Abstract
Objective
The current study's objective was to evaluate the antioxidant activity and the phytochemical composition of the leaves of Rosa abyssinica and Justicia schimperiana.
Methods
The crude extracts (80% methanol) and solvent fractions (aqueous, ethyl acetate and n-hexane) were prepared utilizing cold maceration and solvent fraction methods, respectively. The total alkaloid content (TAC) was quantified for the dried leaf powders. The total phenolic (TPC) and flavonoid (TFC) contents of the crude extracts were also assessed. The antioxidant activity of the crude extracts and solvent fractions was assessed by 2,2-diphenyl-1-picrylhydrazyl (DPPH), 2,2'-Azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) and ferric reducing antioxidant power (FRAP) assays. Analysis using fourier transform infrared (FTIR) spectroscopy was also carried out.
Results
The leaves of R. abyssinica exhibited a higher TAC (8.89%). Both TPC (11.58 ± 0.35 mg GAE/g of extract) and TFC (13.11 ± 0.97 mg QT/g of extract) were observed to be higher in the crude extract of R. abyssinica. Among all tested extracts, R. abyssinica's n-hexane fraction (RANF) showed the highest activity specifically in DPPH (IC50 = 0.24 ± 0.01 mg/ml) and ABTS (IC50 = 0.15 ± 0.03 mg/ml) radical scavenging activity. The aqueous fraction (RAAF) exhibited the strongest activity in FRAP assay (6.49 ± 0.21 mg AA/g of extract). From the Fourier transform infrared (FTIR) spectroscopic analysis, C-H, C = O, O-H and C-O functional groups were identified in the crude extracts of both plants.
Conclusion
These findings provide a basis for future investigation aimed at identifying bioactive compounds and exploring their potential therapeutic applications.
Introduction
An imbalance between free radicals and the body's ability to detoxify them creates oxidative stress. 1 Antioxidants interrupts the oxidation mechanism by scavenge free radicals and other reactive oxygen species (ROS). 2
Experimental studies are revealing the ability of many different plants with phenolic content to prevent the damage caused by oxidative stress.3,4 Rosa abyssinica Lindley and Justicia schimperiana (Hochst. Ex Nees) T. Anders have been reported to be traditionally used in the management of various ailments. Rosa abyssinica Lindley (Rosaceae) locally known as Kega, is among several herbs widely used in Ethiopian folkloric medicine. It is used for the treatment of many diseases, including rheumatic pain, hypertension, scabies, helminthic infection, glandular tuberculosis, diabetes, tapeworm, stomachache and cough.5,6 Experimental studies also revealed the presence of anti-inflammatory, antinociceptive, antidiabetic, antimicrobial and antidepressant-like activity in the plant.6–8
Justicia schimperiana is from the Acanthaceae family and is locally known as sensel in Ethiopia. Its growth habit is shrub. In Ethiopian traditional medicine, it is commonly used to treat jaundice, rabies, asthma, the common cold, stomach cramps, abdominal and intestinal parasites, diarrhea, and wounds.9,10 It has been experimentally demonstrated that this plant exhibits antioxidant, anti-inflammatory, anti-diabetic and wound healing activity.11–13
Despite the widespread use of these plants in Ethiopian traditional medicine, there is limited evidence to support their efficacy and phytochemical composition. Thus far, no studies have documented the antioxidant activity of R. abyssinica leaves and investigations of J. schimperiana have been limited to crude extracts. However, studying solvent fractions provides a better understanding of the distribution of bioactive compounds in polar and nonpolar ingredients of the plant. 14 Additionally, FTIR analysis has not been reported previously for R. abyssinica leaves and has been scarcely reported for J. schimperiana. Therefore, the implementation of this analysis can be an important tool for assessment and prediction of chemical constituents and biological potential of medicinal plants. 15
Hence, considering the high value of these two plants and the increasing demand for new antioxidant agents, it is beneficial to explore the antioxidant capacity and phytochemical composition of the leaves of Rosa abyssinica and Justicia schimperiana.
This research was designed to assess the antioxidant activity as well as the TPC, TFC and TAC of the leaves of these two plants. It is also aimed to determine the various functional groups in the crude extracts using FTIR.
Materials and Methods
Collection and Authentication of Plant Samples
The fresh leaves of Rose abyssinica and Justicia schimperiana were gathered from Addis Ababa, Ethiopia, with specific locations from Yeka sub-city (9° 4'13.13"N: 38°46'28.69"E at elevation 2634 m) and Gulele sub-city (9° 2'30.55"N: 38°46'37.53"E at elevation 2432 m), respectively.The plant specimen authentication was carried out at the National Herbarium, Addis Ababa University, by Mr Melaku Wondaferash with voucher numbers MS001 and MS002 (R. abyssinica and J. schimperiana, respectively).
Plant Crude Extract and Solvent Fraction Preparation
The plant leaf extraction and fractionation were conducted using a method described by Ayalew et al. (2022) 16 The cold maceration technique was used to prepare the crude extracts. The gathered leaves were cleaned and dried under the shade. After that, they were ground using a grinder into a fine powder. After that, 80% methanol was used to soak 600 g of plant powder in a 1:5 mass-to-volume ratio. The mixture was macerated on a shaker for four consecutive days, followed by filtering it using Whatman No. 1 filter paper. A rotary evaporator (40 °C) was used to concentrate the filtrates. The extracts were kept at −4 °C until further use.
To prepare the solvent fraction, the 80% methanol crude extract of both plants was further fractionated using the solvent fractionation method according to. 16 The crude extract (70 g) was fractionated using distilled water, ethyl acetate and n-hexane. Distilled water (280 mL) was used to dissolve the specified amount of crude extract. subsequently, the dissolved extracts were diluted with an equivalent proportion of n-hexane. In order to create a transparent layer, the mixture was placed in a separatory funnel and allowed to stand for hours while being occasionally stirred. The n-hexane component was then removed after two clear layers had formed. Two further macerations of the residue were performed using the same amount of n-hexane and following the same procedure. After collecting the n-hexane filtrates (NF), 280 mL of ethyl acetate was poured onto the residue and a similar procedure was performed. After a clear layer was formed, the ethyl acetate fraction (EF) was removed. The remaining residue was designated as an aqueous fraction (AF). NF and EF were concentrated under reduced pressure using rotary evaporator and the remaining solvent was removed in a hot oven at 40 °C, and the AF was subjected to lyophilizaton. The fractions were then stored at −4 until they were needed. 16
The solvent fractions are named as follows: R. abyssinica 80% methanol crude extract obtained aqueous (RAAF), ethyl acetate (RAEF) and n- hexane (RANF) fractions. J. schimperiana 80% methanol crude extract obtained aqueous (JSAF), ethyl acetate (JSEF) and n- hexane (JSNF) fractions.
Total Phenolic Content (TPC) and Total Flavonoid Content (TFC)
TPC was quantified for the 80% methanol crude extracts obtained from both plants using the Folin–Ciocalteu assay. 17 Folin-Ciocalteu phenol reagent (0.2 mL) and 4.5 mL of distilled water was add to the sample extract (0.1 mL of 1 mg/mL). Then, sodium carbonate (Na2CO3) solution (0.5 mL) was poured into the mixture, which was then incubated for 60 min in the dark at room temperature. The mixtures absorbance was recorded at 725 nm by spectrophotometer. The unit of measurement used to express the TPC result was milligram of gallic acid equivalent per gram of extract (mg GAE/g).
The TFC of the 80% methanol crude extracts of both plants was evaluated by the aluminum chloride colorimetric procedure. Briefly, the sample (1 mg/mL) was diluted with a 2.8 mL distilled water, 1.5 mL ethanol (95%), 0.1 mL aluminum chloride (10%) and 0.1 mL potassium acetate (1 M). The mixture was left to incubate in the dark for 30 min. After that, a spectrophotometer reading was done at 415 nm wavelength to measure the absorbance. The milligram of quercetin equivalent per gram of extract (mg QE/g) was used to display the TFC result. 17
Total Alkaloid Content (TAC)
The TAC was assessed for the two plant leaf powders. Concisely, 10% acetic acid in ethanol was used to dissolve the plant powder (5 g). Afterward, low heat was applied to the mixture while stirring it for 60 min. Once the mixture had cooled to room tempreture, it was filtered with Whatman No. 1 filter paper. Subsequently, the filtrate was evaporated on a hot plate to make ¼ of its volume. After that, a few drops of concentrated ammonium hydroxide solution (NH4OH) were introduced to it and stirred to precipitate the alkaloid fully. Then filter the mixture on the pre-weighted Whatman No.1 filter paper and wash it with 1% ammonium hydroxide solution to remove any soluble impurities. After that, the filter paper was dried in the oven at 40 °C and the weight was measured using a balance. TAC percentage was calculated using the formula below. 18
Antioxidant Activity Test
DPPH (2,2-Diphenyl-1-Picrylhydrazyl) Assay
The DPPH assay was carried out using the protocol stated by Saeed et al (2012).
19
To prepare the DPPH stock, 24 mg of DPPH reagent was dissolved in methanol (100 mL). After that, the solution's concentration was adjusted with methanol to attain nearly 0.98 ± 0.02 at 517 nm absorbance. The experiment was done by combining 0.3 ml of DPPH solution with 0.5 ml of the plant extract at concentrations of 0.2, 0.4, 0.8, 1.2, 1.6 and 2 mg/mL. Ascorbic acid (standard) was measured at a concentration of 0.06, 0.12, 0.24, 0.36, 0.48 and 0.6 mg/ml). The mixture was then left at room temperature in the dark for 45 min. As a control, the blank absorbance value was employed. After that, the spectrophotometer absorbance reading was taken at 517 nm and the antioxidant potential was calculated as follows:
A = Absorbance
ABTS (2,2'-Azino-bis (3-Ethylbenzothiazoline-6-Sulfonic Acid)) Assay
The purpose of the ABTS radical scavenging assay was to determine the ability of the crude extracts and fractions to neutralize ABTS radicals.
20
The ABTS stock solution (7 mM) and potassium persulfate (2.45 mM) were combined in a 1:0.5 ratio to create the ABTS radical, which was then allowed to incubate at room temperature for the entire night in a dark cupboard. The absorbance of the ABTS radical was adjusted with ethanol to be 0.70 ± 0.02 at 734 nm. Then the plant extract (0.1, 0.3, 0.6, 0.9, 1.2 and 1.5 mg/mL) was combined with ABTS radical (3 mL) and the mixture was left to stand at room temperature for 6 min in the dark. Afterward, the spectrophotometric absorbance of the mixture was measured at a 734 nm wavelength. Trolox at the concentrations of 0.06, 0.18, 0.36, 0.54, 0.72 and 0.9 mg/mL served as a standard. The blank reading was taken as a control. The percent inhibition was calculated using the following formula:
A = Absorbance
Ferric Reducing Antioxidant Power (FRAP) Assay
The FRAP assay was undertaken to assess the extract's ability to donate electrons to reduce ferric ions (Fe3+) to ferrous ions (Fe2+). A fresh FRAP reagent was made by combining 10 mM TPTZ solution (24,6-Tripyridyl-s-Triazine) in 40 mM HCl, 20 mM FeCl3. 6H2O (BDH) and 300 mM acetate buffer (pH 3.6) in a 1:1:10 proportion, respectively. The antioxidant activity test was conducted by mixing 900 µL of FRAP reagent (pre-incubated for 10 min) with 90 µL of distilled water and 30 µL of the sample (0.5 mg/mL concentration). Thereafter, the mixture was allowed to react at 37 °C for 10 min. After that, a spectrophotometric of the mixture's absorbance was measured at 593 nm. Milligrams of ascorbic acid per gram of extract (mg AA/g) was used to illustrate the outcome.17,21
Analysis of Fourier Transform Infrared (FTIR) spectra
The 80% methanol extracts of both plants spectra was analyzed with a Fourier transform infrared (FTIR) spectrometer, UK (spectrum 65 FT-IR, PerkinElmer model). The data were obtained using Perkin Elmer Spectrum Software. A total of a hundred scans were performed with 4 cm−1 resolution by employing cosine apodization in the 4000–400 cm−1 region.
Analysis of Statistical Data
JMP Pro 14 software was used to perform descriptive analysis. The IC50 value was analyzed by GraphPad Prism 9. The graphs of DPPH and ABTS were prepared by using Microsoft Excel 2019. However, the FTIR graph was generated by Origin 2012 software. One-way analysis of variance (ANOVA) with Tukey-Kramer HSD was performed to compare the means of FRAP, DPPH and ABTS assays. However, the phytochemical contents (TPC, TFC and TAC) of the two plants were compared using an independent two-sample t-test. Statistical significance was accepted at p ≤ 0.05. Each experiment included triplicate measurements and the data are presented as mean ± standard error of the mean (SEM).
Results
Quantitative Phytochemical Analysis
Total Phenolic Content (TPC) and Total Flavonoid Content (TFC)
In this study, the 80% methanol crude extract of Rosa abyssinica exhibited significantly higher TPC (11.58 ± 0.35 mg GAE/g of extract) and TFC (13.11 ± 0.97 mg QE/g of extract). In contrast, the 80% methanol extract of Justicia schimperiana recorded lower values, with a TPC of 3.05 ± 0.58 mg GAE/g of extract and a TFC of 6 ± 0.24 mg QE/g of extract (Table 1).
Total Phenolic Content and Total Flavonoid Content of Justicia schimperiana (JS) and Rosa abyssinica (RA) Plant Leaves Crude Extracts.

All values are presented as means ± SEM.
The p- values are set for comparison among values within the same raw. Significant difference was considered at p ≤ 0.05.
TFC is presented as “mg QE/g of extract”.
TPC is presented as mg GAE/g of extract.
80% M = 80% methanol.
Total Alkaloid Determination
The total alkaloid analysis of R. abyssinia and J. schimperiana leaf powder was conducted. From 5 g of R. abyssinica leaf powder, 0.44 g of alkaloid was obtained, making up 8.89% of the total weight. In contrast, 5 g of J. schimperiana leaf powder yielded 0.0852 g of alkaloid, which is approximately 1.71% (Table 2).
Total Alkaloid Content of Justicia schimperiana and Rosa abyssinica Plant Leaf Powders.

Both values are presented as means ± SEM.
Significant difference were considered at p ≤ 0.05.
Antioxidant Activity
DPPH (2,2-Diphenyl-1-Picrylhydrazyl) Assay
In the present work, both the crude extracts and solvent fractions of R. abyssinica and J. schimperiana scavenged DPPH radicals in accordance with the dose level (Figure 1 and Figure 2). R. abyssinica's crude extract exhibited better activity (IC50 = 0.3 ± 0.05 mg/mL) than that of J. schimperiana's crude extract (IC50 = 1.34 ± 0.06 mg/mL). Among the solvent fraction, the strongest IC50 value was obtained from RANF (0.24 ± 0.01 mg/mL, followed by RAEF (0.26 ± 0.02 mg/mL) and RAAF (0.26 ± 0.03 mg/mL), which are all obtained from the 80% methanol extract of R. abyssinica (Table 3).
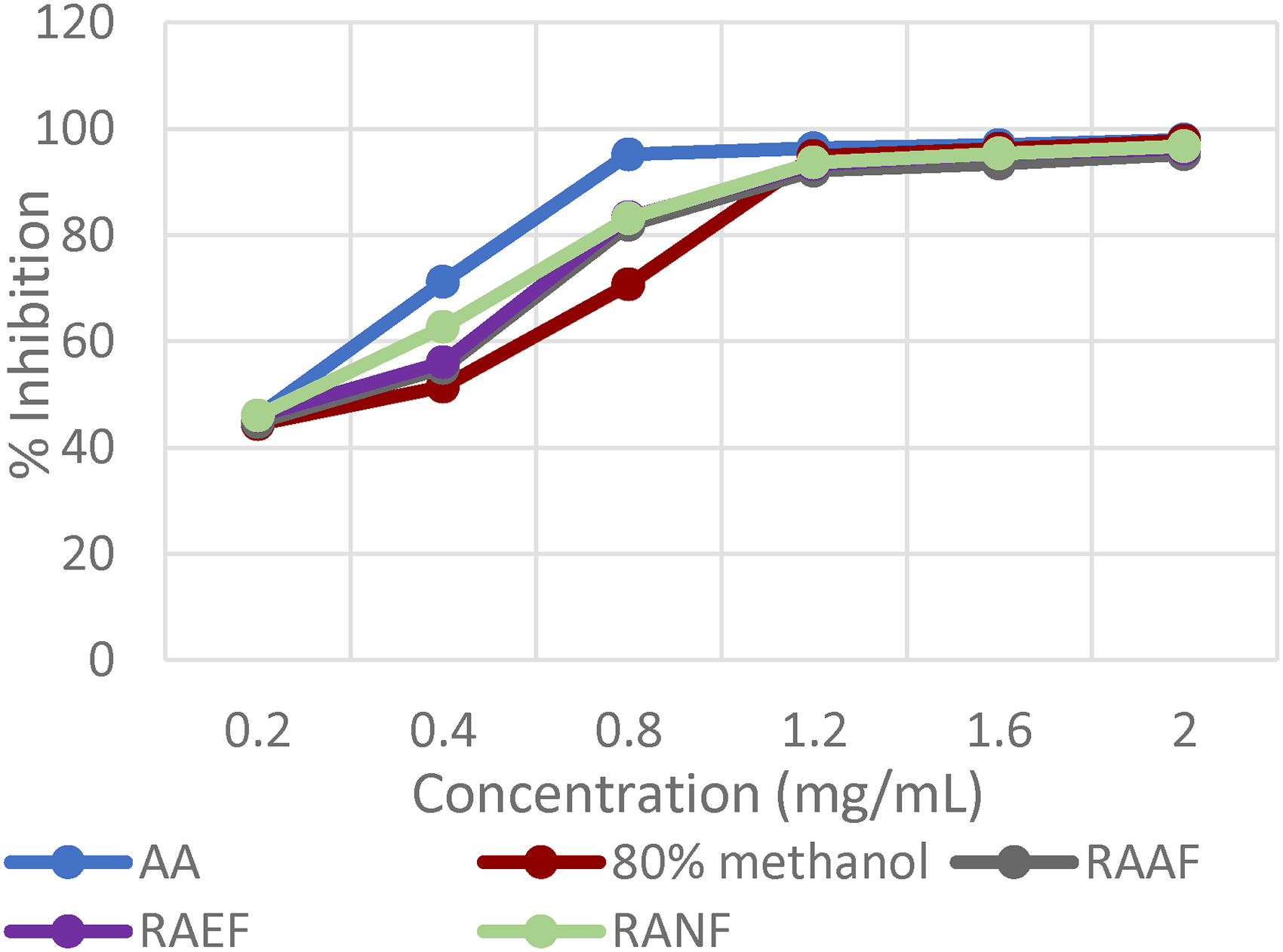
DPPH radical scavanging activity for Rosa abyssinica extracts.

DPPH radical scavanging activity for Justicia schimperiana extracts.
Antioxidant Activity of Rosa abyssinica and Justicia schimperiana Plant Leaves.

Mean ± SEM.
Values in the same column indicated by different letters are significantly differences (p ≤ 0.05).
R. abyssinica 80% methanol crude extract's- aqueous (RAAF), ethyl acetate (RAEF) and n- hexane (RANF) fractions.
J. schimperiana 80% methanol crude extract's- aqueous (JSAF), ethyl acetate (JSEF) and n- hexane (JSNF) fractions.
ABTS (2,2'-Azino-bis (3-Ethylbenzothiazoline-6-Sulfonic Acid)) Assay
All tested extracts of R. abyssinica and J. schimperiana exhibited antioxidant activity in a dose-dependent manner (Figure 3 and Figure 4). The crude extract of R. abyssinica showed higher activity among the crude extracts, with an IC50 of 0.34 ± 0.03 mg/ml whereas, the crude extract of J. schimperiana showed IC50 value of 0.37 ± 0.05 mg/ml. After these two crude extracts were fractionated, the highest activity was observed from RANF (0.15±0.03 mg/mL) obtained from R. abyssinica's 80% methanol extract, followed by RAAF and RAEF with IC50 values of 0.16 ± 0.02 and 0.16 ± 0.03 mg/mL, respectively. In general, the ABTS radical scavenging potency of the fractions can be placed as RANF > RAAF and RAEF > JSEF > JSAF > JSNF (Table 3).

ABTS radical scavanging activity for Rosa abyssinica extracts.

ABTS radical scavanging activity for of Justicia schimperiana extracts.
Ferric Reducing Antioxidant Power (FRAP) Assay
Among the crude extracts, R. abyssinica 80% methanol extract showed 5.11 ± 0.19 mg AA/g of extract which is the highest among all. Among its solvent fractions, 6.49 ± 0.21, 3.38 ± 0.00 and 3.23 ± 0.01 mg AA/g of extract were observed from RAAF, RAEF and RANF, respectively, which were the top three among all (Table 3).
Fourier Transform Infrared (FTIR) spectra Analysis
The FTIR spectra of the 80% methanol crude extracts of Justicia schimperiana and Rosa abyssinica are presented in Figure 5 and Table 4. In Figure 5, J. schimperiana 80% methanol crude extract indicated moderate transmittance. In the extract, the broad band observed at 34.22.16 cm−1 is in the O-H stretching, indicating the occurence of a hydroxyl functional group, confirming the solvent of extraction is an alcohol compound. The peak at 1634.47 cm−1 suggests the existence of a carbonyl (C = C) or aromatic group. The broadband observed at 1013.13 cm−1 in the fingerprint regions is attributed to C-O or C-N stretching vibration.

FTIR spectrum of the crude extracts of the leaves of Rosa abyssinia (RA) and Justicia schimperiana (JS).
FTIR Spectral Data of Justicia schimperiana and Rosa abyssinica Plant Leaves Crude Extracts.
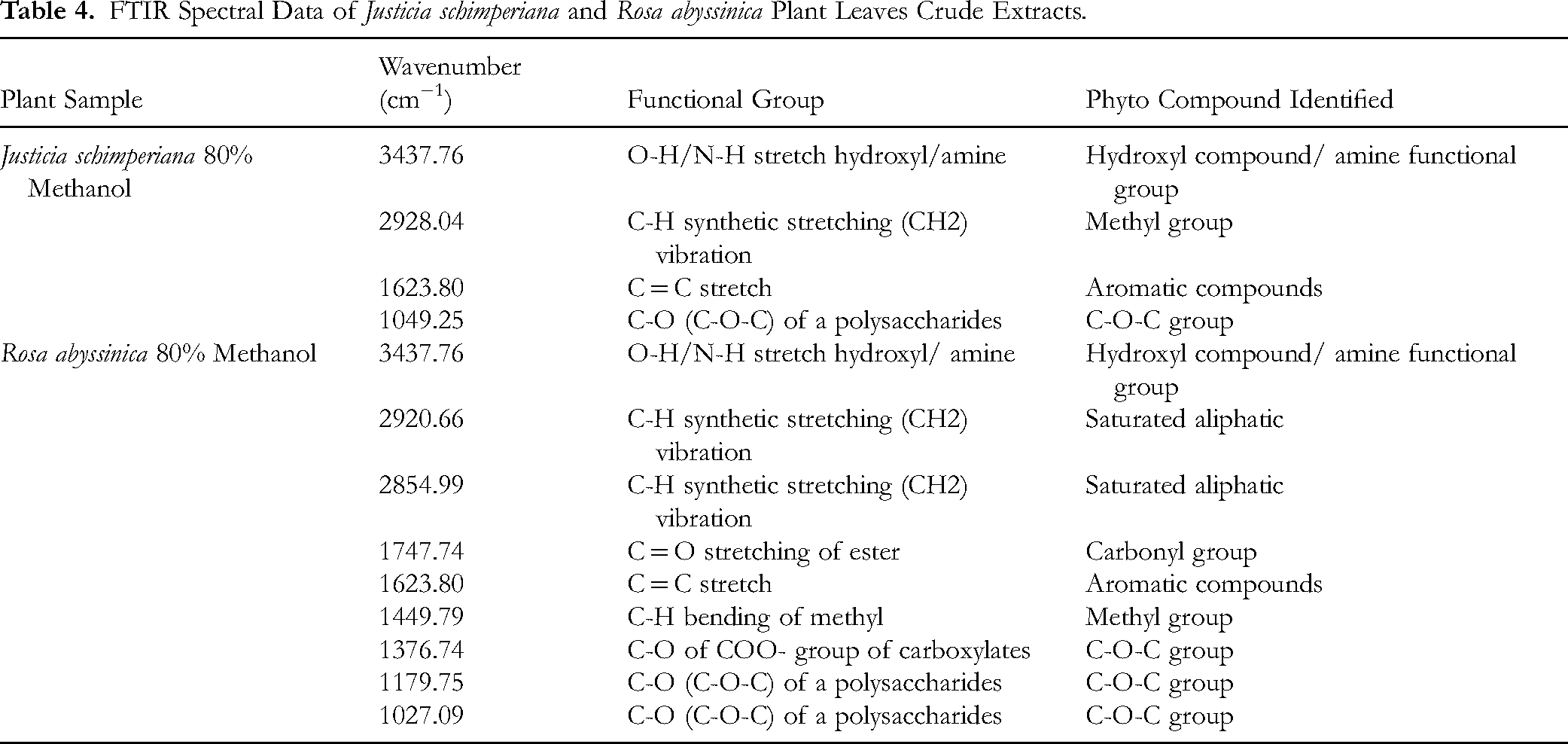
The FTIR spectrum of the 80% methanol crude extract of Rosa abyssinica showed higher transmittance, implying a lower concentration of absorbing compounds. The existence of hydroxyl groups was confirmed by a broad absorption band at 3437.76 cm−1, corresponding to O–H stretching, confirms the presence of hydroxyle group and suggests that the extract orignated from an alcohol-based solvent. The existence of C-O bonds or aromatic groups was also indicated by the peaks observed at 1027.09, 1179.75 and 1376.74 cm−1. Additionally, a transmittance peak at 1623,80 cm−1 reveals the presence of aromatic C = C stretching vibrations (Figure 5 and Table 4).
Discussion
Plants play an important role in the formulation of therapeutic drugs derived from their phytochemicals for treating various illnesses. 22 Plant-based phytochemicals, specifically, phenolics and flavonoids are reported to be important antioxidant agents due to their effectiveness in scavenging most oxidizing molecules. 23 Alkaloids have also been reported to impose antioxidant activity in many different ways. Among the most reported mechanisms is influencing NADPH oxidase which plays a central role in ROS production and the induction of synthesis or translocation of nuclear factor erythroid 2-related factor2 which is involved in cellular defense against oxidative stress. 24
In the present study, quantitative analysis revealed an alkaloid content of 1.71% in the leaves of J. schimperiana (Table 2). This finding is consistent with reports by G/giorgis et al (2022) and Gizaw et al (2022), who also revealed the presence of alkaloids in the leaf extract of the plant.12,25 However, a study by Eyasu et al (2024) reported the absence of alkaloid in the same plant part. 26 Such discrepancies among studies may result from variation in physiological factors, geographical location, environmental conditions and the season of sample collection, all of which strongly influence secondary metabolite biosynthesis. 27
Phenolics and flavonoids are key plant-drived antioxidant compounds, known for their ability to effectively scavenge reactive oxidizing molecules. 4 A previous study reported flavonoid and phenolic content of the leaves of J. schimperina as 82.43 mg QE/100 g of extract and 83.59 mg GAE/100 g of extract, respectively (equivalent to 0.8243 mg QE/g of extract and 0.8359 mg GAE/g of extract). 26 By contrast, our methanolic leaf extract from the same plant species contains 6 ± 0.24 mg QE/g of extract of TFC and 3.05 ± 0.58 mg GAE/g of extract of TPC (Table 1). When normalized to per gram value, the extract shows approximately 7.3-fold higher flavonoid and 3.7-fold higher phenolic content than the previously reported per-gram plant value. As mentioned in the discussion above, the difference likely arises from environmental or physiological factors influencing metabolite accumulation.
The phytochemical profiling of R. abyssinica leaves in the present study confirmed the presence of alkaloids, high total phenolic content and notable flavonoid content (Table 1 and Table 2). Similar findings have been reported for the fruits of the same plant, 28 suggesting that these metabolites are distributed across different plant parts. Expanding phytochemical investigation to include the roots and stems will deepen understanding of the synthesis and distribution of these secondary metabolites in R. abyssinica.
Considering that antioxidants work in different action mechanisms, there is no single universally accepted assay to stand alone in evaluating and qualifying the antioxidant abilities of plant extracts (23). Therefore, the current study used DPPH, ABTS and FRAP assays to evaluate the antioxidant properties of crude extracts and solvent fractions obtained from R. abyssinica and J. schimperiana plant leaves. These three assays are the most frequently used methods in the evaluation of antioxidant ability (1,24,25).
The DPPH assay relies on the DPPH radicals scavenging capacity of the extract, as evidenced by their ability to bleach the purple color of the DPPH solution. 19 In the current study, the J. schimperiana 80% methanol extract showed a 1.34 ± 0.06 mg/mL IC50 value in DPPH radical scavenging activity (Table 3). According to previous research by Feleke et al (2024), the hydromethanolic extract of the root of J. schimperiana is reported to scavange DPPH radical with an IC50 value of 0.14 mg/mL. 11 This result predicts the presence of a better DPPH radical scavenging ability in the root of this plant compared with the leaves. Another study by Emam et al (2022) indicated the 80% ethanol extract from the leaves of J. schimperiana showed 43.3% DPPH radical inhibition at 0.5 mg/mL concentration 13 which aligns with this study's result as presented in Figure 2.
The ABTS radical scavenging potential is evaluated by measuring the decrease in ABTS radical in the presence of a sample that can donate hydrogen. 20 In this study, the 80% methanol crude extract of R. abyssinica exhibited higher TPC and TFC which may contribute to its strong DPPH and ABTS scavenging activty. A similar finding was reported by Singh et al (2016) in which the plant extracts with higher TPC and TFC were observed to have better free radical scavenging potential in DPPH and ABTS assay. 29 Therefore, these results can predict that higher phenolic and flavonoid contents in a sample could enhance free radical scavenging activities.
The present study evaluated FRAP, with results expressed in mg of ascorbic acid per gram of extract (mg AA/g of extract). The quantitative determination of the mg of ascorbic acid in a specific amount of the plant extract provides an indication of the plant's potential as a rich supply of ascorbic acid.
A prior study assessing the antioxidant activity of 30 medicinal plants reported the presence of a strong correlation between DPPH, ABTS and FRAP assays. 30 However, different from the previous studies, a similar pattern was not observed between these three assays in this study. This lack of association suggests that the antioxidant response of the extract depends on the distinct reaction principles and conditions of each assay. DPPH and ABTS primarily assess radical scavenging capacity through hydrogen and electron donation in organic and aqueous systems, respectively. However, the FRAP assay measures the ferric ion (Fe3+) reduction capacity of plant samples to ferrous ion (Fe2+) under acidic conditions. 31 These variation in radical type, solvent polarity and pH can cause different responses among compounds with different reactive property. 32 Consequently, certain antioxidants may perform well in one assay but not in others. This suggests that antioxidant potential can not be captured by a single assay and requires multiple complementary methods.
In the present antioxidant work, the n-hexane fraction (RANF) of R. abyssinica showed the highest DPPH and ABTS radical scavenging capacity, while the aqueous fraction (RAAF) exhibited the strongest reducing power in the FRAP assay (Table 3). This variation may be explained by the polarity-dependent distribution of antioxidant compounds. The n-hexane fraction likely contains nonpolar constituents such as terpenoids and other lipophilic antioxidants that effectively scavenge free radicals, 33 whereas the aqueous fraction may be rich in polar compounds like phenolic acids and the flavonol class of flavonoids, which possess strong ferric reducing potentials. 34 This pattern reflects the chemical diversity of the extract, where compounds with varying polarity and redox potential act through different antioxidant mechanisms. Therefore, further chromatographic profiling is required to determine the specific compounds contributing to these effects.
The FTIR spectra analysis has a significant role in the profiling of functional groups and in predicting bioactive compounds and the chemical composition of herbal medicines. Therefore, it can be used as a reference for selecting medicinal plants with potential therapeutic application. 35
FTRI spectra of R. abyssinica and J. schimperiana identified key functional groups such as O–H, C = O and C–O(Table 4), indicating phenolic, flavonoid and alkaloid compounds along with terpenoids, carboxylic acids and glycosides.36,37 The strong O–H stretching band observed from these plants reflects hydroxyl group responsible for hydrogen donation. 38 Specifically, the broad and intense O–H stretching bands around 3437 cm−1 observed from R. abyssinica explain the higher TPC and TFC obtained from the plant. 39 The C = C vibration confirms conjugated aromatic systems typical of flavonoids and phenolic acids that stabilize free radicals, 40 while the C–H, N-H and C–O–C bands indicate alkaloids, terpenoids and polysaccharides that enhance the overall antioxidant capacity.41,42 Conclusively, the FTIR findings and phytochemical data are in good agreement, confirming that the antioxidant activity of both plants mainly arises from hydroxyl and aromatic-rich phenolic and flavonoid structures.
A previous study by Ahmed et al (2023) also concluded that, FTIR analysis of J. schimperiana leaves showed peaks corresponding to O-H (3748 cm-1), C-H (2917 and 2849 cm-1) and anhydride CO-O-CO (1040 cm-1). 43 In our 80% methanol extract, O-H stretching appeared at 34.22.16 cm-1, C = C at 1634.47 cm-1 and C-O/C-N at 1013.13 cm-1. The difference in peaks positions and intensities likely reflects variation in the extraction solvent composition and efficiency, which preferentially enhances the recovery of polar bioactive compounds. Overall, both studies confirm the presence of alcohol and other oxygen or nitrogen containing functional groups, with our extract showing additional aromatic and C-N features.
Although the present study provides useful data on the antioxidant and phytochemical potential of the leaves of R. abyssinica and J. schimperiana, it has certain limitations. The absence of advanced analytical techniques such as LC-MS/MS, NMR or HPLC-based compound identification restricted the detailed characterization of bioactive constituents. Nonetheless, the finding serves as a valuable baseline for future studies employing more sophisticated analytical and molecular approaches.
Conclusion
This study demonstrates that both Rosa abyssinica and Justicia schimperiana leaf extracts contain significant amounts of flavonoids and phenolic secondary metabolites, contributing to their antioxidant activity. Additionally, the TAC determined from the dried leaf powders further supports the presence of bioactive phytochemicals. The RAAF, RAEF and RANF fractions of R. abyssinica demonstrated the most potent DPPH and ABTS radical scavenging activities. Additionally, the very best ferric ion (Fe3+) to ferrous ion (Fe2+) reducing ability was observed from RAAF. All solvent fractions from Justicia schimperiana exhibited notable antioxidant activity primarily in the ABTS assay, while their performance in the DPPH and FRAP assays was comparatively moderate. FTIR analysis indicated functional groups (C–H, O–H, C = O, C = C) linked to antioxidant activity. Sharper, more intense peaks observed in Rosa abyssinica indicate higher bioactive compound content than J. schimperiana. Overall, Rosa abyssinica demonstrated superior antioxidant potential compared to Justicia schimperiana. Therefore, this study highlights the need for in-depth investigation of the plant's bioactive compounds, including their chemical characterization, mechanism of action and potential therapeutic applications.
Footnotes
Ethical Approval
Ethical approval is not applicable for this article.
Statement of Human and Animal Right
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Author's Contribution
All authors conceptualized and designed the study. Melat Solomon: sample collection, laboratory work and drafting a manuscript; Yalemtsehay Mekonnen: laboratory work, supervision and manuscript commenting and editing; Mujib Abdulkadir: supervision and manuscript commenting and editing. All authors participated in final manuscript approval.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Addis Ababa University and the L'Oréal-UNESCO For Women in Science, Sub-Saharan Africa program 2024 provided support for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Relevant data are available from the authors upon request.




