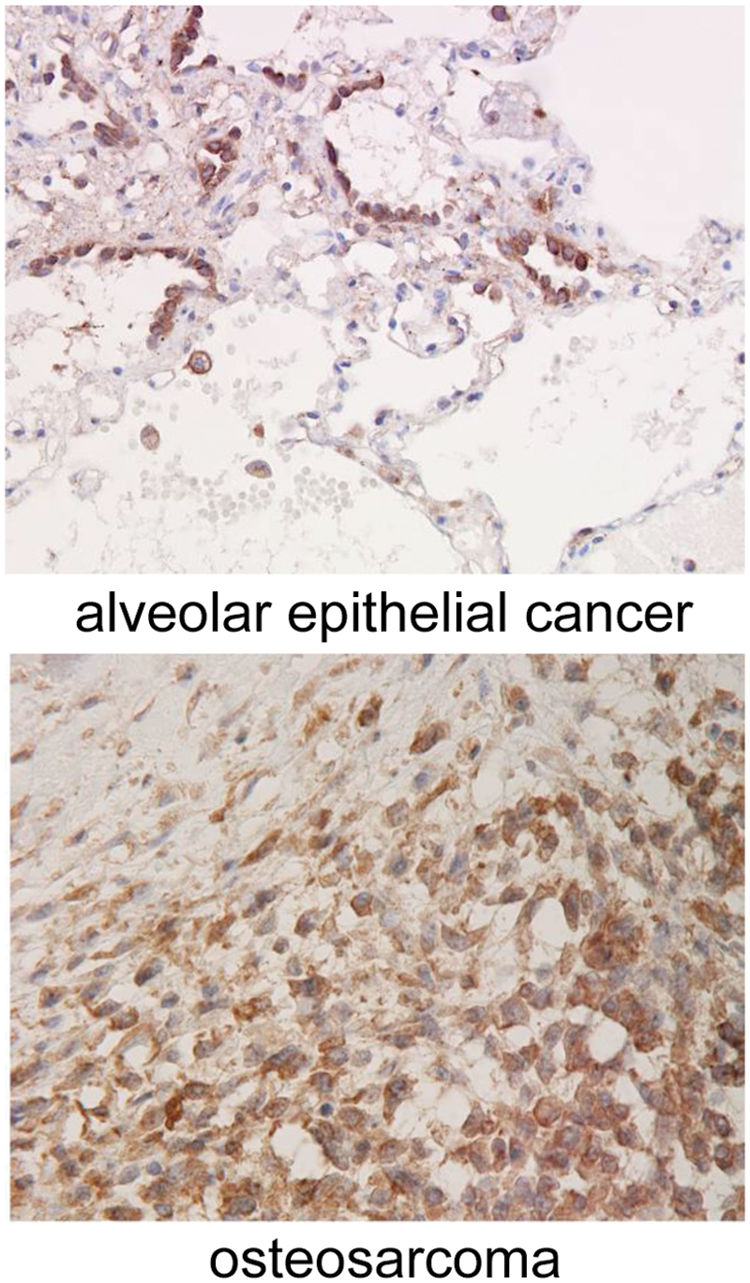
Immunohistochemistry of Crk in human lung bronchioalveolar epithelial cancer (
(
Recent studies demonstrated that CrkII is a likely direct target for miR (microRNA)-126 and that overexpression of miR-126 decreased cell adhesion, spreading, and invasion in lung cancer cells. 13 MiR-126 was significantly down-regulated in gastric cancer tissues compared with matched normal tissues and was associated with clinicopathological features, including tumor size, lymph node metastasis, local invasion, and tumor-node-metastasis (TNM) stage. Ectopic expression of miR-126 in SGC-7901 gastric cancer cells potently inhibited cell growth, migration, and invasion in vitro as well as tumorigenicity and metastasis in vivo. 14
In Crk knockdown cells, the numbers of focal adhesion were decreased, and cells exhibited cytoskeletal disorganization, whereby formation of lamellipodial structures at the leading edges of cells was impaired. Indeed, silencing of Crk by siRNA in human synovial sarcoma cells showed a suppression of hepatocyte growth factor (HGF)-dependent activation of Rac1 and decreased motility, 11 and in glioblastoma cells, Crk knockdown affected the early attachment to laminin. 12 Crk also regulates Rho-dependent phosphorylation of ezrin-radixin-moesin proteins, contributing to direct migration toward hyaluronic acid in the brain (Fig. 3). 15 Increased levels of CrkI mRNA are frequently observed in WHO grade III and IV malignant gliomas, but elevation of both CrkI and CrkII can be observed in only few cases,12,16 indicating that CrkI expression may correlate with poor prognosis of gliomas. Meanwhile, CrkII overexpression was more frequent in higher grade oral squamous cell carcinomas according to the tumor size, lymph node metastasis, and invasive pattern. RNAi-mediated suppression of CrkII reduced the migration and invasion potential, consistent with reduced the expression of Dock180, p130Cas, and Rac1 and the actin-associated scaffolding protein cortactin. 17 Differential expression of CrkI and CrkII emphasizes splicing-dependent control of Crk family in human cancer malignancy.
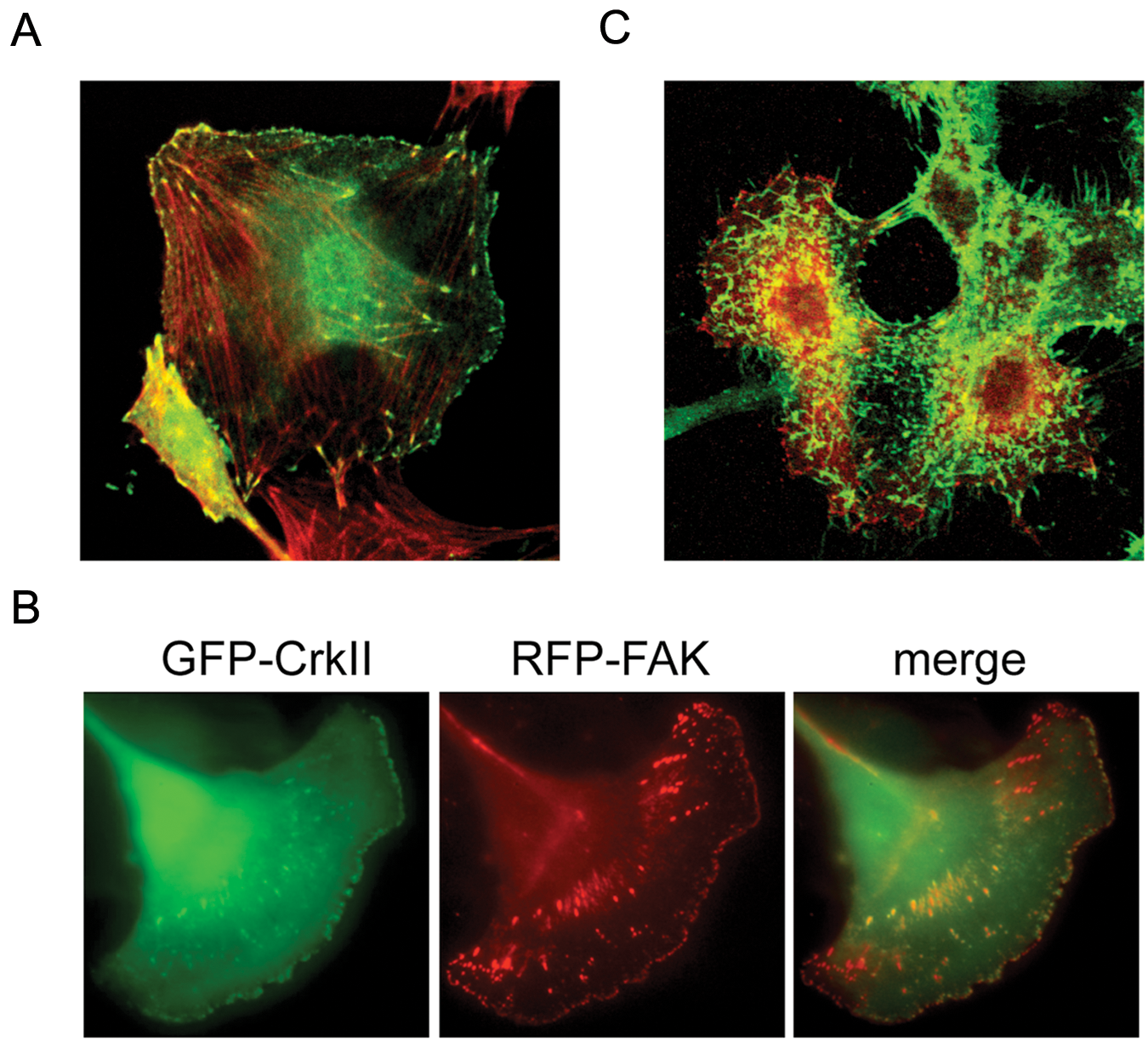
(
Taken together, the above studies suggest that Crk may have a central role in cell invasion and metastasis in highly aggressive motile cancer cells, and they raise the possibility that the Crk family contains appropriate molecular targets for cancer gene therapy. As Crk-depleted cell lines were not lethal, Crk may be an effective therapeutic target for decreasing the metastatic potential of human tumor cells. Here we highlight the significance of Crk proteins and Crk-mediated signaling pathways in the regulation of tumor cell invasion and metastasis and discuss the molecular mechanisms underlying these processes.
Crk-Mediated Signalling Pathways Contributing to Tumor Invasion and Metastasis
Role of Src in Tumor Cell Invasion and Metastasis
Src family kinases (SFKs) are nonreceptor tyrosine kinases involved in various signaling pathways in proliferation, migration, adhesion, survival, and angiogenesis during tumor development. 18 SFKs are composed of 9 family members, Src, Yes, Fyn, Lyn, Hck, Fgr, Blk, Lck, and Frk, that share similar structure and function. Elevated expression and/or activity of SFKs occurs frequently in a variety of human cancers, and hyperactivity of Src is correlated with tumor progression, metastasis, and prognosis. SFKs interact with receptor tyrosine kinases, including epidermal growth factor receptor (EGFR) and VEGF receptor, regulating cell proliferation and gene expression via the Ras/ERK/MAPK pathway and transcription factors such as STAT molecules, respectively. SFKs can also affect cell adhesion and migration via interaction with integrins, actin cytoskeletons, GTPase-activating proteins, scaffold proteins such as p130Cas, paxillin, and focal adhesion kinases (FAK). In fact, activation of EGFR induces Src-dependent phosphorylation of p130Cas, leading to the assembly of a p130Cas/Nck1 complex that promotes Ras-associated protein-1 (Rap1) signaling, followed by enhanced metastasis of pancreatic carcinoma in vivo. 19 Meanwhile, the recruitment of c-Src to the β3 cytoplasmic tail of αvβ3 integrin in pancreatic cancer and other carcinomas evokes c-Src activation, leading to p130Cas phosphorylation and tumor cell survival, that may account for the aggressive behavior of these tumors such as increased lymph node metastases in vivo. 20
We have demonstrated that Crk facilitates tyrosine phosphorylation of c-Met-docking protein Grb2-associated binder 1 (Gab1) at Tyr307 through Src, contributing to the organization of focal adhesions and enhanced cell migration, thereby possibly promoting human cancer development. 21 Indeed, Crk protein is required for the sustained phosphorylation of Gab1 in response to HGF, leading to the enhanced cell motility of human synovial sarcoma cell lines. By mutational analysis, we identified that SH2 domain of Crk is indispensable for the induction of the phosphorylation on multiple Tyr-X-X-Pro motifs containing Tyr307 in Gab1. HGF remarkably stimulated cell motility and scattering of synovial sarcoma cell lines, with the prominent activation of Rac1, extreme filopodia formation, and membrane ruffling. Importantly, the elimination of Crk in these cells induced the disorganization of actin cytoskeleton and complete abolishment of HGF-mediated Rac1 activation and enhanced cell motility. Furthermore, the depletion of Crk remarkably inhibited the tumor formation and its invasive growth in vivo. These results suggest that the sustained phosphorylation of Gab1 via positive feedback loop by Crk and Src in response to HGF contributes to the prominent activation of Rac1, leading to enhanced cell motility, scattering, and cell invasion, which may support the crucial role of Crk in the aggressiveness of human synovial sarcoma. 11
SFKs also can regulate angiogenesis via gene expression of angiogenic growth factors, such as fibroblast growth factor, VEGF, and interleukin-8. Based on these important findings, SFKs are currently being investigated as potential targets for treatment strategies in various cancers, and small-molecule SFK inhibitors have been developed and are undergoing early phase clinical testing. In preclinical studies, these agents have suppressed tumor growth and metastases.
Dasatinib (BMS-354825), a dual Src/Abl kinase inhibitor with potent antiproliferative activity against hematopoietic malignancies harboring BCR-ABL, blocks migration and invasion of human melanoma cells by completely inhibiting SFK kinase activity at low nanomolar concentrations. FAK and p130Cas, in addition to down-regulation of matrix metalloproteinase (MMP)-9, are also inhibited with similar concentrations and kinetics. SFKs and its downstream signaling are thus implicated as having key roles in migration and invasion of melanoma cells. 22
Association between p130Cas and Tumor Invasion/Metastasis
Proteins of the CAS (Crk-associated substrate) family (p130Cas/BCAR1 [breast cancer antiestrogen resistance-1], Hef1/NEDD9/Cas-L, and Efs/Sin and CASS4/HEPL) act as scaffolding molecules to regulate protein complexes controlling cell migration and chemotaxis, apoptosis, cell cycle, and differentiation and, more recently, progenitor cell function. 23 p130Cas was first identified as a highly phosphorylated 130 kDa protein in v-Crk and v-Src transformed cells; it also has been shown to be phosphorylated by FAK or Pyk2/RAFTK/CAK-β kinases in addition to SFKs, and the multiple tyrosine-phosphorylated Tyr-Asp-X-Pro motifs within the p130Cas substrate domain (SD) provide binding sites for numerous proteins including Crk, CrkL, and v-Crk. 24 The p130Cas/Crk complex most frequently contributes to the regulation of cell adhesion, migration, proliferation, and survival of normal cells. In the context of regulating cell adhesion and motility, the p130Cas/Crk complex in turn recruits C3G and DOCK180, a guanine nucleotide exchange factor (GEF) for the small GTPase Rap1 and Rac, respectively. Activated Rac induces membrane ruffling and actin cytoskeleton remodeling and promotes cell migration through ARP2/3 and PAK kinase activation. Re-expression of p130Cas in its deficient mouse embryo fibroblasts transformed by Src promoted an Matrigel invasion, organization of actin cytoskeleton into large podosome structures, MMP-2 activation, and elevated tyrosine phosphorylation of FAK and paxillin, of which all events were strictly dependent on the Tyr-X-X-Pro tyrosine phosphorylations of p130Cas. 25
Aberrant expression of p130Cas proteins, by gene amplification, transcriptional up-regulation, or alteration of protein stability, and the subsequent phosphorylation have been strongly linked to poor prognosis and increased cell invasion and metastasis in cancer as well as resistance to first-line chemotherapeutics in multiple tumor types including breast and lung cancers, glioblastoma, and melanoma.23,26,27 c-Abl phosphorylates Crk at Tyr221, causing disassociation of Crk from p130Cas. The disassembly of Crk/p130Cas complex inhibits cell migration and promotes apoptosis in normal cells. These data illustrate that c-Abl prevents aberrant motility and survival through Crk Tyr221 phosphorylation and modulation of Crk/p130Cas complex and that deregulation of this pathway contributes to cell metastasis. 28
The human HEPL (Hef1-Efs-p130Cas-like) mRNA and protein are selectively expressed in specific primary tissues and cancer cell lines, and HEPL maintains Cas family function in localization to focal adhesions as well as regulation of FAK activity, focal adhesion integrity, and cell spreading. It has been recently demonstrated that up-regulation of HEF1 expression marks and induces metastasis, whereas high endogenous levels of p130Cas are associated with poor prognosis in breast cancer, emphasizing the clinical relevance of Cas proteins. 29
p130Cas in breast cancer
The linkage between p130Cas functionality and breast cancer has been extensively studied. 23,30 Using estrogen-dependent cell lines, a retroviral-insertional mutagenesis screening identified BCAR1 as a principle responsible gene for tamoxifen resistance.30,31 BCAR1 levels positively correlated with HER2 (human EGFR related protein 2)/ErbB2/Neu expression levels. HER2 induces BCAR1/CrkII complex formation, leading to downstream ERK1/2 activation to enhance cellular invasion and proliferation.32,33 Recently, the expression of BCAR1 has shown to be regulated by EGR1 and NAB2 complex in tamoxifen-sensitive and -resistant breast cancer cells. 34 One mechanism underlying BCAR1 confers anti-estrogen resistance and may be accounted for by binding to BCAR3, having indirect GEF activity. BCAR3/BCAR1 complex is highly observed in estrogen receptor (ER)-negative breast cancer cells, and the expression of BCAR3 is required for induction of cyclin D1-linked anti-estrogen resistance. 35 In addition, the BCAR3/BCAR1 complex synergistically activates Src-dependent migration. 36
HER2 overexpression is detected in approximately 25% of invasive breast cancers and is strongly associated with poor patient survival. p130Cas is demonstrated as a crucial mediator of HER2 transformation. p130Cas controls HER2-dependent invasion in 3-dimensional cultures of mammary epithelial cells. Concomitant p130Cas overexpression and HER2 activation enhance PI3K/Akt and Erk1/2 MAPK signaling pathways, which trigger invasion of mammary acini through mTOR/p70S6K and Rac1 activation, respectively. In silico analyses indicate that p130Cas expression in HER2-positive human breast cancers significantly correlates with higher risk for development of distant metastasis, thus underlying the value of the p130Cas/HER2 synergism in regulating breast cancer invasion. 37
High levels of p130Cas SD tyrosine phosphorylation are a common characteristic of ER-breast cancer cell lines. This phosphorylation is an important signaling event in migration, invasion, proliferation, and survival. 38 Disruption of the p130Cas signaling node by expression of phospho-p130Cas SD attenuates the ERK and PI3K/Akt survival pathways in tamoxifen-resistant breast cancer cells, leading to reduced migration and resensitization to tamoxifen. 39 p130Cas is also required for mammary tumor growth and TGFβ-mediated metastasis through regulation of Smad2/3 activity 40 (Fig. 4)
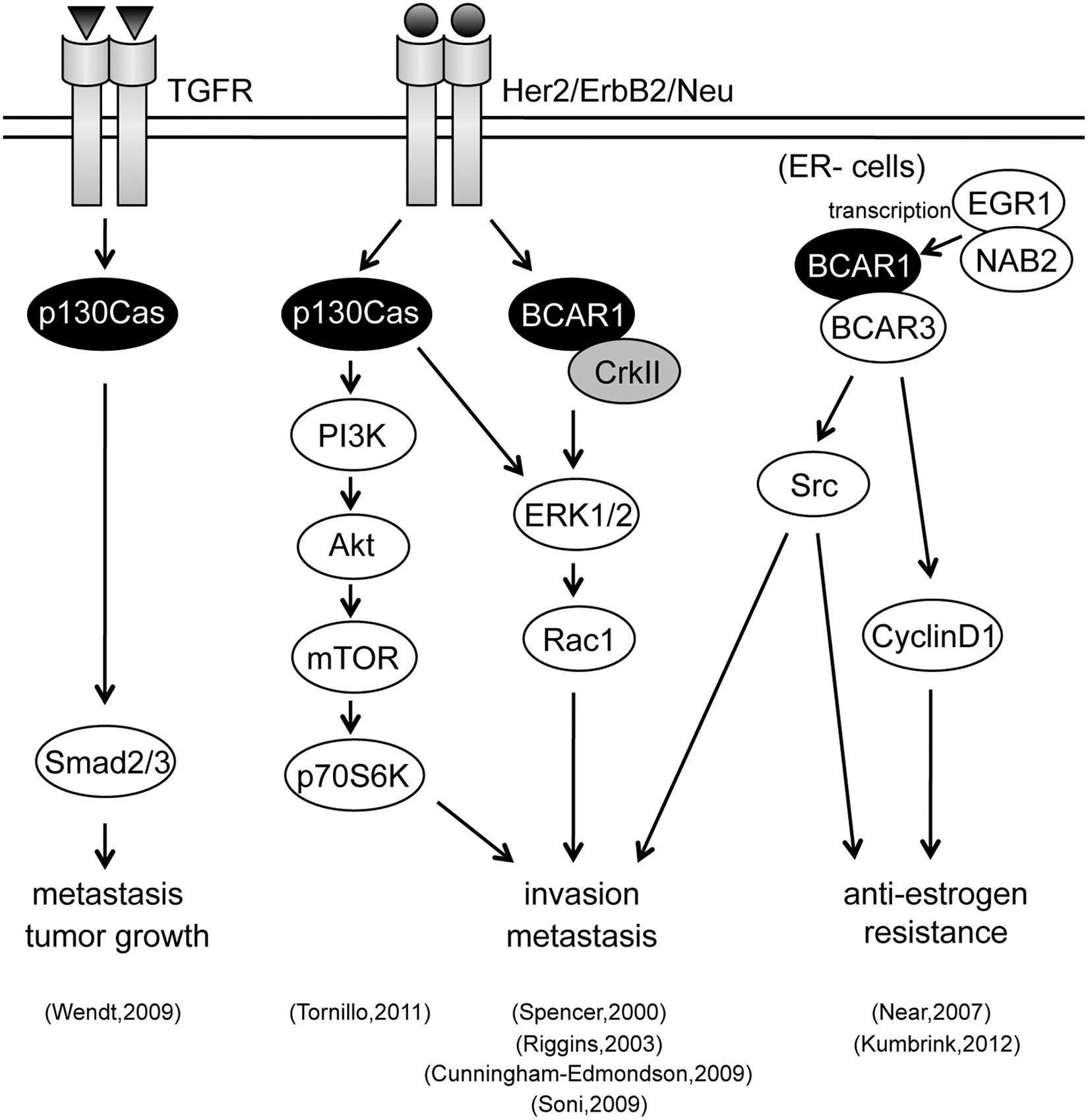
The linkage between p130Cas/BCAR1 protein functionality and breast cancer.
p130Cas in prostate cancer
Prostate cancer metastasis is often associated with poor prognosis. The molecular coupling of Crk to p130Cas serves as a switch that regulates cell migration in several invasive cancer cells. The high expression of CrkI and p130Cas in invasive prostate cancer cells followed by the formation of CrkI/p130Cas complex was demonstrated in the migration and invasion of prostate cancer cells, raising the possibility that CrkI/p130Cas complex could be exploited as potential molecular therapeutic target for prostate cancer metastasis. 41
Involvement of Paxillin in Tumor Metastasis
Paxillin acts as a scaffolding protein, mainly in integrin signaling. 42 Tyrosine-phosphorylated paxillin interacts with v-Crk with high affinity in CT10- transformed fibroblasts and mediates Rho-dependent cell spreading and focal adhesion biogenesis.43,44 Paxillin was also identified as a most remarkably phosphorylated protein in high-metastatic human osteosarcoma HuO9 sublines, and the phosphorylated form is essential for the migration-promoting effect in this sarcoma. The elimination of paxillin by RNAi technique attenuated the motility of high-metastatic cells. These findings suggest that enhanced activity of SFKs and overexpression of paxillin synergistically contribute to the high metastatic potential of human osteosarcoma through the hyperphosphorylation of paxillin. 45
The relationships between paxillin expression and clinicopathological features including metastasis have been investigated in salivary adenoid cystic carcinoma (SACC). High expression of paxillin, in 57.45% of SACC as cytoplasmic staining, was correlated with a more advanced stage and metastasis in SACC, suggesting that paxillin may be a valuable biomarker in advanced SACC and SACC with distant metastasis. 46
The chemokine-CXCL12 and its receptor, CXCR4, recently were shown to play an important role in regulating the directional migration of breast cancer cells to sites of metastasis. CXCL12 induces the tyrosine phosphorylation of FAK, RAFTK/Pyk2, paxillin, Crk, SHP2, and Cbl in addition to the activation of MMP-2 and -9, ultimately resulting in the enhanced chemotaxis, chemoinvasion, and adhesive properties of breast cancer cells. 47
Association of Dock180 and C3G in Tumor Metastasis
Random motility of urinary bladder carcinoma NBT-II cells on collagen critically depends on paxillin phosphorylation at Tyr31 and Tyr118, the binding sites for CrkII. Paxillin/Crk/DOCK180 signaling axis controls collagen-dependent migration mainly through Rac1 activation. Meanwhile, in migrating cells, CrkII constitutively associated with C3G was not sufficient to stimulate the activation of Rap1. Forced activation of the C3G/Rap1 signaling pathway resulted in down-regulation of the paxillin/Crk/DOCK180 complex and reduction of Rac1-GTP, suggesting that Rap1 activation could suppress the Rac1 signaling pathway in epithelial cells. 48 Elevated cytoplasmic expression of Dock180 in ovarian cancer was significantly associated with serous histological type, high-grade cancer and advanced stage, as well as poor overall and disease-free survival. Exogenous expression of Dock180 enhanced cancer cell migration and invasion, whereas the depletion of Dock180 by an siRNA approach retarded them in association with down-regulation of MMP-2. Thus, Dock180 contributes to ovarian carcinogenesis and dissemination and is a potential prognostic marker and therapeutic target. 49
Crk-Mediated Signalling Molecules Contributing to Tumor Metastasis
Crk and p120-Catenin
As a member of adherens junction, p120-catenin (p120ctn) plays a major role in cell adhesions through stabilization of E-cadherin and is transcriptionally downregulated in non-small-cell lung cancer (NSCLC). Transcription factor SP1 is a transcriptional repressor of p120ctn that directly binds to segments (–9 to +36) of the p120ctn promoter, and Crk has identified to be involved in the transcriptional down-regulation of p120ctn. Overexpression of CrkI/II in NSCLC cells down-regulated p120ctn, the effect of which was abrogated by simultaneous silencing of SP1. These findings emphasize the role of Crk in transcriptional repression of p120ctn and potentially the following metastasis in NSCLC. 50
Crk and Abl
Formation and dissolution of intercellular adhesions are important processes during tumor metastasis. Cadherin-mediated intercellular adhesion requires dynamic regulation of the actin cytoskeleton. The Abl family of tyrosine kinases are activated by cadherin engagement, localized to cell-cell junctions, and contribute to the formation of adherens junctions via Rac activation, which evokes a Rho-ROCK-myosin signaling pathway required for the maintenance of intercellular adhesion. Thus, Abl kinases regulate the formation and strengthening of adherens junctions downstream of active Rac, and, importantly, Abl kinases are components of a positive feedback loop that uses the Crk/CrkL proteins to promote the formation and maturation of adherens junctions. 51
Crk and MMP
Both MMP-2 and MMP-9 play critical roles in tumor invasion, but their production is differentially controlled. The promoter region of MMP-9 has the conserved proximal AP-1 binding site, whereas that of the MMP-2 possesses a noncanonical AP-1 site. v-Crk-transformed cells have augmented production of MMP-2 and MMP-9, leading to in vitro invasiveness. AP-1 appears thus to play a critical role in the production of MMP-2 and MMP-9 and invadopodia formation in v-Crk-transformed cells. 52
Crk in Endothelial Cells
Induction of epithelial cell motility is a fundamental morphogenetic event that is recapitulated during tumor metastasis. Intermittent hypoxia (IH) that develops in neovascularized solid tumors has been described to positively influence the tumor growth by modulating the behavior of cancer cells as well as of endothelial cells. NDRG1 and CrkI/II are indeed regulators of endothelial cell migration under IH: silencing of CrkI/II resulted in an increase in endothelial cell migration, whereas the invalidation of NDRG1 decreased it. 53
Summary
Over the past several decades, considerable effort has been directed toward identifying the function of the Crk family of proteins and its mediated signaling. Crk contributes to the generation, derivation, and amplification of diverse signaling pathways, regulating numerous biological and pathological behaviors. The Crk-mediated signaling pathways that particularly operate in invaded cells during the development and pathological conditions associated with cancer metastasis appear to be extremely intricate and variable. The spatiotemporal dynamics of Crk per se and its mediated signaling assemblies may be critical to achieve these processes. Further effort should be made to explore and elucidate when and how Crk recruits specific effector proteins that are coupled with the migration machinery.
Footnotes
Acknowledgements
The authors thank all members of the Tanaka laboratory who participate in Crk research and dedicate this review to the late Dr. Hidesaburo Hanafusa.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This paper was in part supported by grants-in-aid from the Ministry of Education, Culture, Sports, Science, and Technology of Japan.
