Abstract
Background
This study explored the phytochemical composition, Gas Chromatography-Mass Spectrometry (GC-MS), Fourier Transform Infrared spectroscopy (FT-IR), and antioxidant properties of the crude methanol extract (CRUME) and its fractions from
Methods
Qualitative and quantitative phytochemical analyses were performed on CRUME and its methanol (MEFUC), ethyl acetate (EAFUC), and dichloromethane (DIFUC) fractions. Antioxidant assays, including Thiobarbituric reacting substance (TBARS), 2,2-diphenyl-1-picrylhydrazyl (DPPH), ferric reducing antioxidant power (FRAP), and total antioxidant capacity (TAC), were conducted. FTIR analysis was used to identify functional groups, and GC-MS analysis identified bioactive compounds in the methanol fraction. Molecular docking and drug-likeness assessments were conducted to evaluate hepatoprotective potential and safety profiles.
Results
Phytochemical screening revealed diverse classes in varying concentrations. Antioxidant assays showed maximum TBARS scavenging activity for CRUME (73.67 ± 4.16% at 250 µg/mL), DIFUC (91.67 ± 0.16% at 250 µg/mL), EAFUC (81.33 ± 0.16% at 125 µg/mL), and MEFUC (69.33 ± 0.58% at 500 µg/mL), comparable to BHT (74.33 ± 0.16% at 15.53 µg/mL). DPPH activity peaked at 500 µg/mL for CRUME (98.00 ± 0.70%), DIFUC (68.33 ± 2.08%), and EAFUC (61.67 ± 5.51%), while MEFUC showed peak activity at 15.53 µg/mL (89.33 ± 1.16%), similar to ascorbic acid (64.19 ± 1.34% at 31.25 µg/mL). FTIR revealed multiple functional groups; GC-MS identified 39 bioactive compounds. Docking studies highlighted 10 compounds with strong hepatoprotective potential. Drug-likeness and ADMET profiling indicated no signs of hepatotoxicity, immunotoxicity, cytotoxicity, or mutagenicity.
Conclusion
The methanol leaf extract of
This is a visual representation of the abstract.
Keywords
Introduction
The increased prevalence of hepatic disorders, due to growing exposure to toxins, infections, and metabolic diseases, requires investigation into alternative therapeutic approaches.1,2 In recent years, medicinal plants have been identified as important sources of bioactive compounds with hepatoprotective potential.
Oxidative stress is a significant factor in disease development, notably liver illness, 6 highlighting the importance of effective antioxidant defense mechanisms. The antioxidant capacity of the extracts was evaluated using laboratory assays including Total Antioxidant Capacity (TAC), Ferric Reducing Antioxidant Power (FRAP), and 1,1-diphenyl-2-picrylhydrazyl radical (DPPH) scavenging capacity. Lipid peroxidation, a key indicator of oxidative stress, was measured by the Thiobarbituric acid reacting substances (TBARS) assay to determine inhibition levels. 7 These concentration-dependent antioxidant profiles allow for efficacy comparison with common free radical inhibitors such as BHT and ascorbic acid. Phytochemical composition and bioactive compounds were identified using Gas Chromatography-Mass Spectrometry (GC-MS), and functional groups were analyzed by Fourier Transform Infrared (FTIR) spectroscopy. Molecular docking analysis then facilitated the identification of phytochemicals with strong binding affinities to drug (hepatoprotective) target proteins. Finally, pharmacokinetic investigations built upon docking findings to strengthen the overall results.
Materials and Methods
Plant Materials
The leaves of
Preparation of Plant Extracts
Six kilogrammes (6 kg) of
Fractionation of the Methanol Extract
The methanol crude extract was mixed with silica gel mesh 60-120, passed into a chromatography column, and separated into the constituent components using methanol, dichloromethane, and ethyl acetate solvents. Thin-layer chromatography was applied to separate the phytoconstituents of the fractions. This was followed by gas chromatography-mass spectrometry to identify different substances within the sample. Fourier-transform infrared spectroscopy was done to elucidate their structures.12,13
Phytochemical Analysis of Uvaria chamae Extract and Fractions
A preliminary phytochemical examination was conducted using the protocols established by Harborne. 14
Evaluation of in vitro Antioxidant Assays of Uvaria chamae Extract and Fractions
To assess the antioxidant activity of the extract and its components, the following recognized antioxidant models were employed:
Assessment of antioxidant capacity uses the thiobarbituric acid-reactive species (TBARS) photometric assay as per the Buege method. 15 A modified thiobarbituric acid-reactive species (TBARS) assay was employed to quantify the lipid peroxides generated from egg yolk homogenates as lipid-rich substrates.
Assessment of Antioxidant Capability via the Ferric Reducing Antioxidant Power (FRAP) Photometric Test
Ferric reduction involves the conversion of Fe3+ to Fe2+ by antioxidants in an acidic environment. 15 The sample's reduction power was assessed according to the methodology outlined by Sahreen et al. 16
The evaluation of antioxidant capacity was conducted using the Total Antioxidant Capacity (TAC) photometric test as described by Pellegrini et al. 17 The total antioxidant capacity assay was conducted using the phosphomolybdate technique.
Assessment of antioxidant capability using the 2,2-diphenyl-1-picrylhydrazyl radical (DPPH) spectrophotometric method. The DPPH free radical scavenging activity of the sample was assessed using the method of Gyamfi et al. 18 with minor modifications.
To Assess the in silico Drug Likeness of the Phytochemicals in the Most Active Fraction of Uvaria chamae Leaves
Protein Retrieval and Preparation
The crystal structures of SOD (PDB ID: 5VF9) and Catalase (PDB ID: 1DGH) were obtained from the Protein Data Bank (PDB). The protein architectures were optimized using Discovery Studio software. This involved removing water molecules and irrelevant ligands, fixing missing atoms or residues, and correcting bond orders and protonation states (Gasteiger charges and polar hydrogen), utilizing AutoDock technologies. The proteins were constructed by including Gasteiger charges and polar hydrogen, utilizing AutoDock technologies. 19
Ligand Retrieval and Preparation
The crystal structures of chosen chemicals from
Prediction of Active Sites
Following the creation of protein-ligands, the receptor grid for the proteins was established at the centroid of the co-ligand. The grid box was created by selecting the amino acid residues located at the active site. The analysis of the active location was conducted utilizing Biovia Discovery Studio software.
Molecular Docking
Prior to docking, both proteins and ligands were saved in PDBQt format. Molecular docking was conducted with AutoDock Vina Software. 21 The two-dimensional diagrams and three-dimensional surface representations of the protein-ligand interactions were produced utilizing Discovery Studio software and Pymol software, respectively. The physicochemical properties, lipophilicity, solubility, pharmacokinetics, and Lipinski drug-likeness of the examined compounds were assessed using the SwissADME Server. 22
Chemicals and Reagents
All chemicals utilized in this investigation were of analytical grade and sourced from May and Baker, England; BDH, England; and Merck, Darmstadt, Germany. The reagents utilized for all assays were commercial kits and products from Randox, USA; QCA, Spain; Teco (TC), USA; and Biosystem Reagents and Instruments, Spain.
Statistical Analysis
The presented results are expressed as the mean ± standard deviation (SD) from three independent experiments (n = 5). To assess the statistical significance of the obtained data, a One-way Analysis of Variance (ANOVA) was employed. Mean comparisons were conducted using the Duncan multiple test. The statistical analysis was executed with GraphPad Prism version 8.4, and a confidence interval of p < .05 was established to determine statistical significance.
Results
Quantitative Phytochemical Analyses of Crude Extracts, Methanol Fraction, Ethylacetate Fraction, and Dichloromethane Fraction of Uvaria chamae
The result of the quantitative phytochemical analysis of
Quantitative Phytochemical Analyses of Crude Extracts, Methanol Fraction, Ethylacetate Fraction, and Dichloromethane Fraction of
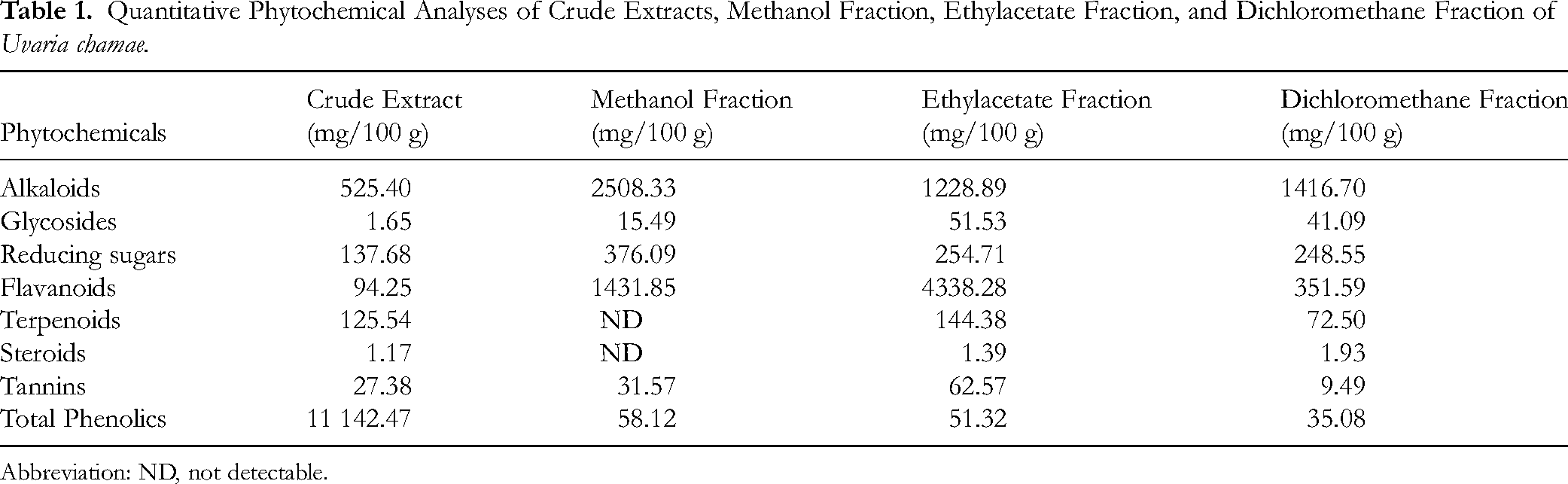
Abbreviation: ND, not detectable.
Determination of Thiobarbituric Acid-Reacting Substances (TBARS) (DIFUC), Ethyl Acetate Fraction (EAFUC), and Methanol Fraction (MEFUC) of Uvaria chamae Leaves
The result (as shown in Table 2) revealed that various concentrations (15.53, 31.25, 62.5, 125, 250, 500 µg/mL) of crude methanolic extract and fractions of
Determination of TBARS Scavenging Activities of Crude Extract, Dichloromethane Fraction, Ethyl Acetate Fraction, Methanol Fraction of

*Results are expressed in Means ± SD (n = 5). Mean values with different letters as superscripts down the column are considered significant at
Determination of Ferric ion Reducing Power (FRAP) Concentration of Crude Extract, Dichloromethane Fraction, Ethyl Acetate Fraction, Methanol Fraction of Uvaria chamae Leaves
The result (as shown in Table 3) revealed that various concentrations of (15.63, 31.25, 62.5, 125, 250, 500 µg/mL) of crude methanolic extract and fractions of
Determination of FRAP Concentration of Crude Extract, Dichloromethane Fraction, Ethyl Acetate Fraction, Methanol Fraction of

*Results are expressed in Means ± SD (n = 5). Mean values with different letters as superscripts down the column are considered significant at
Furthermore, the total absorbances are: CRUME (17.41), DIFUC (19.01), EAFUC (13.94) and MEFUC (21.44).
Determination of Total Antioxidant Capacity (TAC) Concentration of Crude Extract, Dichloromethane Fraction, Ethyl Acetate Fraction, Methanol Fraction of Uvaria chamae Leaves
The result (as shown in Table 4) revealed that various concentrations of (15.63, 31.25, 62.5, 125, 250, 500 µg/mL) of crude methanolic extract and fractions of
Determination of TAC Concentration of Crude Extract, Dichloromethane Fraction, Ethyl Acetate Fraction, Methanol Fraction of

*Results are expressed in Means ± SD (n = 5). Mean values with different letters as superscripts down the column are considered significant at
Furthermore, the total absorbances are: CRUME (84.33), DIFUC (82.33), EAFUC (64.99) and MEFUC (38.66).
Determination of DPPH Scavenging Activities of Crude Extract, Dichloromethane Fraction, Ethyl Acetate Fraction, Methanol Fraction of Uvaria chamae Leaves
The result (as shown in Table 5) revealed that various concentrations of (15.63, 31.25, 62.5, 125, 250, 500 µg/mL) of crude methanolic extract and ractions of
Determination of DPPH Scavenging Activities of Crude Extract, Dichloromethane Fraction, Ethyl Acetate Fraction, Methanol Fraction of

*Results are expressed in Means ± SD (n = 5). Mean values with different letters as superscripts down the column are considered significant at p < 0.05.
Fourier Transform Infrared Spectroscopy (FTIR) of Eluents of Methanol Fraction of Uvaria chamae Leaves
As in Figure 1, the Fourier Transform Infrared Spectroscopy (FTIR) of eluents of methanol fraction of

Fourier transform infrared spectroscopy (FTIR) of eluents of methanol fraction of
C–Br (Halogen of Bromine) appeared at wavelength of 690.8796 cm−1, C–Cl (Halogen of Chlorine) appeared at wavelength of 1030.659 cm−1, C–O (Ether CO Symmetrical stretch) appeared at wavelength of 1236.255 cm−1, H2C=CH (Ethene C=C anti symmetrical stretch) appeared at wavelength of 1397.019 cm−1, R–CO (Ester CO symmetrical stretch) appeared at wavelength of 1721.021 cm−1, SCN (Thiocyanate SCN stretch) appeared at wavelength of 1950.449 cm−1 COOH (Carboxylic COO stretch) appeared at wavelength of 2083.711 cm−1, R2C=O (Carbonyl compound CO anti-symmetrical stretch) appeared at wavelength of 2278.126 cm−1, C=N (Nitrile CN anti-symmetrical stretch) appeared at wavelength 2512.308 cm−1, C–H (Methylene C–H symmetrical stretch) appeared at wavelength 2738.125 cm−1, R3CHOH (I alcohol OH stretch) appeared at wavelength of 3014.040 cm−1, R2CHOH (2 alcohol OH stretch) appeared at wavelength of 3346.233 cm−1 and R3CHOH (3 alcohol OH stretch) appeared at wavelength of 3542.772 cm−1.
Gas Chromatography-Mass Spectrophotometry (GC-MS) of Eluents of Methanol Fraction of Uvaria chamae Leaves
As in Table 6, the results of Gas chromatography-Mass Spectrophotometry (GC-MS) of eluents of methanol fraction of
Gas Chromatography-Mass Spectrophotometry (GC-MS) of Eluents of Methanol Fraction of

In silico Drug Likeness of the Phytochemicals in Methanol Fraction of Uvaria chamae Leaves Showing the Target Proteins
As in Figure 2, the 3 D view of the target proteins used in the docking which were catalase (PDB IDGH) and SOD (PDB ID: 5VF9) were shown.

3D (surface) view of target proteins (catalase in orange color while SOD in blue color) and active site region in white color.
In silico Drug Likeness of the Phytochemicals in Methanol Fraction of Uvaria chamae Leaves Showing the Chosen Phytochemicals
As in Table 7, the GC-MS identified phytochemicals against the target proteins were assessed to determine the binding affinities. 10 phytochemicals with the lowest binding affinities for the 2 target proteins were identified and used for the docking. These phytochemicals includes Cyclohexadecane1,2,diethyl, Ethyl oleate, Heptadecane,2,6,10,14 tetramethyl, 2,4, di-tert-butylphenol, Heptadecane 7 methyl, Dodecane 2,6,10- trimethyl, 1,2,Benzene dicarboxylic acid butyl-2-ethyl hexyl ester, 1-Hexacosene, Dodecane, 2, 6, 11- trimethyl and o-cymene.
Binding Affinities of Studied Phytocompounds of Methanol Fraction of

In silico Drug Likeness of the Phytochemicals in Methanol Fraction of Uvaria chamae Leaves Showing the 2D Active Sites in the Catalase Enzyme and the Binding of the 10 Phytochemicals to the Active Sites
As in Figure 3, the 2D view of the active sites of the catalase enzyme with the pattern of binding of the 10 phytochemicals to these active sites were shown.

Active site interaction (2D diagram) of studied phytocompounds of methanol fraction of
In silico Drug Likeness of the Phytochemicals in Methanol Fraction of Uvaria chamae Leaves Showing the 2D Active Sites in the Superoxide Dismutase (SOD) Enzyme and the Binding of the 10 Phytochemicals to the Active Sites
As in Figure 4, the 2D view of the active sites of the SOD enzyme with the pattern of binding of the 10 phytochemicals to these active sites were shown

Active site interaction (2D diagram) of studied phytocompounds of methanol fraction of
In silico Drug Likeness of the Phytochemicals in Methanol Fraction of Uvaria chamae Leaves Showing the Pharmacokinetics and Drug Likeness Properties of the 10 Studied Phytocompounds
As shown in Table 7, the Pharmacokinetics and drug likeness properties of the 10 studied phytocompounds were showed that Cyclohexadecane1,2,diethyl; ethyl oleate; Heptadecane 2,6,10,14-tetramethyl; 2,4-di-tert-butylphenol; Heptadecane-7-methylhexyl ester; Dodecane-2,6,10-trimethyl; 1,2-benzenedicarboxylic acid butyl-2-ethyl; 1-Hexacosene; Dodecane-2,6,11-trimethyl and o-cymene are not hepatotoxic, immunotoxic, cytotoxic and mutagenic to cells. All the 10 studied phytocompounds are not carcinogenic except ethyl oleate and o-cymene.
As shown in Table 8 the
Pharmacokinetics and Drug Likeness Properties of Studied Compounds.

Legend: MW = Molecular weight (g/mol); NoHA = Number of hydrogen bond acceptors; NoHD = Number of hydrogen bond donors; ClogP = Calculated partition coefficient (lipophilicity); VON = Violation of drug-likeness rules; Toxicity class = Predicted acute toxicity category (III = moderately toxic, VI = non-toxic) (Active = predicted toxic; Inactive = predicted non-toxic).
Discussion
The practice of using plant materials for disease treatment is not peculiar to Africa but also found in several other parts of the world. However, most plants used in traditional medicine for various diseases have not been scientifically proven.
23
Phytochemical screening identified some phytochemical compounds at varying concentrations. This list includes alkaloids, glycosides, reducing sugars, flavonoids, terpenoids, steroids, tannins, and phenols. These compounds, including phenolic acids, flavonoids, and terpenoids, are already extensively documented to be involved in antioxidant activity.24–26 The findings of this study are in line with the report of Onyekwelu et al,
27
which showed that
Phenolic Content and Antioxidant Activity
Antioxidants are crucial for health and disease prevention because they counteract free radicals, which cause oxidation, leading to chronic diseases such as heart disease, diabetes, and various cancers.
28
As indicated in Tables 1 and 2, the ethyl acetate fraction of
TBARS Assay
The TBARS assay is crucial in evaluating antioxidant defense systems as it measures lipid peroxidation products like malondialdehyde (MDA), a marker of oxidative stress. Table 2 highlights that the dichloromethane fraction of
FRAP Assay
The FRAP assay is an important method in evaluating antioxidant defense thiol systems based on the ability of compounds to reduce Fe3⁺ to Fe2⁺ ions. From Table 3, the methanol fraction of
Total Antioxidant Capacity (TAC)
The TAC assay evaluates the cumulative antioxidant capacity of all antioxidants present in a sample, providing a comprehensive measure of their combined efficacy. As shown in Table 4, the crude extract of
DPPH Assay
The DPPH assay is a key method for evaluating the free radical scavenging ability of antioxidants, providing insight into their capacity to neutralize oxidative stress. Table 5 highlights the superior antioxidant potential of the
In-Silico Drug-Likeness Analysis
The
GC-MS and FTIR Findings
GC-MS analysis identified 31 phytochemicals, of which several possess antioxidant activity, including o-cymene and ethyl oleate. FTIR spectra revealed functional groups such as carboxylic acids, phenols, and esters, which contribute to the antioxidant properties of the extracts. These findings further support the potent antioxidant bioactivity potential of
Limitations of the Study
Limited Scope of Biological Evaluation
The study primarily focused on in vitro antioxidant assays (DPPH, FRAP, TBARS, TAC) and
Restricted Extraction and Fractionation Methods
Lack of funding restricted the fractionation to only a few solvent fractions (methanol, ethyl acetate, dichloromethane, and crude extract). Other alternative solvents or extraction techniques, such as supercritical CO₂ or ultrasonic extraction, would have been included as these might possibly yield different or more bioactive profiles.
Conclusion
This study underscores the therapeutic potential of
Footnotes
Acknowledgments
The authors are grateful to the Staff of the postgraduate Laboratory, Department of Biochemistry, University of Nigeria Nsukka for their technical assistance.
Ethical Considerations
Ethical approval is not applicable.
Consent to Participate
Not applicable.
Consent to Publish
Consent to publish is not applicable.
Authors’ Contributions
CUV: Conceptualization, Methodology, Data Curation, Writing original Draft, Writing review and editing. OCE: Methodology, Data Curation, Investigation, Writing review and editing. ESI: Conceptualization, Methodology, Data Curation, Writing review and editing, Supervision. AOE: Methodology, Data Curation, Investigation, Writing review and editing. PEJ: Conceptualization, Methodology, Data Curation, Writing original Draft, Writing review and editing, Supervision. All authors read and approved the final manuscript for publication.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data have not been previously deposited on any public repository. All the data generated in this study have been included in the manuscript.
