Abstract
Introduction
Since its inception, maybe even before the advent of medicinal science, human society has relied heavily on plants as a main source of medicine.1-3 Many essential components that are used as medication for the treatment and prevention of a wide range of ailments are abundant in nature. 3 Due to the presence of phytochemical components, medicinal plants are helpful for the prevention and treatment of human illnesses.4,5 Numerous medications are derived from bioactive phytomolecules. Medicinal plants are the source of medications such as quinine and artemisinin for malaria, narcotic painkillers like morphine, anticancer agents like vincristine, and cardioactive medicinal products like digoxin. The first step in discovering leads from plant sources is to investigate the phytochemical and pharmacological properties of plant samples as their traditional applications. 6
In traditional phytomedicine practices, the Fabaceae family plays a significant and advantageous role in addressing a wide array of health conditions. 7 Its therapeutic effects span multiple biological systems, highlighting its estrogenic, antibacterial, antioxidant, antifungal, and insecticidal properties. 7 The utilization of Fabaceae plants encompasses the treatment of various ailments, including polymenorrhea, anemia, ulcers, menorrhagia (especially during pregnancy), upper respiratory difficulties, bronchitis, gland inflammation, rheumatic arthritis, sleeplessness, melanogenesis inhibition, and regulation of energy consumption and metabolism. 7 Within the diverse genera belonging to the Fabaceae family, including Rhynchosia, Indigofera, Vachellia, Senna, and Albizia, there is widespread utilization of plants for addressing a spectrum of 134 health concerns. These remedies particularly focus on issues related to female sexually transmitted infections, reproductive health, gastrointestinal disorders, and respiratory ailments. 8 Traditional medicines derived from these genera primarily originate from shrubs, trees, and herbs, with roots, leaves, bark, and fruits being the most commonly utilized plant components. 8 Additionally, the genus Cynometra stands out for its significant contribution to traditional medicine, encompassing species like C. brachyrrhachis, C. cauliflora, C. capuronii, C. hankei, C. manii, and C. ramiflora. 9 For example, in Tanzania, the roots of C. brachyrrhachis are predominantly employed to combat fungal infections, while in Madagascar, the leaf decoction of C. capuronii is utilized for treating yellow fever. C. cauliflora demonstrates versatile applications, with its leaves being used for addressing diarrhea, hyperlipidemia, and diabetes, its fruits for alleviating loss of appetite, and its seed oil for managing skin conditions. Moreover, C. manii exhibits potential in cancer treatment. 9
Cynometra ramiflora L., a member of the Fabaceae family, is employed in traditional medicine to address various conditions, including diabetes, hypertension, hypercholesterolemia, and gout. 10 Leaves are utilized for their anti-herpetic properties, while the roots serve as a purgative agent (an aid in bowel movement). The oil derived from the seeds is incorporated into lotions for treating skin diseases. 11 Beyond its traditional applications, C. ramiflora has become a focus of contemporary research over the past decade, particularly through bioassay-guided activity analysis. Various extracts of C. ramiflora have demonstrated antinociceptive, antibacterial, 11 antioxidant,10,12 cytotoxic, 13 antihyperglycemic. 14
As a result of global phytochemical research efforts, numerous secondary metabolites with diverse pharmacological properties have been isolated. Plant-derived bioactive compounds are recognized as a vital reservoir of lead compounds, offering a foundation for the development of novel therapeutic agents across various major disease areas.15,16 While extensive studies have investigated various species within the Fabaceae family, there is a notable scarcity of phytochemical investigations on the species C. ramiflora. This research is undertaken with the objective of exploring the in-vitro cytotoxicity, antioxidant, thrombolytic, anti-inflammatory, as well as the in-vivo antidiarrheal, hypoglycemic, and analgesic potential of C. ramiflora leaves. The study includes a comprehensive phytochemical screening to identify chemically distinct and structurally diverse secondary metabolites from this plant species through the Gas Chromatography-Mass Spectrometry (GC-MS) technique. As per our best knowledge, this report represents the first instance of a GC-MS phytochemical investigation of C. ramiflora, shedding light on the chemical composition of this plant species.
Materials and Methods
Collection of Plant Materials
Leaves (4.5 kg) of Cynometra ramiflora were collected at July 2022, from Sundarbans, Bangladesh. A voucher specimen for C. ramiflora Family: Fabaceae have been deposited at Bangladesh National Herbarium, Mirpur Road-1, Dhaka for future references (DACB 93485).
Solvent Extraction of the Plant Materials
1 kg leaves powder of C. ramiflora was soaked in 5L of methanol in an airtight container for 15 d with occasional shaking and stirring at room temperature. The solvent filtered through a cotton plug from the plant materials. It was then concentrated by evaporating solvent with a Rotary Evaporator (Machine model) at low temperature and pressure. Finally, 25 g of methanolic crude extract (MESF) were collected from the plant materials.
Fractioning of the Extract
The modified Kupchan partitioning previously developed by Van Wagenen et al. was used to fractioning the MESF. 17 The crude extracts (5 g), which diluted in 10% aqueous methanol, were partitioned using petroleum ether (PESF), dichloromethane (DCMSF), ethyl acetate (EASF), and aqueous fractions (AQSF) (Figure S1). By letting the solvents evaporate at normal temperature, the partition fractions were concentrated and this function was done for three times to fractionate and yield more amount.
Chemicals and Reagents
The current study utilized premium-grade reagents and chemicals often applied in laboratory settings. Folin-Ciocalteau reagent, gallic acid, Tween 80, and ascorbic acid were purchased from Merck in Darmstadt, Germany, while 2,2-diphenyl-1-picrylhydrazyl (DPPH) was procured from Sigma-Aldrich in St. Louis, MO, USA. Diclofenac sodium, normal saline, loperamide, and glibenclamide were provided by Square Pharmaceuticals Ltd in Dhaka, Bangladesh. Morphine was acquired from Gonoshasthaya Pharma Ltd, while vincristine was obtained from Beacon Pharma Ltd, and streptokinase was supplied from Incepta Pharma Bangladesh Ltd.
Phytochemical Analysis Through the GC-MS
The analysis of phyto-constituents in C. ramiflora leaves extract utilized a Shimadzu auto-sampler coupled with a GC-MS-QP2010 ultra instrument from Japan. A 5 MS/HP column (30 m, 0.25 mm, and 0.25 μm) was employed, with highly pure helium serving as the mobile phase at a linear speed of 39 cm/s and a circulation rate of 1.12 mL/min. The oven temperature exhibited a constant increase at an average rate of 10 °C per minute, ranging from 110 °C to 280 °C. The needle temperature was set at 250 °C, and a 50 µL injection volume was introduced in splitless mode (ratio of 10:1). The detector voltage was maintained at 0.94 kV. The ion source, MS transfer line, and ion source ambient temperatures were all sustained at 200 °C and 250 °C, respectively. Full-scan mass spectra within a (m/z) range of 85 to 500 were recorded at a speed of 10 000 u/s. Identification of peaks and chemical constituents was achieved through a search in the National Institute of Standards and Technology (NIST) collection.
Antioxidant Evaluation
Total Phenolic Content
The assessment of Total Phenolic Content (TPC) in leaves extractives was conducted through the utilization of the Folin-Ciocalteu test.
18
To 1 mL of each extractive (1 mg/mL), 2.5 mL of Folin-Ciocalteu reagent (previously diluted tenfold with distilled water), and 2.5 mL of Na2CO3 solution (7.5% w/v) were introduced. The combination underwent a 30-min incubation period at room temperature in a dark environment. Following this, the absorbance at 760 nm was quantified using a UV-Vis spectrophotometer. A calibration curve for the gallic acid standard was established, plotting absorbance against diverse concentrations (100, 50, 25, 12.5, 6.25, 3.12, 1.56, 0.78, and 0.39 µg/mL) of gallic acid solutions. The quantification of TPC in the tested samples was expressed in milligrams of gallic acid equivalent (GAE) per gram of the extractives. This TPC assay underwent 3 repetitions. The equations describing the calibration curve for gallic acid are as follows:
DPPH Scavenging Testing
The determination of the antioxidant potency of leaves extractives was accomplished through the DPPH assay.
19
Initially, 2 mL of each methanolic solution containing the test materials, spanning various concentrations achieved through serial dilution (500, 250, 125, 62.5, 31.25, 15.62, 7.81, 3.90, 1.95, and 0.97 µg/mL), were thoroughly blended with 3 mL of a freshly prepared methanolic DPPH solution (0.004% w/v). This mixture was then subjected to a 30-min incubation period in darkness at room temperature. Subsequently, absorbance readings were taken at 517 nm using a UV-Vis spectrophotometer, with methanol serving as the blank. The percentage of inhibition (I%) of the DPPH free radical was determined using the formula:
20
Cytotoxicity
The assessment of cytotoxicity was conducted employing the brine shrimp lethality assay. 21 Brine shrimp eggs, specifically Artemia salina, underwent a 24 h incubation in homemade seawater to yield nauplii, with continuous oxygen supply throughout the hatching process. The hatched shrimp were lured towards the light source via a perforated dam before being selected for the experimental procedure. Nauplii were visually inspected, and a count was conducted before transferring them to test tubes containing 10 mL of simulated seawater. To establish various concentrations, a serial dilution technique was employed (400.0, 200.0, 100.0, 50.00, 25.00, 12.50, 6.25, 3.12, 1.56, and 0.78 µg/mL). All test samples were placed in vials and dissolved in 200 µL of pure dimethyl sulfoxide (DMSO) to generate stock solutions, which were subsequently applied to the designated test tubes. The test tubes were then left undisturbed for 24 h, and survivor counts were conducted thereafter. Vincristine sulfate served as the positive control. For the negative control groups, 100 µL of DMSO was introduced into 3 pre-marked glass test tubes containing 10 mL of simulated seawater and 10 shrimp nauplii. After 24 h, the vials underwent inspection with a magnifying glass, and survivor counts were recorded. Percent (%) mortality was calculated for each dilution, and the concentration-mortality data underwent statistical analysis using a log-linear regression graph. The effectiveness of the plant product, or the concentration-mortality relationship was typically expressed as the LC50 value.
Thrombolytic Activity Investigation
The thrombolytic potency of all extractives was calculated as per the previous study's procedure, where streptokinase (SK) was used as the positive control.
22
An Eppendorf tube containing 15,000 000 I.U. of lyophilized Altepase (Streptokinase) from a commercial source (Beacon Pharmaceutical Ltd) was obtained, and 5 mL of sterile distilled water was added and thoroughly mixed. For in-vitro thrombolysis, 30 000 I.U. of this solution employed as a positive control. In various vials, 10 mg of methanolic extractuves were taken and dissolved in 1 mL of distilled water. Healthy participants gave aliquots of venous blood (5 mL) that divided among 10 separate sterile Eppendorf tubes and incubated at 37 °C for 45 min. Each tube contained 0.5 mL of blood. The serum was entirely withdrawn after clot formation without disrupting the clot, and clot weight was calculated as per the below equation.
%clot lysis = (Weight of the lysis clot/Weight of clot before lysis) × 100
Anti-Inflammatory Effects
Crude methanolic extractives were tested for anti-inflammatory effectiveness using two different methods: heat-induced membrane stabilization and hypotonicity-induced membrane stabilization. Membrane stabilizing behavior in hypotonic and heat-induced solutions has been observed through methods known to utilize the capacity of the Shimadzu UV spectrophotometer at 540 nm to obtain the absorbance values of the supernatant fluids. 23
Hypotonic Solution-Induced Hemolysis
Centrifuge tubes were filled with acetylsalicylic acid (0.10 mg/mL), serving as the standard drug. These tubes were then filled with 5 mL of a 10 mM sodium phosphate buffer solution (pH 7.4) and 50 mM NaCl, constituting a hypotonic solution. Additionally, 0.50 mL of erythrocyte (RBC) suspension was added, followed by incubation at room temperature for 10 min. Subsequent centrifugation at 3000 rpm for approximately 10 min, decantation, and filtration of the soluble supernatant were carried out. The absorbance for each tube was measured, and the following equation was employed to measure the membrane stabilization:
Heat-Induced Hemolysis
Two sets of centrifugation tubes were prepared, one containing an extract or fraction of 1 mg/mL along with a 30 μL erythrocyte suspension and 5 mL of isotonic buffer. The other set mirrored the first, except for the absence of crude extract or fraction. After gently inverting the tubes, one set was incubated in a water bath at 54 °C for 20 min, while the second set was kept in an ice bath at 0 to 5 °C. Centrifugation of these combinations at 1300 rpm for 3 min allowed the determination of supernatant absorbance. The percent (%) inhibition was calculated using the following equation.
Where OD1 is the unheated test sample, OD2 is the heated test sample, and OD3 is the heated control sample.
Investigational Animal
The healthy, aged 4-5 weeks, weighing 25-30 g, and adult albino mice (Swiss) were picked from the animal section of the International Centre for Diarrheal Diseases and Research in Bangladesh (ICDDR,B). Prior to initiating the animal investigation, the mice were kept in polypropylene cages under certain environmental conditions: a 25 ± 2 °C average temperature, 55% relative humidity, and a light/dark cycle of 12 h each. During this period, the mice were nourished with ICDDR,B mice foods and water. 24 As per the previous study, animals were categorized into 5 categories: Group I, Group II, Group III, Group IV, and Group V. Here Group I was always negative control, Group II was always positive control, Group III contained the 200 mg/kg dose of MESF. Group IV contained 400 mg/kg dose of MESF, and Group V contain 600 mg/kg dose of MESF. 25 A total of 100 mice were used in the study, with 25 allocated for the each in-vivo experiment. Each experimental group consisted of 5 mice, and the allocation of mice to these groups was done randomly. 25
Evaluation of Anti-Diarrheal Activity
The effectiveness of methanolic extracts of C. ramiflora from the leaves was evaluated in a mouse model with diarrhea induced by castor oil.26,27 The loperamide 50 mg/kg was employed as a positive control, whereas the negative control group was given a solution containing only 1% Tween 80. After a 30-min interval of providing both extract and standard dosages, each animal received 0.5 mL of castor oil orally via a feeding needle and was subsequently placed in its allotted cage. The floor was meticulously inspected for a duration of 4 h to evaluate the efficacy of the tested chemicals in avoiding diarrhea. It was wrapped with absorbent material to maintain the visibility of feces stains. Every hour, data was gathered after the administration of castor oil, and the extent of fecal suppression caused by the plant extract was assessed using the given equation.
Evaluation of the Hypoglycemic Activity
The hypoglycemic efficacy of the extractives was assessed through an oral glucose tolerance test following the previous investigation.
28
Animals were administered by sugar solution via a feeding needle. Subsequently, after 5 min, each animal in the negative control, positive control, Group III-IV received their correspondence doses, while glibenclamide (10 mg/kg body weight) was standard and 1%Tween-80 in normal saline was administered as negative control, respectively. Glucose level in blood was monitored using diabetic strips at 30 min intervals over a span of 1.5 h. The percentage reduction in blood glucose levels was computed using the following formula:
Evaluation of Central Analgesic Activity
The central analgesic efficacy of C. ramiflora extractives was assessed using the tail immersion method. 29 By reducing the potency of the supplied morphine (15 mg/mL) using saline water, the positive control morphine dose (2 mg/kg, subcutaneous) was produced. 30 The extract being analyzed was administered orally to the mice through a feeding syringe. Throughout the experiment, the tail of the mouse was immersed in hot water at a temperature of 55 °C. The pain response time, which refers to the duration for each mouse to move its tail away from the hot water, was evaluated at many time intervals: before (0 min), as well as at 0, 30, 60, and 90 min after the delivery of the analyzed samples.
Evaluation of Peripheral Analgesic Activity
The analgesic effectiveness of C. ramiflora methanolic extracts on the peripheral level was assessed by the acetic acid-induced writhing method.
31
Every set of animals was exposed to pain induction by the application of glacial acetic acid at 48 mg. The experimental groups, namely Group-I, Group-II, Group-III, and Group-IV, received oral dosages of aspirin 24 mg as a positive control. The negative control group was administered a solution containing Tween 80 and acetic acid. The number of writhes was evaluated at a 30-min interval after the intraperitoneal injection of acetic acid. The reduction writhing percentage was determined by using the following methodology:
Statistical Analysis
The experimental results obtained from the in-vitro and in-vivo examinations were presented as average number whenever feasible. The in-vivo data underwent assessment through Student's t-test utilizing Microsoft Excel 2016 (United States). Significance in statistics was assigned to p ≤ .05.
Results
Identification of Phytoconstituents by GC-MS Analysis
A total of 47 phytoconstituents were predominantly found in the MESF based on their GC-MS analysis. The spectrum obtained from GC-MS analysis is available as supplemental Figure S2. Among the isolated compounds, Tetratriacontyl pentafluoropropionate (9.25%) was the most abundant compound. After that, Pentatriacontane were predominant (8.75%) (Table 1), while Loliolide (0.50%), Hexadecanoic acid (0.50%), 2-hydroxy-1-(hydroxymethyl) ethyl ester (0.50%), Octadecane, 5-methyl- (0.50%), and Thiophene, tetrahydro-, 1,1-dioxide (0.48%) were the least amount compounds (Table 1). Two bioactive plant steroids such as γ-sitosterol and ergost-5-en-3-ol (3beta, 24R) were also detected from the MESF. Furthermore, vitamin E and its derivatives such as γ-tocopherol, and phytol were identified from the MESF fractions. According to GC-MS analysis, all identified compounds’ names, retention time, % area, molecular formula, molecular weight, and PubChem CIDs, were tabulated in Table 1. In addition, the reported bioactivities of these identified compounds were also tabulated with their corresponding references (Table 1, Figure S2).
List of detected phytochemicals through the GC-MS approaches of the methanolic extract of Cyanometra ramiflora.

Cytotoxicity
LC50 of positive control (Vincristine sulphate) was 0.45 μg/mL. Among the all extractives of C. ramiflora, the highest brine shrimp lethality was given by DCMSF (LC50 = 11.05 μg/mL), then followed by MESF (LC50 = 33.15 μg/mL), EASF (LC50 = 49.38 μg/mL), and the lowest potent was PESF (LC50 = 139.68 μg/mL) (Figure 1).

LC50 value of the methanolic extract C. ramiflora at brine shrimp lethality assay.
Thrombolytic Properties
Compared to the control group, MESF showed the most potent thrombolytic property (52.74%), followed by PESF (51.56%), EASF (47.28%), and DCMSF (46.95%). In contrast, the positive control SK (30 000 I.U.) showed 66.67% of the clot lysis after 90 min of incubation at 37 °C (Figure 2).

Thrombolytic properties of the C. ramiflora leaves methanolic extract.
Antioxidant Properties
Total Phenolic Content
The highest phenolic content was found in EASF (98.75 mg of GAE/g of extractives). PESF, AQSF, and MESF demonstrated approximately the same quantities of phenolic content (40.88, 40.56, and 40.25 mg of GAE/g of extractives). However, among all the extractives, DCMSF showed the lowest level of phenolic content (36.13 mg of GAE/g of extractives) (Figure 3).

Total phenolic content of different methanolic extractives of C. ramiflora.
DPPH Scavenging Testing
Figure 4 shows the IC50 values for the plant extractives’ ability to quench DPPH radicals. Among the methanolic extractives, at the DPPH scavenging test, MESF, DCMSF, and EASF showed better activity (IC50 = 17.71, 21.82, and 36.29 µg/mL) than the standard EDTA (IC50 = 75.94 µg/mL). The outcomes concluded that the PESF scavenging properties were lower (IC50 = 107.14 µg/mL) than those of the other fractions.
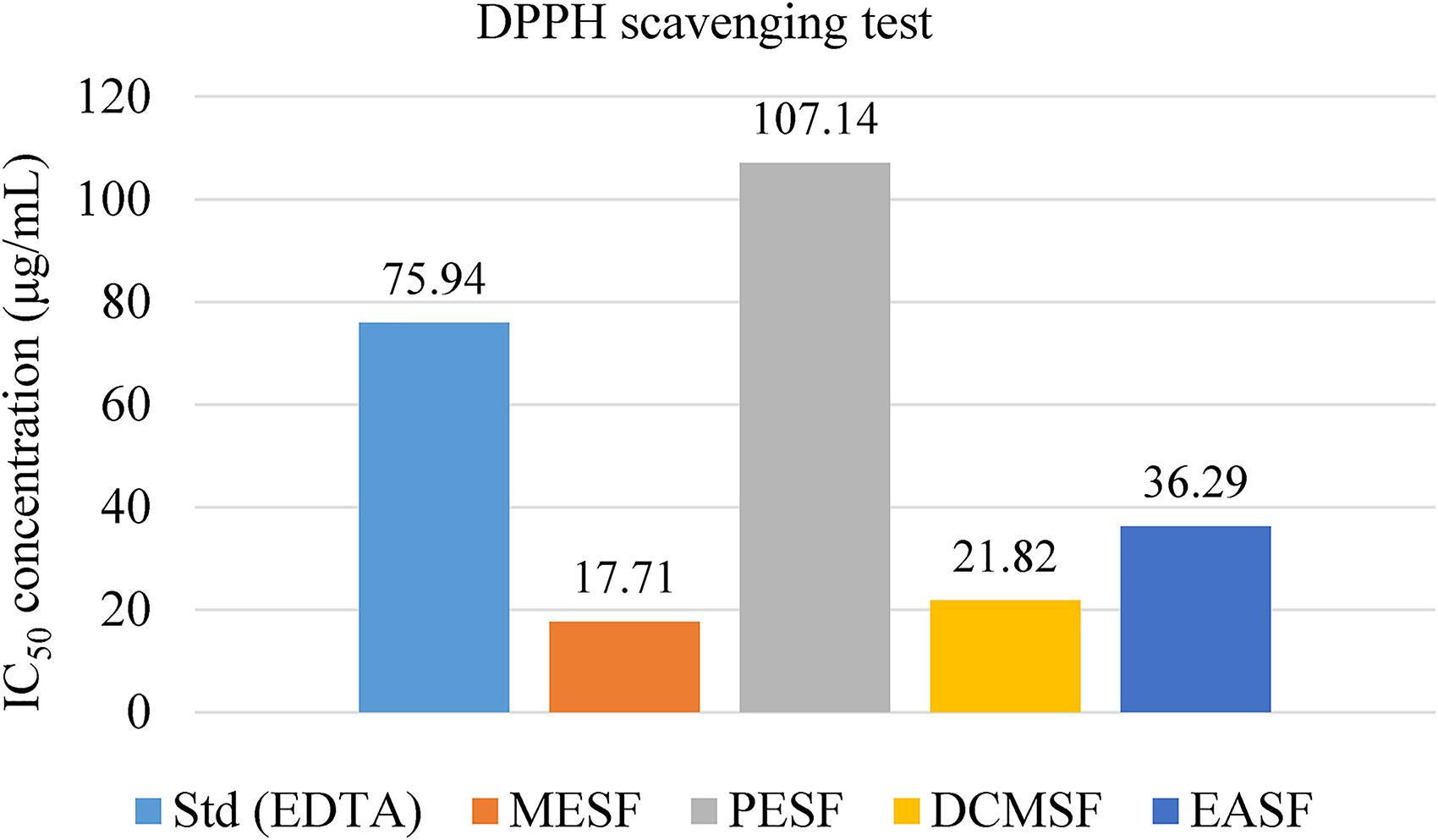
Antioxidant properties of C. ramiflora leaves methanolic extract through DPPH scavenging approach.
Anti-Inflammatory Activity
Hypotonic Induced Membrane Stabilizing Property
The outcomes in the membrane stabilizing investigation of methanolic extract and its different fractionates of leaves of C. ramiflora were effective, as the extractives prevented the lysis of erythrocytes induced by hypotonic solution. Among them, the EASF was the most effective (64.71%), and the DCMSF was the least potent (23.44%) for hemolysis, while the STD group acetyl salicylic acid exhibited 74.21% inhibition of hemolysis at normal conditions (Figure 5).

% inhibition of hemolysis of different extractives of leaves of Cynometra ramiflora.
Heat and Cold Solution Induced Membrane Stabilizing Property
The various methanolic extractives from the leaves of C. ramiflora demonstrated significant protection against the lysis of the human erythrocyte membrane induced by both heat and cold solutions. In comparison to the standard acetyl salicylic acid (0.10 mg/mL), the protective effect was observed and is depicted in Figure 6. Specifically, EASF showed a 7.88% inhibition, MESF exhibited an 18.25% inhibition, DCMSF displayed a 39.47% inhibition, and PESF demonstrated a substantial 69.92% inhibition of hemolysis in red blood cells (Figure 6).

% inhibition of hemolysis of different extractives of leaves of Cynometra ramiflora.
Anti-Diarrheal Activity
The positive control group (STD) was medicated with Loperamide at 50 mg/kg and demonstrated a remarkable and statistically significant reduction in diarrheal feces at 1 to 4 h interval (p < .001). When compared to the STD, experimental groups, treated with MESF at varying doses (200, 400, and 600 mg/kg), showed moderate anti-diarrheal dose and time-dependent effects on the reduction of diarrheal feces. MESF at 600 mg/kg showed notable effects at 4 h interval of 37.5% diarrhea reduction, while STD demonstrated a 75% reduction (Table S1 and Figure 7).

Antidiarrheal activity of the C. ramiflora leaves methanolic extract.
Hypoglycemic Property
Overall, these findings suggest a time-dependent effect of the investigational treatment on blood glucose levels in mice. Group III (200 mg/kg) showed a decreasing trend in blood glucose levels over time, with the highest reduction observed at 90 min (65.80%). Group IV (400 mg/kg) exhibited fluctuating reductions, with the maximum at 90 min (72.53%). Group V (600 mg/kg) demonstrated reductions at 0, 30, and 60 min, with a notable decrease at 60 min (12.93%), followed by a slight increase at 90 min (63.73%) (Table S2 and Figure 8).

Hypoglycemic activity of the C. ramiflora methanolic extract.
Evaluation of Central Analgesic Property
The positive control group (STD) showed the gradually increment of average time of tail immersion (Table S3), indicating a strong analgesic effect (p < .001). MESF at varied dosages (200, 400, and 600 mg/kg) produced dose and time-dependent analgesic impacts in the experimental groups. When comparing to the control, these groups had considerably longer tail immersion periods, for instances, 600 mg/kg MESF exhibiting promising outcomes at 90 min (5.38 s, p < .001) (Table S3 and Figure 9).
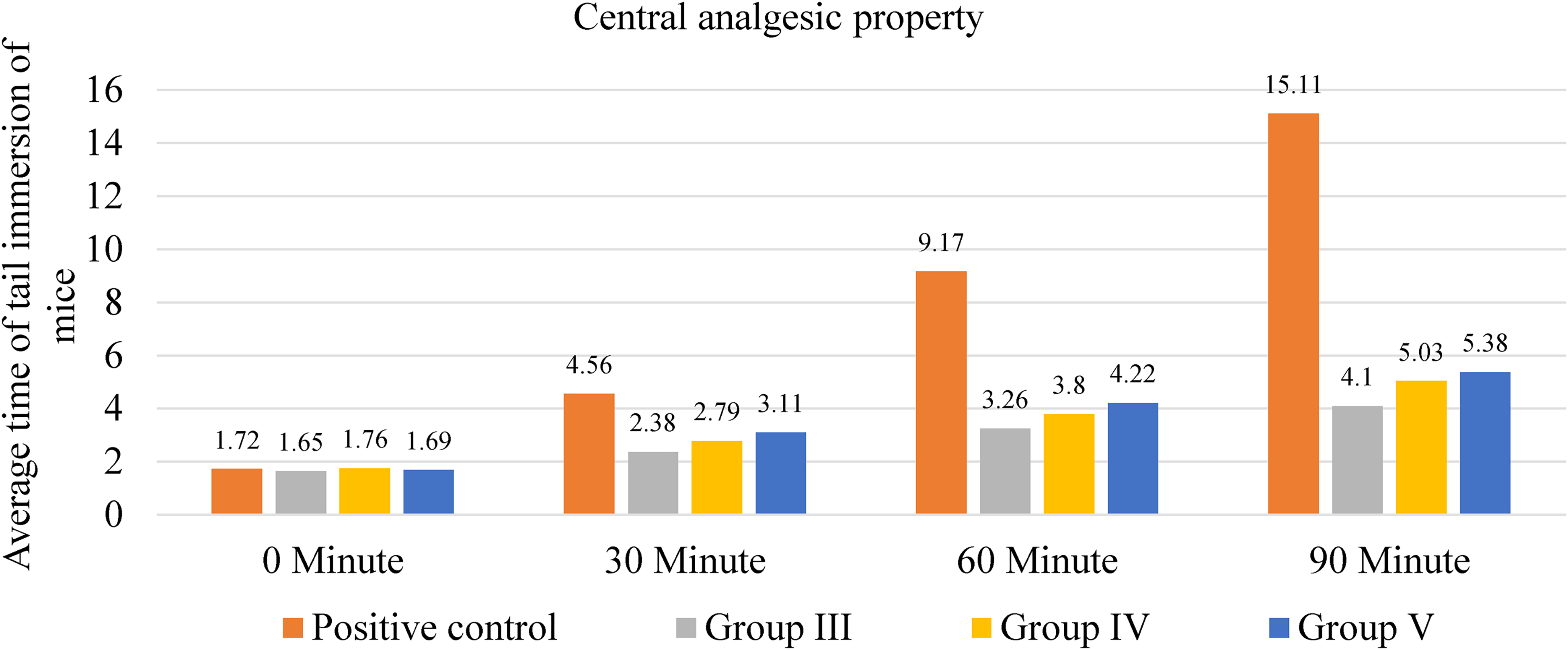
Central analgesic property of the methanolic extract of C. ramiflora.
Evaluation of Peripheral Analgesic Property
The positive control group, which received 50 mg/kg of acetyl salicylic acid sodium, displayed considerably reduced inhibition at 83.14% (p < .001). MESF administration at different dosages (200, 400, and 600 mg/kg) led to dose-dependent inhibited impacts, with percentages of 19.10%, 61.79%, and 68.53%, respectively. Compared to the STD group, 400 and 600 mg/kg doses showed a moderate level of peripheral analgesic properties (Table S4 and Figure 10).

Peripheral analgesic property of the methanolic extract of C. ramiflora.
Discussion
The present research examined the chemical components within the leaves methanolic extract of C. ramiflora. This analysis revealed a range of potentially valuable bioactive elements. These elements could play a crucial role in driving the diverse bioactive effects observed in the leaves extract and its different solvent-based fractions. The GC-MS analysis of the MESF expressed the availability of several phytoconstituents, such as organic acids (Nonahexacontanoic acid, Tetradecane, 4-methyl-), esters (Carbonic acid, eicosyl vinyl ester, Dodecyl cis-9,10-epoxyoctadecanoate, 3-Cyclopentylpropionic acid, 2-chlorophenyl ester), aliphatic hydrocarbons (Cyclononane, 1,1,4,47,7-hexamethyl-, Vitamin E, phytol), ketones (1-Cyclohexyl-2-methyl-prop-2-en-1-one, 2(5H)-Furanone, 5-(7-hydroxy-6-methyloctyl)-, Spiro[4.5]decan-7-one, 1,8-dimethyl-8,9-epoxy-4-isopropyl-), alcohols (E,E,Z-1,3,12-Nonadecatriene-5,14-diol), amine (Tranylcypromine), phenols (Phenol, 2,6-dimethoxy-4-(2-propenyl)-, 1,2,4-Benzenetriol, Catechol, 4-Methyl catechol), steroids (γ sitesterol, ergost-5-en-3-ol, (3beta, 24R)), and tocopherols (Vitamin E, γ-tocotrienol, phytol) (Table 1). These 47 compounds from the MESF may contribute to the medicinal activities of the leaves. From the previous literature study, most of the detected compounds in this study have antioxidant, antibacterial, and anticancer effects (Table 1). The availability of unsaturated or phenolic compound especially may play a role for antioxidant properties of the MESF.57,58 Another molecule phytol was also identified from this plant which have potential anticancer and antioxidant activities that are matched with the MESF bioproperties. 59 Another preceding study demonstrated 4 different compounds of the MESF such as glutinol, glutinone, β-sitosterol, ethyl 4- ethoxy benzoate. 9
Free radicals and oxidants are harmful to the body because they are produced by both natural cellular processes and external causes such as emissions, smoking, medicines, and radiation. The buildup of free radicals exceeds the body's ability to remove them, resulting in oxidative stress. This mechanism has a crucial role in the onset of degenerative and chronic illnesses such as cardiovascular disease, autoimmune disorders, carcinoma, rheumatoid arthritis, cataracts, ageing, and neurological diseases. The human body uses a variety of strategies to resist oxidative stress, including the creation of antioxidants. These antioxidants can be produced naturally by the body or obtained from food sources or supplementation. 60 Some previous studies revealed the potent antioxidant properties of the C. ramiflora leaves, such as the stem bark ethanol extract, which showed the highest antioxidant activity with an IC50 value of 41.90 ppm at the DPPH radical scavenging assay. 10 Also, earlier studies showed that the IC50 of the methanolic extract from the stems of C. ramiflora in the DPPH test was around 31.62 µg/mL. 61 Another research found that the ethyl acetate fraction of a C. ramiflora seed methanol extract had high antioxidant and anti-lipid peroxidation properties, while a methanol extract of C. ramiflora bark had minimal toxicity against mouse fibroblasts. 9 The antioxidant investigation of the current study also revealed potent antioxidant activity of the C. ramiflora leaves, such as MESF, DCMSF, and EASF (IC50 = 17.71, 21.82, and 36.29 µg/mL, respectively). A study revealed that the antioxidant activity of C. ramiflora was found to vary depending on the polarity of the fractions. Specifically, the IC50 values for the semi-polar and polar fractions were 91.26 and 183.57 ppm, respectively, indicating differing levels of antioxidant potency. 62 Conversely, the nonpolar fraction did not exhibit any antioxidant activity. 62 Interestingly, the ethanol extract demonstrated superior antioxidant potency compared to the fractions, with an IC50 of 55.44 ppm. 62 Some compounds detected through the GC-MS technique, such as 9,12-Octadecadienoic acid, methyl ester, hexadecanoic acid, 2-hydroxy-1-(hydroxymethyl)ethyl ester, carbonic acid, eicosyl vinyl ester, and tetracontane, 3,5,24-trimethyl, may be responsible for such antioxidant properties.36,40,42,46,48 Further investigation is needed to find out the responsible antioxidant phytochemicals of C. ramiflora.
Due to the antiproliferative nature of the medicinal plants, which is a significant concern, a low-cost and dependable method, the brine shrimp (Artemia salina) lethality bioassay was used to detect the cytotoxicity of C. ramiflora leaves' methanolic extractives. 6 Previous some studies indicated strong cytotoxicity activities of C. ramiflora. Such as evaluation using an MTT assay against HeLa, T47D, and WiDR cell lines, while the results showed the leaves' ethanolic extract of C. ramiflora was the most active against WiDR cell lines (IC50 = 69.66 ppm). 10 The 3 major compounds had been isolated, including apigenin, caffeic acid, and 3-(23,4-trihydroxyphenyl)-7-hydroxycoumarin, from Cynometra ramiflora Linn extracts and identified through the NMR spectra. 10 Furthermore, another study showed the cytotoxic activity of a methanolic bark extract of C. ramiflora against different cell lines, such as HT29 and MDA-MB-435S (IC50 = 1.79 and 2.35 mg/mL, respectively). 11 Another study based on brine shrimp lethality bioassays found that chloroform and methanol leaves extracts of C. ramiflora showed strong cytotoxicity (LC50 = 8.27 and 4.61 µg/mL, respectively). 59 Further earlier investigation demonstrated both methanolic leaves and stems extracts had potent cytotoxicity against brine shrimp nauplii (LC50 = 4.61 and 1.59 µg/mL, respectively). 61 Nearly same potentcy of C. ramiflora leaves' methanolic extract against brine shrimp has also been detected in this study, for example, DCMSF, MESF, and EASF (IC50 = 11.05, 33.15, and 49.38 μg/mL, respectively). Various phyto-constituents detected through the GC-MS technique such as γ-sitesterol, ergost-5-en-3-ol,(3beta, 24R), cyclotetradecane,1,7,11-trimethyl-4-(1-methylethyl)-, may be accountable for the anticancer properties of the MESF and its extractives.53,63,64
The ability of drugs to stabilize the erythrocyte membrane extends to the stabilization of lysosomal membranes, suggesting potential anti-inflammatory properties. This is attributed to the structural resemblance between the two membranes, leading to alterations in cell mediator activity.23,65 In this context, the EASF and MESF derived from C. ramiflora leaves demonstrated noticeable anti-inflammatory activity by preventing erythrocyte hemolysis under hypotonic conditions. Several anti-inflammatory phyto-molecules, such as loliolide and cyclotetradecane, 1,7,11-trimethyl-4-(1-methylethyl)-phyto-molecules, were detected through the GC-MS technique, potentially responsible for this response.34,53 Further exploration is required for a comprehensive understanding of the anti-inflammatory activity of C. ramiflora.
Thrombosis stands as a prominent contributor to morbidity and mortality in various vascular disorders. 66 The conversion of fibrinogen to fibrin in the presence of activated thrombin leads to thrombus or blood clot formation. Tissue plasminogen activator (t-PA), released from damaged tissues, transforms plasminogen into plasmin. Fibrinolytic medications, activating t-PA, are administered to patients with blocked veins or arteries, exerting a thrombolytic effect. 67 Reducing platelet aggregation is protective against specific heart disorders, such as atherosclerosis. 68 In the present study, nearly all extractives of C. ramiflora displayed significant thrombolytic activity. This discovery may contribute to the development of recombinant variants of first-generation thrombolytic agents, addressing issues related to systemic fibrosis and bleeding complications.69,70
Natural pain relievers are being looked into as alternatives to synthetic drugs because they have fewer side effects. 71 In this investigation, the findings suggest that the plant's methanolic extract elicited a slight reduction in pain perception at the central analgesic level (Table S3 and Figure 9). However, peripheral analgesic assessments unveiled a notable alleviation of acetic acid-induced pain sensation in mice (Table S4 and Figure 10). Though analgesic compounds were lacking in the GC-MS technique, the presence of the excessive antioxidant compounds among the MESF phytochemicals may be responsible for such analgesic properties. Previous studies imply that inflammation and oxidative stress are closely connected pathophysiological processes, with one readily caused by the other. 72 Therefore, the presence of antioxidant molecules may reduce the pain of the experimental mice, especially in the peripheral analgesic investigation technique.
The current study also found significant antidiarrheal effects of the MESF. Castor oil induces diarrhea primarily through 3 mechanisms: producing nitric oxide, increasing gastrointestinal membrane calcium permeability, triggering prostaglandin production, resulting in increased fluid and electrolytes in the intestine, and stimulating peristalsis. 73 The main compound in castor oil, ricinoleic acid, has been documented to disrupt the intestinal barrier through the generation of prostaglandins and stimulation of peristaltic activity, potentially resulting in diarrhea.74-76 Antioxidant molecules have significant prostaglandin inhibition activity. 77 In this current study, MESF showed moderate antidiarrheal potency (Figure 7), while its antioxidant investigation showed strong activity of some extractives such as MESF, DCMSF, and EASF, as well as several antioxidant compounds detected from this GC-MS analysis, which may be responsible for the inhibition of castor oil-induced gut inflammation and diarrhea effects.
Diabetes mellitus, a metabolic disease, is a worldwide public health concern. As a result, the need for safe and effective anti-diabetic chemicals derived from organic sources is increasing by the day. 6 Because of their low toxicity, low cost, and ubiquitous availability, herbal treatments have the potential for widespread use in hyperglycemia management. Numerous studies have shown the blood sugar-lowering effects of plant extracts. 6 As a result, the present study focuses on the early assessment of C. ramiflora's hypoglycemic effects in mice. Our results indicated that the MESF moderately decreased blood sugar levels following glucose-induced hyperglycemia in mice (Figure 8 and Table S2). Therefore, the notable hypoglycemic effect of C. ramiflora observed in this study could be attributed to the identification of phytochemicals through the GC-MS technique. This study represents a pioneering exploration of the plant extract; hence, there are no preceding findings concerning the hypoglycemic characteristics of this plant. Numerous studies have suggested that oxidative stress is intricately linked to the origin and physiological mechanisms of diabetes mellitus. 78
Conclusions
The phyto-chemical analysis of leaves extract from C. ramiflora revealed 47 compounds identified through GC-MS spectrometry methods. The separated botanical metabolites could serve functional roles for medicinal applications. During biological screening, the dichloromethane fraction of C. ramiflora exhibited the highest cytotoxicity in comparison to the standard vincristine sulfate. Particularly the methanolic and petroleum ether displayed thrombolytic activity exceeding 50%. Additionally, the methanolic and dichloromethane fractions exhibited considerable antioxidant characteristics. Nevertheless, during the anti-inflammatory test, different sections demonstrated varied results in 2 distinct types of tests. The in-vivo investigation findings indicated that the leaves extract of this plant species possesses moderate anti-diarrheal and central analgesic properties. The leaves methanolic extract demonstrated noteworthy glucose-lowering capability after 90 min. Consequently, the current pharmacological investigation disclosed the efficacy of C. ramiflora as a promising reservoir of potential bioactive compounds suitable for innovative drug exploration and therapeutic advancements. Further investigations are essential to identify the bioactive components in C. ramiflora extracts responsible for these biological activities.
Limitations of the Study
In the present investigation, the identification of phytochemicals utilized the GC-MS technique instead of isolation and purification methods. To improve this study, it could benefit from employing the nuclear magnetic resonance approach for detection of the isolated compounds. Additionally, computational chemistry might prove valuable in comprehending the mechanism of action of bioactive phyto-molecules following their in-vivo and in-vitro analysis.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241257377 - Supplemental material for Multifaceted Chemico-Pharmacological Insights into Cynometra ramiflora L.: Unveiling its GC-MS, Cytotoxic, Thrombolytic, Anti-Inflammatory, Antioxidant, Anti-Diarrheal, Hypoglycemic, and Analgesic Potentials
Supplemental material, sj-docx-1-npx-10.1177_1934578X241257377 for Multifaceted Chemico-Pharmacological Insights into Cynometra ramiflora L.: Unveiling its GC-MS, Cytotoxic, Thrombolytic, Anti-Inflammatory, Antioxidant, Anti-Diarrheal, Hypoglycemic, and Analgesic Potentials by Md. Abdus Samadd, Md. Rifaet Hossain, Mohammad Abdullah Taher, Mohammad Mahmudul Hasan, Shariful Haque, Mashruf Islam Rafid, Md. Solaiman Hossain, Md. Ashraful Islam and Mala Khan in Natural Product Communications
Footnotes
Acknowledgments
The authors acknowledge the support of lab-facility of the State University of Bangladesh, and BRiCM.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The Animal Ethics Committee of the State University of Bangladesh thoroughly scrutinized and approved the ethical principles and methodologies utilized in the research (2023-04-04/SUB/A-ERC/001).
Statement of Human and Animal Rights
The study adhered to the protocols for ethically handling animals in investigations as delineated by the Federation of European Laboratory Animal Science Associations (FELASA).
Statement of Informed Consent
This article does not involve human participants, so informed consent is irrelevant.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.









