Abstract
Background
Diabetes mellitus is a chronic metabolic disorder characterized by persistent hyperglycemia and associated complications. Natural plant-based therapies have gained increasing attention due to their potential efficacy and safety.
Objective
This study aimed to chemically characterize of AE fruit methanol (AEM), hexane (AEH), and water (AEW) extracts and evaluate their anti-diabetic activity using both
Methods
AE extracts were prepared and chemically analyzed using standard phytochemical and GC/MS analysis.
Results
GC-MS analysis identified 14 compounds in the AEM and 12 in the AEH known to possess anti-diabetic activity. AEM and AEH enhanced GLUT4 translocation in L6-GLUT4myc muscle cells, with AEM showing the highest activity. Oral AEM treatment reduced blood glucose levels in diabetic mice from 525 ± 46 to 234 ± 56 mg/dL after 5 weeks. Both extracts exhibited antioxidant activity, with AEM achieving 69% inhibition in the DPPH assay.
Conclusion
The findings demonstrate that chemically characterized AE extracts possess significant anti-diabetic activity in both
Introduction
Diabetes mellitus is a chronic disease associated with genetic disposition, ageing, obesity and lack of physical activity. 1 The predominant form of diabetes is type II, usually initiated by insulin insensitivity leading to hyperglycemia and hyperinsulinemia. Prolonged diabetes eventually leads to severe health complications, especially cardiovascular disease, diabetic nephropathy and retinopathy, fatigue, thirst, frequent urination, and blurred vision. 2 Balancing blood glucose level is the main strategy to prevent diabetic complications. 3
Increased glucose levels in the circulation trigger insulin secretion from the pancreas. Insulin is transcribed and expressed in the pancreatic beta-cells. It is then exported to the body through the portal circulation to the liver, where the liver's hepatocytes partially clear it with over 50% of the insulin on the first pass. 4 The leftover insulin leaves the liver through the hepatic vein and travels to the heart via the venous system. 5 Through arterial circulation, insulin is delivered to the rest of the body. Insulin helps to produce vasodilation all along the arterial tree. At the level of microvasculature, insulin leaves the bloodstream and enters muscle, hepatocytes and adipocytes, where it promotes glucose transporter-4 (GLUT4) translocation to the plasma membrane (PM) and glucose absorption. Insulin journey is terminated by degradation in the kidney. 6
Binding of the insulin to its receptors on the PM induces the translocation of glucose transporter-4 (GLUT4) from intracellular vessels to PMs, which results in increased diffusion of glucose in muscle, liver, and adipose tissues. 7 Once binds to its receptor (insulin receptor, IR), it activates the IR tyrosine kinase activity toward autophosphorylation by inducing structural rearrangement of the transmembrane domains to bring them into close proximity with each other, and the consequent activation of the IR tyrosine kinase toward phosphorylation of its major substrates IRS1, 2. Phosphorylation sites on IRS1,2 constitute entropic information to attract class I Phosphoinositide 3-kinase (PI3K-I), which rapidly generates membrane domains enriched in Phosphatidylinositol 34,5-trisphosphate (PIP3). PIP3 attracts the pleckstrin homology (PH) domain of Protein kinase B (PKB), also known as Akt, which makes the protein available for phosphorylation. Activated Akt1,2 migrates to the cytosol and intracellular membranes, where it phosphorylates AS160, a substrate of 160 kD. The phosphorylation of AS160 inhibits its ‘GTPase-activating protein’ (GAP) activity; hence, insulin signaling leads to inactivation of an inhibitor of Rab GTPases that regulate vesicle fission, destination, and fusion and ultimately GLUT4 translocation to the PM.8,9 Parallel to the insulin signaling pathway, muscle contraction leads to increased cellular [AMP]/[ATP] ratio and deprivation of oxygen and glucose. These conditions trigger the AMP-activated protein kinase (AMPK) leading to augmentation of GLUT4 trafficking to the PM.8,9
Insulin resistance and the subsequent development of type 2 diabetes are associated with aberrant GLUT4 translocation to the PM, thereby contributing to hyperglycemia.
9
Numerous plant species have been explored for their potential to manage hyperglycemia.10–12 Searching for bioactive phytochemicals possessing anti-diabetic action can aid in treating diabetes especially when its mechanism of action is described.
Okra fruit has long been used as a dietary medicine to treat heart and liver disorders, hypertension, obesity and diabetes.16–18 Moreover, AE extracts were previously reported to possess polyphenolic compounds, flavanols derivatives, riboflavin, thiamine, folic acid, niacin, carotene, vitamin C and amino acids.19,20 However, comprehensive experimental validation of its anti-diabetic effects, particularly through integrated
Materials and Methods
Materials
Mice standard food purchased from Envigo, Israel. STZ (Streptozotocin) (Sigma, S0130), polyclonal anti-myc (A-14) (Sigma, C3956), the MTT reagent (Sigma, 475989), methoxyamine hydrochloride, pyridine, N-methyl-N-(trimethylsilyl)-trifluoroacetamide (MSTFA), and other standard chemicals were purchased from Sigma Aldrich (St. Louis, MO, USA). Rat L6 muscle cell line, stably expressing myc-tagged GLUT4 (L6-GLUT4myc cells) were purchased from Kerafast (Boston, MA, USA). α-MEM (modified Eagle's medium) standard culture medium (Sigma, M4526
Plant Extract Preparation
Silylation Derivatization
A solution of methoxyamine hydrochloride (MeOX) in pyridine (40 µL of 40 mg/mL) was introduced to each dried sample (10 mg) along with 10 µL of a ribitol standard solution (0.2 mg/mL). The mixture was shaken at 37 °C for 1.5 h. Following this, 90 µL of MSTFA was added to the sample, and the mixture was shaken vigorously for an additional 30 min at 37 °C. The prepared samples were stored in glass vials (2 ml) with micro-serts (200 µL) and promptly sealed. A 1 µL aliquot of each derivatized sample was injected into a GC/MS for analysis. 21
Gas Chromatography-Mass Spectrometry Analysis
GC/MS analysis was performed on Agilent 6850 GC (Agilent, Palo Alto, CA, USA), equipped with Agilent 5975C single quadrupole MS, CTC-PAL RSI 85 auto-sampler, and HP-5MS (5% phenyl-polymethylsiloxane) capillary column (0.25 µm × 30 m × 0.25 mm) (Agilent, Palo Alto, CA, USA). The following conditions were applied: injector temperature, 250 °C; initial temperature, 50 °C for 5 min; gradient of 5 °C/min until 180 °C; gradient of 10 °C/min until 270 °C and a hold time of 10 mi., and increasing to 320 °C. The MS parameters were set as follows: source temperature, 230 °C; transfer line, 325 °C; quadrupole: 150 °C; Detector: 325; positive ion monitoring; and electron ionization (EI)-MS measurement at 70 eV. Helium was used as a carrier gas, at 0.6 mL/min. 16
Identification of Components
The percentage composition of the samples was computed from the GC peak areas, Agilent MassHunter software was used for data analysis, including alignment, annotation, and peak area calculation. Upon annotation all peaks from each sample, total peak area was calculated, followed by specifying the percentage peak area for each. Library searches were conducted using the National Institute of Standards and Technology (NIST14) GC/MS library and mass spectra from the literature. 21 For compound annotation, a minimum match factor of 70% was set, and all sugars, amino acids, fatty acids, sterols, tocopherols, and phenolic acids were identified by comparing their retention time and mass spectra to those of commercial standards (Merck company).
DPPH Scavenging Activity
Stock solutions at a concentration of 1 mg/ml in methanol were prepared from Trolox, AEM and AEH extracts. Each one of these stock solutions was diluted in methanol to prepare the working solutions with the following concentration: 2, 5, 10, 20, 50 and 100 µg/ml. DPPH solution was mixed with both methanol and with each of the above-mentioned working solutions at 1:1:1 ratio. In addition, a negative control solution was prepared by mixing DPPH solution with methanol in 1:1 ratio, while Trolox was used as a positive control. All these solutions were incubated for 30 min at room temperature in a dark cabinet. The optical density of these solutions was determined spectrophotometrically at a wavelength of 517 nm (Jenway 72000, Cole-Palmer, Vernon Hills, IL, USA). The antioxidant activity of Trolox and 
Cell Culture
L6 rat muscle cell line stably expressing myc epitope at the first exofacial loop of GLUT4 (L6-GLUT4myc cells; Catalog no. ESK202-FP) were purchased from (Kerafast, Boston, MA, USA) and grown in a cell culture incubator at 37 °C and 5% CO2. Myoblasts were maintained in
Cytotoxic Assay
L6-GLUT4myc cells were seeded in 96-well plates at a density of 20 000 cells per well, left for one day and then incubated with 
Determination of Surface GLUT4myc
The GLUT4myc relative distribution on the PM of intact L6 muscle cells was assayed as previously described.
23
Briefly, cells were grown in 24-well plates (4 × 104 cells per well), left for one day, after which
Animals’ Treatment
C57BL/6 mice were purchased from Jackson Laboratory (ME, USA). 14-week-old male and weighing approximately 35 g were used and allowed to adapt for 1 week before the study. Mice had free access to chow diet (24% protein, 4.7% fat, 40% carbohydrate, and 4% crude fiber) and water and were maintained at 25 ± 2 °C with 12/12 h light/dark cycles. All animal experiments conducted in accordance with the recommendation of the Animal Care and Use Committee of Arab American university. Sex-specific differences in glucose metabolism and insulin sensitivity are well documented and influenced by hormonal and molecular factors. Accordingly, only male mice were used in this study to reduce variability and ensure consistency in metabolic assessments. 24
STZ was intraperitoneally injected at a dose of 65 mg/kg (in 50 mM citrate buffer, pH 4.5) the same volume of 50 mM sodium citrate was injected into the control group (Vehicle). After STZ injection, blood samples were obtained from the tail vein, and glucose levels were monitored using a glucometer (Abbott, Abbott Park, IL, USA). Mice with blood glucose levels >200 mg/dL were selected and separated into two groups: Diabetic none treated group and diabetic treated with AEM group (n = 6). AEM, 100 mg/kg body weight, was administered orally by gavage daily for 5 weeks. This concentration was chosen based on comparable extracts previously tested in our laboratory. 22 The same volume of water-methanol was administered to the vehicle control normal and diabetic group. Bodyweight, food and water intake were monitored daily. Blood glucose levels were monitored weekly. 22 The mice treatment steps are illustrated in the below picture.

Statistical Analysis
The data were expressed as mean ± SEM. One way ANOVA test was used to investigate statistically significant differences. A level of
Results
This study focused on testing the chemical composition, antioxidant activity, cytotoxicity, and antidiabetic activity
Toxicity of A. esculentus Extracts
Three distinct AE extracts were prepared in water, methanol and Hexane. The extract's toxicity was evaluated in rat L6 muscle cells using MTT assay. Extract concentration that led to more than 10% cell death was considered as toxic. The water extract was viscous and toxic at low concentrations compared with the methanol and hexane extracts. AEW was non-toxic up to 0.125 mg/ml (Figure 1A) as it led to more than 15% cell death when cells were exposed to 0.25 mg/ml. AEM was safe for more than 1 mg/ml, as cell viability was 111 ± 7.5% when treated with 1 mg/ml of the extract (Figure 1B). AEH considered safe up to 0.25 mg/ml as cell viability was 101 ± 8.5% but dropped drastically to 9 ± 8.5% at 0.5 mg/ml (Figure 1C).
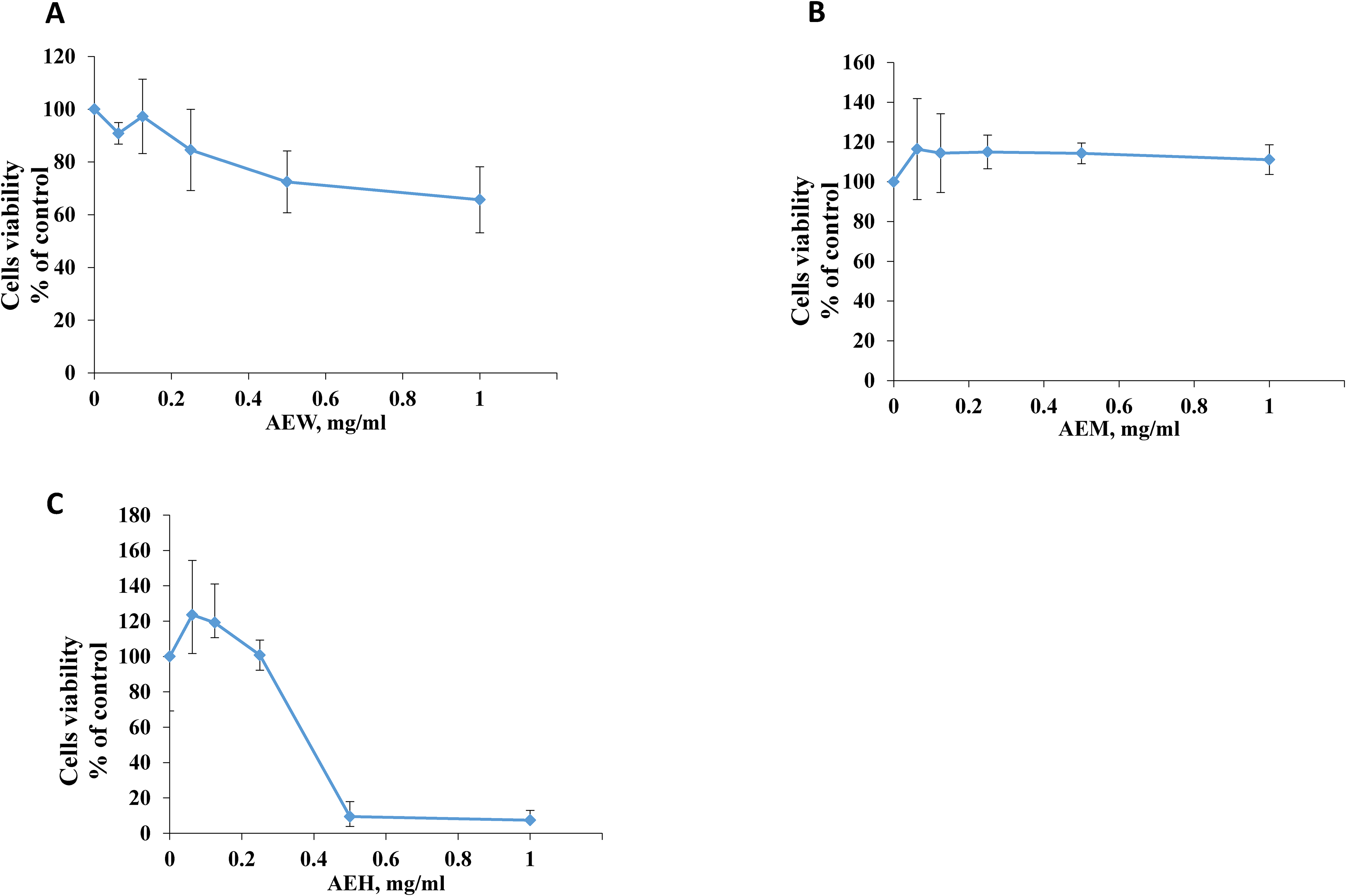
Effect of AE extracts on cell viability assessed by MTT assay. L6-GLUT4myc cells (20,000 cells/well) exposed to AEW (A), AEM (B) and AEH (C) for 24 h and cells viability was tested. Values represent as a % of untreated control cells (mean ± SEM), n = 3.
Effects of A. esculentus Extracts on GLUT4 Translocation
GLUT4 is the main glucose transporter in muscle, and it is continuously recycled between intra cellular vesicles and the PM. Insulin, muscle contraction and other stimuli, shifts GLUT4 toward the PM leading to increased glucose uptake. AEW, AEM and AEH effect on GLUT4 translocation to the PM was evaluated. L6-GLUT4myc myoblasts exposed to two different safe non-toxic concentration of each extract for 24 h without and with insulin as described in the methods section. AEW did not enhance GLUT4 translocations in cells treated with up to 0.125 mg/ml. It even seems that AEW slightly decreased GLUT4 translocation (statistically not significant) especially in the presence of insulin from 128 ± 5% to 92 ± 22 (Figure 2A). AEM was the most effective in augmenting GLUT4 translocation to the PM. GLUT4 translocation increased from 100% and 134 ± 12% in control cells without and with insulin respectively, to 216 ± 40 and 249 ± 30 when cells were exposed to 0.25 mg/ml in the absence and presence of insulin respectively. GLUT4 translocation increased even more when cells were exposed to 0.5 mg/ml and reached 240 ± 81% and 263 ± 88% without and with insulin respectively (Figure 2B). AEH was less effective as GLUT4 translocation was increased to only 124 ± 21% and 174 ± 29% without and with insulin respectively when cells were treated with the highest nontoxic concentration of AEH, 0.125 mg/ml (Figure 2C).

Effect of AE extracts on Glucose transporter type 4 (GLUT4) translocation to the plasma membrane. L6-GLUT4myc cells (150,000 cells/well) were exposed to AEW (A), AEM (B) and AEH (C) for 24 h. At the last 20 min of exposure to AE extracts, cells were either treated without (-) or with (+) 100 nM insulin, and the relative amount of GLUT4 at the PM was quantified using an antibody coupled colorimetric assay. Values are expressed as the mean ± SEM, relative to untreated control cells, of three independent experiments conducted in triplicate. *p < 0.05 significant as compared with (a) - ins control group, (b) + ins control group.
This result suggests a possible additive efficacy between
Effect of AEM on Diabetic Mice Mass and Blood Glucose Levels
The effect of AEM on blood glucose levels in the STZ-injected mice is depicted in Figure 3(A). The treatment of diabetic C57BL/6 mice with AEM for 5 weeks led to significant reductions in blood glucose levels compared to diabetic control group (p < 0.05). The effect was significantly appreciated after 1 week of administration. At day 36, the blood glucose level in the diabetic control group mice was 455 ± 86 mg/dL compared to 227 ± 73 mg/dL in AEM treated mice, respectively (Figure 3A). Blood glucose levels in non-diabetic mice were around 100 mg/dL with and without AEM all along the experiment phase.

The effect of oral administration of 100 mg/kg AEM on blood glucose levels (A) and body weight (B) of STZ-induced diabetic mice. Values are expressed as mean ± SEM. *p < 0.05, significant as compared with diabetic vehicle treated group.
AEM did not affect the control non-diabetic mice mass, which was 32 to 34 gr during the experiment period. As expected, diabetic mice mass was reduced from 34 ± 1 gr on the first day of the experiment to 23 ± 2 gr on day 42. However, AEM was able to partially restore the diabetic mice mass. Diabetic mice mass treated with AEM decreased only from 34 ± 2 to 27 ± 1 gr (Figure 3B).
A. esculentus Extracts DPPH Scavenging Activity
The effect of potential antioxidants on DPPH is attributed to their hydrogen-donating ability. 25 The antioxidant activity of AEM and AEH extracts was evaluated by the DPPH scavenging activity assay. AEM and AEH scavenging activity was tested up to 100 µg/mL and reached 69 ± 1.2% and 36 ± 1%, respectively (Figure 4). None of the extracts led to maximal scavenging like Trolox, but AEM was more efficient in DPPH scavenging, in correlation with its higher content of antioxidant compounds (Table 1, see discussion for more details).

DPPH radical scavenging activity of AEM and AEH. Trolox was used as a positive control. Data are presented as a mean ± SEM of three independent triplicates.
Gc/MS Phytochemical Profile of

Phytochemical Analysis of A. esculentus Extracts
Our GC/MS analysis results revealed overall 62 detected compounds in
14 compounds in the AEM and 12 in the AEH are known to possess anti-diabetic activity (highlighted in bold, Table 1). 2-Pyrrolidinone and docosanoic acid found in AEM and AEH as well as 10-heptadecenoic acid and 1-monopalmitin found only in AEH are reported to inhibit α-glucosidase activity which is beyond the scope of this manuscript. 17 compounds were reported to treat diabetic animals, enhance the insulin signaling pathway and augment GLUT4 translocation to the PM and thus glucose disposal (references in Table 1). Three out of them namely, myo-Inositol, malic acid, and stearic acid are present in both extracts. Five were detected in AEH only: pentadecanoic acid, 9-hexadecenoic acid, phytol, α-linolenic acid and octadecanoic acid. Eight were detected only in AEM: pyroglutamic acid, trigonelline, ferulic acid, N-acetyl-D-glucosamine, caffeic acid, γ-tocopherol, stigmasterol and β-sitosterol. The total percentage of these 17 compounds was 5.75% of the total area in AEM and only 0.567% of AEH. This could explain why AEM extract was more potent in increasing GLUT4 translocation to the PM (Figure 2). Palmitic Acid (PA) possess 37.25% and 1.02% of AEH and AEM detected phytochemicals respectively. The antidiabetic activity of PA is controversial (refer to the discussion for more details).
Discussion
Recently, there is increased interest in the search for organic nutrients and traditional medicinal herbs including antidiabetic and antioxidant medicinal plants. These herbs’ medical activity is attributed to some phytochemical classes especially flavonoids, polyphenols, terpenoids and carotenoids for reducing the damaging effects of free radicals especially in patients suffering from diabetes mellitus.
48
In this regard
Table 1 summarizes several phytochemical compounds present in AEM and AEH extracts such as phenols and alkaloids. Higher quantities of phytochemical contents were presented in AEM compared to AEH. These findings were supported by the reports from previous studies, which revealed the presence of alkaloids, flavonoids, carbohydrates, phenols, proteins, tannins, terpenoids, and sterols in these fractions.54,55
This study reports the presence of phenolic compounds in
While antioxidant activity may contribute to metabolic protection under diabetic conditions, the antidiabetic effects observed in this study are more directly linked to modulation of glucose homeostasis pathways. Specifically,
Whole-body insulin-mediated glucose homeostasis is associated with GLUT4 recycling between the PM and intracellular vesicles and its expression levels.60,61 The connection between insulin resistance and GLUT4 has received increasing attention, and some intriguing findings have been reported.
62
Insulin-resistant glucose transport in adipocytes from obese and diabetic subjects is connected with lower GLUT4 mRNA and protein expression, according to research by Berger et al, Cushman et al, and Sivitz et al,.62–64 These findings confirm GLUT4's role in insulin-dependent glucose homeostasis. Thus, a modest upregulation of GLUT4 expression serves as a useful target for the management of insulin resistance in patients. We have recently revealed that the expression of GLUT4 in L6 cells exposed to AEM was also considerably increased (Data not published). AEM and AEH demonstrated significant activation of GLUT4 translocation into PMs in the current
The difference of GLUT4 translocation increasing effect between fractions is attributed to the difference in the compounds’ composition in each fraction. Some of these phytochemicals are suggested to increase GLUT4 expression, transport activity and translocation to the PM as detailed in Table 1. GC/MS analysis for AEM revealed several phytosterols such as stigmasterol and β-sitosterol, phenolic acids like ferulic acid and caffeic acid, fatty acids like palmitic acid, nitrogen-containing compounds like N-Acetyl-D-glucosamine and sugars like. myo-inositol. These compounds are effective in increasing GLUT4 translocation to the PM.21,53 Wang et al showed that stigmasterol improves GLUT4 basal expression in cells and partially rectify the GLUT4 translocation defect caused by insulin, thereby reducing the severity of insulin resistance in KK-Ay animals.
44
Ponnulakshmi et al showed that β-sitosterol enhances glycemic control by activating IR and GLUT4 in adipose tissue of high-fat and sucrose-induced type-2 diabetic rats. Results from
Our GC/MS analytical results show that extracts from
Myo-inositol accounted for 1.7% of AEM and 0.052% of AEH. Derivatives of inositol are naturally occurring substances in both plants and animals which, according to reports, support insulin's activity in promoting glucose uptake by moving GLUT4 to the PM, especially in muscle tissues. 68 When given orally to C57BL/6 mice, myo-inositol stimulates GLUT4 translocation in skeletal muscle, decreasing postprandial plasma glucose and insulin levels. 66
Stearic acid accounted for 0.1% of AEM fraction and 2.9% of AEH fraction. Stearic acid serves as a potent PTP1B inhibitor, possibly causing an enhancement in the insulin receptor signaling to stimulate glucose uptake into adipocytes. 69
Trigonelline accounted for 0.021% of the AEM fraction. It was shown to activate PPAR-γ, and thus decrease insulin resistance and diabetic nephropathy in T2DM rats. 30 Trigonelline also enhanced insulin sensitivity in soleus muscle by upregulating GLUT4, pT308-Akt, and insulin receptor autophosphorylation, ultimately leading to decrease in blood glucose and insulin. 70
Ferulic acid accounted for 0.014% of AEM extract. A previous study reported that ferulic acid stimulates the absorption of glucose and increases the production of glycogen in insulin-resistant muscle cells. Further mechanistic research revealed that ferulic acid significantly increases the activity of the transferrin receptor-containing endosomal compartment through the PI3K/atypical protein kinase C-dependent pathway.
71
Moreover, trans-ferulic acid also increases AMPK activation in a time- and dose-dependent way and increases the phosphorylation of acetyl-CoA carboxylase (ACC), indicating that it might encourage the oxidation of fatty acids; as such it might possess antilipidemic and antidiabetic activity. Trans-ferulic acid also raised glutathione levels while lowering intracellular reactive oxygen species (ROS) and nitric oxide (NO) brought on by hyperglycemia. Interestingly, it also upregulated the glucose transporters, GLUT2 and GLUT4 gene expression, and inhibited c-Jun N-terminal protein kinase (JNK1/2) by lowering the phosphorylation level in tested cells under high glucose conditions in hyperglycemia-induced human HepG2 liver cells and rat skeletal muscle L6 cells.
72
In alloxan-induced diabetic mice, ferulic acid (10 mg/kg body weight) increased the levels of the antioxidant enzymes glutathione peroxidase (GPx) and super oxide dismutase (SOD) and decreased the levels of glucose in the kidney and serum.
73
In an independent study oral ferulic acid (0.05 g/kg) treatment markedly raised plasma insulin levels and decreased blood glucose in C57BL/KsJ db/db mice.
74
These results are in line with our results reported here for the antidiabetic activity of the AEM extract
Pyroglutamic acid accounted for 0.112% out of the AEM fraction. Previous study demonstrated that oral glucose tolerance and serum insulin levels were both reduced by diet containing 0.05% pyroglutamic acid in Goto-Kakizaki (GK) rats and KK-Ay mice. Rats fed with pyroglutamic acid exhibited higher ability to tolerate glucose. Moreover, their Glucose-6-Phosphatase gene expression was suppressed in the GK-Pyroglutamic acid group compared to the GK-control group. 27
Caffeic acid accounted for 0.028% of the AEM fraction. Caffeic acid is known for its anti diabetic activity. It enhanced the GLUT4 protein expression in adipose tissue. 75 Oral administration of caffeic acid resulted in a significant enhancement of serum insulin level and a decrease in blood glucose level of diabetic rat models. 76 Moreover, our group reported recently that caffeic acid binds to amylase and glucosidase and inhibits their activity, thus decreasing polysugars digestion and absorption to the body. 77 In this study, blood glucose level in diabetic mice fed AEM was significantly reduced. This could be in part due to inhibiting digestion enzymes in addition to stimulating GLUT4 activity and reducing the oxidative state of the animals.
Stigmasterol was present at 0.122% of AEM fraction. Wang and colleagues reported that oral glucose tolerance and insulin resistance were improved in KK-Ay mice who received oral stigmasterol (50 mg/kg/day) for four weeks. Moreover, this treatment also lowered their fasting blood glucose and blood lipid indexes, including cholesterol and triglycerides. More telling and in line with our reported results here, stigmasterol increased in GLUT4 expression in white adipose tissue, skeletal muscle, and L6 cells. 44
β-Sitosterol was detected at 0.221% of AEM fraction. It was reported that diabetic rats treated with up to 20 mg/Kg β-Sitosterol for 21 days had significantly lower serum glucose levels compared with diabetic untreated rats. 78 One more study reported that diabetic rats treated with β-sitosterol exhibited improved glycemic control and activated GLUT4 and IR in the adipose tissue. 46 These results are in line with our reported results here for the AEM extract as it significantly augmented GLUT4 translocation to the PM and dramatically reduced hyperglycemia in diabetic mice. AEM activity could be in part attributed to β-Sitosterol as well as to the above reported phytochemicals.
Palmitic Acid (PA) possessed 37.25% and 1.02% of AEH and AEM area respectively. The antidiabetic activity of PA is controversial. PA acts as anti diabetic agent in short term treatments (60 min),79,80 but it causes insulin resistance in long term treatments (16-24 h).81,82 In this study, L6 muscle cells were exposed to AE extracts for 24 h. The high content of PA in AEH could explain its low GLUT4 enhancnet compared to cells exposed to AEM.
Based on the present
In summary, this study revealed that
Conclusion
This study demonstrates that
Supplemental Material
sj-docx-1-npx-10.1177_1934578X261433112 - Supplemental material for In Vivo and in Vitro Evaluation of the
Anti-Diabetic Activity of Chemically
Analyzed Abelmoschus esculentus Extracts
Supplemental material, sj-docx-1-npx-10.1177_1934578X261433112 for
Supplemental Material
sj-pptx-4-npx-10.1177_1934578X261433112 - Supplemental material for In Vivo and in Vitro Evaluation of the
Anti-Diabetic Activity of Chemically
Analyzed Abelmoschus esculentus Extracts
Supplemental material, sj-pptx-4-npx-10.1177_1934578X261433112 for
Footnotes
Acknowledgements
We are thankful to Almaqdisi and AAUP Research Foundations for providing financial support.
Ethical Approval
All animal experiments were conducted in accordance with the recommendations of the Animal Care and Use Committee of the Arab American University. The study protocol was reviewed and approved by the Institutional Animal Care and Institutional Review Board (IRB) of the Arab American University. No human subjects were involved in this study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Arab American University Palestine (AAUP) Research Foundation and the Almaqdisi Program (French–Jerusalem Joint Program; Consulat Général de France à Jérusalem).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. No human participants were involved in this study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
