Abstract
Objective
Senecio asirensis, an endemic species of Saudi Arabia, has been traditionally used to treat various ailments. The present study aimed to investigate the phytochemical composition, antimicrobial activity, and cytotoxic potential of Senecio asirensis fractions, and to identify the main compounds in the extract that are the most active.
Methods
The antimicrobial activity was evaluated using disc diffusion assay. Moreover, the cytotoxic activity evaluated using the MTT assay in various cancer cell lines (MCF-7, HepG2, and A549) and validated by molecular docking for the major compound of the most active fraction. Finally, gas chromatography and a mass spectrometer (GC-MS) analysis was carried out to identify the chemical compounds in hexane extract of Senecio asirensis.
Results
The hexane fraction exhibited significant antimicrobial activity against Gram-positive bacteria (S. aureus) and yeast organisms (C. albicans and C. tropicalis), while chloroform and butanol fractions showed no activity against the tested microbial strains. Moreover, in the cytotoxic activity assay, notably, the hexane fraction showed a significantly higher cytotoxic effect on MCF-7. The GC-MS analysis of the hexane fraction revealed the presence of 26 phytoconstituents, with nonacosane being the most abundant compound. Therefore, the molecular docking was employed to validate the cytotoxic activity for the major compound of the most active fraction (nonacosane) against human DNA topoisomerase IIa.
Conclusion
These findings provide a scientific basis for the traditional use of S. asirensis in medicine and suggest that its bioactive compounds have potential therapeutic applications in anticancer treatments. Further research is needed to isolate and characterize other active compounds and to explore potential synergistic effects among them.
Introduction
In practically every element of human life, including nutrition, the environment, culture, society, and religion, medicinal plants are thought to be indispensable. Among these, the usage of therapeutic plants dates back approximately 60,000 years.1–3 Additionally, traditional herbal preparations have been essential in the treatment and prevention of numerous diseases since ancient times.1,4 In recent years, there has been a surge in interest in natural products with therapeutic activities due to their potential to counteract the deleterious effects of free radicals and their low toxicities compared to synthetic antioxidants widely utilized in food products, cosmetics, and drugs.5–7 These natural products have garnered significant attention for their antimicrobial properties, which are attributed to their abundant antioxidant content.8,9 Furthermore, the global emergence of antimicrobial resistance and the evolution of multi-drug-resistant (MDR) isolates have complicated treatment strategies, necessitating the development of new classes of therapeutic agents to combat infectious diseases. 10
Natural products also hold considerable potential as anticancer drugs, with 51% of currently available treatments for this pathology being directly or indirectly derived from natural sources. 11 Senecio, one of the largest genera in the Asteraceae family, comprises over 1500 species of herbs, shrubs, vines, and trees with a vast global distribution. 12 Traditional medicine has employed extracts from the leaves and roots of various Senecio species to treat fever, cough, stomach pain, gastric ulcers, diabetes, skin wounds, and as vasodilators, antiemetics, and anti-inflammatory agents. 13
Senecio asirensis, an endemic species to Saudi Arabia, is found in limited locations such as Jabal Fayfa, Raida, Bal Lasmar, Jabal Samdah, Bal Jurshi, and Tannouma. Other Senecio species, including Senecio bojeri, S. flavus, S. glaucus, S. hadiensis, S. haggariensis, S. lyratus, S. schimperi, S. sumarae, and S. vulgaris, are distributed across various regions of Saudi Arabia. Some Senecio species are highly toxic due to the presence of pyrrolizidine alkaloids (PAs). 14 These species produce furanoeremophilanes, which are commonly isolated and characterized for their medicinal purposes. 15 Additionally, certain Senecio species exhibit gastroprotective effects and have been employed in the treatment of sexually transmitted infections. 16
This study aims to evaluate the antibacterial and cytotoxic properties of the methanol extract and fractions obtained from the aerial parts of S. asirensis. It is part of our ongoing research on medicinal plants in Saudi Arabia and seeks to validate the traditional uses associated with this particular plant. In vitro and in silico techniques are used in the evaluation.
Materials and Methods
Plant Collection and Preparation of the Extracts and Fractions
The leaves and stems of S. asirensis were collected during the autumn season (2023) from the southern region of Saudi Arabia. The aerial parts were dried in artificial heat at 45 °C and coarsely powdered (300 g) before extraction with ethanol (2000 mL) using a soxhlet apparatus. The liquid extract was concentrated under vacuum using rotavapor to get 61 g of crude semi-solid ethanol extract. Further suspension in distilled water produced 6 g of crude extract, followed by partitioning with solvents of different polarities such as n-hexane, chloroform (CHCl3), and n-butanol (BtOH) to yield 10 g, 15 g, and 20 g of dried extract, respectively.
Phytochemical Analysis
Gas Chromatography/Mass Spectrometry Analysis
An Innowax FSC column with a particle size of 0.25 mm and a film thickness of 0.25 m was used as the stationary phase, and helium was used as the mobile phase (0.8 mL/min). It was injected in 0.1 L with a split ratio of 40 : 1. The oven temperature of the gas chromatography (GC) was initially set at 60 degrees Celsius for 10 min, then increased to 220 degrees Celsius at a rate of 4 degrees Celsius per minute, held constant for 10 min, and then increased to 240 degrees Celsius at a rate of 1 degree Celsius per minute. Both the injector and the transfer line were set to 250 and 280 °C, respectively. A scan mass range of 35-450 was used for MS detection at 70 eV.
Constituent Identification
The Adams 17 and Wiley 18 gas chromatography and a mass spectrometer (GC-MS) compounds mass spectral libraries were used to compare similar mass spectra found for BVF constituents. The characterizations of compounds were performed by comparing the RT (retention time) with genuine reference standards under the same above-indicated conditions. 19
Biological Evaluation
Determination of the Cytotoxic Activity (MTT Assay)
The MTT assay was employed to evaluate the antiproliferative effects of S. asirensis fractions against lung (A549), liver (HepG2), breast (MCF-7) cancer cells, and non-tumor endothelial (HUVEC) cells. 20 Briefly, different concentrations of each fraction were added to the cells (5 × 104/well) after 24 h of incubation in a 96-well plate, and doxorubicin was used as a positive control, while dimethyl sulfoxide (DMSO) was used as a negative control. After incubation for 48 h, 10 μL of the MTT solution (5 mg/ml, prepared in phosphate-buffered saline) was added to each well. After that, the cells were incubated for another 4 h. In order to dissolve the formazan crystals, the excess medium was removed from the wells and replaced with 100 L of acidified isopropanol. Optical densities were determined by a microplate spectrophotometer (BioTek, USA) at 570 nm. The OriginPro 8.5 software was used to calculate the IC50 (50% inhibition of cell growth).
Antimicrobial Activity
Bacterial Strains
In the current study, we examined the antimicrobial activities of six distinct microbial strains using various fractions of S. asirensis. These included two Gram-negative strains, namely Escherichia coli (ATCC 25922), Salmonella typhimurium (ATCC 14028) and two Gram-positive namely Staphylococcus aureus (ATCC 29213), Enterococcus faecalis (ATCC 29212), as well as two yeast strains, Candida albicans (ATCC 61922) and Candida tropicalis (ATCC 66029).
Disc Diffusion Assay
Extracts of S. asirensis were evaluated for antimicrobial activity using Mueller Hinton Agar (MHA) plates. Inoculations were performed in nutrient broth and incubated overnight at 37 °C to adjust turbidity to 0.5 McFarland standards, yielding a final inoculum of 1.5 million CFU/ml. 21 Standard microbial culture broth was used for lawn culture of MHA plates. An extract of plants in DMSO was prepared at a concentration of 50 mg/ml. A 6-mm cork-borer was used to bore six wells into the inoculated medium. A 50-liter bottle was filled with each well's extracts. The bacteria strains were tested with ciprofloxacin 10 g as a positive control, while DMSO used as negative control. Following diffusion at room temperature for 30 min, plates were incubated for 18-24 h at 37 °C. Plates were observed for the presence of a clear zone around the well, which signified the presence of antimicrobial activity. Observation and measurement of the diameter of inhibition zone were carried out.
Molecular Docking
The three-dimensional coordinates of DNA topoisomerase IIa (PDB Id: 1ZXM) were obtained from the PDB database (http://www.rcsb.org/pdb/). Molecular docking was carried out using Autodock-Vina as implemented in PyRx-0.8.
22
The Lamarckian genetic algorithm was employed as the scoring function.
23
Before the molecular docking process, the protein was prepared by eliminating water molecules, hetero-atoms, salts, and adding polar hydrogen atoms. Additionally, Kollman United-atom type charges were assigned. The protein format was then converted from pdb to pdbqt, followed by energy minimization using the built-in tools in PyRx. The two-dimensional structure of nonacosane was retrieved from PubChem (CID_12409) and transformed into a pdbqt format compatible with AutoDock using PyRx. The ligand's energy was minimized using the Universal Forcefield (UFF), and Gasteiger partial charges were applied. Non-polar hydrogens were combined, and rotatable bonds were defined. Molecular docking was performed within a grid-box with dimensions of (32.8 × 27.4 × 31.5) Å, centered at coordinates (36.4 × 0.9 × 34.5) Å. All other docking parameters were set to the software's default values. The conformation of nonacosane with the lowest binding energy was selected for further analysis in Discovery Studio 2020 (BIOVIA).
24
The dissociation constant (Kd) of nonacosane for DNA topoisomerase IIa was calculated based on the binding energy (ΔG) using the relation.
25
Results
Phytochemical analysis
The chemical profile of S. asirensis using GC-MS analysis (Figures 1 and 2) and the chemical content, retention times, and area percentages of the S. asirensis hexane fraction are displayed in Table 1 according to their elution on the HP Innowax column. GC-MS analysis identified approximately 26 phytoconstituents. The most abundant constituents were nonacosane (39.4%), methyl alpha-ketopalmitate (25.1%), and heptacosane (10.7%). The remaining compounds in the S. asirensis hexane fraction are listed in Table 1.

Chemical structure of the major compound (Nonacosane) in S. aseransis hexane fraction.
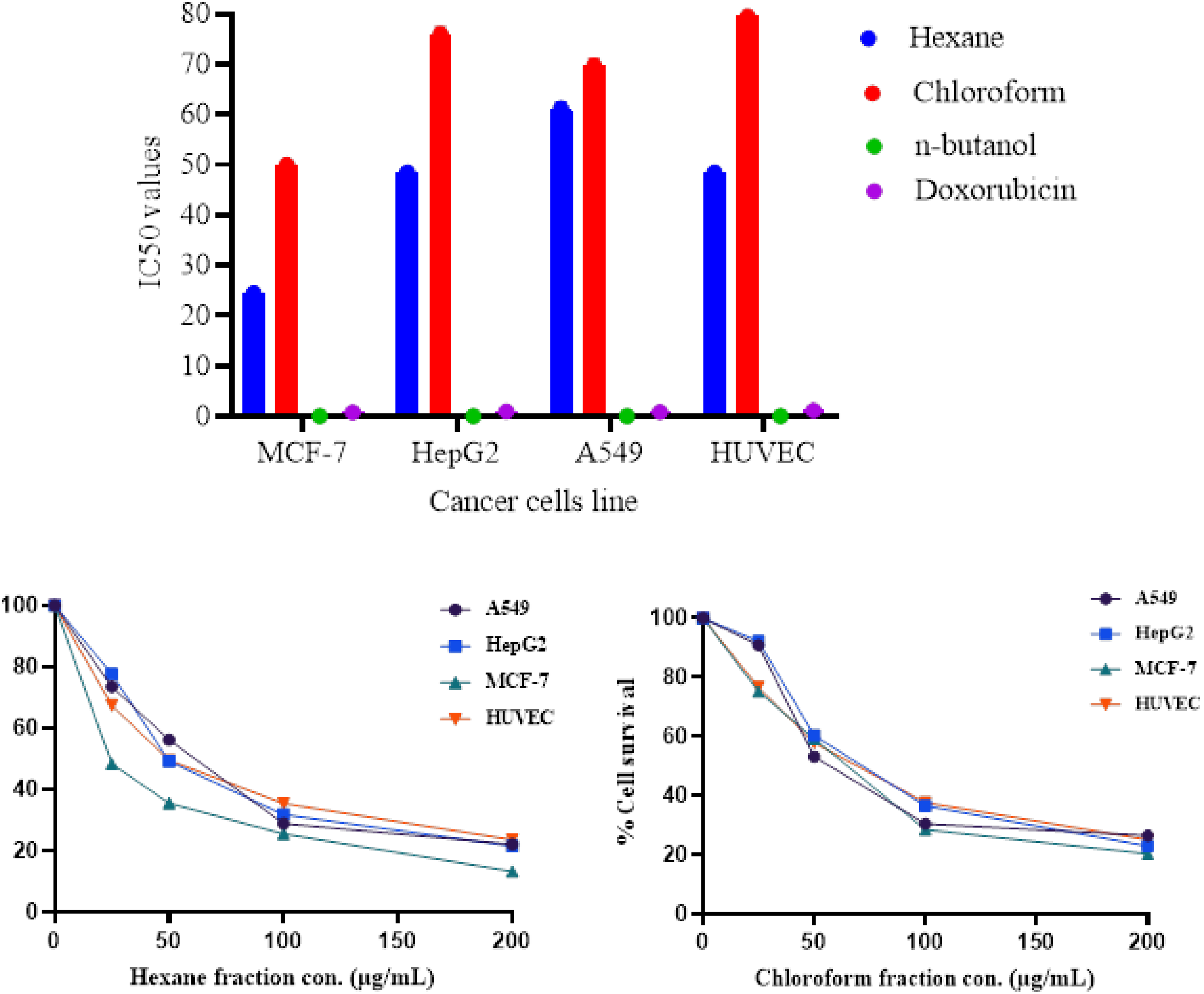
Effect of hexane and chloroform of S. aseransis on cell viability after 48 h treatment.
Phytochemical Compounds Identified in Hexane Fraction of S. aseransis.

Antimicrobial Activity
A disc diffusion assay was performed to assess the antimicrobial activity of S. aseransis fractions. We observed that the S. aseransis hexane fraction showed antimicrobial activity against Gram-positive bacteria (S. aureus) with an inhibition zone of 11 mm. S. aseransis hexane fraction also showed strong activity against yeast organisms (C. albicans and C. tropicalis), with inhibition zones of 10 mm and 13 mm, respectively. However, the chloroform and butanol fractions showed no activity against all microbial strains (Table 2).
Antimicrobial Activities of the Different Fractions of S. asirensis.

NA: no activity
Cytotoxic Activity
MTT assay was performed to assess the reduction in cell viability of S. aseransis fractions. We observed that S. aseransis fractions induced a concentration-dependent reduction in cell viability in all tested cell lines after 48 h of exposure, with a significantly higher response in MCF-7 cells compared to the remaining cells. In MCF-7 cells, the hexane fraction exhibited a higher viability reduction effect (IC50 = 24.5 μg/mL) compared to the chloroform fraction (IC50 = 50 μg/mL), while n-butanol does not show any activity as shown in Table 3 and Figure 2. Results are expressed as means ± SD for three replicate values for each concentration.
IC50 Values of Different Fractions of S. asirensis Against MCF-7, HepG2, A549, and HUVEC Cells.

Molecular Docking
Human DNA topoisomerase IIa is a crucial enzyme involved in the regulation of DNA topology and structure during various cellular processes, including replication, transcription, and chromosome segregation. It plays a pivotal role in maintaining the integrity of the DNA double helix by introducing transient breaks in the DNA strands, allowing them to unwind or supercoil, and then resealing the breaks. This enzymatic activity is essential for DNA replication, repair, and recombination. In our investigation, we conducted molecular docking experiments involving DNA topoisomerase IIa and nonacosane. The outcomes of our study revealed that nonacosane formed a binding interaction within the central cavity of DNA topoisomerase IIa (Figures 3a,b). These findings align with previous observations involving various natural compounds, including aminocoumarins, quinolones, chlorojanerin, and rutin. This suggests that these compounds occupy the ATP-binding site of the enzyme, functioning as competitive inhibitors of the ATP-binding region of topoisomerase IIa. The DNA topoisomerase IIa-Nonacosane complex exhibited stability through five hydrophobic (alkyl) interactions with specific residues, namely ILE125 (5.06 Å), PRO126 (4.15 Å), VAL137 (5.04 Å), ILE88 (4.97 Å), and ILE217 (4.44 Å) (Table 4). Additionally, the protein-ligand complex displayed van der Waals interactions with multiple other residues, including ASN91, ALA92, ASP94, ASN95, ARG98, ASN120, ILE141, PHE142, THR147, SER148, SER149, ASN150, THR159, GLY161, GLY164, ALA167, LYS168, and THR215 (Figure 3c). The calculated binding free energy for the DNA topoisomerase IIa-nonacosane complex was −5.9 kcal mol−1, corresponding to a dissociation constant of 2.12 × 104 M−1 (Table 4).

Molecular docking of nonacosane to the active site of DNA topoisomerase IIa. (a) Ribbon diagram of the binding mode, (b) 3D representation of binding mode, and (c) Molecular interaction between DNA topoisomerase IIa and nonacosane. The distances are in Å.
Interaction Parameters for DNA Topoisomerase IIa and Nonacosane.

To assess the docking algorithms’ capability in predicting the conformation of the protein-bound ligand, we conducted re-docking of the co-crystallized ligand (Nonacosane) in DNA Topoisomerase IIa. This step was undertaken to validate the accuracy of the docking procedure. From figure 4, the two almost superimposed conformation between the conformation of the re-docked ligand and the conformation of the original ligand in the DNA Topoisomerase IIa enzyme pocket, with a very low value of RMSD is 2.1867 Ǻ less than 3 Å. So this means that the efficiency of AutoDock-Vina as implemented in PyRx-0.8 software for performing molecular docking is good.
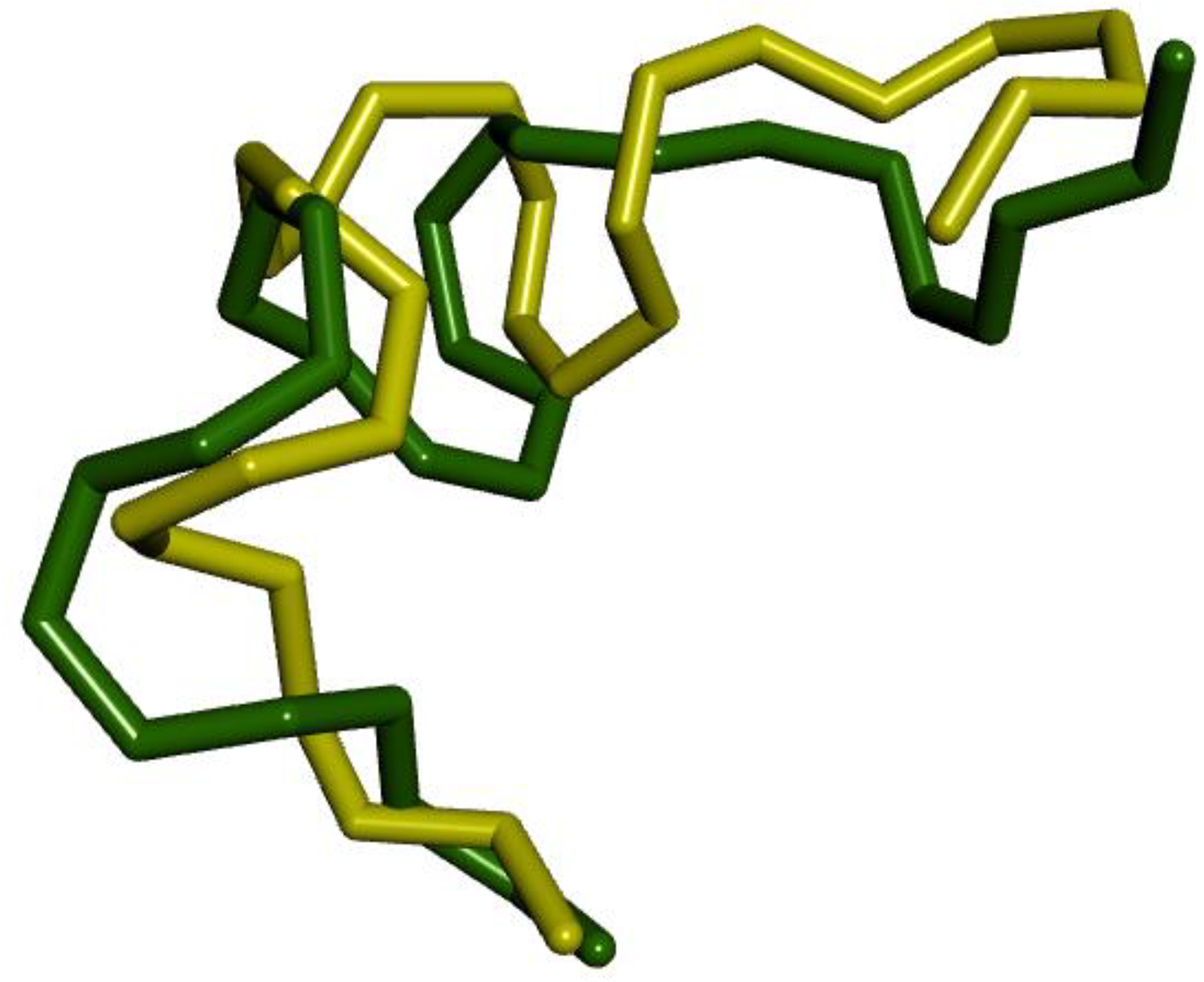
Re-docking poses with RMSD value it 2.1867 Å (Green = Original, yellow = Docked).
Discussion
The present study aimed to evaluate the phytochemical composition, antimicrobial activity, and cytotoxic potential of S. asirensis fractions. Our findings provide insights into the therapeutic properties of this plant and offer a scientific basis for its traditional use in medicine.
Phytochemical analysis using GC-MS revealed the presence of 26 phytoconstituents in the S. asirensis hexane fraction, with nonacosane, methyl alpha-ketopalmitate, and heptacosane being the most abundant compounds (39.4%, 25.1%, and 10.7%, respectively) (Table 1). These compounds have been reported to exhibit various biological activities, including antimicrobial and anticancer properties. In a study by Kaur and Arora, 26 nonacosane exhibited antimicrobial activity against S. aureus and E. coli. Similarly, heptacosane has been shown to display antibacterial and antifungal activities. 27 Furthermore, a study by Mishra et al identified methyl alpha-ketopalmitate as a potent inducer of apoptosis in leukemia cells. 28 The presence of these bioactive compounds in S. asirensis might be responsible for the observed therapeutic effects in our study.
The antimicrobial activity of S. asirensis fractions was assessed using the disc diffusion assay. The hexane fraction of S. asirensis exhibited significant antimicrobial activity against Gram-positive bacteria (S. aureus) and yeast organisms (C. albicans and C. tropicalis), while the chloroform and butanol fractions showed no activity against the tested microbial strains. The observed antimicrobial activity of the hexane fraction is consistent with other studies that have reported potent antimicrobial activities for hexane fractions in plant extracts, such as Azadirachta indica, 29 Syzygium aromaticum, 30 and Zingiber officinale. 31 These findings suggest that the hexane fraction of S. asirensis contains bioactive compounds with antimicrobial potential, which could be harnessed for the development of novel antimicrobial agents to combat MDR pathogens. According to recent study, essential oils high in phytol, heptacosan, nonacosan, and pentacosan exhibit stronger antibacterial properties, especially against Escherichia coli. 32 According to Konovalova et al, a significant proportion of alkanes in the forms of pentacosane, heptacosane, and nonacosane are present in essential oils that have potent antibacterial properties. 33 The most prevalent phytoconstituent found in ethanolic extracts of Cayratia trifolia and Moringa oleifera, according to Khan and Javaid, is nonacosane, which has potent anti-inflammatory, antioxidant, and cytotoxic effects. 34 Subsequent investigation revealed that nonacosane possessed the strongest antimicrobial potential against a variety of bacteria, such as Salmonella typhi, Escherichia coli, Enterobacter faecalis, Staphylococcus aureus, and Klebsiella pneumonia.35,36 It also possesses great anti-diabetic potential. Nonacosane has potential qualities that could make it useful in a range of commercial applications. 37 Not even essential oils’ tiny ingredients are insignificant. Through their synergistic effects, they can contribute significantly, even in little amounts, to the biological activity of essential oils. It is well known for its exceptional pharmacological, therapeutic, antioxidant, anticancer, and antibacterial effects against a variety of pathogens. Tetratriacontane has even been found in essential oil with a small amount. 38
The cytotoxic activity of S. asirensis fractions was evaluated using the MTT assay in various cancer cell lines. A concentration-dependent reduction in cell viability was observed in all tested cell lines after 48 h of exposure to S. asirensis fractions. Notably, the hexane fraction exhibited a significantly higher cytotoxic effect on MCF-7 cells (IC50 = 24.5 μg/mL) compared to the chloroform fraction (IC50 = 50 μg/mL), while the n-butanol fraction showed no activity (Table 1). The cytotoxic activity of the S. asirensis hexane fraction is in line with other studies on plant extracts, where hexane fractions have shown potent cytotoxic effects against cancer cell lines, such as MCF-7, HepG2, and A549. 27 A study by Moghadamtousi et al reported a similar cytotoxic effect for the hexane fraction of Piper betle against MCF-7 cells (IC50 = 29.4 μg/mL). 6
These results of the present study indicate that the S. asirensis hexane fraction possesses potential anticancer activity, which may be attributed to the presence of bioactive compounds identified through GC-MS analysis. However, an extensive class of natural compounds is represented by the PAs. More than 660 of them can be found in a broad range of plants belonging to various botanical groups. Asteraceae, especially the Senecioneae tribe, was one of the most affected families. 39 Regretfully, these chemicals have teratogenic, genotoxic, and acute toxic effects on both people and animals. Prolonged consumption of PA can have detrimental effects, regardless of the source contaminated plants, hay, straw, silage, grasses, medications, cereals, pollen, or honey. It can sometimes result in lethal poisoning in animals. Exposure to PAs in humans is linked to both acute and chronic hepatotoxicity, which can lead to hazardous outcomes such as ovarian illness, especially in children.39,40 Many organizations have created or put out policies pertaining to the highest concentrations of PAs in dietary items. In order to protect consumer health, the European Commission recommends that the daily allowance of PAs (with a 1,2 unsaturated necine skeleton) with their N-oxides in oral herbal products (infusions and teas) not exceed 10 μg, and that the daily allowance for herbal extracts not exceed 1 μg. 41 Specifically, the European Union has established a 150 μg/kg maximum limit for PAs in teas and flavored teas. Although studies are being conducted to evaluate the dangers, China has not yet set limitations for PAs in food. 42
Conclusion
In conclusion, our findings demonstrate that the hexane fraction of S. asirensis contains bioactive compounds with antimicrobial and cytotoxic properties which was confirmed by molecular docking study, supporting its in-vitro cytotoxic activity. Further research is warranted to isolate and characterize all active compounds responsible for these activities, which could lead to the development of new therapeutic agents for treating infectious diseases and cancer. It is also essential to explore the potential synergistic effects of the identified compounds, as the combination of these phytochemicals might enhance their efficacity.
Footnotes
Acknowledgments
The authors extend their appreciation to Deanship of Scientific Research at Princess Nourah bint Abdulrahman University, through the Research Groups Program Grant no (RGP-1443-0043).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable to this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Deanship of Scientific Research at Princess Nourah bint Abdulrahman University, through the Research Groups Program Grant no (RGP-1443-0043).

