Abstract
Background
Innula cappa (Buch.-Ham. ex D. Don) DC (Compositae) is used in folkloric medicine to treat diabetes and other ailments. The current study aimed to assess the anti-hyperglycemic and antioxidant effects of methanolic extract and solvent fractions of I. cappa leaves as well as bioassay guided isolation of active compounds from the active extract.
Method
The α-amylase and α-glucosidase inhibition of the extracts was evaluated by colorimetric assays, and the anti-diabetic activity was tested on alloxan-induced diabetic mice model. Moreover, flame emission and atomic absorption spectrometry techniques were employed to analyze the chemical composition of the plant. Compounds were identified and isolated from the leaf latex of I. cappa using FTIR, GCMS, silica gel column chromatography, and their structures were determined using 1H-NMR, 13C-NMR, and ESI-MS spectral data.
Results
The results showed that the ME showed significant α-amylase, α-glucosidase inhibitory hyperglycemic activities, significantly improving body weight, glucose tolerance, lipid profiles, and hematological markers. The UV-FTIR spectra revealed diagnostic peaks corresponding to several functional groups whereas GC-MS spectral analysis revealed 35 phytochemical components with different retention times in the ME of I. cappa. Spectroscopic isolation of the ME led to the isolation 5 compounds including ursolic acid, benzoic acid, 2-hydroxybenzoic acid, stigmasterol and β-sitosterol.
Conclusion
ME exert significant antihyperglycemic and antioxidant effects demonstrating the medicinal value of I. cappa and emphasize its significance as unexplored natural reservoirs of phytogenic chemicals that require further investigation using cutting-edge analytical techniques.
Introduction
Diabetes mellitus (DM) is characterized by chronic hyperglycemia, abnormalities in lipid, protein, carbohydrate metabolism, and irregularities in insulin action or secretion or both are. 1 Polydipsia, polyuria, polyphagia, weight loss, occasionally obscured eyesight, and an increased risk of infectious diseases are all common side effects of hyperglycemia. The increase in reactive oxygen species brought on by hyperglycemia gradually results in serious consequences. 2 Many approaches are being researched as alternatives to the treatment of DM in order to improve the effectiveness of medication, including herbal and nanoparticulate systems. Hyperlipidemia, which can be brought on by a number of different metabolic disorders, affects about 40% of people with DM. 3 Chronic oxidative stress brought on by hyperglycemia also weakens the antioxidant defense system, leading to increased apoptosis and defective insulin gene expression. 4 Most diabetic drugs in clinical practice have poor glucose control and are associated with lactic acidosis liver and renal problems. 5
Both preventative and curative pharmaceutical preparations for humans heavily rely on medicinal plants. Herbal medicines are the only affordable source of healthcare, especially for the poorest patients. 6 Furthermore, because of their quality, safety, effectiveness, low side effects, and ease of access, herbal medications are becoming increasingly popular in both developed and developing nations. The plant-derived products are important sources of bioactive compounds, such as antioxidants, and are used to produce new treatment agents for DM. 7 Ethnobotanical research reports that over 800 plants are used as traditional diabetes therapies in underdeveloped nations because of their effectiveness, affordability, and absence of side effects. 8 Although plant-based sources have the potential to be used as antidiabetic drugs, the scientific community has not given them enough attention. It has recently been recognized that a nutritious diet that includes appropriate herbs can help lower the prevalence of diabetes. 9
Innula cappa (family compositae) is used in the Ayurvedic medicinal system for the treatment of bronchitis, diabetes, fever, hypertension, and rheumatism. 9 Several compounds including isoalantolactone, germacranolide, β-sitosterol, and lupeol isolated from I. cappa demonstrated a variety of biological actions against diseases caused by oxidative stress, including cancer, diabetes, inflammation, and neurological disorders. 10
Literature studies have demonstrated that I. cappa has the ability to remove heat and harmful substances, hemostasis and analgesia. It is used to treat fever and chills, thoracoabdominal discomfort, pharyngitis, irregular menstruation, postpartum hemorrhage and palpitations. 11 Chemical composition of I. cappa shows that it is mainly composed of flavonoids, phenols, terpenoids, organic acids, and volatile oils. 12 A number of biological characteristics of I. cappa leaves have been reported by prior pharmacological studies including anti-inflammatory, antibacterial, analgesic, antioxidant, anticancer, immunomodulatory, and antidiabetic activities.13–15
From the literature, it is evident that it is used against various diseases because of its rich phytochemical contents. Additionally, not much research has been carried out regarding its potential to have antidiabetic effects. Thus, this study sought to determine the scientific basis for utilization of I. cappa leaves by traditional practitioners in management of diabetes by administering 70% methanol extracts to mice with alloxan-induced diabetes. Furthermore, an effort was made to identify specific bioactive compounds that may be responsible for the antidiabetic activity and elucidate the structure of pure isolated compounds using advance chromatographic and spectroscopic experiments with a hope to find some novel and interesting structure that will add the number of known natural compounds from this medicinally important plant species.
Material and Method
Plant Material
The fresh leaves of I. cappa were collected before the flowering stage in May 2023 from Chagharzai region (72° 34.53 E/72.63 N) of District Buner, KPK Pakistan. The plant was identified by botanist from the Department of Botany Abdul Wali Khan University Mardan, and the voucher specimen number/accession number AWKB-0523 was subsequently deposited in the Herbarium of the Botany department. The clean leaves were then dried in air drying oven for 48 h at 40 °C. The dried leaves were pulverized into powder using a blender and stored at 4 °C. The plant leaves were employed in current investigation because the recipes are traditionally prepared as hot water decoctions. 16
Preparation of Extract and Fractionation
The finely powdered I. cappa leaves (500 g) were mixed with methanol (70%) for the preparation of ME by cold maceration process for a period of 72 h. The ME was prepared by maceration process and concentrated using rotary evaporator (Heidolph Laborota 4000, Germany) at a temperature of 60 °C. The semisolid ME was subsequently fractionated with n-hexane, chloroform and ethyl acetate liquid-liquid partitioning method to produce fine extracts. The extracts were collected and preserved in a desiccator containing anhydrous sodium sulphate to remove traces of water in them at 4 °C until used for further studies. No preservatives were added. Further analyses used the ME. The percentage yield of the crude ME was calculated to be 3.17%. This stock was used to make new preparations each time they were required. 16
Experimental Animals
The study was carried on sixty healthy adult male Swiss albino mice weighing 25–30 g, which were obtained from Veterinary Research Institute (VIR) Peshawar, Pakistan. The animals were housed in polypropylene cages layered with husk and kept in a semi-natural light/dark condition (12 h light/12 h dark). The animals were allowed free access to water and standard pellet diet as well as standard laboratory and environmental conditions (temperature of 25 to 35 °C and relative humidity of 50% to 55%). Research was conducted in compliance with the globally recognized guidelines for the handling and application of laboratory animals. 17
Induction of Diabetes
In this study a single dose of intraperitoneal injection of alloxan (Sigma Aldrich, Germany) Albino mice were injected intraperitoneally with with alloxan (150 mg/kg i.p) (Sigma Aldrich, Germany) dissolved in 0.1 M citrate buffer (pH = 6.5) was used to induce diabetes in overnight fasted mice. After 72 h of alloxan injection, the mice were checked for BGL from the tail vein using glucometer (Accu-check, Roche diagnostic USA). Only the mice with FBG ≥ 200 mg/dL, were considered to be diabetic and included in the study. 18 The mice were allowed to free acess to 10% glucose solution to avoid early hypoglycemia.
Experimental Design
Forty eight mice were selected for the initial investigations in the experiments. Six normal healthy mice were chosen randomly for control group. Forty two diabetic-induced mice were selected and six mice were randomly assigned for each group for the study.
19
Group I - NS (control mice orally administered with distilled water) Group II - DC (Alloxan-induced mice administered orally with distilled water) Group III - Gb (Alloxan-induced mice administered orally with glibenclamide 10 mg/kg) Group IV - ME (Alloxan-induced mice administered orally with methanolic extract 250 mg/kg) Group V - Hex (Alloxan-induced mice administered orally with n-Hexane extract 250 mg/kg) Group VI - Chl (Alloxan-induced mice administered orally with chloroform extract 250 mg/kg) Group VII - EA (Alloxan-induced mice administered orally with ethyl acetate extract 250 mg/k) Group VIII - AE (Alloxan-induced mice administered orally with aqueous extract 250 mg/kg) All the treatments were started 72 h after alloxan injection and continued for 15 days.
Determination of Blood Glucose Levels
The BGLs of mice were measured before and then throughout the experiment using a fractional tail amputation technique using one-touch electronic glucometer with glucose test strips (Accu-ChekActive, Roche Diagnostics, Germany). Blood was withdrawn from the mice on the day 4, 7, 11 and 15th after diabetes-induction for assessment of blood glucose. 19 The alteration in the body weight was observed throughout the treatment in the experimental animals.
Biochemical Analysis
The blood samples for biochemical analysis was collected via retro-orbital plexus after 15 days of treatment from mice starved overnight under diethyl ether anesthesia before sacrificing and was then left to coagulate for 30 min. The serum was separated by centrifuging the material for 10 min at 25 °C at 3000 rpm. It was then investigated for the estimation of biochemical parameters such as lipid profile (TC, TG, LDL and HDL), liver and kidney function tests (ALT, AST, ALP, urea and creatinine). These biochemical parameters were measured through Reflotron Plus biochemical analyzer (Roche Diagnostics, Germany) and commercially available Reflotron strips.20,21
Assessment of Toxicity in Mice
The preliminary toxicological experiments were conducted in accordance with OECD-423 to ascertain whether plant extracts had any harmful effects. 22 The overnight fasted mice were given oral doses of extracts (500, 1000, 2000, and 2500 mg/kg) for toxicity assessment. The general behavior of experimental mice was observed regularly to ensure that the mice showed no overt behavioral problems, such as shivering, drowsiness, trembling, contractions, piloerection, loss of weight, agitation, or paralysis. 23
Ethical Considerations
The experimental procedure was approved by the Abdul Wali Khan University Animal Ethics Committee. The experimental protocols were executed after a week of acclimatization in accordance with the National Research Council Committee, Guidelines for Care and Use of Laboratory Animals in Biomedical Research (USA). 24
Antidiabetic Enzymatic Assays
The α-amylase and α-glucosidase inhibitory activities of plant extracts were assessed based on standard protocols. 25 The findings were presented as percentage of inhibition.
Elemental Analysis
The ME obtained from I. cappa leaves was subjected to the elemental analysis using SEM along with an energy dispersive X-ray spectrometer (EDX) (Model SPG No. 1; Unicam, Cambridge, UK) at the appropriate temperature, wave-length, and lamp current for each element following the method. 26 The following elements’ reference standards were obtained from Sigma-Aldrich (USA): Zn, Na, K, Ca, Mg, Cu, Ni, Fe Mn, Co, and Cr. The manufacturer's guidelines were followed for setting the operating parameters for the functioning elements.
UV-Vis Spectrophotometric Analysis
The ME was dissolved in a ratio of 1:10 (w/v) in methanol. It was filtered through Whatman (1) filter paper and the extract was centrifuged at 12,000 rpm for 10 min and again filtered through Whatman (1) filter paper, further diluted in a ratio of 1:10 with methanol and then scanned from 200 to 800 nm on Beckham Coulter DU 720 ultraviolet–visible (UV–Vis) spectrophotometer (Beckham Coulter Inc., USA) using methanol as the blank. 27
FT-IR Spectroscopy
FTIR characterization of the dry crude extract (1 mg) was performed at room temperature using a Nicolet 6700 FTIR spectrophotometer (Thermo Scientific, USA). The spectral resolution was set at 4 cm−1 and the scanning was done from 400 to 4000 cm–1. The analysis was repeated twice for spectrum confirmation. The spectral resolution was tuned to 4 cm−1 at a rate of 16 nm/s. The infrared spectra obtained from the sample were interpreted by comparing the spectral data with references from the identification of functional groups contained in the sample. 28
Thin Layer Chromatography and Column Chromatography
The active fraction of ME was dissolved in methanol in a ratio of 1: 20 (w/v). A spot of the solution was carefully applied onto TLC plates and left to dry. The plates were developed in prepared solvent systems of varying ratios of different solvent systems to allow the compounds in the spot to move upwards by capillary attraction. The positions of the different compounds were observed under UV light at 254 nm and 365 nm. The starting mobile phase considered was 100% n-hexane followed by ethyl acetate and then methanol. The migration behavior of the separated substances (visible spots) was measured and expressed as a retardation factor (Rf value) in order to qualitatively assess the plates. The separated components (visible spots) were observed under both visible and ultraviolet (UV) light. The solvent ratios giving the best separation of the compounds in the extract were used for CC.
The active extract (5 g) was subjected to silica gel (60-120 mesh) normal phase CC (7.5 × 100 cm column). Silica gel (100 g) was mixed with hex/EA (3:7) solvent system to form a homogenous suspension (slurry) and stirred using a glass-stirring rod to remove bubbles. The silica gel slurry was then poured into the column. The sample to load on the column was prepared by mixing and grinding 0.5 of the extract with 0.5 g of silica gel into fine green powder. The sample was then introduced into the column and eluted with the solvent system established through TLC profiling. For each eluent system, equal fractions were collected and fractions with similar TLC profiles were pooled together and concentrated to dryness by rotary evaporation. 26
GC-MS Analysis
The active fractions obtained were reconstituted in dichloromethane: methanol (1:1, v/v) mixture, filtered through 0.45 μm filters and transferred to 2 mL vials one at a time for GC-MS analysis. The ME was analyzed using a gas chromatograph (Agilent USB-393752) fitted with a capillary column and a FID detector as per specification previously published. 27 The peaks were recognized by comparing the mass spectra of the GC peaks with the National Institute of Standards and Technology's version 11 databases.
By comparing the retention times of the samples with those of previously identified compounds in the literature and using spectrum data obtained from NIST, Wiley, and NIST libraries, the components of the samples were identified. Furthermore, for additional authentication, the mass spectra's fragmentation pattern was compared to previously released data. 27
NMR Experiments
The defatted ME (0.1 g) was dissolved in 0.7 ml of methanol-D4 (containing TMS, Cambridge Lab. USA) to prepare the NMR sample in a 5 mm NMR tube. A determined quantity of DMSO was applied as an internal standard. 1H NMR spectra were acquired on a 400 MHz on a JEOL Lambda 400 NMR spectrometer (9.4 Tesla) and on a 500 MHz Varian (11.75 Tesla). 13C NMR spectra were acquired at the corresponding frequencies: 100 MHz (9.4 Tesla) and 125 MHz (11.75 Tesla). The same spectral parameters were used for both instruments following standard protocols. 28
Statistical Analysis
The data analysis was conducted using IBM SPSS statistics for Windows, version 21 (IBM Corp., Armonk, N. Y. USA). Using a mixed-design ANOVA, the association between and within groups was determined in addition to the mean and standard error. A -value of less than 0.05 indicated the statistical significance level results.
Results
Study of Acute Oral Toxicity
The LD50 value of I. cappa extracts were tested up to dose of 2000 mg/kg and it was discovered that the mice were not killed or shown any signs of toxicity at any of the doses tested, The LD50 value was found to be greater than 2000 mg/kg, which, in accordance with the Hodge and Sterner scale, falls within the practically harmless range.
Antidiabetic Activity of I. cappa in Alloxan-Induced Diabetic Mice
The results indicated that FBGL during experimental period (day 0-15) were considerably higher in diabetic control group as compared to normal control. Significantly decreased BGLs were observed in glibenclamide group (10 mg/kg) from day 0 to day 15 (413.2 ± 2.3-202.3 ± 1.4) and ME (250 mg/kg) (411.3 ± 2.4-210.4 ± 2.5) However, in the case of other fractions, no obvious glucose-lowering efficacy was observed in alloxan-induced diabetic mice as shown in Table 1.
Hypoglycemic Activity of ME and Solvent Fractions of I. cappa Leaves in Alloxan-Induced Diabetic Mice.

Values are expressed as mean ± SEM n = 6 in each group. * P < 0.05; **P < 0.01; compared with diabetic control. (NS = Normal saline, DC = Diabetic control, Gb = Glibenclamide, ME 1= Methanolic extract 1, ME 2= Methanolic extract 2, Hex = n-Hexane, Chl = Chloroform, EA = Ethyl acetate, AE = Aqueous extract)
The BW of DC group was significantly reduced compared to the normal control. The animals treated for with glibenclamide (10 mg/kg) and I. cappa ME (250 mg/kg) showed substantial (P < 0.01) rise in body weight. Body weight began to gradually increase on day three following treatment with ME comparable to Gb. However, in case of other fractions, no significant restoration in body weight was seen, suggesting that they had no sufficient effect on body weight as shown in the Table 2.
Effect of ME and Solvent Fractions of I. cappa Leaves on Body Weight in Alloxan-Induced Diabetic Mice.

Values are expressed as mean ± SEM n = 6 in each group. * P < 0.05; **P < 0.01; compared with diabetic control. (NS = Normal saline, DC = Diabetic control, Gb = Glibenclamide, ME 1= Methanolic extract 1, ME 2= Methanolic extract 2, Hex = n-Hexane, Chl = Chloroform, EA = Ethyl acetate, AE = Aqueous extract)
Serum lipid (TC, TG, HDL, and LDL) levels measured on day 0 did not reveal any statistically significant changes between the groups. When Gb and ME were administered to diabetic mice, the levels of blood TC, TG, and LDL were reduced (p˂0.05, p˂0.01) while the levels of HDL were increased (p˂0.05, p˂0.01) in a dose-dependent way in comparison to the diabetic control group. Other fractions had less obvious effects and slightly altered lipid profile. The antihyperlipidemic effect of ME and fractions are depicted in Table 3.
Effect of ME and Solvent Fractions on Lipid Profile.

Values are expressed as mean ± SEM n = 6 in each group. * P < 0.05; **P < 0.01; compared with diabetic control. (NS = Normal saline, DC = Diabetic control, Gb = Glibenclamide, ME 1= Methanolic extract 1, ME 2= Methanolic extract 2, Hex = n-Hexane, Chl = Chloroform, EA = Ethyl acetate, AE = Aqueous extract); (TC = Total cholesterol, TG = Triglycerides, LDL = Low-density lipoprotein cholesterol, HDL = High-density lipoprotein)
The serum AST, ALT and ALP levels in the blood were considerably higher in the mice of DC group than normal control group suggesting liver damage. In comparison to the DC group, the glibenclamide group (10 mg/kg) exhibited a significant decrease in these enzymes levels. Similarly a dose-dependent decrease in ALP, ALT and AST was observed in mice treated with ME (250 mg/kg). Serum creatinine and urea levels were also significantly improved by the ME (250 mg/kg) in diabetic mice as shown in Table 4.
Biochemical Analysis of ME and Solvent Fractions of I. cappa Leaves.

Values are expressed as mean ± SEM n = 6 in each group. * P < 0.05; **P < 0.01; compared with diabetic control. (NS = Normal saline, DC = Diabetic control, Gb = Glibenclamide, ME 1= Methanolic extract 1, ME 2= Methanolic extract 2, Hex = n-Hexane, Chl = Chloroform, EA = Ethyl acetate, AE = Aqueous extract: ALT = alanine aminotransferase, AST = aspartate aminotransferase, ALP = Alkaline phosphatase
α-Amylase Inhibition Activity (%)
The α-amylase enzyme's inhibitory activity was examined for test formulations in various solvents. Significant inhibition was shown by ME, Chl, and EA formulations at 500μg/ml as illustrated in Table 5. Maximum activity (64.8 ± 1.96) was observed for ME formulation in phosphate buffer saline as compared to acarbose (78 ± 1.9).
α-Amylase Inhibitory Activity of ME and Solvent Fractions of I. cappa Leaves.

(Result are expressed as mean ± SEM (*p < 0.05, **p < 0.01)
(Me = Methanolic extract, Hex = n-Hexane, Chl = Chloroform, EA = Ethyl acetate, AE = Aqueous extract)
α-Glucosidase Inhibitory Activity (%)
The ME showed greatest inhibition (70.6 ± 2.7) of α -Glucosidase activity at 500μg/ml compared to standard acarbose (83.5 ± 2.8). The effects of various formulations on the inhibitory activity of α-glucosidase at various concentrations are shown in Table 6.
α-Glucosidase Inhibitory Activity of ME and Solvent Fractions of I. cappa Leaves.
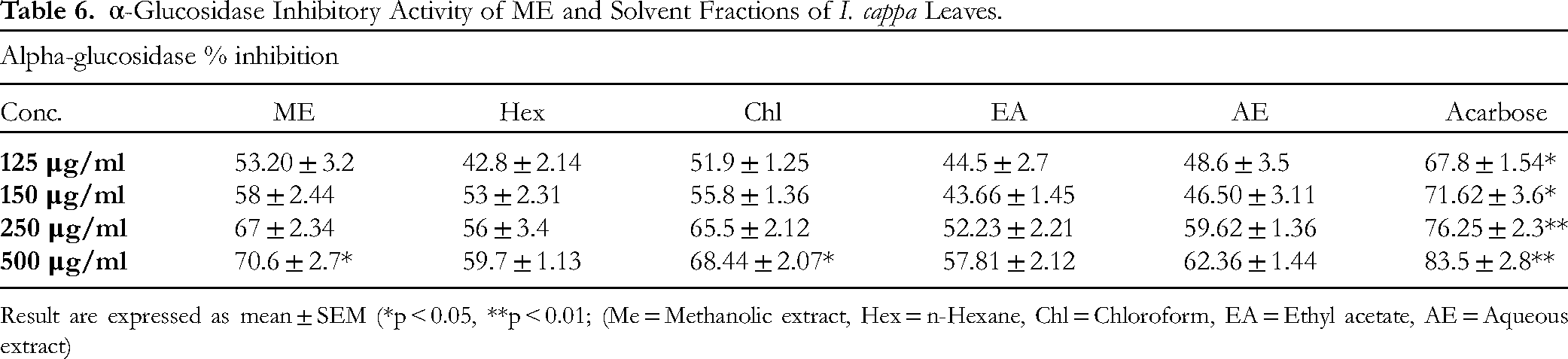
Result are expressed as mean ± SEM (*p < 0.05, **p < 0.01; (Me = Methanolic extract, Hex = n-Hexane, Chl = Chloroform, EA = Ethyl acetate, AE = Aqueous extract)
Chromatographic Study with ME
The CC study was carried out with the active ME to separate the eluates namely, F1–12 (F1 4.2% w/w), F13- F24 (F2 3.42% w/w), F25- F34 (F3 2.51% w/w), F35- F44 (F4-4.78% w/w), F45- F53 (F5-3.82% w/w), F54- F61 (F6-4.68% w/w), F62-F69 (F7-4.71% w/w) and F70-F83 (F8-4.44% w/w) using different solvents on polarity basis. A total of 83 fractions of 50 ml each were collected. Each fraction was then screened for their in vivo antidiabetic activities. The eluates with same Rf value were pooled together to afford 8 major fractions (MetA-MetH) and evaporated to dryness.
The pooled fractions of ME, F1–12, F13- F24, F25- F34, F35- F44, F45- F53, F54- F61, F62-F69 and F70- F83 were named as MetA, MetB, MetC, MetD, MetE, MetF, MetG and MetH respectively as shown in Table 7. The pooled eluates were tested in fasting blood glucose level in alloxan induced diabetic mice.
Column Chromatography Profile of Leaf Extracts I. cappa.
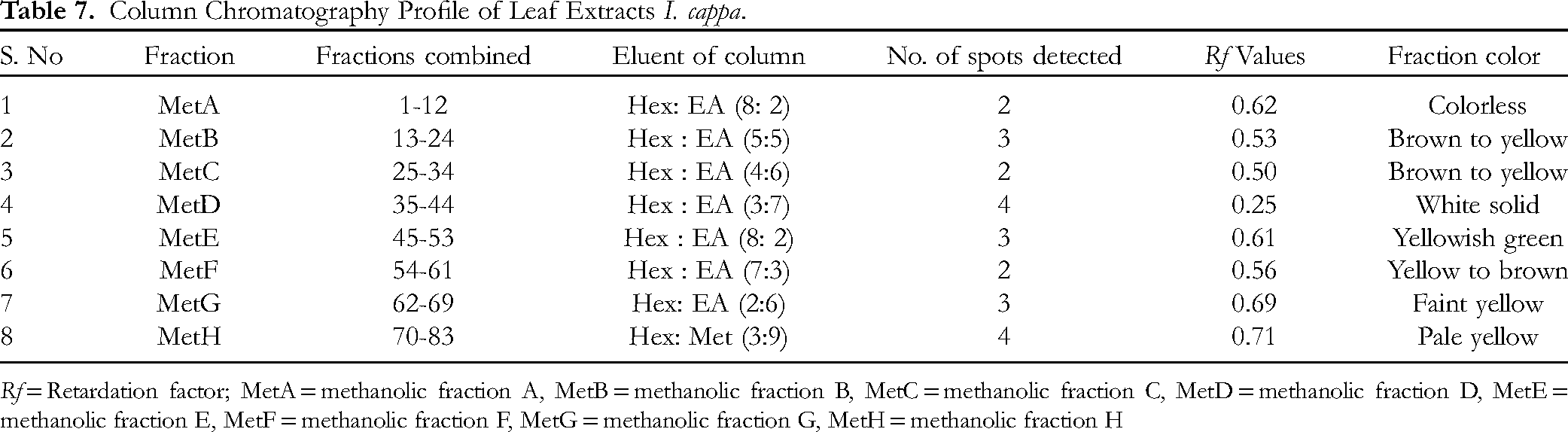
Rf = Retardation factor; MetA = methanolic fraction A, MetB = methanolic fraction B, MetC = methanolic fraction C, MetD = methanolic fraction D, MetE = methanolic fraction E, MetF = methanolic fraction F, MetG = methanolic fraction G, MetH = methanolic fraction H
Effects of Various Isolated Fractions ME (100 mg/kg), and Glibenclamide (50 mg/kg) on Blood Glucose in Alloxan Induced Diabetic Mice
From the study it was observed that the fraction “MetD” showed significant (P < 0.05) decrease in blood glucose but the other fractions did not show significant effect of blood glucose when compared with normal control. The results of the effect of various isolated fractions of ME (100 mg/kg) on the BGL in alloxan induced diabetic mice are shown in Table 8.
Effect of Isolated Fractions of ME in Diabetic Mice.

Values indicate mean ± SEM (n = 6), *p < 0.05, **p < 0.01 compared with normal control.
(MetA = methanolic fraction A, MetB = methanolic fraction B, MetC = methanolic fraction C, MetD = methanolic fraction D, MetE = methanolic fraction E, MetF = methanolic fraction F, MetG = methanolic fraction G, MetH = methanolic fraction H)
Characterization of Bioactive Fraction and Structure Elucidation
The inactive and less active fractions were discarded. Finally, the active fraction elute was selected for the structural characterization by UV-FTIR, GC-MS and NMR (1H & 13C) analysis.
Ultraviolet–Visible Spectrum of the MetD
The phytochemicals in the active fraction MetD were confirmed by spectroscopic scanning for characteristic peaks in the ultraviolet and visible light regions. The scan was to establish if compounds containing σ-bonds, π-bonds, and lone pair of electrons, chromophores and aromatic rings were present. The spectrum for the extract showed peaks at 660, 356, 305 and 237 nm with different absorption values as shown in Figure 1.
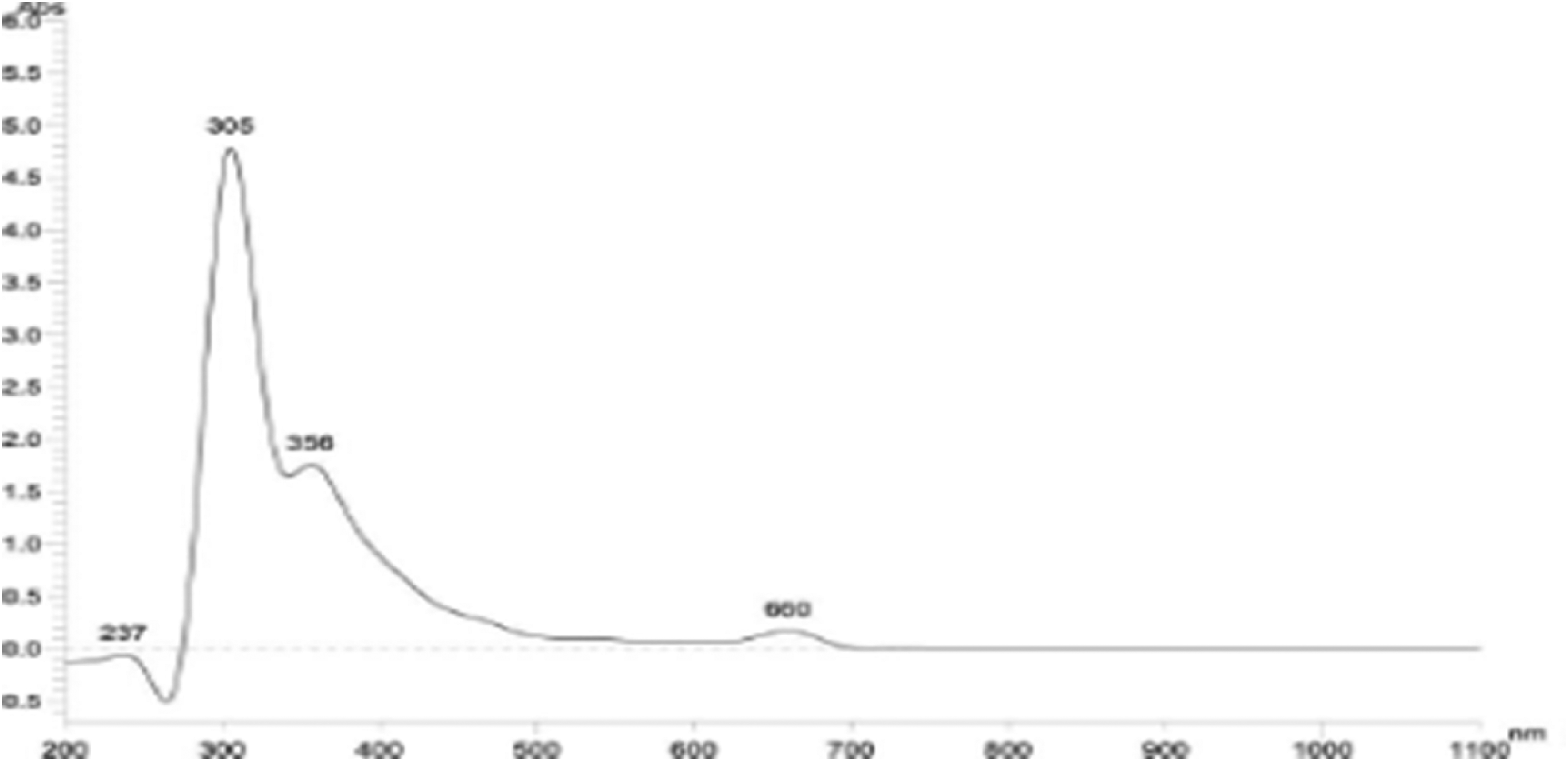
Uv spectrum of MetD of I. cappa leaves.
FTIR Spectroscopic Analysis of MetD
The FTIR spectrum of the active fraction revealed peaks at distinct wavelengths which corresponds to various functional groups of phytocompounds as depicted in Figure 2 and Table 9. The FTIR spectra indicate the presence of functional groups such as hydroxyl, carbonyl, ketone and ethereal linkage. Therefore, it was further investigated with other sophisticated analytical techniques to confirm the presence of the functional groups and to elucidate the structure.

FTIR Spectrum of MetD.
FTIR Profile of MetD.

Elemental Analysis
The existence of major, minor and trace elements that are pharmaceutically active has been established by elemental analysis of active ME using the ED-XRF technique as shown in Table 10.
XRF Analysis of MetD.

Elemental mean value (Mean ± SEM)
GC-MS Chromatogram Analysis
In-depth phytochemical identification, the ME (MetD) was further evaluated by GCMS. The GCMS chromatogram of MEtD showed 35 peaks of compounds. The GCMS chromatogram of compounds was identified by comparing their peak retention time, height (percent), and patterns of mass spectral fragmentation to those of known compounds present in the National Institute of Standards and Technology library (NIST). In GCMS, most of the compounds related to the classes like fatty acids, steroids, vitamins, and esters. The identified compounds from MEtD are described in Figure 3 and Table 11.

GC-MS chromatogram of MetD.
GC-MS Analysis of ME of I. cappa.

MF = Molecular formula, MW = Molecular Weight, RT = Retention Time, PB = Probability
Identification and Structure Elucidation of MetD Fraction
The results of the present study showed that the

1H NMR spectral elucidation of the compounds (
Spectroscopic Data of Isolated Compounds from I. cappa.

13C-NMR Data for Ursolic Acid
13CNMR (125 MHz, DMSO-d6) δ 36.9, 27.4, 72.2, 39, 56, 19, 33.5, 40, 46.5, 37, 24, 115, 135, 44, 29, 24, 47.4, 53, 39, 38.6, 31, 36.2, 28.70, 16.5, 15.8, 17, 23.7, 17.9, 17.5, 22.
13C-NMR Data for Benzoic Acid
13CNMR (125 MHz, DMSO-d6) δ 128.4, 131.2, 127.8, 133.4, 124.9, 130.8, 180.0.
13C-NMR Data for 2-Hydroxybenzoic Acid
13CNMR (125 MHz, DMSO-d6) δ 122.7, 133.5, 113.8, 162.3, 114.9, 129.4, 180.0, 37.8, 53.8, 137.6, 36.2, 28.7, 119.1, 163.3.
13C-NMR Data for Stigmasterol
13CNMR (125 MHz, DMSO-d6) δ 37.2, 31.7, 71.1, 42.3, 140.8, 121.7, 31.9, 36.1, 50.2, 50.2, 36.5, 19.8, 39.7, 45.9, 56.9, 24.3, 28.9, 56.2, 12.2, 19.4, 40.4, 21.1, 138.3, 129.3, 51.2.
13C-NMR Data for β-Sitosterol
13CNMR (125 MHz, DMSO-d6) δ 37.2, 31.7, 71.1, 42.3, 140.8, 121.7, 31.9, 36.1, 50.2, 36.5, 19.8, 39.7, 45.9, 56.9, 24.3, 28.9, 56.2, 12.2, 19.4, 40.4, 21.1, 138.3, 129.2, 51.2, 34.0, 19.06, 21.2, 25.40, 13.06.
Discussion
The current investigations were carried out to ascertain the antioxidant and antihyperglycemic effect I. cappa in alloxan-induced diabetic mice as well as bioassay guided isolation and characterization of compounds from the active extract (ME). Alloxan induces diabetes by producing ROS, which cause the pancreatic beta-cells to rapidly degrade, resulting in hyperglycemia leading to free radicals production due to glucose auto-oxidation.29,30 Oral glucose tolerance test studies showed that the BGL of alloxan-induced diabetic mice was increased to a significant level. Administration of ME (250 mg/kg) significantly lowered the mean BGLs resulting in hypoglycemia. The presence of phytocompounds and antioxidants in the plant might be responsible for lowering glucose levels which is in line with the previous studies.31,32 Numerous studies have demonstrated that medicinal plants with hypoglycemic properties work through a variety of processes, such as increasing insulin production, improving target cell insulin sensitivity, and promoting the regeneration of the β-cells in the pancreatic islets of Langerhans. 33 According to several writers, the antidiabetic action of flavonoids, steroids, terpenoids, and phenols is caused by their presence. 34 Flavonoids have also been shown to function as insulin secretagogues and repair injured beta cells in rats with diabetes induced by alloxan. 35
In addition to elevated glucose levels, it is typified by a changed lipid profile due to abnormally active fat mobilization from adipose tissues and uncontrolled lipolytic hormonal effects on fat storage. 36 Diabetic animals showed a different lipid profile than normal mice, with considerably higher concentrations of TC, TGs, and LDL and lower concentrations of HDL. The administration of ME resulted in a considerable improvement in the altered lipid profile, with a significant decrease in TC, TG, and LDL and an increase in HDL. Improved hepatic glycogenesis, lipolytic hormone modulation, and inhibition of lipid peroxidation consumption could be the likely mechanism of lipid profile correction. 37 Our result is consistent with studies that a number of plants have this type of anti-hyperlipidemic effect.38,39
Increased levels of the liver enzymes ALT, ALP, and AST are considered to be reliable markers of hepatotoxicity and are indicative of liver cell destruction. These enzymes are increased in diabetic mice due to damage to the hepatic cells. 40 Furthermore, increased ROS levels are mostly responsible for the inflammatory damage to liver cells. Previous studies have demonstrated that elevated levels of AST, ALP, and ALT are linked to diabetic situations, including those caused by alloxan in experimental mice. 41 Mice with diabetes who received ME of leaves showed liver and renal architectural restoration that was on line with earlier studies.42,43
Inhibition of intestinal pancreatic α-amylase and α-glucosidase activities results in delayed carbohydrate digestion of absorbable monosaccharides leading to a drop in postprandial hyperglycemia.44,45 The ME in both the cases caused significant inhibition of these enzymes compared to standard acarbose. The study is consistent with pervious research works.46,47
The GC-MS spectral data of the ME tentatively identified a number of chemicals that were determined to be efficacious in terms of their antidiabetic and antioxidant potentials. Literature studies have shown that some of the compounds have been the subject of both pharmacological and toxicological studies. In diabetic context Pyrrolidine, 2-Methoxy-4-vinylphenol, Dasycarpidan-1-methanol acetate, 9-Octadecenoic acid, Oleic acid, Phloroglucinol, O,O'-bis (acetyl)-, n-Hexadecanoic acid, Docosanoic acid, à-D-Glucopyranoside, methyl (CAS), β-Sitosterol and Stigmasterol are utilized either singly or in conjunction as hypoglycemic agents.48–54 The compound groups detected using UV-Vis and FTIR spectroscopy corresponded with the secondary metabolites that were previously discovered through traditional phytochemical screening. 55 Spectroscopic techniques, in particular NMR analysis, were used to determine the structural elucidation of isolated molecules. Using NMR analysis, the ME was found to include 2-hydroxybenzoic acid, β-sitosterol, stigmasterol, ursolic acid and benzoic acid, which have been linked to the antihyperglycemic and antihyperlipidemic effects. Literature studies have demonstrated that the flavonoid quercetin shows antihyperglycemic action through the regeneration of pancreatic islets cells. 55
Benzoic acid regulates blood sugar levels by inhibiting lipid peroxidation, increasing the absorption of glucose, secreting insulin, and blocking enzymes like α-glucosidase and α-amylase. 56 2-hydroxybenzoic acid and showed impressive hypoglycemic effects, reducing BGLs and HbA1c levels by a variety of mechanisms, such as enhanced secretions of incretin hormone-like GLP1 and enzyme inhibition.57–61 Ursolic acid, β-sitosterol and Stigmasterol have the ability to reduce blood sugar and act as antioxidants. 62 The outcomes of the current study are in line with the previous research works,63,64 analyzing the influence of these phytocompounds on antidiabetic and antioxidant activities.
The methodological quality of alloxan was one of the difficulties that affected our research throughout the testing procedures. As a result of its restricted ability to accurately reflect, early loss of pancreatic β-cell mass in mice was detected. Due to intricate relationships between many tissues and organ systems in a living animal, it was difficult to determine dose selection and the mechanism of action of a test chemical or intervention as the end-result (such as a decrease in blood glucose). This exploratory study's lack of testing at varied extract concentrations prevented it from fully demonstrating the I.cappa antidiabetic effect, which is still study vulnerability. Other limitations included the usage of terminology while looking for evidence and the structural clarification of the compounds.
Conclusion
The present study concluded that the ME of I. cappa showed potential antidiabetic and antioxidant activity in alloxan-induced diabetic mice, which justifies its usage as an antidiabetic agent in folklore medicine
Footnotes
Abbreviations
Acknowledgements
The authors are very grateful to the authority of the Pharmacology lab Department of Botany Abdul Wali Khan University for their cooperation and the facilities provided for conducting the biological experiments.
Author Contributions
Shahid Rahman and Gul Jan: conceptualization, software, validation, investigation, resources, writing—original draft preparation, visualization, supervision, and project administration. Farzana Gul Jan: methodology, formal analysis and data curation. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The animal study was reviewed and approved by the administrations of Department of Botany (Pharmacology Lab) and Department of Pharmacy, Abdul Wali Khan University Mardan, Pakistan. The study was conducted in accordance with the Guide for the Care and Use of Laboratory Animals. 65
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the institutional Animal Biosafety and Bioethics Committee of the Department of Botany (N0.0225/BOTAWK), Abdul Wali Khan University Mardan, Pakistan.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
