Abstract
The vascular endothelial growth factor (VEGF) and its receptor (VEGFR) have been shown to play major roles not only in physiological but also in most pathological angiogenesis, such as cancer. VEGF belongs to the PDGF supergene family characterized by 8 conserved cysteines and functions as a homodimer structure. VEGF-A regulates angiogenesis and vascular permeability by activating 2 receptors, VEGFR-1 (Flt-1) and VEGFR-2 (KDR/Flk1 in mice). On the other hand, VEGF-C/VEGF-D and their receptor, VEGFR-3 (Flt-4), mainly regulate lymphangiogenesis. The VEGF family includes other interesting variants, one of which is the virally encoded VEGF-E and another is specifically expressed in the venom of the habu snake (
Keywords
Introduction
Angiogenesis, the formation and maintenance of blood vessel structures, is essential for the physiological functions of tissues and is important for the progression of diseases such as cancer and inflammation.1,2 In recent decades, a variety of signaling molecules, such as VEGF-VEGFRs, ephrin-Eph receptors, angiopoietin-Tie, and the Delta-Notch system, have been identified as playing important roles in angiogenesis. Among these, vascular endothelial growth factors (VEGFs) and receptors (VEGFRs) regulate both vasculogenesis, the development of blood vessels from precursor cells during early embryogenesis, and angiogenesis, the formation of blood vessels from pre-existing vessels at a later stage 3 (Fig. 1). The VEGF family of genes contains at least 7 members, including the viral genome–derived VEGF-E, whereas the VEGFR family of genes has 3 to 4 members depending on the vertebrate species.4,5 VEGF-A and its receptors VEGFR-1 and VEGFR-2 play major roles in physiological as well as pathological angiogenesis, including tumor angiogenesis. VEGF-C/D and their receptor VEGFR-3 can regulate angiogenesis at early embryogenesis but mostly function as critical regulators of lymphangiogenesis. 6
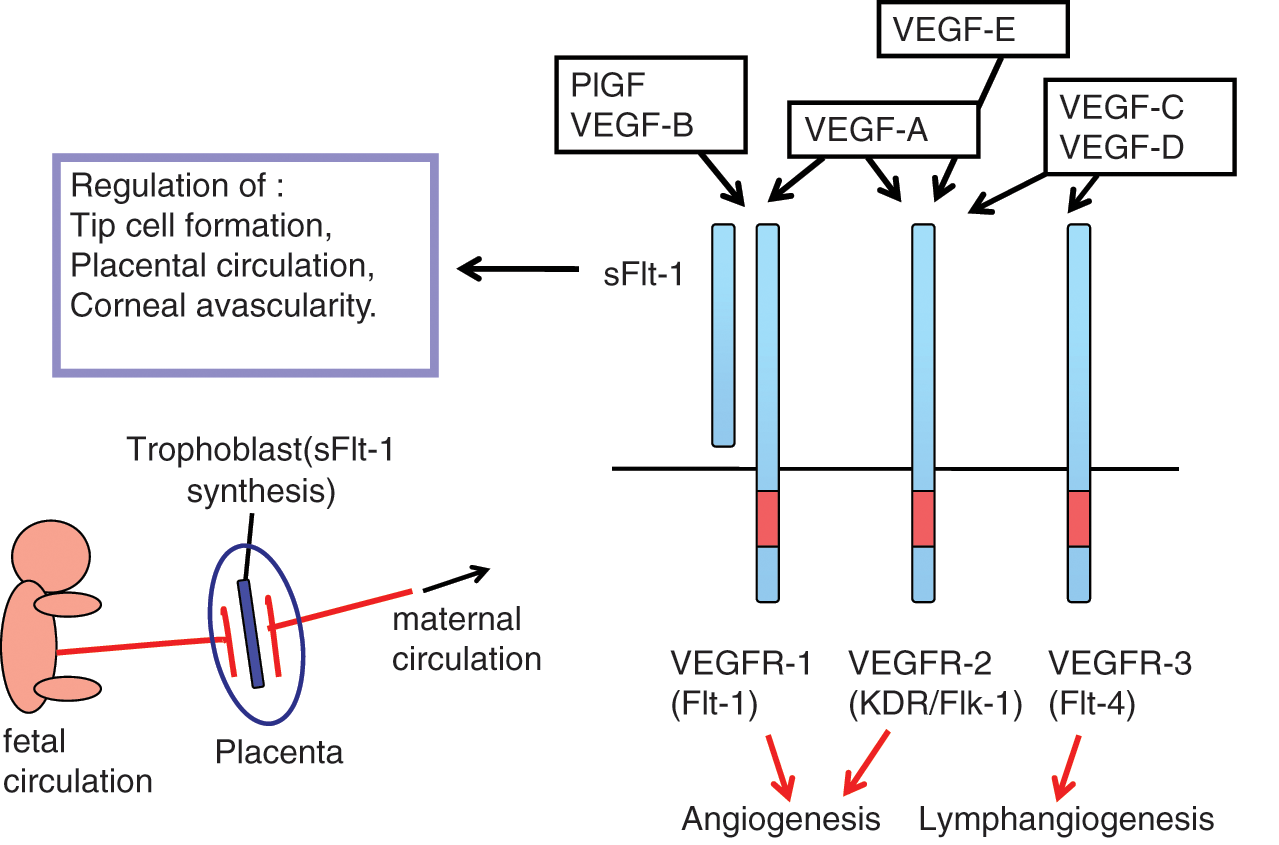
The VEGF and VEGFR system. VEGF-A and its receptors, VEGFR-1 and VEGFR-2, play a major role in vasculogenesis and angiogenesis. In addition, sFlt-1, a soluble form of VEGFR-1, is expressed in various cells such as trophoblasts and negatively regulates angiogenesis.
VEGF-A has a variety of functions, including pro-angiogenic activity, vascular permeability activity, and the stimulation of cell migration in macrophage lineage and endothelial cells. Recently, anti–VEGF-VEGFR drugs such as an anti–VEGF-A neutralizing antibody and multikinase inhibitors have been developed and widely used for the treatment of major solid tumors.7,8 The clinical efficacy of these medicines has been well evaluated; however, none of them provide a complete cure for cancer patients. The molecular basis of the refractoriness in some tumors and the acquisition of resistance to these medicines should be extensively studied to develop more efficient anti-angiogenic therapies.
On the other hand, VEGFs have pro-angiogenic potential for the maintenance of various tissues at physiological levels and for the formation of new blood vessels to overcome ischemic diseases. The utility of VEGF family members in pro-angiogenic medicine, together with the possible side effects, should be characterized in more detail for clinical applications.
Structure and Function of the VEGF Family
VEGF, also known as VEGF-A, is a protein with vascular permeability activity that was originally purified from a fluid secreted by a tumor.
9
A few years later, a protein with angiogenic activity was independently purified and named VEGF.
10
Molecular cloning, however, revealed that these 2 proteins were identical and encoded by a single gene.
3
The VEGF family includes VEGF-A, VEGF-B, VEGF-C, VEGF-D, PlGF (placental growth factor), VEGF-E (Orf-VEGF), and
VEGF-A
Through alternative splicing, the VEGF-A protein contains subtypes, such as peptides of 121, 165, 189, and 206 amino acids in humans. 3 Except for VEGF-A121, the other peptides have a basic stretch near the carboxyl terminus. The basic stretch of VEGF-A165 has a weak affinity for acidic materials such as heparin/heparan sulfate and to neuropilin-1, a membrane protein involved in neuronal cell regulation and a coreceptor for VEGF-A. The basic stretch of VEGF-A189 has a strong binding affinity to heparin/heparan sulfate, and thus, most of the VEGF-A189 molecules appear to be localized on the cell surface or in the extracellular matrix.
The
Among subtypes of VEGF-A, VEGF-A165 is most important both quantitatively and qualitatively. Maes
VEGF-A binds to and activates both VEGFR-1 and VEGFR-2, promoting angiogenesis, vascular permeability, cell migration, and gene expression.
5
In addition, Lee
PlGF and VEGF-B
These molecules bind to and activate only VEGFR-1. As will be described later, VEGFR-1 has the ability to bind tightly to its ligands but has a weak tyrosine kinase activity, generating signals weaker than VEGFR-2. Both
VEGF-C and VEGF-D
These 2 members of the VEGF family are produced as premature forms and are cleaved by proteases such as furin in both the amino- and carboxyl-terminal portions.
19
After processing, these molecules develop a higher affinity for VEGFR-3, which is expressed on lymphatic endothelial cells and stimulates the receptor for lymphangiogenesis. In addition, these proteins have a weak affinity for VEGFR-2, activating angiogenesis to some extent. VEGF-C is expressed during embryogenesis, whereas VEGF-D is expressed after birth during adult stages. This difference in gene expression is thought to be a major cause for lethality in
VEGF-E, an Angiogenic Protein Encoded in the Pro-Angiogenic Orf Virus Genome
The Orf virus, a parapoxvirus infecting sheep, goats, and sometimes humans, is known to induce angiogenesis at sites of infection on the skin. In 1994, Lyttle
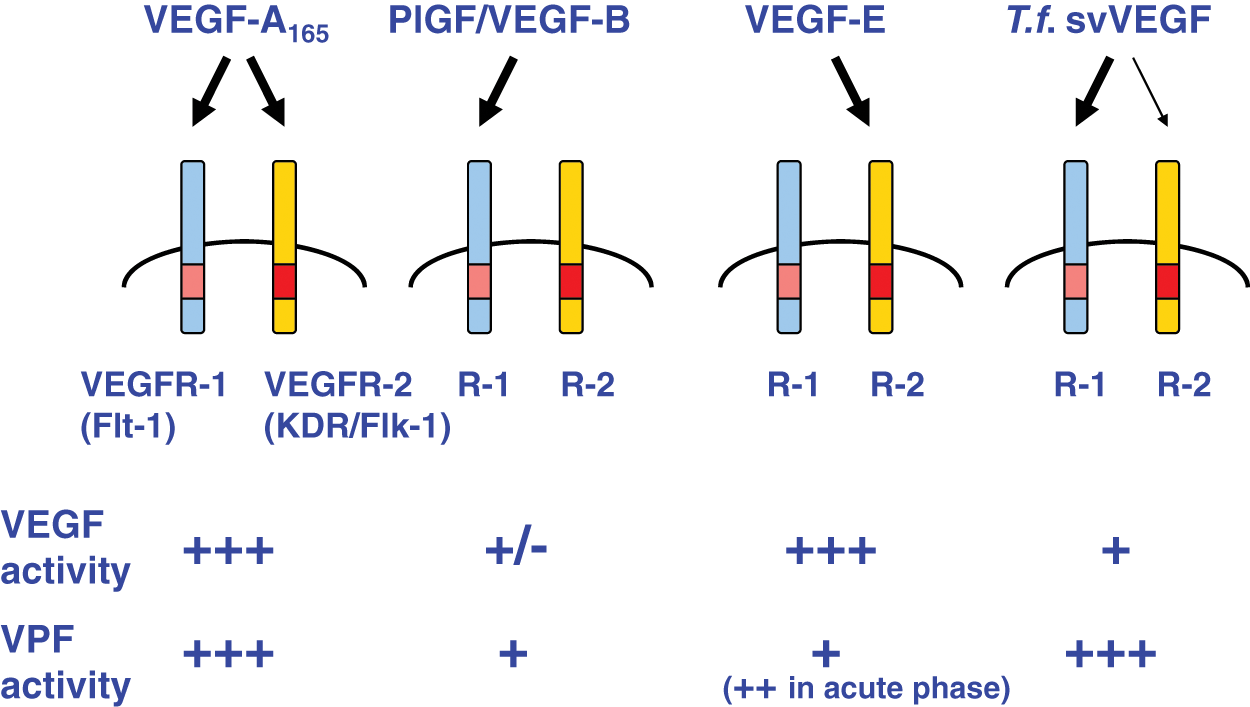
Unique activation of VEGFRs by VEGF-E and
Because the human genome does not contain the original
T. flavoviridis svVEGF, a VEGF-Like Molecule Secreted in Snake Venom
Snake venom contains a variety of molecules that attack target animals both directly (as toxins) and indirectly (as toxin-promoting materials). From
Structure of VEGFRs
VEGFRs are typical tyrosine kinase receptors (TKRs) carrying an extracellular domain for ligand binding, a transmembrane domain, and a cytoplasmic domain, including a tyrosine kinase domain 4 (Fig. 1). The overall structure of VEGFRs is similar to that of the PDGFR family members; however, these 2 receptor families have clear differences: the PDGFR extracellular domain contains 5 immunoglobulin (Ig)–like domains, whereas VEGFRs bear 7 Ig-like domains. Both TKRs share a tyrosine kinase domain with a long kinase insert (KI) of 60 to 70 amino acids; however, the amino acid sequences in the KI of these 2 TKRs are very different from each other. The KIs in PDGFR family members contain 1 or 2 Tyr(Y)-x-x-Met(M) motifs as autophosphorylation sites, and these motifs have been shown to be strong binding sites for the SH2 domain of the p85 subunit in the PI3-kinase complex and to activate the PI3K pathway. These autophosphorylation sites were demonstrated to be crucial for the cell growth signal mediated by PDGFR and for the cell transformation signal mediated by v-Fms, an activated form of M-CSFR (a member of the PDGFR family). However, none of the VEGFRs contain this Y-x-x-M motif in their KI region or in the carboxyl-terminal region, indicating that the downstream signaling from VEGFRs may be different from that of the PDGFR family. 5
Signaling of VEGFRs
VEGF-A binds to and activates VEGFR-1 (Flt-1) and VEGFR-2 (KDR/Flk-1 in mice). VEGFR-1 has a high affinity for VEGF-A (Kd = 1~10 pM), which is one order higher than that of VEGFR-2, whereas its tyrosine kinase activity is approximately 10-fold weaker than that of VEGFR-2.
26
The major pro-angiogenic signal is generated from the ligand-activated VEGFR-2. Within the KI or carboxyl-terminal region, TKRs have tyrosine autophosphorylation sites, which are important for the downstream signal. Unlike most of the TKRs that activate the Ras pathway or PI3K pathway, we found that the PLCγ-PKC-MAPK pathway is highly activated in VEGF-bound VEGFR-2 and used as a crucial signal for endothelial proliferation. An SH2 domain of PLCγ specifically binds to the 1175-PY site of VEGFR-2 (1173-PY in mice) and further activates PKC, particularly the PKCβ pathway.27,28 An 1175-phenylalanine (F) mutant of VEGFR-2 significantly decreases the MAPK pathway under stimulation with VEGF and cannot efficiently activate the endothelial proliferation signal. Furthermore, mice with an amino acid knock-in at the 1173 site from Y to F (
Taken together, these results strongly suggest that the PLCγ-PKC-MAPK pathway initiated from the VEGFR-2 1175-PY site plays a pivotal role in pro-angiogenic signaling from VEGFR-2. A spontaneous mutant of zebrafish carrying the lethal circulatory system abnormality was shown to have a mutation in the fish
VEGFR-1 has a much weaker kinase activity than VEGFR-2, and the signaling cascade is not fully understood. The 1169-Y on VEGFR-1 corresponding to 1175-Y on VEGFR-2 is a PLCγ activation site from VEGFR-1. However, we found that 1169-PY is not a major autophosphorylation site on VEGFR-1. Consistent with this finding, direct pro-angiogenic activity from VEGFR-1 is usually weak or undetectable. 34
In addition to vascular endothelial cells, VEGFR-1 is expressed on macrophage lineage cells and facilitates migration of these cells. Recently, we have shown that a scaffold protein RACK1 is involved in this migration signal and that the VEGFR-1-RACK1-PI3K-Akt pathway appears to be important for this signal. 35
The biological functions of VEGFR-1 have been the topic of several studies. Fong
VEGFR-3 has a typical tyrosine kinase like other VEGFRs, and upon stimulation with VEGF-C, the PKC pathway and Ras pathway were reported to be activated for lymphangiogenesis. However, it remains to be clarified which autophosphorylation site(s) on the tyrosine residues in VEGFR-3 is responsible for these pathways and is critical for lymphangiogenesis.
Unique Characteristics of sFlt-1
The VEGFR-1 (Flt-1) gene expresses 2 mRNAs: one is a long form of approximately 8 kb, and the other is a short form of 2.5 to 3.0 kb. 44 The short mRNA is highly expressed in normal placenta, encoding a soluble form of Flt-1 known as sFlt-1.44,45 The sFlt-1 contains 6 Ig-like domains with a short, 31 amino acid–long tail derived from the 5′ region of intron 13 and exhibits a strong binding ability to VEGF-A, PlGF, and VEGF-B.45,46
Within the placenta, trophoblasts located between the fetal and maternal blood vessel systems preferentially express sFlt-1 (Fig. 1). Thus, an interesting possibility is that sFlt-1 functions as a biochemical barrier between fetal and maternal circulation in the placenta by suppressing excess angiogenesis and abnormal vascular permeability. From this model, the level of sFlt-1 should be controlled at an appropriate physiological range because overtrapping of VEGF may cause severe problems in the circulatory system of the placenta. Interestingly enough, abnormal overexpression of sFlt-1 in the placenta was observed in a major disease in the field of obstetrics. In 2003, Maynard
A podocyte-specific knockout of the
sFlt-1 was also found to be expressed in corneal epithelial cells. 50 This strongly suggests that sFlt-1 suppresses angiogenesis near the lens and maintains the transparency of the eye.
Anti–VEGF/VEGFR Therapy and Anticancer Therapy
The VEGF-VEGFR system is unique in that it consists of a very limited number of molecules that play a central role in angiogenesis. The major ligand (VEGF-A) is a single gene product, and it utilizes only 2 TKRs (VEGFR-1 and VEGFR-2), although neuropilin-1 is used as a coreceptor. Other ligands, such as PlGF, VEGF-C, and VEGF-D, and the receptor VEGFR-3 appear to be partly involved in pathological angiogenesis, such as tumor vasculature. On the other hand, tumors metastatic to lymph nodes express higher levels of VEGF-C/D, suggesting that the VEGF-C/D and VEGFR-3 system plays an important role in lymph vessel–dependent tumor cell migration into lymph nodes. The angiopoietin-Tie system is also involved in pathological angiogenesis, but the details of its role in the process of carcinogenesis are not fully understood.
On the basis of these results, anti–VEGF-VEGFR drugs such as anti–VEGF-A neutralizing antibody and tyrosine kinase inhibitors have been developed, and bevacizumab (anti–VEGF-A humanized monoclonal antibody) has been approved for the treatment of colorectal, breast, lung (non–small cell type), and renal cancers as well as for glioblastoma patients.3,51 Multikinase inhibitors such as sorafenib and sunitinib are now approved for renal and hepatic cancer patients.
In addition to these medicines, others that target the VEGF-VEGFR system, including VEGF-Trap (a fusion protein of VEGFR-1 and VEGFR-2 ligand-binding domains), anti–VEGFR-1 or anti–VEGFR- 2 neutralizing antibody, soluble VEGFR-3, VEGFR-1 or VEGFR-2 peptide vaccine therapy, 52 and anti-PlGF antibody,53,54 have been developed and are undergoing preclinical and clinical trials.
The Molecular Basis of Anti-Angiogenic Therapy
In 1993, Kim
However, in clinical trials, treatment of cancer patients with an anti–VEGF-A antibody alone did not produce significant suppression of tumor growth, except for renal cancer. In contrast to murine tumor transplantation models, the growth rate of tumors in patients is usually slower, and tumor angiogenesis may develop more slowly compared with the murine tumor models. In clinics, the vasculature in tumors might be more stable than that in the murine system and less sensitive to VEGF-A blockade alone.
Another hypothesis regarding the efficacy of the anti-VEGF antibody and anti-VEGFR tyrosine kinase inhibitor on tumor growth in patients is termed “vascular normalization,” whereby the absorption of VEGF-A induces a transiently normalized vascular structure, more stabilized and well covered with pericytes with lower vascular permeability. 56 These conditions may result in a lower tissue pressure within tumors, having a better diffusion of anticancer drugs. It is probable that both vascular normalization and suppression of new tumor angiogenesis can occur in parallel within the tumors in patients treated with anti–VEGF-VEGFR drugs (Fig. 3).
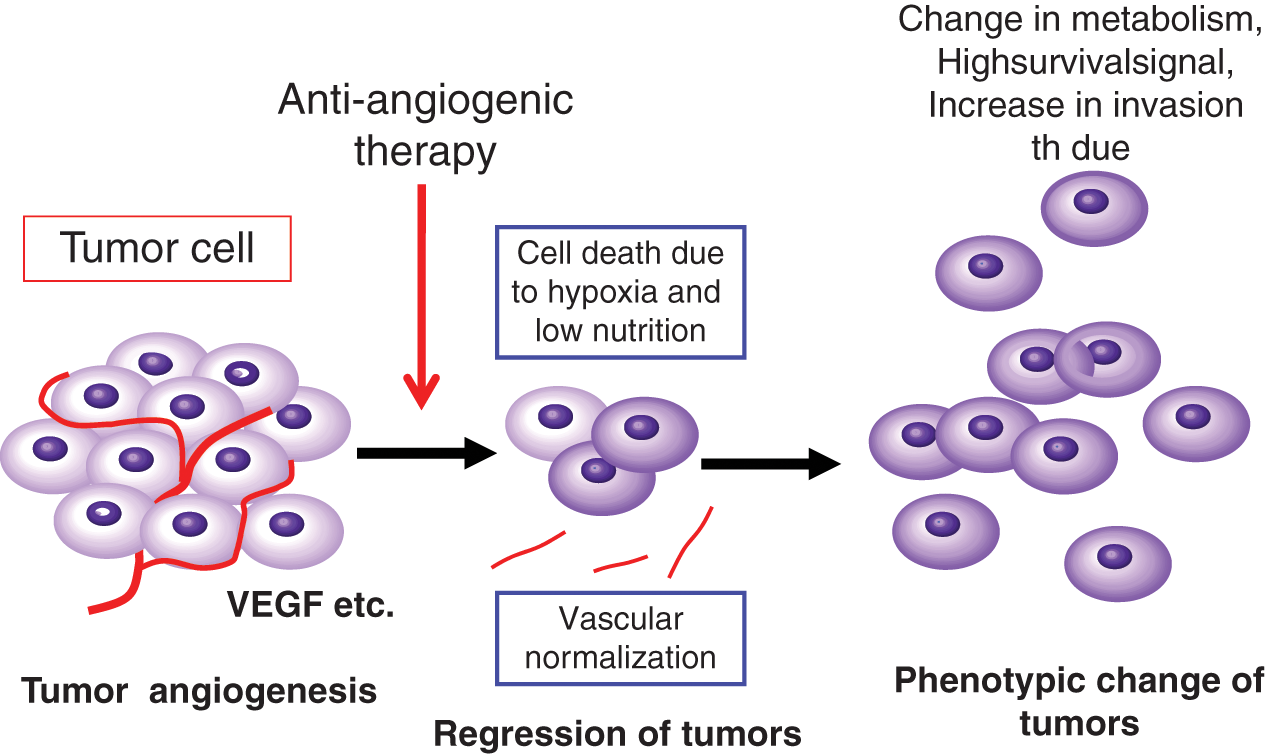
A possible response of tumor cells to anti-angiogenic therapy: a model. A direct suppression of tumor angiogenesis and “vascular normalization” results in the suppression of tumor growth. However, after long-term therapy, tumor cells under hypoxia and low nutrition double stress acquire a resistant phenotype.
Side Effects and Refractoriness to Anti-Angiogenic Therapy
A variety of side effects, such as hypertension, renal dysfunction, proteinuria, thrombosis, bleeding, and arrhythmia, have been reported in patients under anti–VEGF-VEGFR therapy.7,8 Among these, the frequency of hypertension and proteinuria is higher than that of others, suggesting a direct relationship with the blockage of VEGF-A in tissues. A decrease in the level of VEGF-A in the kidney could induce damage to vascular endothelial cells in glomeruli, and such a dysfunction of glomerular microvasculature may cause proteinuria. However, the molecular basis of hypertension under VEGF-VEGFR blockage remains to be clarified.
Whether tumor cells acquire refractoriness or resistance to anti-angiogenic therapy after long-term treatment is an important question. In clinical trials, the efficacy of anti–VEGF-VEGFR therapy on the increase in survival time seems sometimes inconsistent. Survival time did not increase stably during the course of treatment, and in some trials after a long period, the efficacy appears to decrease, suggesting a resistance or refractoriness of tumors to this treatment. In addition, in some preclinical and clinical trials, glioblastoma showed an enhanced invasiveness after anti-angiogenic therapy 57 (Fig. 3).
Many experimental models could be introduced and studied to understand this resistance. Casanovas
Pro-Angiogenic Therapy
Ischemic heart failure and cerebral attacks with thrombosis or bleeding are major diseases in humans. Furthermore, recent studies strongly suggest that some degenerative diseases, such as neuronal degeneration, are due to lower circulation as well as lower VEGF-VEGFR signaling in neuronal cells.60,61
For the treatment of ischemic heart and brain diseases, pro-angiogenic therapy could be useful because these diseases are essentially due to poor circulatory conditions. Among the VEGF family, VEGF-A plays a crucial role in blood vessel formation in embryogenesis and the earlier stages after birth. However, in adult stages, VEGF-A stimulates not only VEGFR-2 but also VEGFR-1 (Flt-1), which enhances the migration of inflammatory cells such as macrophages, resulting in inflammation and hypervascular permeability. Several articles have previously reported that K14 promoter–driven VEGF-A transgenic mice exhibit severe inflammation with edema in dermal tissues with angiogenesis, this being a model of psoriasis vulgaris, which is a chronic inflammatory skin disease. 62 On the other hand, we have shown that the VEGFR-2–specific ligand VEGF-E induces well-organized blood vessels with pericyte coverage and maintains normal vascular permeability. No clear inflammatory reaction was observed in VEGF-E transgenic mice. K14-PlGF only showed a very limited angiogenic response.63,64
In the case of motoneuron degeneration, such as the amyotrophic lateral sclerosis (ALS) rat model, Storkebaum
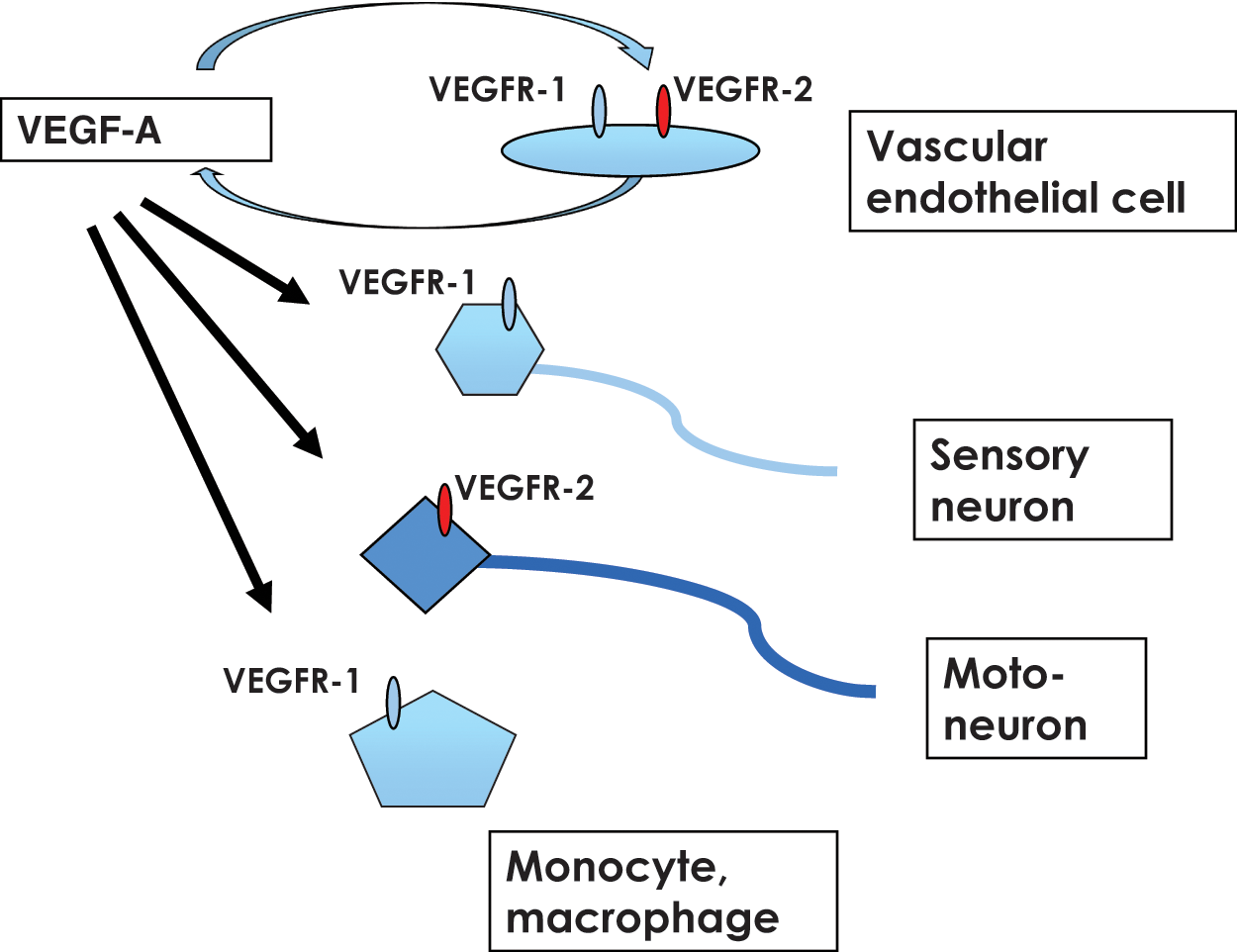
A pro-angiogenic therapy using the VEGF-VEGFR system. Recent studies suggest that sensory neurons express VEGFR-1 and motoneurons express VEGFR-2. These receptors are biologically functional, and therefore, an appropriate ligand, such as VEGF-E, can be used for pro-angiogenic therapy as well as for neuron protection therapy.
Taken together, stimulation of VEGFR-2 and VEGFR-1 with VEGF-A or VEGFR-2 alone with VEGF-E is a possible pro-angiogenic therapy. VEGF-E is a particularly attractive molecule because it does not enhance an inflammatory reaction, whereas VEGF-A might through the activation of VEGFR-1. Although the
Conclusions
Phylogenetically, VEGF-A is highly conserved from fish to mammals. Furthermore, 3 VEGFR systems with a soluble form of VEGFR-1 are maintained from amphibians to mammals. These findings suggest that VEGF-VEGFR is not only used for the formation of blood vessels but also for the maintenance of various tissues such as neuronal tissue. The VEGF-VEGFR system is closely linked to other angiogenesis regulatory systems such as angiopoietin-Tie and Delta (Dll4)–Notch systems for tight regulation of angiogenesis. More extensive studies on the VEGF-VEGFR system and its relationship with other regulators are necessary and should open new fields toward developing better strategies to treat cancer and other diseases.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by a Grant-in-aid Special Project Research on Cancer-Bioscience [grant number 17014020] from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
