Abstract
Mood disorders are increasingly recognized as systemic conditions involving immune dysregulation and gut–brain axis disturbances. While probiotics and prebiotics have been extensively studied, the mechanistic and therapeutic roles of postbiotics—the bioactive metabolites produced by microorganisms—remain insufficiently synthesized in psychiatric research. This review addresses this gap by examining how key postbiotic compounds, including short-chain fatty acids (SCFAs), gamma-aminobutyric acid (GABA), serotonin, dopamine, tryptophan metabolites, and oxytocin, modulate neuroimmune pathways implicated in depression and related disorders. Mechanistically, postbiotics influence histone deacetylase activity, G-protein–coupled receptor signaling, cytokine production, microglial activation, and blood–brain barrier integrity. Emerging clinical data suggest adjunctive benefits in stress, anxiety, and depressive symptoms, though dose-response relationships and long-term outcomes remain underexplored. Overall, postbiotics represent a promising mechanistically targeted strategy for modulating the gut–brain–immune axis; however, rigorous clinical trials are required to validate therapeutic efficacy and optimize formulation and dosing strategies.
Introduction
Mood disorders such as depression and anxiety are among the most prevalent and debilitating mental health conditions worldwide, posing significant challenges to global health systems. 1 Despite advances in pharmacological and psychotherapeutic treatments, a substantial proportion of patients remain refractory to conventional interventions, necessitating the exploration of novel therapeutic avenues. In recent years, the gut-brain axis has emerged as a critical focus in neuropsychiatric research, offering a new lens through which to understand and potentially treat mood disorders. Emerging studies on neuroprotective agents, such as chinonin from traditional herbs, highlight anti-inflammatory and antioxidant mechanisms that intersect with gut-brain pathways.2,3 Additionally, molecular axes such as ESR2–miR-10a-5p–BDNF in postpartum depression highlight the role of synaptic plasticity in mood regulation. 4
The gut-brain axis (GBA) is a complex, bidirectional communication network that links the central nervous system (CNS), enteric nervous system (ENS), and gastrointestinal tract. This network involves intricate signaling via neural (vagus nerve), endocrine (hypothalamic-pituitary-adrenal axis), immune, and metabolic pathways. Central to this axis is the gut microbiota, the trillions of microorganisms residing in the gastrointestinal tract, which play a pivotal role in maintaining host homeostasis, including immune regulation, metabolic balance, and even behavioral and emotional states. 5
Disruptions in the gut microbiota—collectively termed dysbiosis—have been increasingly implicated in the pathophysiology of mood disorders. Alterations in microbial composition and function can influence neurotransmitter production, immune-inflammatory responses, intestinal permeability, and stress reactivity, all of which are known to contribute to the onset and maintenance of depressive and anxiety symptoms.
Traditionally, research has focused on probiotics (live microorganisms that confer health benefits) and prebiotics (non-digestible food components that promote beneficial bacterial growth) as modulators of the gut-brain axis.6–8 However, a new class of microbial-based interventions—postbiotics—has gained attention for its potential advantages in stability, safety, and targeted bioactivity.
Postbiotics are non-viable microbial fragments, cell remnants, or fermentation-produced metabolites that exert physiological benefit to the host. They are comprised of short-chain fatty acids (SCFAs), microbial peptides, extracellular polysaccharides, cell wall components, enzymes, vitamins, and other bioactive compounds. Postbiotics offer long-term, direct modulation of the gut-brain axis compared to the colonization needs of probiotics and inconsistent prebiotic fermentation, although they cannot remodel the microbiota in the long term. They can exert anti-inflammatory, antioxidant, neuroprotective, and immunomodulatory effects that are increasingly recognized as relevant to mental health.9–14 The novelty of postbiotics as a new class of drug and the lack of comprehensive reviews of their applications in neuropsychiatry underscore the need for this review to gather the current data and guide future research
Interestingly, postbiotics interact with different domains of the gut-brain axis. They modulate immune activity by reducing chronic low-grade inflammation; regulate neurotransmitter synthesis and signaling; and enhance intestinal barrier integrity, thereby limiting endotoxemia.15,16 These multi-factorial effects demonstrate the therapeutic potential of postbiotics as adjuvants or alternatives to mood disorder treatment.
Recent integrative investigations have further clarified the bidirectional relationship between neuroinflammation, metabolic signaling, and mood regulation. Emerging systems biology approaches demonstrate that microbiota-derived metabolites influence neural circuitry, neurotrophic signaling (eg, BDNF pathways), and inflammatory cascades implicated in depressive pathology. 17 These findings reinforce the translational relevance of targeting microbial metabolites rather than solely modifying microbial composition.
This review aims to provide a current summary of the evidence related to the role of postbiotics in mood regulation. It reviews the biochemical and molecular mechanisms through which postbiotics influence the gut-immune-brain axis, evaluates preclinical and clinical evidence, and outlines possible avenues for future research and clinical use in the field of psychobiotics.
Mental Disorders
Mental disorders encompass a heterogeneous group of conditions characterized by dysregulation of cognition, emotion, behavior, and stress adaptation. Major depressive disorder (MDD), bipolar disorder, generalized anxiety disorder, and stress-related disorders are strongly associated with immune activation, altered cytokine profiles (eg, IL-6, TNF-α), microglial activation, and disrupted gut barrier function. Accumulating evidence suggests that gut microbiota alterations contribute to neurotransmitter imbalance, HPA-axis dysregulation, and systemic inflammation. 18 Accordingly, microbiota-targeted strategies—including probiotics, prebiotics, and postbiotics—have been explored as adjunctive approaches in psychiatric care.
Depression
Depression and anxiety disorders are prevalent mental health conditions that affect millions globally. Genetic variations in oxytocin receptor and serotonin transporter genes have been linked to anxiety disorders, while imbalances in neurotransmitters like dopamine, serotonin, and norepinephrine are associated with depression.19,20 Depression manifests as persistent low mood, loss of interest, and other symptoms like guilt, despair, and changes in appetite and sleep patterns. Anxiety, on the other hand, is characterized by stress, fear, and physical symptoms without a clear cause. The World Health Organization states that 280 million people worldwide suffer from depression, with women being 50% more likely to experience it compared to men. 21 Various factors contribute to the development of depression and anxiety, including neurotransmitter imbalances, family history, stressful life events, and hormonal changes. Treatment options typically involve medication, psychotherapy, and lifestyle changes. 18 There is a complex relationship between anxiety and depression, with both conditions affecting the gut microbiome. 22 Chronic stress can result in increased cortisol levels, which affect neurotransmitters such as dopamine and serotonin, both of which are associated with depression. Additionally, the levels of Brain-Derived Neurotrophic Factor (BDNF) are significant in stress-related depression. 19
The gut microbiota has recently been recognized as a crucial factor in the gut-brain axis, playing a significant role in shaping stress responses and mental health outcomes.18,22,23 Alterations in the gut microbiota have been linked to a range of mental and neurological disorders. Research has demonstrated variations in the fecal microbiota of individuals suffering from depression compared to healthy individuals, with particular bacterial strains such as Lachnospiraceae and Oscilibacter being associated with depressive symptoms. 24 These findings suggest that gut bacteria influence mental health through the production of neuroactive metabolites—including valeric acid, GABA, short-chain fatty acids, serotonin precursors, and modulators of BDNF signaling.6,25
Psychosis
Psychosis is a mental health disorder defined by a disconnection from reality, encompassing symptoms such as delusions, hallucinations, disorganized thought processes, and challenges in differentiating between what is real and what is imagined. It can be a sign of various mental health conditions or medical issues and requires professional assistance for accurate diagnosis and treatment. Therapies modulating oxytocinergic signaling show promise in regulating anxiety, depression, and schizophrenia. 26 Positive symptoms of psychosis, caused by excess dopamine in the mesolimbic tract, may also involve imbalances in glutamate, production of gamma-aminobutyric acid (GABA), acetylcholine, and N-methyl-D-aspartate (NMDA) glutamate receptor function. 27 Antipsychotic medications are commonly used to manage psychotic episodes and disorders, tailored to each individual's needs.
Probiotic supplementation can influence gut microbiota, reduce inflammation, and modulate immune responses. Imbalances in gut microbiota are linked to increased pro-inflammatory cytokines and the severity of psychotic symptoms. Probiotics may benefit patients with schizophrenia by increasing BDNF levels and supporting neuroplasticity and cognitive function. In addition, studies have shown improvements in fecal microbiota and alleviation of negative symptoms in patients with schizophrenia after probiotic treatment.28,29
Several studies have demonstrated alterations in the gut microbiome and its metabolites in individuals with schizophrenia, with consistent findings of reduced levels of SCFAs, such as butyrate and propionate. These compounds exhibit anti-inflammatory and neuroprotective effects, acting through the gut–brain axis to influence brain function and behavior. 30 In a recent human case–control study, Kowalski et al 31 found that schizophrenia patients had significantly lower fecal concentrations of SCFAs and indole derivatives (from microbial tryptophan metabolism), the latter being linked to worsened negative symptoms and cognitive deficits. Preclinical models further support these findings, revealing that postbiotics such as butyrate, propionate, and lipopolysaccharide (LPS) fragments may exert more stable and rapid effects compared to live probiotics, highlighting their therapeutic potential. 32
Clinical trials have explored the effects of dopamine, serotonin, and oxytocin modulation for cognitive impairment in schizophrenia/psychosis. Probiotics, including Lactobacillus acidophilus, Bifidobacterium bifidum, Bifidobacterium lactis, and Bifidobacterium longum, have shown positive effects on symptom severity and metabolic profiles in patients with chronic schizophrenia.33,34
Mania
Manic illnesses, characterized by periods of intense and elevated mood, energy, and activity, can lead to impulsive and risky behavior. Treatments typically involve a combination of medication and support services. A combination of environmental and genetic factors, imbalances in brain chemistry, and medical disorders may cause manic episodes.35,36 Depression is often linked to stress-related systems, while mania is associated with energy and mitochondrial dysfunction. 37
Studies in mice models suggest that diet and microbiota can influence manic-like behaviors. For example, intervention with nitrated products altered certain bacteria, leading to manic-like behaviors. 38 Additionally, feeding probiotics to rats increased excitability in enteric sensory neurons. At the same time, fermented medium from specific bacteria decreased excitability in enteric nervous system neurons, indicating specificity in bacterial effects on the enteric nervous system (ENS). 39
Administering specific probiotic strains to mice has been found to reduce mania-like behaviors and induce lasting alterations in the expression of GABA receptors within the central nervous system. 40 In humans, probiotic supplementation has reduced hospital readmissions for mania in bipolar disorder patients, suggesting translational potential.41,42
Improved mood was observed in healthy volunteers consuming probiotic-containing drinks. 43 Female mice fed with specific probiotics exhibited more frequent grooming activity, potentially influenced by oxytocin. 44
Postbiotics from certain bacteria have shown benefits similar to probiotics in heat-stressed birds.45,46 Sodium butyrate, as a postbiotic, improved animal sociability and memory. 47 Co-administration of α-lactalbumin with sodium butyrate was effective in generating antidepressant-like activity in animal models of idiopathic autism. 25
In humans, administration of heat-inactivated Lactobacillus gasseri CP2305 has been associated with reduced anxiety scores and improved sleep quality in randomized trials, supporting translational relevance. 48
How Postbiotics Manage Happiness
The mechanism by which probiotics may influence mood is not yet fully understood, but several potential pathways have been proposed, as illustrated in Figure 1. One potential mechanism involves the gut-brain axis, a bidirectional communication network linking the gut and the brain. Probiotics can alter the composition of the gut microbiota, which in turn can result in variations in the production of neurotransmitters and neuroactive compounds, ultimately impacting brain function and mood. For example, certain probiotic strains have been shown to increase the production of GABA, a neurotransmitter that has calming effects on the brain and can help reduce anxiety and improve mood. 49
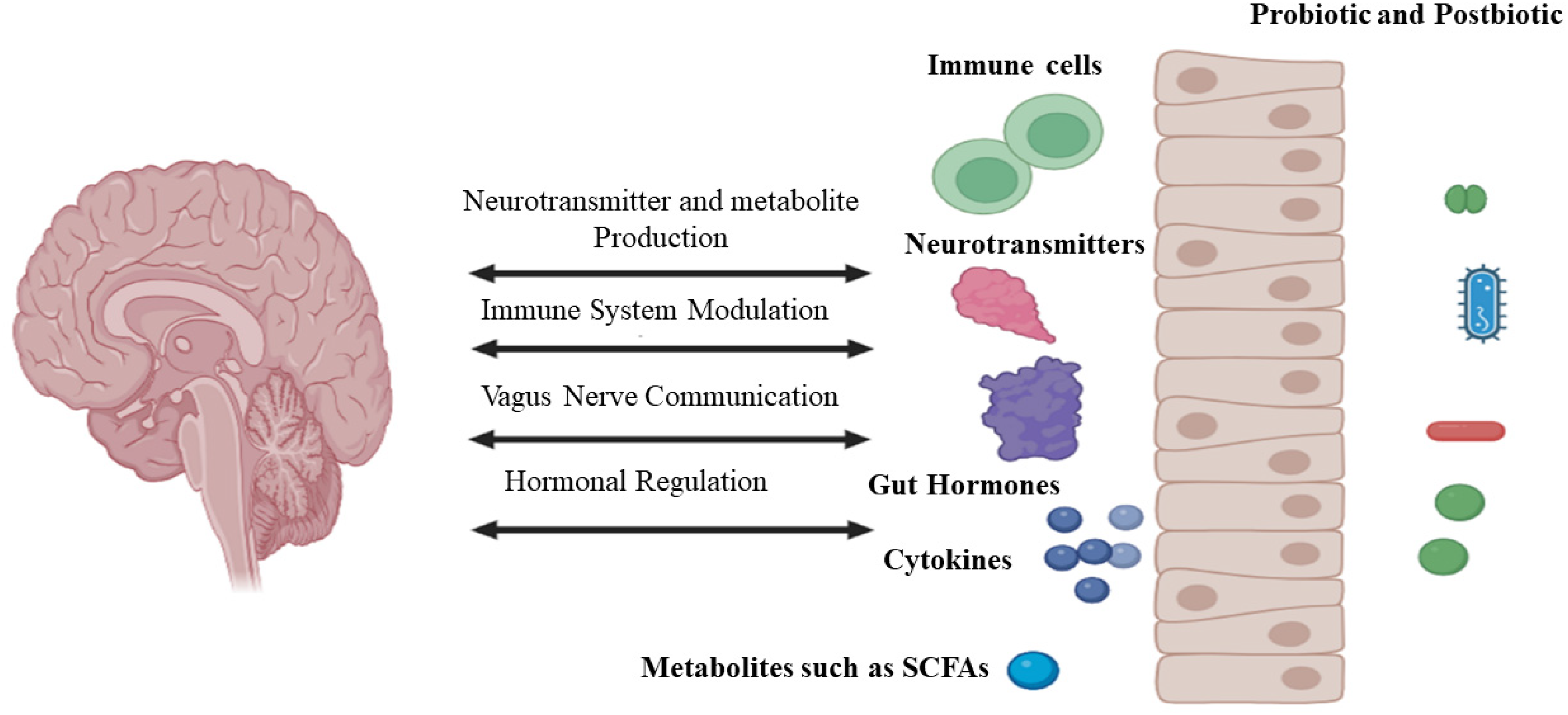
Mechanistic pathways of postbiotics in modulating mood via the gut-brain-immune axis.
Moreover, probiotics and postbiotics may exhibit anti-inflammatory properties that could influence mood regulation. Chronic inflammation has been associated with mood disorders such as depression, and probiotics have been demonstrated to lower markers of inflammation in the body.50–52 By decreasing inflammation, probiotics may help improve mood and overall well-being.
Additionally, probiotics can impact the production of short-chain fatty acids (SCFAs) in the gut. These SCFAs, which are generated by specific gut bacteria during the fermentation of dietary fibers, have been found to positively affect mood and cognition. Acting as signaling molecules, SCFAs communicate with the brain and influence pathways related to mood. 53
Postbiotic Metabolites Responsible for Happiness
Short Chain Fatty Acids (SCFAs)
SCFAs, produced through the fermentation of indigestible carbohydrates by intestinal bacteria, impact both non-immune and immune cells in the intestine. They act as an energy source for the gut microbiota and intestinal epithelial cells, ensuring the integrity of the mucosal barrier. Moreover, SCFAs play a crucial role in both innate and intestinal immunity. As histone deacetylase (HDAC) inhibitors, they exhibit anti-inflammatory properties, promote the differentiation of regulatory T (Treg) cells, and help reduce the severity of colitis.54,55 SCFAs influence both non-immune and immune cells within the intestine, playing a crucial role in maintaining intestinal homeostasis. They have the potential to signal the brain, indicating their significant role in the gut-brain axis and overall health. 56 Next-generation probiotics (NGPs) represent a promising class of microorganisms that have been extensively studied for their biotherapeutic properties, particularly as efficient producers of short-chain fatty acids (SCFAs). For instance, Akkermansia muciniphila, a key NGP from the Verrucomicrobia phylum, degrades mucins to generate SCFAs like butyrate and propionate, enhancing intestinal barrier integrity and reducing inflammation, with studies showing its potential in alleviating metabolic disorders and obesity-related mood disturbances through endocannabinoid system regulation. 57 Similarly, Faecalibacterium prausnitzii, from the Firmicutes phylum, ferments carbohydrates to produce high levels of butyrate, promoting anti-inflammatory effects, Treg cell induction, and intestinal homeostasis, with evidence linking its depletion to inflammatory bowel diseases and depressive symptoms, suggesting therapeutic applications in mood disorders via SCFA-mediated immunomodulation.58,59 These NGPs, unlike traditional probiotics, offer disease-specific benefits and warrant further clinical trials to validate their SCFA production in human neuropsychiatric interventions. 60 Table 2 presents probiotic species, including NGPs.
Individual postbiotic compounds exert their effects on mood disorders through both distinct and overlapping neuroimmune pathways. Among SCFAs, butyrate has emerged as a key immunometabolic mediator, acting primarily through histone deacetylase (HDAC) inhibition, activation of G-protein–coupled receptors (FFAR2 and FFAR3), and promotion of regulatory T cell differentiation. These effects result in reduced systemic and central inflammation, attenuation of microglial activation, and preservation of gut and blood–brain barrier integrity—processes closely linked to the pathophysiology of depression and bipolar spectrum disorders.70,71 In contrast, neurotransmitter-related postbiotics such as GABA exert more direct neuromodulatory effects by engaging GABA_A and GABA_B receptors expressed on enteric neurons, vagal afferents, immune cells, and microglia, thereby influencing cytokine production, hypothalamic–pituitary–adrenal axis activity, and limbic network signaling. 72
Although both SCFAs and neurotransmitter-related postbiotics converge on immune modulation and neuroinflammation attenuation, their mechanisms differ in primary targets. Butyrate predominantly acts via epigenetic regulation (HDAC inhibition) and immunometabolic pathways, whereas GABA primarily modulates neuronal excitability and vagal signaling. Overlap occurs at the level of cytokine regulation, microglial activation, and hypothalamic–pituitary–adrenal (HPA) axis modulation, suggesting a complementary rather than redundant interaction within the gut–brain–immune axis.
GABA
GABA is a primary inhibitory neurotransmitter within the central nervous system. It plays a pivotal role in reducing neuronal excitability and regulating muscle tone, helping to maintain balance and prevent overexcitation of neurons. 73 Dysfunctions in the central GABA system are linked to anxiety disorders, while positive GABA receptor modulators have anxiolytic effects. 74 Gut microbiota-produced GABA can influence the CNS via the enteric nervous system, impacting the gut-brain axis. GABA also exhibits various health benefits, such as antidepressant, anti-diabetic, anti-hypertensive, neuroprotective, cardiovascular-regulating, and anti-cancer properties. 75 Probiotic lactic acid bacteria (LAB) synthesize GABA through the enzyme glutamate decarboxylase, contributing to intracellular pH homeostasis.76,77 GABA enhances growth hormone function, increases plasma concentration, and promotes protein synthesis in the brain. 78 Interaction with Neuroimmune Pathways: GABA modulates neuroimmune pathways by reducing microglial activation and cytokine release, distinct from butyrate's metabolic focus but overlapping in anti-inflammatory effects on mood disorders like anxiety. 47
Dopamine
Dopamine is indeed a vital neurotransmitter, playing an essential role in the brain's reward system. It significantly influences feelings of pleasure and motivation, contributing to our ability to experience happiness and drive. Its effects extend to various aspects of behavior and cognition, including mood, attention, and learning. When a person is physically attracted to another, the activation of dopamine, serotonin, and oxytocin production increases, leading to a reduction in pain and regulation of emotions by disconnecting the amygdala, which is responsible for negative emotions like fear.27,79–81
Interestingly, more than 50% of dopamine is generated in the gut by various bacteria such as Staphylococcus aureus, Escherichia coli, Bacillus cereus, Bacillus mycoides, Bacillus subtilis, Proteus vulgaris, and Serratia marcescens. Enterococcus species have been discovered to produce dopamine in a gastrointestinal-like environment when the dopamine precursor L-3,4-dihydroxyphenylalanine (L-dopa) is present. This production of dopamine by specific gut bacteria, such as Enterococcus faecium, can affect the host by influencing dopaminergic pathways.82–84
Serotonin
Moving on to serotonin, also known as 5-hydroxytryptamine (5-HT), plays a crucial role in managing happiness and regulating various physiological functions. 79 Serotonin, produced and released by Enterochromaffin (EC) cells in the human gut, plays a crucial role in regulating the contractile frequency during peristalsis and metabolism. Postbiotics, which are metabolic byproducts of probiotic bacteria, can enhance luminal serotonin levels by affecting the expression of enzymes and receptors involved in serotonin synthesis.85,86
The gut microbiota influences serotonin levels in the brain by regulating the expression of serotonin receptors, transporters, synthesis enzymes, and metabolic enzymes. Research has demonstrated that mice with human gut microbiota exhibit higher colonic serotonin levels compared to germ-free mice. Additionally, certain postbiotics derived from probiotic bacteria, like acetate and butyrate, have been shown to increase serotonin levels in the colon.82,87–89
Tryptophan
Tryptophan, an essential amino acid crucial for synthesizing neurotransmitters like melatonin, niacin, and serotonin, plays a vital role in mood, cognition, and behavior. While not produced in the human body, tryptophan is acquired through diet or protein breakdown. It serves as a building block for serotonin and nicotinamide adenine dinucleotide (NAD/NADH) via the kynurenine pathway.90,91 Probiotic supplementation has been linked to changes in serotonin levels, with studies showing decreased fecal tryptophan and increased serum serotonin after probiotic intake. 86
Moreover, probiotics have been associated with reduced depression symptoms by lowering the kynurenine/tryptophan ratio, leading to improved mood scores. In particular, Lactobacillus plantarum DR7 supplementation enhanced the serotonin pathway while stabilizing the dopamine pathway over 12 weeks compared to a placebo. This was evidenced by decreased expressions of enzymes involved in dopamine metabolism and increased expressions of serotonin-related proteins. 56
Generally, tryptophan's involvement in neurotransmitter synthesis underscores its impact on mental health, with probiotics like Lactobacillus strains influencing serotonin levels and potentially alleviating depressive symptoms through modulation of the tryptophan metabolism pathway.86,92,93
Oxytocin
Oxytocin, a neuropeptide hormone produced in the brain, plays a significant role in managing happiness and overall well-being. Known for its involvement in love, trust, and bonding behaviors, oxytocin also reduces94,95 anxiety and stress by interacting with the stress axis. 96 Additionally, oxytocin promotes wound healing and muscle health, contributing to physical resilience and lowering the risk of obesity-related issues. 97
In terms of mental health, oxytocin acts as an anxiolytic, decreasing stress levels and promoting feelings of enjoyment. Studies have shown that consuming probiotics like Lactobacillus reuteri can increase oxytocin levels in the brain through a vagus nerve-mediated mechanism, highlighting the gut-brain axis and the influence of gut bacteria on systemic health.94,95 In addition to Lactobacillus reuteri, strains such as Lactobacillus rhamnosus and Bifidobacterium longum have demonstrated oxytocin-mediated or social-behavior–modulating effects in preclinical studies. 98
Furthermore, oxytocin stimulates reward centers in the brain, leading to a more positive outlook and increased prosocial behavior. Supplementing with Lactobacillus reuteri has been shown to enhance oxytocin expression in the hypothalamus, activate the dopamine reward system, and improve nurturing behaviors in animal models.99–101
By modulating oxytocin levels, probiotics like Lactobacillus reuteri can impact various aspects of health, including mental, social, and physical well-being. 101 The findings suggest that oxytocin plays a crucial role as a postbiotic in promoting overall health and quality of life.
Microbiota-Brain Signaling
Bacteria-brain signaling encompasses the communication and interaction between the gut microbiota and the brain. The gut-brain axis is a two-way communication system that includes the central nervous system (CNS), the enteric nervous system (ENS), and the gut microbiota. This axis, made up of trillions of bacteria residing in the gastrointestinal tract, can influence brain function and behavior through various signaling pathways. This communication is thought to impact several aspects of mental health, such as anxiety, depression, stress response, and cognitive function. Dysbiosis, or an imbalance in the gut microbiota, has been linked to specific mental health disorders.102,103
Neurotransmitter Production
The gut-brain axis involves communication between the gut and brain through neurotransmitters. Key neurotransmitters produced in the gut include serotonin, GABA, acetylcholine, and dopamine. They influence gastrointestinal function and can impact mood, behavior, and disorders like IBS and depression.104,105 Understanding these interactions is important for developing new treatments for gut-brain axis disorders.
Microbial Metabolites
Microbial metabolites like SCFAs play a key role in the gut-brain axis by influencing communication between the gut and the brain. SCFAs can act as signaling molecules that impact neurotransmitter production, immune responses, and inflammation, affecting mood regulation and gut-brain barrier function. Other metabolites, like indole, also influence neurotransmitter synthesis and gut motility. Understanding these interactions is crucial for developing therapies to improve gastrointestinal and mental health.45,106–109
Immune System Modulation and Inflammation Control
The gut microbiota plays a pivotal role in the development and regulation of the host immune system, exerting systemic effects that extend to the central nervous system (CNS). Alterations in gut microbial composition—commonly referred to as dysbiosis—have been increasingly linked to neuropsychiatric disorders such as depression and anxiety, in part through their modulation of immune and inflammatory pathways.
The immune system and gut microbiota engage in constant bidirectional communication. Commensal microbes can influence both innate and adaptive immunity by promoting immune tolerance, regulating dendritic cell and macrophage activity, enhancing gut barrier integrity, and modulating the secretion of cytokines and antimicrobial peptides.110–113 Importantly, chronic low-grade inflammation and immune dysregulation have been identified as key contributors to the pathophysiology of mood disorders. Elevated levels of pro-inflammatory cytokines such as IL-6, TNF-α, and IL-1β have been observed in patients with depression and anxiety, contributing to altered neurotransmitter metabolism, impaired neuroplasticity, and HPA axis dysregulation.
Postbiotics can directly influence these immunoinflammatory processes. SCFAs, such as butyrate, acetate, and propionate, have demonstrated potent anti-inflammatory properties. They suppress the activation of nuclear factor kappa B (NF-κB), inhibit histone deacetylases (HDACs), and promote the differentiation of regulatory T cells (Tregs), thereby reducing systemic inflammation. Butyrate also supports intestinal epithelial health by enhancing mucosal barrier integrity and limiting microbial translocation that might otherwise trigger immune responses. For example, butyrate interacts with neuroimmune pathways by reducing microglial activation, distinct in its metabolic HDAC inhibition but overlapping with GABA's effects on cytokine suppression. 114
Tryptophan metabolites generated by microbial metabolism, such as indole derivatives, also interact with the aryl hydrocarbon receptor (AhR) to modulate immune cell activity and cytokine profiles. Some postbiotic components have been shown to suppress microglial activation and neuroinflammation in preclinical models of depression.
The vagus nerve acts as a critical mediator in the microbiota-immune-brain axis. Postbiotic molecules can stimulate afferent vagal pathways, leading to central modulation of immune function through the cholinergic anti-inflammatory reflex—a mechanism involving acetylcholine-mediated suppression of pro-inflammatory cytokine release by macrophages. 115
It should be noted that while butyrate-driven pathways predominantly target immune regulation and epigenetic control of inflammatory signaling, and GABA-related mechanisms primarily modulate neural excitability and neuroimmune communication, both pathways converge on the shared outcome of dampening neuroinflammation and restoring emotional and stress-related homeostasis. 71
Collectively, these mechanisms underscore the role of postbiotics as key immunomodulatory agents capable of mitigating neuroinflammation and restoring immune balance, factors that are essential for improving mood and emotional regulation. Understanding these interactions offers novel therapeutic opportunities in targeting the immune-inflammatory axis in mood disorders.
Vagus Nerve Communication
The vagus nerve acts as a vital communication route between the gut and the brain. It facilitates the transmission of signals from gut bacteria to the brain, and vice versa, playing a pivotal role in the gut-brain axis. This pathway underscores the intricate relationship between the gut microbiota and brain function, highlighting the bidirectional nature of their interactions. This nerve sends signals from the gut to the brain, informing it about the state of the gut environment, such as the presence of harmful pathogens or beneficial nutrients. In response, the brain can modulate various physiological and behavioral responses to maintain homeostasis.116–118
The vagus nerve serves as a crucial conduit for communication between the gut and the brain. It enables the transmission of signals from gut bacteria to the brain, and vice versa, emphasizing its essential role in the gut-brain axis. This pathway highlights the complex, bidirectional relationship between the gut microbiota and brain function.110–112
Hormonal Regulation
Gut bacteria can influence hormone production and signaling, such as affecting the release of stress hormones like cortisol or regulating appetite-regulating hormones like leptin and ghrelin.
Leptin, a hormone produced by fat cells, plays a crucial role in regulating energy balance and appetite. Gut bacteria can modulate leptin levels, influencing the hunger signals that are sent to the brain. 119 Ghrelin, often referred to as the “hunger hormone,” stimulates appetite and drives food intake. The gut microbiota can affect ghrelin production, thereby influencing sensations of hunger and fullness. 120 Cortisol is a hormone produced by the adrenal glands in response to stress. When cortisol levels become dysregulated, it can contribute to mood disorders and impair cognitive function. 121 Gut bacteria can influence cortisol production through interactions with the hypothalamic-pituitary-adrenal axis. 122 Insulin, a hormone produced by the pancreas, plays a key role in regulating blood sugar levels. The gut microbiota can influence insulin sensitivity and glucose metabolism, potentially contributing to metabolic disorders such as diabetes. 123 Oxytocin, often called the “love hormone” or “bonding hormone,” plays a pivotal role in social bonding, trust, and emotional regulation. Interestingly, gut bacteria may influence the production and release of oxytocin, which in turn can affect social behavior and emotional well-being. 124
Challenges and Futures of Postbiotics
Challenges and Limitations
Postbiotics present major challenges and limitations for mood disorder applications. Strain-associated metabolite profiles introduce composition heterogeneity, exacerbated by the absence of standardized production protocols and regulatory processes, which complicate formulation, labeling, and clinical acceptance.13,125 Host-specificity, based on microbiota diversity and genetic polymorphism, restricts extendability to populations. 126 Variations in gut microbiota composition across geographic regions, dietary patterns, age groups, and genetic backgrounds may influence responsiveness to postbiotic interventions. Personalized microbiome profiling may therefore enhance generalizability and therapeutic precision.
Most studies are either preclinical or small-scale, which complicates the evidence base. This leads to variability in strains, dosing, chemicals, and endpoints, increasing the risk of reproducibility issues and publication bias. 19 Human trials are scant, with poor translation from animal models because of microbiota heterogeneity, limiting evidence for durable efficacy and safety. 62 Whereas postbiotics have a good safety profile with minimal side effects comparable to probiotics (eg, gas or bloating), long-term negative effects in vulnerable populations necessitate ongoing monitoring. 125
Regulatory frameworks for postbiotics vary internationally. Currently, most are classified as functional food ingredients or dietary supplements rather than pharmaceutical agents. Standardization of production, metabolite quantification, and quality control remains an unmet need.
Advances
Advances in postbiotics involve precision-formulated metabolites for specific applications (eg, butyrate-enriched extracts) and multi-omics technology-based integration toward biomarker identification, thereby enhancing therapeutic specificity and efficacy in mood disorders. 115 In contrast to prebiotics and probiotics, postbiotics possess distinctive advantages, including enhanced stability that negates the requirement for live cell viability and enhanced safety for immunocompromised patient. 127 Their non-viability favors directed bioactivity, permitting direct delivery of metabolites such as butyrate to neuroimmune pathways without transcending colonization barriers. 128 These characteristics render postbiotics a potential adjunctive modality for personalized treatments of mood disorders, as seen by clinical and mechanistic evidence (Tables 1 and 2). Evidence from clinical trials supports postbiotics’ efficacy, such as Lactobacillus gasseri CP2305, improving mental state and sleep in stressed individuals. 61 Pilot RCTs show postbiotic blends reduce anxiety symptoms in healthy adults. 129 Combined with antidepressants, postbiotics enhance efficacy in depression. 130 Dose-dependent effects have been demonstrated primarily in preclinical models. For example, butyrate concentrations between 1–5 mM exhibit anti-inflammatory and neuroprotective effects, whereas concentrations exceeding 10 mM may lose efficacy or induce cytotoxic responses in vitro. Human therapeutic ranges remain undefined, highlighting the need for pharmacokinetic and dose-optimization trials. 63
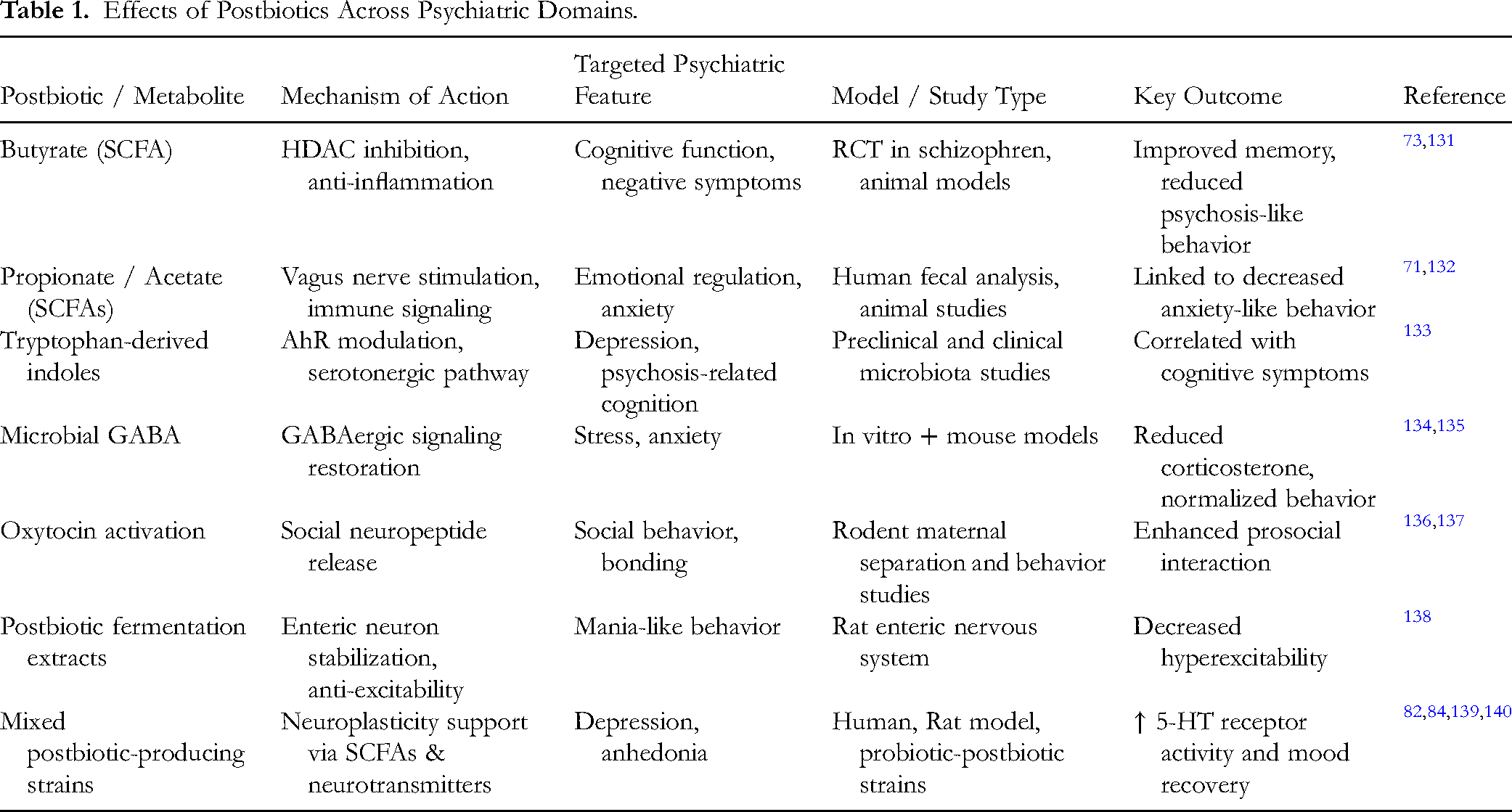
Effects of Postbiotics Across Psychiatric Domains.
Probiotic species, Including Next-Generation Probiotics (NGPs), Postbiotic Molecules, and Neuroprotective Effects.

While preclinical studies consistently demonstrate anti-inflammatory and behavioral benefits, translational extrapolation remains cautious. Rodent models allow controlled microbiota manipulation but do not fully recapitulate human microbiome diversity, environmental exposures, or psychiatric heterogeneity. Therefore, clinical validation is essential before definitive therapeutic claims can be established.
Future Directions
Large-scale, placebo-controlled clinical trials with standardized postbiotic products and psychiatric outcomes will be the subject of future studies to establish efficacy and safety. Long-term (>6-12 months) longitudinal studies are required to evaluate sustained psychiatric outcomes and relapse prevention.
Biomarker identification (eg, microbiota composition, cytokine profile) will inform targeted interventions in personalized medicine concepts. Multi-omics strategies (metabolomics, transcriptomics, microbiomics) will deliver systems-level insights into the neuropsychological action of postbiotics. 115
Randomized controlled trials investigating heat-killed Lactobacillus gasseri CP2305 and other postbiotic preparations report improvements in stress-related symptoms, sleep quality, and mild depressive features. 141 However, most trials involve small sample sizes and short durations (<12 weeks), limiting conclusions regarding sustained efficacy.
Preliminary evidence suggests synergistic effects when postbiotics are combined with conventional antidepressants, potentially enhancing anti-inflammatory action and reducing treatment resistance. Larger combination trials are warranted.
Future mechanistic studies should integrate transcriptomics, epigenomics, and metabolomics to clarify how postbiotics regulate microglial phenotypes, synaptic plasticity, and neurotrophic signaling networks.
Comparison with Probiotics and Prebiotics
Unlike probiotics, postbiotics do not require viability to exert biological effects, potentially offering improved safety and stability. Compared with prebiotics, which indirectly modulate microbial composition, postbiotics deliver defined bioactive molecules with predictable mechanistic targets. However, they may lack the ecological benefits associated with live microbial colonization. Thus, each strategy presents distinct advantages and limitations.
Safety Considerations
Postbiotics are generally considered safe due to the absence of live organisms. Reported adverse effects are mild and include transient gastrointestinal discomfort. Long-term safety data remain limited, particularly regarding high-dose SCFA supplementation.
Study Limitations
This review is limited by reliance on heterogeneous preclinical models, small-scale clinical trials, and variability in postbiotic formulations. Lack of standardized dosing and outcome measures restricts cross-study comparisons.
Conclusion
Postbiotics represent a mechanistically grounded approach to modulating the gut–brain–immune axis in mood disorders. By targeting inflammatory cascades, epigenetic regulation, neurotransmitter signaling, and barrier integrity, these microbial metabolites offer a biologically plausible adjunct to conventional psychiatric therapies. Although preliminary clinical findings are promising, robust randomized trials, standardized formulations, and long-term safety evaluations are essential before routine clinical integration. Advancing precision microbiome-based psychiatry will require multidisciplinary collaboration and systems-level investigation.
Footnotes
Acknowledgements
None.
Author Contribution Statement
Conceptualization: [Aziz Homayouni-Rad] Methodology: [Saba Kamalledin Moghadam, Aziz Homayouni-Rad] Investigation: [Aziz Homayouni-Rad, Alireza Asghari, Saba Kamalledin Moghadam] Data curation: [Jalil Houshyar, Hossein Alikhah] Formal analysis: [Babak Daneshfard, Saba Kamalledin Moghadam] Writing — original draft: [Saba Kamalledin Moghadam, Babak Daneshfard] Writing — review & editing: [All authors] Visualization: [Saba Kamalledin Moghadam] Supervision: [Alireza Asghari, Aziz Homayouni-Rad, Babak Daneshfard]
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Not applicable.
