Abstract
Introduction
In the aim to investigate the possible influence the variation of the food composition on ruminant performances, several approaches was based on medicinal plants for their beneficial effects on digestibility. Our study was oriented to determine the phytochemical screening, the antioxidant activity, and the
Methods
Colorimetric analyses were used for the quantification of phenolic compounds, and the antioxidant activity of azerole extracts was evaluated using DPPH-free radical scavenging activity. The
Results
Leaves contained the highest levels of mineral matter, neutral detergent fibre (NDF), and nitrogen matter (TNM) when compared to those of berries. Furthermore, the DPPH assay showed that
Conclusion
Data showed that azerole berries and leaves contained an important level of total fiber, nutrients, and mineral matter. The phytochemical analyses demonstrated a strong correlation between the richness of phenolic compounds and their antioxidant capacity. Indeed, yellow azerole berries and leaves can be recommended as an additive in goat feed rations to improve the animals’ quality.
Introduction
The field of animal nutrition has been considered as a subject of several studies that aim to improve the quality of the ration in order to increase the absorption of nutrients and optimize the productivity and quality of animal products. Indeed, the health and performance of animals have historically been managed by a variety of conventional chemical products, including antibiotics, antiparasitic and anti-inflammatory medications, and chemical food supplements. However, due to their detrimental effects and side effects, a number of these products have been outlawed in several countries. 1
In this context, the use of herbal products, rich in active substances, has shown several beneficial effects on ruminants such as improving the efficiency of nutrient use or boost milk production.2,3
Generally, leaves, fruits, and flowers of this plant are used to treat several pathologies, such as cardio-vascular diseases including heart failure, atherosclerosis, hypertension, and hyperlipidemia. In addition, azerole extracts are characterized by antispasmodic, diuretic, and anti-diarrheal activities. 5
Phytochemical screening of the species testifies to its richness in antioxidants, especially polyphenols and flavonoids. 6
In Tunisia,
Interestingly, leaves of several trees and shrubs have important nutritional values and can improve the poor forage quality of diets, especially during dry periods. 8 Precisely, they represent an important gazing resource in arid and semi-arid regions of the world. 8
As is known, goats have the capacity to valorize the lowest-productive forage areas; they are better suited to browsing in shrubs, trees, and scrub. They are characterized by their ability to digest woody species because their rumen is more buffered and contains more protozoa and bacteria. 9
In this context, the purpose of this research is to valorize
Results
Physiochemical Analysis
Table 1 presents the dry matter and phytochemical contents of
Physicochemical parameters (%DM) of Crataegus azarolus fruits and leaves.

Data are represented as an SD means (n = 3). Means in the same column with no common index differ significantly
Parietal Constituents and Lipid Contents in Leaves and Fruits
Table 2 presents variation in contents of parietal constituents of fruit and leaf substrates. Indeed, the leaf contained the highest contents of neutral detergent fibre, acid detergent fibre, lignin detergent acid, lignin, hemicellulose, true crude cellulose, and lipid.
Variation of Parietal Constituents and Lipid Contents (%DM) in Crataegus azarolus Fruits and Leaves.

Data are represented as mean ± SD (n = 3). ADF, Acid detergent fiber; NDF, Neutral detergent fiber; TRC, True crude cellulose; ADL, lignin detergent acid; HC, hemicellulose.
Phenolic Compounds Determination
According to Table 3, the phytochemical screening revealed the richness of
Phytochemical Screening of Cartaegus azarolus Fruits and Leaves Aqueous Extracts.

Data are represented as mean ± SD (n = 3). Means in the same row without a common upper index differ significantly (
DPPH• Scavenging Activity
The antioxidant capacity test showed that CABAE and CALAE exhibited important DPPH• free radical scavenging activity (RSA), expressed by their ability to neutralize the DPPH• radical. Interestingly, the inhibition percentages of
Dose Response of Antioxidant Capacity and Determination of the Inhibitory Concentration 50 (IC50) of

Data are expressed as mean ± SD (n = 3). Means in the same column without a common upper index differ significantly (
ABTS•+ Scavenging Activity
The antioxidant capacity assessed using the ABTS method reveals that
Inhibitory Concentration 50 (IC50) and Concentration Response Effect of CABAE, CALAE, and Butylated Hydroxytoluene (BHT) Against (ABTS•+).

Data are expressed as mean ± SD (n = 3). Means in the same column without a common upper index differ significantly (
In Vitro Ruminal Fermentation and Digestibility Parameters Assessment
In the present study, we further looked at the effect of
The
In fact, for both substrates, the gas production curve represents 3 phases (Figure 1). The first one, characterized by a minimum production speed (lag phase “a”), is explained by the microbiota's adaptation to the substrate. The second represents an increase in gas production level (substrate degradation phase) or exponential gas production (phase “b”) where gas production attained its maximum volume. Finally, the third one was a stationary phase where the gas production speed was constant, which constitutes the fermentation levels of phase “b” (the end of the fermentable fraction “c”).

Interestingly, gas production depends on the carbohydrate nature, the characteristics, and the degradation degree of the substrate. On the other hand, fruits were characterised by the highest variable, (“b”). Whereas, the leaves showed the highest level of fermentable fraction degradation (“c”). However, the volume of gas at 24 h (Gp) and the total volume of gas emitted (y) represents a variation depending on the substrate used. The highest volumes were observed on the leaves (51.50 ± 0.50 and 59.16 ± 0.28, respectively) (Table 6).
Effect of Crataegus azarolus Fruits and Leaves on the in Vitro Digestibility Parameters in Goats.

Data are represented as mean ± SD (n = 3). a: Volume of gas produced from the easily fermentable soluble fraction (ml); b: Volume of gas produced from the insoluble potentially fermentable fraction (ml); c: The rate of gas production(ml/hour); Gp: The gas volume production after 24 h of incubation in ml from 300 mg dry sample and Y: Volume of total gas produced
Table 7 represents the ruminal fermentation parameters results, such as digestibility of organic matter (DOM), metabolizable energy (ME), and volatile fatty acids (VFA). Importantly, the leaves were characterized by the highest DOM (83.47% ± 4.53). Likewise, for the ME and the VFA (9.55 ± 0.07 Kcal/kg DM and 1.17 ± 0.01 mmol/syringe, respectively).
Effect of Crataegus azarolus Fruits and Leaves on the Digestibility of Organic Matter (DOM), Metabolizable Energy (ME), and Concentration of Volatile Fatty Acids (VFA) in Goats.

Data are represented as mean ± SD (n = 3). DOM, Digestibility of organic matter; ME, metabolizable energy; VFA, Concentration of volatile fatty acids.
Discussion
The aim of this study is to determine the chemical analysis of
The physicochemical analyses revealed that leaves and berries contained considerable levels of mineral matter such as calcium, magnesium, iron, and nitrogen, with significant variations. In fact, our results corroborate the reports of Bouadra et al
10
and Sammari et al,
11
on
The obtained results showed that the leaves were characterized by higher levels of total lipids and total nitrogen matter compared to fruits. In the same context, several studies showed that fruits were richer in non-structural carbohydrates and contained low levels of lipids and proteins. However, leaves were characterized by abundant amounts of protein.14,15
Generally, primary metabolite contents such as lipid and protein were controlled by several factors, including genetics, climate, and geography. These fluctuations can also be explained by the season, the cultivation techniques, and the fruit maturing stages.16–18
The parietal constituent's determination demonstrated that leaves were the richest in total fiber as compared to fruits, which was previously reported.13,19 Total fiber content in plants showed numerous seasonal, stationary, and genetic discrepancies. 20
The chemical analysis showed that CALAE and CABAE contained considerable concentrations of phenolic compounds, of which the leaves contained the highest level. Similarly, these findings have been described by Belkhir et al 6 and Mraihi et al. 21
The highest level of phenolic compounds observed in leaves can be explained by their exposure to several factors, such as UV rays, to ensure the protection of membranes, chlorophylls, and other sensitive organelles. 7
Several reports characterized the secondary metabolite profile of azerole fruit with a few disparities, 7 which are correlated with chemotypes and morphotype diversity. 22
The high-performance liquid chromatography coupled to electrospray ionization tandem mass spectrometry (LC-ESI-MS) analysis of the CABAE resulted in the identification of 5 phenolic compounds. which are 3 phenolic acids (quinic acid, gallic acid, and p-coumaric acid) and 2 flavonoids (quercetin and Kampherol). Additionally, quinic acid and p-coumaric acid were found to be the major compounds, while quercetin was found to be the major flavonoid.
11
The high-performance liquid chromatography (Shimadzu HPLC) analysis of
Moreover, identification of phenolic compounds in several varieties of
Generally, the phenolic profile has also been shown to vary depending on the polarity of the extraction solvent, maturity, plant, and collection season. More importantly, the biosynthesis of polyphenols, flavonoids, and tannins has a qualitative and quantitative variation due to the variation in the physiology of the plant and the collection period. 26
The
In this respect, the antioxidant capacity varies significantly depending on the active ingredient composition, species, and the analyzed part. 29 Thus, the relatively large anti-radical activity of the various extracts of the azerola tree can be explained in part by their richness in flavonoids, 30 known for their potent antioxidant activity. 31
Previously, other studies reported that
The effect of
On the other hand, the high digestibility of these substrates was correlated in part to their small particle size. In fact, ruminal feed degradation and total tract nutrient digestibility can both be accelerated by smaller pasture particles. 34 According to Thomas and Russell 35 and Stojanovic et al, 36 this effect is triggered by the increased surface area that is created by forage size reduction, which allows quicker and simpler microbial adhesion and increases cellulose degradation.
The obtained results showed that the phenolic contents of fruits and leaves enhance rumen digestibility parameters, which is in line with the reports Benchaar and Greathead. 37 In the same context, Giger-Reverdin et al 38 demonstrated that goats’ ability to graze on tannin-rich shrubs and to detoxify tannins, polyphenols, and other secondary metabolites is much greater than that of other ruminants. The highest level of digestibility identified in leaves can be explained by their richness in total fat and nitrogenous matter. In fact, the supplementation in protein and fat promotes ruminal fermentation, microbial growth, total VFA concentration, and the digestibility of nutrients.39,40 Similarly, it has been demonstrated that protein supplementation in a low-quality and poor ration can improve feed usage and productive performance in ruminants. 41
On the other hand, data indicated that leaf digestibility, which is the richest in parietal constituents, was higher than that of fruits. This can be explained by the goats’ rumens micobiota composition, characterized by type B protozoa (
Indeed, Kara et al
8
demonstrated that total
The ruminal fermentation parameters (DOM, ME, and VFA) were predicted from the gas volume produced after 24 h of incubation. More importantly, the data analysis showed that the leaves had the highest levels of metabolizable energy, digestibility of organic matter, and volatile fatty acids. In this respect, Kara et al
8
demonstrated that
These results may be linked in part to the composition of microflora and microbial growth at the rumen level in goats. 44 In fact, goats are characterized by their ability to digest the most woodiest plants. Thus, the digestibility of dry matter and parietal constituents in a highly lignified rationed goat is higher than that of other small ruminants. This allows the goats to maintain a maximum rate of ruminal fermentation. 45
VFA represents the main forms of energy available after digestion, representing 70% to 80% of the total energy absorbed by ruminants.
46
In addition,
Typically, goats are the most adaptive ruminants to various and difficult pastures. This is explained by several physiologic particularities of their digestive system, such as the large area of nutrient absorption in their intestinal epithelium, the ruminal plasticity, and the huge salivary glands, which also reinforce their capacities for digestibility. 48
Limitations and Future Perspectives
The present study demonstrated the beneficial effect of
Conclusion
The current study underlines the chemical analysis of
Materials and Methods
Reagents and Chemicals
The chemical reagents (Folin-ciocalteu, sodium carbonate, quercetin, aluminum trichloride, vanillin, DPPH (2,2-diphenyl-1-picrylhydrazyl), BHT (butylated hydroxytoluene), hydrochloric acid, sulfuric acid, chloroform, NaOH, KH2PO4, sodium lauryl sulfate, CuSO4, gallic acid, quercetin, tannic acid, catechin, ethanol and methanol) were purchased from Sigma Chemical Co. (Sigma-Aldrich GmbH, Steinheim, Germany).
Plant Material and Growing Area
The fresh vegetal matter was washed with distilled water, and the leaves and edible parts of fruits were dried at 40 °C using an air-ventilated oven type BINDER. Then, crushed in a blender (RETSCH SK 300 type cross beater) to obtain a powder of 6 mm diameter, which was used in chemical analysis.
Mineral and Organic Matter Contents
The dry matter was determined after drying fresh samples (leaves and fruit) in a ventilated oven (Type BINDER) at 105 °C until a constant weight was reached (DM). The mineral content was obtained after calcining the DM at 550 °C in a muffle furnace (Tony Guller Orselina Zurich MOD L 51/5) for 4 h from a test portion of the sample until the white or gray. 50 The organic matter content is determined according to the following formula: % OM = 100% MM, where OM: organic matter and MM: mineral matter. Every sample was analyzed in triplicate.
Total Nitrogenous Matter Assessment
The dosage of total nitrogen has been realized according to the Kjeldahl method,
51
and samples were analyzed in three replicates. The procedure includes three steps. Firstly, the mineralization using Kjeldahl mineralizer type BEHR LABORTECHNIK, in an acidic medium by the addition of sulfuric acid with a catalyst (CuSO4). Secondly, the distillation of ammonia nitrogen (NH4+) to ammonia (NH3) by the addition of NaOH (10N) and adding it to a boric acid solution (4%) including two colored indicators (Methyl red and Bromothymol blue) using semi-automatic nitrogen distiller type VELP Scientfica. Finally, a titration with H2SO4 (0.1N) reached the end point. However, the total nitrogen matter (TNM) was determined according to the following formula: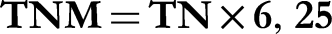
Parietal Constituents’ Determination
The determination of fibre contents was determined using fiber test type RAYPA, according to Van Soest et al
52
Total parietal constituents, which represent the NDF fraction (Neutral Detergent Fibre), were extracted using an NDS solution. The recovered fraction represents cellulose, hemicellulose, and lignin. Likewise, the lignocellulosic fraction was determined by the addition of ADS solution. The lignin content represents the ADL fraction (Acid Detergent Lignin), and all samples were analyzed in three replicates. The other parameters were determined using the following formula:
Total Lipid Quantification
Lipid extraction was established according to the method of Bligh and Dyer. 53 Briefly, 1 g of sample powder was mixed with hot distilled water for 10 min to inactivate phostholipases. After cooling, the mixture was filtrated through Whatman filter paper, and plant material was recovered and completely crushed using a porcelain mortar and pestle. Chloroform methanol solution (2:1; v/v) was added gradually, giving a total volume of 15 ml solvent/g dry matter. Finally, the obtained homogenate was allowed to settle, and the chloroform layer containing the total lipids was recovered and placed in a Binder ventilated oven at 60 °C during 48 h. All samples were analyzed in three replicates.
Extracts Preparation
For each sample, the filtrate phase was evaporated in a ventilated oven (type BINDER) at 45 °C, and its residue was mixed with 3 mL of distilled water. Indeed,
Total Polyphenol Assessment
The dosage of total polyphenols was established according to the Folin-Ciocalteu method described by Dewanto et al 54 For each assay, 100 µL of azerole extracts were mixed with 2 mL of a sodium carbonate Na2CO3 (2%) solution, freshly prepared, and incubated for 5 min. The mixture was added to 100 µL of Folin-Ciocalteu (1N) and incubated for 30 min at room temperature in the dark. The absorbance was determined using a GENESYSTM 8 Unicam benchtop visible spectrophotometer at a wavelength (λ) = 750 nm. The absorbance was read at 750 nm. The total polyphenol content was determined by a calibration curve of gallic acid used as a standard. The polyphenol concentrations were determined in milligrams of gallic acid equivalent per gram of dry matter (GAE/g DM). All samples were analyzed in three replicates.
Total Flavonoid Contents
The quantification of total flavonoids was carried out by the method of aluminum chloride (AlCl3) reported by Dawno et al 54 Briefly, an intake of 250 μL of extract was mixed with 75 μL of NaNO2 (5%) and kept in the dark for 6 min at room temperature. Then, 150 μL of AlCl3 (10%) was added to the mixture and incubated for 5 min. This preparation was added to 500 μL of NaOH (1 M), and the final volume was increased to 2.5 mL with distilled water. The absorbance was determined using a GENESYSTM 8 Unicam benchtop visible spectrophotometer at a wavelength (λ) = 510 nm. Total flavonoid contents were determined by a quercetin calibration curve, and the concentrations were expressed in milligrams of quercetin equivalent per g of dry matter (mg EQ/g DM). All samples were analyzed in three replicates.
Total Tannin Determination
The dosage of total tannins was performed by Folin-Ciocalteu, previously described by Ribarova et al 55 In fact, 100 µL of the extract was added to 500 μL of Folin-Ciocalteu, 7.5 mL of distilled water, and 1 mL of Na2CO3 (35%), and the distilled water was adjusted to 10 mL. The mixture was stirred and kept in the dark at room temperature for 30 min. Similarly, different concentrations of tannic acid were carried out under the same operating conditions as the samples used as a standard to quantify total tannins. The absorbance was determined using a GENESYSTM 8 Unicam benchtop visible spectrophotometer at a wavelength (λ) = 725 nm. The results were expressed in milligrams of tannic acid equivalent per g of dry matter (mg TAE/g DM). All samples were analyzed in three replicates.
Condensed Tannin Quantification
Condensed tannin contents were measured calorimetrically according to the method of Sun et al. 56 Briefly, for each assay, 50 μL of the extract was added to 3 mL of vanillin (4%) in methanol, including 1.5 mL of concentrated sulfuric acid. The mixture was incubated for 15 min at room temperature in the dark. The absorbance was determined at 500 nm, and all samples were analyzed in three replicates. The content of condensed tannins was determined from the linear calibration curve, using catechin as a standard at increasing concentrations (0, 10, 50, 100, 200, 400, 800, and 1000 μg/mL). The results were determined from the linear regression equation of the calibration curve: y = 0.0543x and expressed in mg EC/g DM.
All samples were analyzed in three replicates.
Free Radical-Scavenging Activity
The 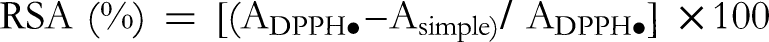
Antioxidant Activity by the (ABTS•+) Test
The ABTS method was used to assess the antioxidant activity of 
As a positive control, BHT was produced in the same quantities as the test extracts. The extract concentration needed to scavenge 50% of the (ABTS•+) was defined as the RSA results that were shown as IC50 values (µg/mL).
In Vitro Ruminal Fermentation
The ruminal fermentation of azerole berries and leaves was performed using the
Organic matter digestibility (OMD), metabolizable energy (ME), and volatile fatty acid level (VFA) were determined according to Makkar
59
and Menke et al
61
methods using the following formulas:


Statistical Analysis
The data were analyzed using statistical analysis by the variance according to the GLM procedure of the software SAS (2002)
62
and compared by the Duncan multiple rank test, and a 
For digestibility parameters, the kinetics of gas production were analyzed using non-linear regression according to the model of Orsskov and Macdonald.
51
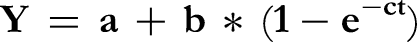
Footnotes
Acknowledgements
The financial support of the Tunisian Ministry of Higher Education and Scientific Research and the Institution of Agricultural Research and Higher Education is acknowledged
Authors’ contributions
H.S. and S.J. performed the experiments, analyzed the data, and wrote the article. A.A. and H.S. helped in data processing and analyses. H.S. supplied materials and reagents and assisted in the planning of experiments.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval to report this case was obtained from the “Institut Pasteur de Tunis” (JORT472001) “Comité d'Ethique Bio-medicale (CEBM)” accepted the procedure. The Tunis University local ethics committee's guidelines for the use and care of animals, as well as the National Institutes of Health's suggestion, were followed in all experimentations and sacrifice procedures.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with “Comité d'Ethique Bio-medicale (CEBM)” from the Tunis Pasteur Institute (JORT472001) approved protocols.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
