Abstract
Introduction
A millet grain diet reduces the absorption of cholesterol and fat, facilitates emptying of the stomach, and may improve gut health.
Objective
This study examined the effects of pearl millet and its ethanolic extract on the gut microbiota, intestinal structure, and serum metabolome in obese rats fed a high-fat diet.
Methods
This study examined the impacts of pearl millet grain (MPG) and its ethanolic extract (MPGethanolE) on gut health by investigating alterations in the gut microbiota and serum metabolome of obese rats fed a high-fat diet. Rats were divided into eight groups, based on the treatments they received: control, high-fat diet (HFD), HFD + MPGethanolE (25 mg/kg), HFD + MPGethanolE (50 mg/kg), HFD + MPGethanolE (100 mg/kg), HFD + MPG (10%), HFD + MPG (20%), and HFD + MPG (30%).
Results
The results showed that the fecal samples from MPG-treated groups exhibited a significant difference in pH, which varied from 6.5 to 4.5. Furthermore, higher dosages of MPG and MPGethanolE altered the composition of the gut flora, leading to significant increases (p < .05) in the abundance of Akkermansia muciniphila (19.01%, 18.45%, and 20.39% vs 0.00% in controls), while the lowest dose of millet ethanolic extract was found to increase the abundance of Lactobacillus spp. (31.71%, 32.89%, and 33.09% vs 20.39% in controls). In contrast, the abundance of Bifidobacterium spp. remained unchanged under the interventional diets throughout the experimental period.
Conclusion
Both MPG and MPGethanolE diets were found to improve the intestinal architecture, including the villus shape, and considerable differences in metabolomics profiles were observed between the untreated and treated groups. Hence, these alterations may be mediated by putative prebiotic-like dietary impacts. Nevertheless, further studies are needed to establish the impacts of these grains and their extracts on gut metabolites, such as SCFAs, as well as host metabolism.
Introduction
Obesity is described as an abnormal or excessive accumulation of body fat that may serve as a prominent risk factor for many chronic conditions, including metabolic syndrome (MetS). MetS is a pathological condition driven by several risk factors, such as systemic hypertension, atherogenic dyslipidemia, and insulin resistance. These interrelated factors can lead to prothrombotic and proinflammatory conditions, which are often associated with atherosclerosis and cardiovascular disease. 1 Obesity and MetS are associated with obesogenic high-calorie and low-fiber diets. Hence, diet is one of the most significant modifiable factors through which these multifactorial disorders may be prevented. Millet, for instance, is a well-known traditional, nutritious staple food that is rich in fiber, protein, and other essential nutrients, including iron, zinc, magnesium, selenium, and vitamins. 2 Furthermore, millet has been suggested to improve the composition of the gut microbiome, leading to beneficial modulation of host dietary responses.
The symbiotic microorganisms found in the human intestine are referred to as the gut microbiota,3–5 comprising a complex and dynamic microbial community including bacteria, viruses, archaea, and eukaryotes. This community maintains a symbiotic relationship with the gut mucosa and plays important metabolic, immune, and gut-protecting roles in healthy people. 6
The diverse gastrointestinal microbiota is primarily composed of bacteria belonging to three major phyla: Firmicutes (Gram-positive bacteria), Bacteroidetes (Gram-negative bacteria), and Actinobacteria (Gram-positive bacteria). The phylum Firmicutes contains over 200 genera, including Lactobacillus, Bacillus, Clostridium, Enterococcus, and Ruminicoccus, while the phylum Bacteroidetes contains both the genera Bacteroides and Actinobacteria.7–9
Numerous endogenous and exogenous factors have been shown to impact the composition of the gut microbiota, including diet.4,10–13 Long-term consumption of a high-fat diet (HFD) can cause gut microbiota imbalance (dysbiosis) and metabolic disorders (obesity or diabetes), 14 and has been shown to generate low-grade chronic intestinal inflammation in rats. 15 A high-fat diet is thought to be a risk factor for the development of type 2 diabetes (T2D), cardiovascular disease (CVD), and central nervous system problems. 16
Pearl millet (PM) grains have a high fiber content, at around 7.8% per 100 g. 17 In the human gastrointestinal tract, host amylase enzymes mostly break down starches. However, some fibers—such as galactooligosaccharides, fructooligosaccharides, resistant starches, inulins, and pectin—are poorly digested. Not all dietary fibers act uniformly on the gut microbiota, with differences in structure and fermentability influencing microbial shifts and metabolic outcomes. Recent studies have shown that fibers with varying chain lengths produce distinct microbiota patterns and metabolic benefits in high-fat-diet models, underscoring the importance of fiber characteristics in obesity-related gut modulation effects.18,19 Therefore, millet—which is rich in complex fibers—may exert unique gut-modulating effects through its specific fermentable components. Members of the gut microbiota (including Firmicutes, Bacteroidetes, Bifidobacterium, and Prevotella species) can ferment and depolymerize dietary fiber to produce short-chain fatty acids (SCFAs) such as acetate, propionate, and butyrate. 20 The health benefits derived from SCFA synthesis are represented by a concurrent decrease in luminal pH, which inhibits the growth of harmful microbes and promotes nutritional absorption.21,22 However, millet grains are rich in phenolic components—including phenolic acids, flavonoids, polymeric tannins, and anthocyanins 23 —which have low bioavailability, and thus, limited usefulness regarding promoting health. Due to the chemical complexity and greater molecular weight of polyphenols, in addition to the lack of esterase enzymes in the human stomach, they cannot be readily absorbed in the small intestine, with only around 5%–10% of the total dietary polyphenols ultimately being absorbed. Therefore, a significant number of consumed polyphenols enter the colon and are reduced to corresponding low-molecular-weight metabolites by colonic bacteria, thus influencing the gut bacterial composition in an interesting manner.24,25
Although the dietary ingestion of millet has been linked to improved gut health and lipid metabolism, it is unclear how millet and its ethanolic extracts affect the gut microbiota, intestinal structure, and host metabolism in the context of high-fat-diet-induced obesity. In particular, its prebiotic-like effects on microbes such as A. muciniphila and Lactobacillus spp. and impacts on the serum metabolome remain poorly understood. This study aims to address these gaps through gut microbiota profile, intestinal morphology, and metabolomic analyses. In particular, the study aimed to examine the impacts of consuming pearl millet grain and its ethanolic extract on alterations in the gut microbiota, intestinal structure, and serum metabolome in obese rats fed a high-fat diet.
Materials and Methods
Preparation of Raw Powder and Ethanolic Extract
Pearl millet (PM) (Pennisetum glaucum L.) grains were purchased from a local market in the Al Baha region, Saudi Arabia, and ground into a fine powder using a commercial grinder (SANYO, Tokyo, Japan). The flour PM whole-grain (MPG) powder was stored in a dry, clean place for later use. To prepare the PM grain powder ethanolic extract (MPGethanolE), 2 kg of the PM flour was suspended in 2.5 L of ethanol (70% purity) with continuous shaking for 3 h. This procedure was repeated 3 times. Then, the obtained extract was filtered using filter paper (Whatman No. 1) and concentrated using a rotary vacuum (±50 °C, 450 mm Hg). The dried residues were placed in tightly closed glass tubes and stored at −20 °C for later use. 26
The doses of millet grain (MPG) and its ethanolic extract (MPGethanolE) were selected based on human equivalent intake, calculated using body surface area normalization, to reflect realistic dietary consumption levels in humans. At the time of the experiments, MPG powder and MPGethanolE residues were dissolved in 0.1% DMSO as a carrier solvent. 27
Experimental Design, Animals, and Diets
Forty-eight adult male Wistar rats (150 ± 20 g, ten weeks old) were obtained and maintained in the Experimental Animal Care Center at King Saud University, Riyadh, Saudi Arabia. The rats were housed in controlled ambient conditions (22 ± 2 °C, 50% humidity, 12/12 h light/dark cycle) for one week of acclimatization, during which they were freely allowed access to food and drinking water.
Both the control diet (3.85 kcal/g) (Cat. No. D12450H) and HFD (3.85 kcal/g) (Cat. No. D12451) were purchased from Research Diets (NJ, USA). The HFD was designed to induce obesity, IR, and NAFLD after feeding for 8 consecutive weeks. The energy obtained from fat, protein, and carbohydrates for each diet is shown in Table 1. The ingredients of both diets can be obtained from the company website. The reporting of this animal study conforms to the ARRIVE 2.0 guidelines. 28
Energy Intake for Both Control Diet and HFD.
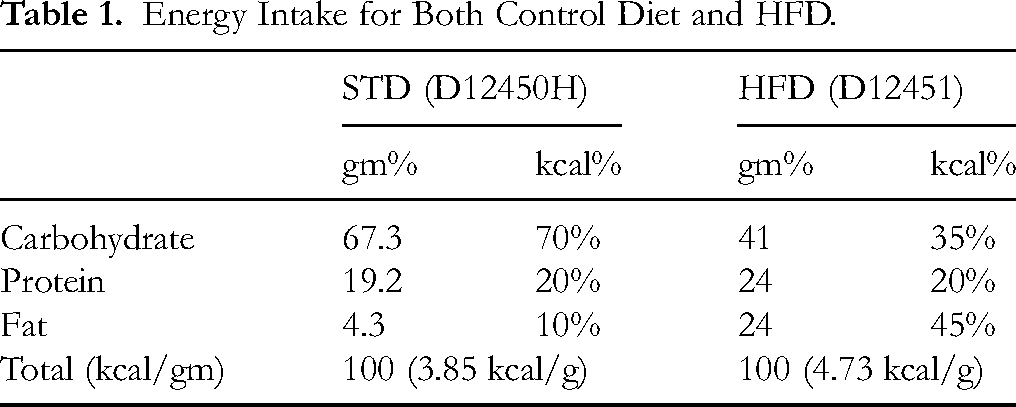
Rats were divided into 8 groups (n = 6/group). One was a control group, in which rats were given 0.25 mL of 0.1% DMSO (vehicle) orally. The other 7 groups of HFD-fed rats were treated as follows: 1 group was only fed an HFD (0.25 mL of 0.1% DMSO) orally; 3 groups were fed an HFD and concomitantly orally administered MPG powder solution (0.25 mL) at a concentration of 10%, 20%, or 30%; the other 3 groups were fed an HFD and concomitantly orally administered MPGethanolE solution (0.25 mL) at concentrations of 25, 50, or 100 mg/kg. All treatments were administered daily via gavage using a stainless-steel feeding cannula by a well-trained vet for 8 consecutive weeks. Throughout the experimental period, the rats’ body weight and food intake were recorded weekly. The doses of millet ethanolic extract (MPGethanolE) used in this experiment were adapted from Luqman et al, 29 whereas whole-grain doses were adapted from Khan et al 30
The experimental protocol (Ref. No.: KSU-SE-21-49) was approved by the Research Ethics Committee at King Saud University, Riyadh, Saudi Arabia.
Blood and Intestinal Tissue Sample Collection and Preparation
All animals were fasted overnight, then anesthetized with an intraperitoneal dose of ketamine hydrochloride (90 mg/kg) and xylazine hydrochloride (10 mg/kg) according to the method of Kim et al 31 Blood (1 mL) was directly collected via cardiac puncture into plain tubes. The tubes were then centrifuged (3000 rpm; 10 min) at room temperature to collect the serum. All serum samples were collected in new tubes and stored at −20 °C for future biochemical analysis. Rats were then euthanized using the cervical dislocation protocol. Intestinal tissues were collected on ice and cut into smaller pieces. Parts of the intestine were placed in 10% buffered formalin for further analysis.
Intestinal Microscopic Observation
Intestine tissue samples fixed in 10% buffered formalin were dehydrated in ethanol at increasing concentrations (70-100%) and cleared with xylene. The tissues were then embedded in paraffin wax, sliced into 4 µm thick sections, stained with hematoxylin and eosin, and examined under a light microscope (Olympus Optical, Tokyo, Japan). 32
Fecal Sample Collection
At the end of the experiment, feces were collected from each group and stored at −80 °C in sterile, disposable plastic centrifuge tubes until further analysis.
Fecal Sample Analysis
DNA Extraction
Fecal DNA was isolated from all samples using the Purelink microbiome DNA purification kit (Cat No A29790, Waltham, MA, USA), following the manufacturer's instructions. The purified DNA was stored at −20 °C for later use. The DNA purity and concentration were ensured using a Genova nanodrop (Italy) at A260/A280. 33
Real-Time Quantitative PCR (qPCR)
Real-time qPCR was conducted as described in, 6 with a few modifications, using the Applied Biosystems 7500 Real-time PCR System running software. Amplification and detection were performed using GoTaq® RT-qPCR (Promega, USA). All amplifications were performed in triplicate in a final volume of 20 μL containing 10 μL of 2xSYBR Green PCR Master Mix, including ROX as a passive reference, 900 nM of each primer (Table 2), and 2 μL of template DNA. The PCR protocol was as follows: an initial cycle of 95 °C for 10 min, followed by 40 cycles at 95 °C for 15 s, and 1 min at 60 °C.
Primers Used in Real-Time qPCR Reactions.

Fecal pH Determination
The gut contents were thawed on ice, and approximately 400 mg of the gut contents were transferred to a clean tube. HPLC-grade water was then added at a 10:1 ratio. Next, the mixture was homogenized for 2 min at 1200 rpm, then centrifuged for 10 min at 4 °C at 3509 g. Subsequently, the gut water pH was measured using a pH meter (Mettler Toledo, USA). 39
Metabolite Extraction and GC-MS Analysis
The metabolite extraction and GC-MS analysis were conducted according to an earlier study, 40 with some modifications. Serum samples (100 μL) collected in Eppendorf tubes were first extracted with 5 μL of 4 mg/mL ribitol dissolved in MilliQ water (Millipore, USA), as an internal standard to make up for any metabolite loss during the extraction procedure. Initially, 450 μL of a 3:1 (v/v) acetonitrile/methanol mixture was added to each sample and vortexed for 2 min. The samples were then left undisturbed at room temperature for 10 min. Subsequently, the samples were centrifuged at 12 000 rpm and 4 °C for 20 min, and the supernatant was collected into new Eppendorf tubes. The second extraction was performed using the remaining residues by mixing 200 μL of a methanol/water solution (8:1 v/v), and the supernatant was collected after centrifugation. Both supernatants were then combined and centrifuged for 5 min, after which the final supernatant was transferred to a new Eppendorf tube. Samples were air-dried with a heat block set at 30 °C for 24 h. The derivatization protocol was first performed by adding 50 μL of 2% methoxyamine HCL to pyridine (Sigma-Aldrich, Taufkirchen, Germany), after which samples were incubated for one hour at 37 °C. Following incubation, 100 μL of N-methyl-N-(trimethylsilyl)-trifluoroacetamide (MSTFA) with 1% trimethylchlorosilane (TMCS) (Sigma-Aldrich, Germany) was added, and the mixture was incubated for 30 min at 70 °C. Samples were then centrifuged for 1 h at room temperature at 12 000 rpm and transferred to GC-MS glass vials. All additional analytical standards were air-dried and derivatized in a similar manner. Three replicates were used for each optimization step.
The GC-MS analysis was performed using an Agilent Technologies 5973N GC/MS (USA) equipped with an HP-5MS capillary 54 column (30 m × 0.250 mm i.d.; 0.25 μm film thickness; Agilent J&W Scientific). One microliter of the derivatized samples was injected, using helium as the carrier gas at a constant flow rate of 1.1 mL/min. The temperatures at the inlets and the MS source were 250 and 230 °C, respectively. The oven temperature was maintained at 75 °C for 4 min before being raised to 280 °C at a rate of 4 °C/min and maintained for 1.56 min.
The data processing and metabolite identification were carried out. The GC-MS data were obtained using the GC/MSD Chemstation software (Agilent). Following acquisition of the total ion chromatogram, the mass spectra were determined. The National Institute of Standards and Technology (NIST) 08 mass spectrum library was used to identify the obtained metabolite peaks, and the extracted compounds were aligned and normalized using the internal standard ribitol. For feature identification, peaks with a similarity index greater than 80% were considered. The Human Metabolome Database (HMDB) was used to identify metabolites of microbial origin.
Statistical Analysis
Statistical analyses were performed using the GraphPad Prism analysis software (Version 8, California, CA, USA). Comparisons between animal groups were performed via one-way ANOVA followed by Tukey's t-test. The alpha level for determination of statistical significance was set at 0.05. A heatmap at the genus level was generated using GraphPad Prism (Version 8, CA, USA). The proportions of genera in every group were determined using Microsoft Excel 2010.
Results
Improvement in Intestine Histology
Following 8 weeks of dietary intake, it was noted that the intestines of the control rats exhibited a normal tubular structure of the duodenum, characterized by concentric layers (Figures 1A and 2A). However, the intestinal samples obtained from HFD-fed rats showed severe damage in the villi structure, with loss of the epithelium and normal structure of the lamina propria. In addition, there was an increase in inflammatory cell infiltration, combined with an increase in the thickness of both the submucosa and the muscularis propria. Moreover, the serosa layer was found to be damaged (Figures 1B, C and 2B). Intestinal tissues obtained from rats in the HFD + MPGethanolE (25 and 50 mg/kg) groups showed improvements in the villi structure, epithelium, and lamina propria, as well as the thickness of the submucosa and muscularis propria (Figure 1C and D). Furthermore, the intestinal tissues obtained from rats fed with HFD + MPGethanolE (100 mg/kg) and HFD + MPG powder (10%, 20%, and 30%) showed a normal tubular duodenal structure, with normal layers and villi structure (Figures 1E, F and 2D–F).

Intestinal histology following HFD + MPGethanolE intake. (A) Control rat sample, showing the normal structure of the duodenum with a tubular structure of concentric layers. The mucosa (M) lines the lumen and is formed into villi (V) and intervening crypts (C). The villi consist of epithelium (E) supported by the lamina propria (LP). Beneath the mucosa lies the submucosa (SM) and the muscularis propria (MP). The adventitia (A) is the outer layer, which is referred to as the serosa (S) in those parts of the intestine covered by mesothelium. (B) HFD-treated rat sample, showing severe damage in the V structure (long black arrow) with the loss of E and the normal structure of the LP, as well as increased inflammatory cell infiltration (short thin black arrow), increased thickness of the SM and MP (arrowhead), and damaged S (short thick arrow). (C) HFD + MPGethanolE (25 mg/kg)-treated rat sample, showing an improvement in the V, E, and LP structures. However, increased numbers of damaged V were dominant (thick black arrow), with increased thickness of the SM and MP (arrowhead). (D) HFD + MPGethanolE (50 mg/kg)-treated rat sample, showing a normal tubular duodenal structure (thin black arrow) with little damage in the V structure (thick black arrow). All other layers look normal. (E, F) HFD + GPMethanolE (100 mg/kg)-treated rat samples, showing normal V structure (thin black arrow) and normal duodenal layers.
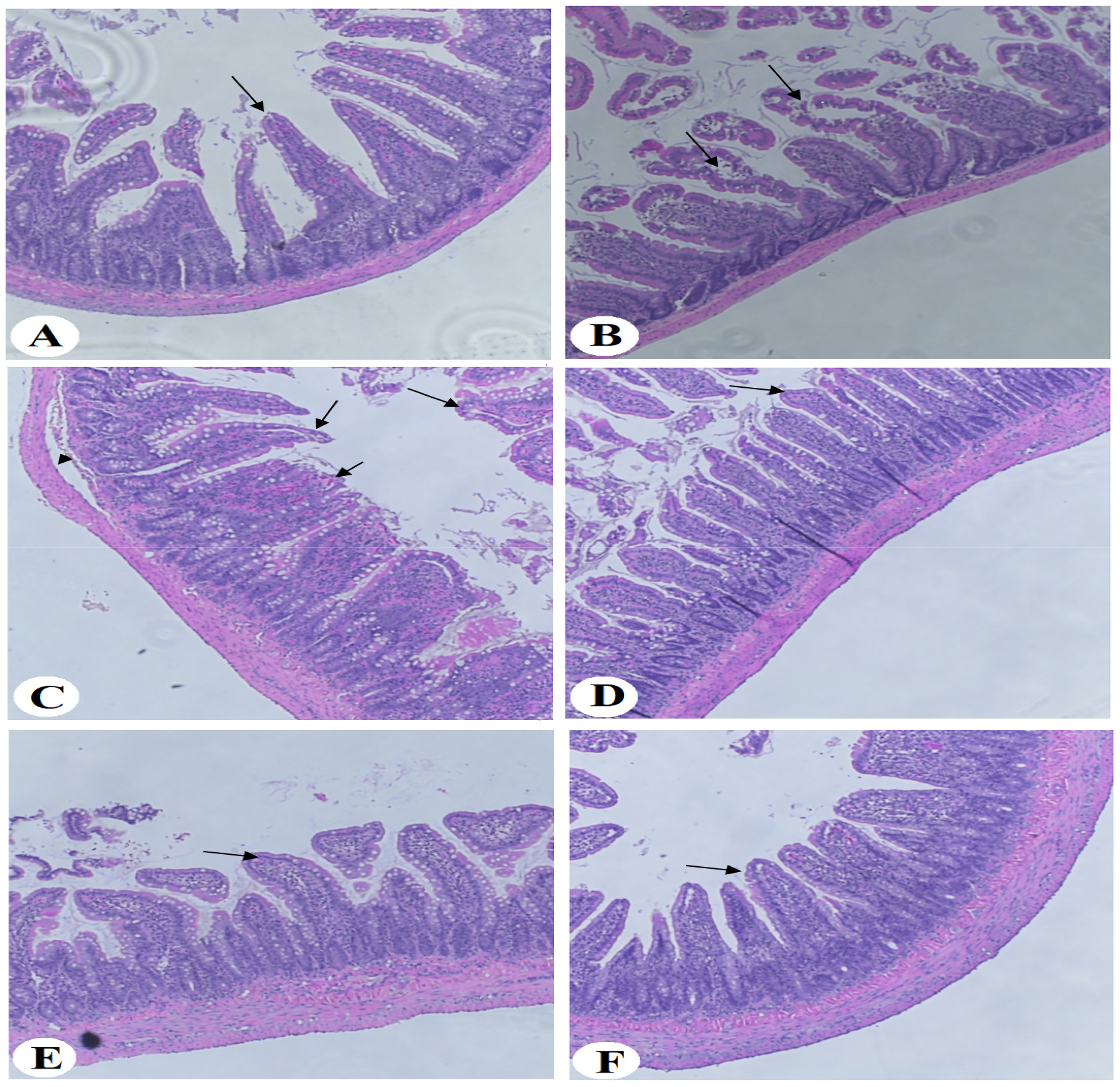
Intestinal histology following HFD + MPG powder intake. (A) Control rat sample, showing the normal structure of the duodenum tubular structure of concentric layers. All layers, including the villi, appear normal. (B, C) HFD-treated rat samples, showing some normal villi but severe damage in most of the other villi (V) structures (long black arrows) and detached SM layer (arrowhead). (D–F) Samples from rats treated with HFD + MPG powder (10%, 20%, and 30%, respectively), showing normal tubular duodenal structure with normal layers and V structure (thin black arrow).
Fecal pH Changes
The pH values of the fecal samples were found to range from 6.5 to 4.5, with statistically significant differences between groups (p < .05) (Figure 3A and B). The lowest pH value was found in the HFD + MPGethanolE (100 mg/kg) group, while the highest value was observed in the control group. In the HFD + MPGethanolE groups, fecal pH was significantly decreased, compared with that of the HFD control group (p < .05), at all tested doses (25 mg/kg, pH 6.1 ± 0.1; 50 mg/kg, pH 5.8 ± 0.1; 100 mg/kg, pH 4.5 ± 0.1), with the highest dose showing the most significant reduction. Fecal pH was also significantly decreased in the HFD + MPG groups compared with the HFD control (p < .05), in a dose-dependent manner (10%, pH 6.3 ± 0.1; 20%, pH 5.5 ± 0.1; 30%, pH 4.8 ± 0.1), with the 30% diet showing the lowest pH value. These results indicate significant acidification of the gut environment with increasing supplementation of millet grain and ethanolic extract.
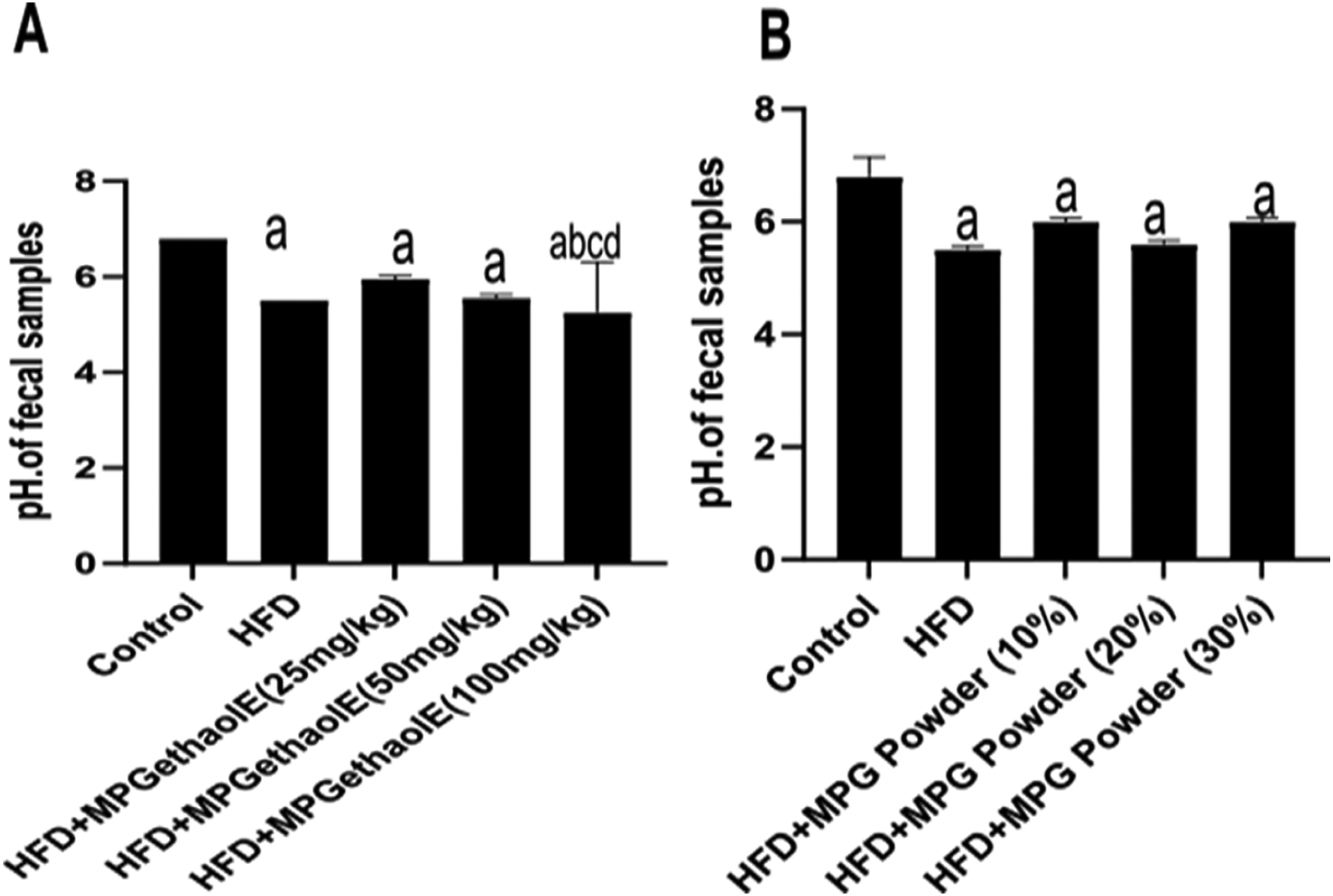
Fecal pH changes in rat groups. Data were analyzed via one-way ANOVA followed by post hoc Tukey's t-test, are presented as mean ± SD, and were considered significantly different at p < .05. (A) HFD + MPGethanolE and (B) HFD + MPG at different concentrations (10, 20, and 30%). a: significantly different versus the control rats; b: significantly different versus HFD; c: significantly different versus HFD + MPGethanolE (25 mg/kg) or MPG powder (10%); d: significantly different versus HFD + MPGethanolE (50 mg/kg or MPG powder (20%). No significant differences in pH values were observed between the control and both diet regime patterns. Both HFD + MPGethanolE and HFD + MPG powder exhibited similar pH values (5.7-5.8), compared with 6.5 for the control, except for HFD + MPGethanolE (100 mg/kg), which showed the lowest pH value of 4.5.
Gut Microbiota Composition Diversity
The intestinal microbiota composition in all groups is shown in Figure 4A–F, while the gut bacterial composition at the family and phylum levels is shown in Figure 5B and C. Firmicutes and Verrucomicrobia revealed a significant difference between the treated groups and the HFD or control groups (p < .05); in contrast, Actinobacteria showed no significant difference between the HFD and treated groups (p > .05). The HFD + MPGethanolE (100 mg/kg), HFD + MPG powder (10%), and HFD + MPG powder (20%) groups showed significant (p < .05) increases in Akkermansia muciniphila (19.01%, 18.45%, and 20.39%, respectively, vs 0.00% in the control group) and marked increases in Lactobacillus spp. (31.71%, 32.89%, and 33.09%, respectively, vs 20.39% in the control group) (p < .05). Notably, the highest abundance of Lactobacillus spp. was found in the HFD + MPGethanolE (25 mg/kg) group. The bacterial community profiles are illustrated using a heatmap, which shows the differences in the abundances of three key genera. At the genus level, the HFD group showed a relatively low abundance of Lactobacillus spp. Similarly, Akkermansia muciniphila was found to be absent in the control, HFD, HFD + MPGethanolE (25 mg/kg), HFD + MPGethanolE (50 mg/kg), and HFD + MPG powder (30%) groups, while Clostridium spp. was absent in all experimental groups (Figure 5).

Relative abundance of bacteria in animal groups. Relative abundances of Bifidobacterium spp. (A,B), Lactobacillus spp. (C, D), and Akkermansia muciniphila (E, F). Bifidobacterium spp. was noted to be present in high concentrations throughout all experimental groups. Lactobacillus spp. was absent in the HFD group, and was mainly increased under the HFD + MPGethanolE (25 mg/kg) treatment. Akkermansia muciniphila was noted to be present only in the HFD + MPG powder (10% and 20%) and in the HFD + MPGethanolE (100 mg/kg) groups. Data were analyzed via one-way ANOVA followed by post hoc Tukey's t-test, are presented as mean + SD, and were considered significantly different at p < .05 a: significantly different versus control rats; b: significantly different versus HFD; c: significantly different versus HFD + MPGethanolE (25 mg/kg) or MPG powder (10%); d: significantly different versus HFD and HFD + MPGethanolE (50 mg/kg) or MPG powder (20%).
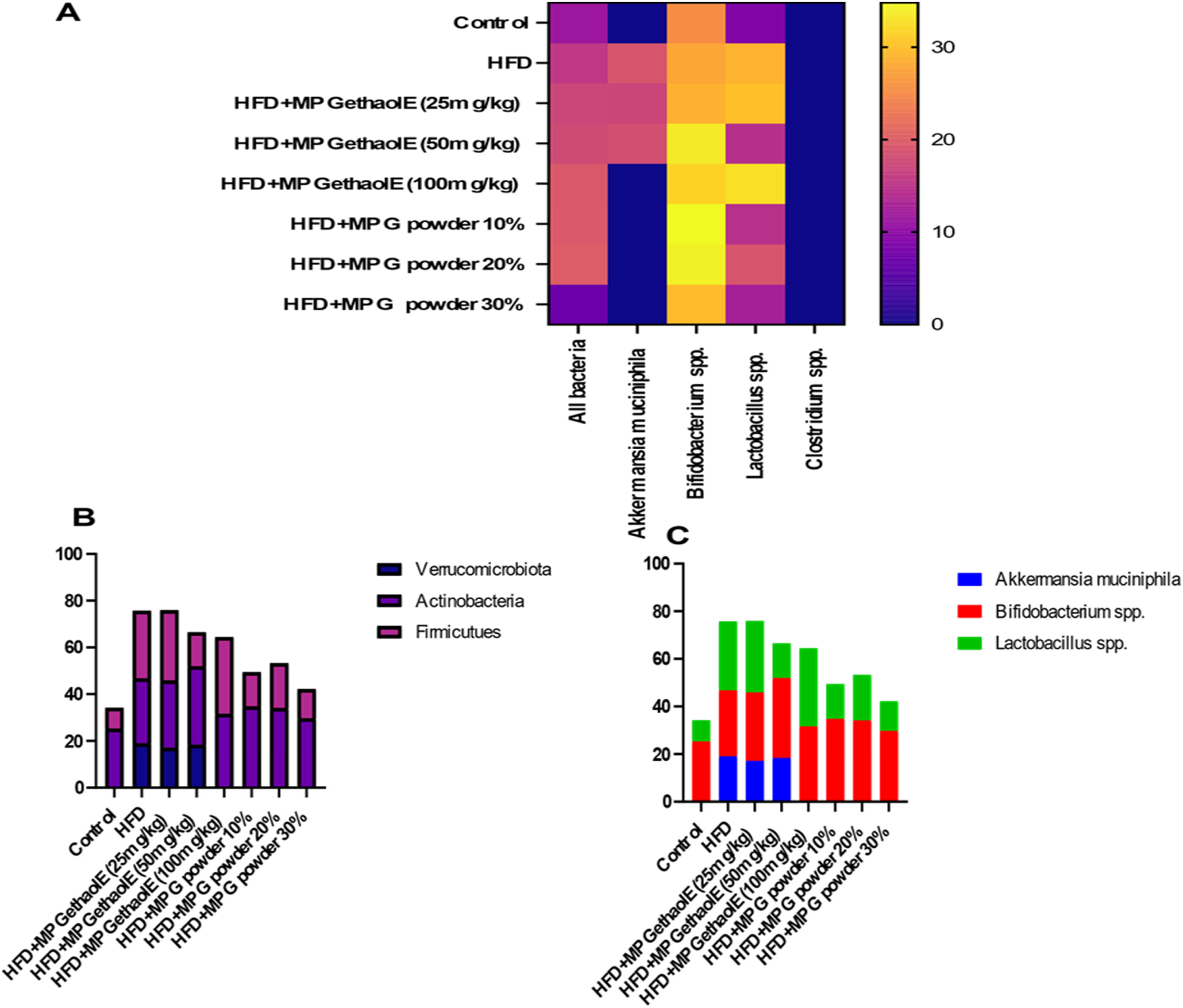
Heatmap of the gut microbiota in all animal groups. (A) Gut microbiota composition at the phylum level; (B,C) gut bacterial composition at the family level in response to intake of millet grain and its ethanolic extract at different dosages. At the family level, beneficial bacteria (Bifidobacterium spp. and Lactobacillus spp.) were dominant, with the significant appearance of Akkermansia muciniphila—particularly in the groups treated with ethanolic extract at 10, 20, or 30%—indicating the ability of millet grain to act as a prebiotic agent and restore gut homeostasis.
Alteration of Metabolites in Serum
Initially, a two-step solvent extraction procedure for serum samples was performed utilizing acetonitrile/methanol and methanol/water, followed by trimethylsilylation derivatization, even though methanol by itself is considered an effective solvent for extracting metabolites from body fluids. Metabolomics profiling was performed via untargeted GC-MS analysis. The metabolomic alterations induced in sera via dietary interventions of millet whole grains and millet extracts in HFD rats were thereby obtained, and the results are depicted in Table S1. A total of 74 metabolites—including lipids, medium-chain fatty acids, fatty acid esters, other esters, ether, fatty aldehyde, carboxylic acid derivatives, amino acid derivatives, and metabolites of bacterial origin and other metabolites—were revealed in the present study. To conduct differential metabolomics analysis among the study groups, principal component analysis (PCA) was performed and a hierarchical clustering analysis heat map using normalized (auto-scaled) peak intensity (concentration) data was obtained. Considerable differences between the metabolomics profiles characterizing the control, MPGethanolE, and MPG groups were observed on the PCA score plots (Figure 6), with each group showing clearly defined distribution zones. The principal components of the metabolomics response significantly differed between the control, HFD, MPGethanolE, and MPG groups, with the changes due to administration of millet and its extract primarily affecting the principal components. The distribution of serum metabolites between all groups was also assessed via hierarchical clustering. Serum metabolite concentrations associated with different doses of MPG showed significantly different frequency patterns between the groups (Figure 7).

PCA score plot for serum metabolomic alterations. Considerable differences between the metabolomic profiles of the control, MPGethanolE, and MPG groups can be observed on the PCA score plots. 1–8 denote the animal groups: 1, control; 2, HFD; 3, HFD + MPGethanolE (25 mg/kg); 4, HFD + MPGethanolE (50 mg/kg); 5, HFD + MPGethanolE (100 mg/kg); 6, HFD + MPG (10%); 7, HFD + MPG (20%); 8, HFD + MPG (30%).

Serum metabolite distribution heatmap between all groups. Serum metabolite concentrations associated with different doses of MPG revealed different frequency patterns between groups.
Discussion
Millet grains and polyphenols have been shown to have prebiotic-like effects on the gut microbiota. 24 Therefore, in this study, we tested the impacts of millet grains and their ethanolic extract on the gut microbiota in obese rats fed a high-fat diet. Ultimately, our findings show that the gut flora composition changed after treatment with MPG and MPGethanolE, when compared with that of the control group. Our results emphasize the ability of millet to enhance the intestinal and villous architectures, and that the potential prebiotic-like effects of both interventional diets mediates these changes.
In this study, the administration of an HFD led to the accumulation of lipid droplets in the rat intestinal mucosa, associated with significant changes in organ architecture characterized by alterations in the typical villus conformation and an increase in inflammatory cells (Figures 1 and 2). This result is in line with the study of Sferra et al, 41 who emphasized that an HFD not only can cause hepatic steatosis but also intestinal steatosis, which may induce inflammation via the generation of reactive oxygen species (ROS) in response to chronic HFD consumption. 42 Some Lactobacillus strains cannot resist the oxidative stress resulting from an HFD, 43 which could justify their absence in HFD rats in the present study. Bifidobacterium species were found in all groups—including the control group—demonstrating their capacity to endure oxidative stress and alleviate the intestinal damage caused by ROS formation, mediated by the stimulation of significant antioxidant activity in the gut and production of antioxidant enzymes. 44
Other studies have also suggested that excessive fat consumption is linked to considerably higher levels of lipopolysaccharides (LPSs) in the blood,45–47 and that the diet can induce mast cell activation in the intestinal mucosa, resulting in the production of histamine and other inflammatory mediators that can affect intestinal permeability, thus affecting the gut microbiota composition. Interestingly, LPSs and other pathogen- or damage-associated molecular patterns have been shown to increase in the circulation under conditions of increased gut permeability, thus stimulating the immune system and cytokine production,48,49 as well as inducing inflammation. 50 In our previous study, 27 low LPS levels were observed in all treated groups, which could be interpreted as being due to the high abundance of commensal bacteria—particularly Bifidobacterium spp., Lactobacillus spp., and Akkermansia muciniphila—thus maintaining the gut balance (Figure 4).
Another important observation in this study is that the consumption of millet grains and ethanolic extract improved the intestinal histology in rats in a dose-dependent manner (Figures 1 and 2). Akkermansia muciniphila can benefit the host intestinal epithelial structure by increasing the rate of mucus turnover via the production of SCFAs from degraded mucin—the preferred energy source for the host epithelium, which synthesizes and secretes mucus. 51 The observed improvement in intestinal architecture may also be attributed to the phytochemical constituents of millet, which have been shown to have high antioxidant activity52,53 and to ameliorate intestinal oxidative stress in both humans and mice (Shapiro et al, 2007). 54 The antioxidant characteristics of phenols are attributable to their ability to scavenge free radicals and modulate antioxidant enzymes, 42 resulting in repair of the intestinal structure.
The administration of finger millet (whole grain or bran) has been shown to improve inflammatory biomarkers in mice fed a high-fat diet for 12 weeks via decreasing Interleukin-1 beta (IL-1β) concentrations and adipose inflammation, as well as decreasing the expression of genes associated with inflammation such as nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kB), macrophage inflammatory protein-1 alpha, TNF-a, and monocyte chemoattractant protein-1. 55 These changes could be driven by the fibers present in millet, which may directly influence the gut immune system by inhibiting the proinflammatory Toll-like receptor (TLR) 2-1 pathway in human dendritic cells (DCs) and mouse macrophage cells. 11
Many dietary nutrients, including oligosaccharides and simple sugars, act as substrates for microbial metabolism in the host, and influence fecal pH due to acid generation through fermentation. In gastrointestinal disorders such as colorectal cancer, inflammatory bowel disease, and constipation, the colonic pH and abundances of gut microbes are typically found to depart from normal levels. 56 The major metabolic end-product of carbohydrate-fermenting bacteria, such as Lactobacillus or Streptococcus, is lactic acid. Notably, lactic acid production increases the acidity of feces. In a previous work, 57 culturing Lactobacillus using broccoli puree decreased the pH from 6.51 to 4.28 via stimulation of glycolysis; as such, glycolysis acid products may contribute to acidification of the intestinal contents. A decrease in pH (eg, to below a pH of 5.5) is very beneficial in the gut, as it decreases the proliferation of pathogenic bacteria such as Bacteroides spp. and Escherichia coli. 14 This is in agreement with our study, with the decrease in fecal pH (Figure 3) suggesting the production of some acids after either fiber or polyphenol fermentation. 58 This, in turn, led to the absence of pathogenic Clostridium spp., as well as the persistence of beneficial Bifidobacterium spp., Lactobacillus spp., and Akkermansia muciniphila (Figures 4 and 5). The abundance of Bifidobacterium remained stable throughout the experiment. The same growth level was observed pre- and post-treatment with all diet inventions; Lactobacillus was also noted as being present in all animal groups, although to a lesser extent in the control group compared with the other groups, and disappeared completely in the HFD group. Akkermansia muciniphila showed a different pattern of growth, being absent in all groups except the HFD + MPG (10% and 20%) and MPGethanolE (100 mg/kg) groups. These findings are in line with Metzler-Zebeli et al, 59 who reported a decrease in the fecal pH in pigs and an increase in Lactobacillus spp. after feeding pigs with resistant starch, leading to significant production of SCFAs. Moreover, tea polyphenols have been found to increase fecal acidity and modulate gut flora, which was mainly attributed to the fermentation of polyphenols by the gut microbiota and the generation of SCFAs as end-products. 60 Overall, an acidic fecal pH was found to induce beneficial modifications in the gut bacterial composition, with a remarkable increase in the commensal bacterial species and an absence of pathogenic bacteria.
In the present study, dietary supplementation with pearl millet whole grain (MPG) and its ethanolic extract (MPGethanolE) induced measurable shifts in key microbial taxa: the relative abundance of Akkermansia muciniphila rose to 19.01%, 18.45%, and 20.39% in the three treatment groups, compared with 0.00% in the HFD control; meanwhile, Lactobacillus spp. increased to 31.71%, 32.89%, and 33.09% versus 20.39% in the control (p < .05). These increases suggest a clear prebiotic-like effect of millet and its extract, increasing the abundance of beneficial bacteria known to be linked with improved gut barrier function and metabolic regulation; for example, Akkermansia has been shown to correlate inversely with obesity and metabolic dysregulation in humans. 61 The substantial increase in Lactobacillus further supports the notion that the components of millet favor the proliferation of probiotic-type microbes.
Our results also indicate the dominance of Firmicutes at the phylum level. The microbiota of the millet ethanolic extract (25 mg/kg) group contained the highest proportion of Lactobacillus spp., further suggesting that the millet extract has a prebiotic-like effect. This concurs with the findings of Corrêa et al, 62 who demonstrated the role of phytochemicals in stimulating the growth of beneficial bacteria (eg, Lactobacillus spp.) and hindering the proliferation of pathogenic bacteria (eg, Clostridium spp.). Although the small intestine does not absorb 90%–95% of polyphenols, microbes in the large intestine can break down these compounds, thus modulating the gut bacterial population 63 and thereby alleviating dysbiosis, highlighting the potential of polyphenols as therapeutic and nutraceutical agents. 64 The blooming of Lactobacillus has been observed to benefit the host in different ways, such as limiting pathogen development through the generation of acetic acid and lactic acid, which, in turn, reduces pathogenic bacterial growth or epithelial invasion, increases intestinal barrier function, and modifies the activity of the immune system. 65 Moreover, an in vitro study 66 has indicated that Lactobacillus also exhibit adhesiveness features—thus preventing pathogenic bacteria from adhering to host cells—which may explain the hindered growth of pathogenic Clostridium spp. in our samples.
Decreased levels of Akkermansia muciniphila have been reported to correlate with increased energy intake and adiposity, and to be associated with a diet low in fiber, 51 which was similarly observed in the group of rats fed an HFD in this study. In contrast, the groups of rats fed with higher doses of millet grains and millet ethanolic extract showed increases in the abundance of Akkermansia muciniphila, which could be explained by their high fiber and phenol contents.17,23 Fibers are reduced by colonic microorganisms to simple compounds that can more easily be taken into the circulation and exhibit antioxidant activities in the gut environment. 67 Cranberry polyphenols have also been shown to increase the quantity of Akkermansia muciniphila, helping to reduce obesity, insulin resistance, and intestinal inflammation. 68 Another study 69 revealed that treatment with oral inulin for 45 days significantly boosted the abundance of Akkermansia muciniphila. In addition, the administration of aloe and oyster extract had a considerable anti-inflammatory effect, which was linked to the increased abundance of Akkermansia muciniphila. 70 This means that future dietary treatments employing compounds comprising mainly phenols and fibers to enhance the growth of Akkermansia muciniphila, have high potential for weight control in obese individuals and for regulating the gut bacterial composition. 71
In an in vitro study conducted by Connolly et al, 72 the abundance of Bifidobacterium spp. was significantly increased during thick-oat-flake fermentation, which was attributed to the β-glucan content. Conversely, our interventional diet—particularly that comprising millet as whole grains—negatively affected Bifidobacterium, even though millet contains a considerable amount of β-glucan fiber. 73 This could be due to the low dosage intake, as well as variable effects in relation to the native microbiota. 74 In this context, it would have been beneficial if the amount of β-glucan from the Saudi variety of millet had been quantified. More research is therefore necessary to evaluate and compare the effects of PM grains on the gut microbiota due to differences in the amount of β-glucan fiber—the main influencer of a healthy microbiota. 72 The amount of β-glucan can be affected by several factors, including climatic conditions and the quality of the agricultural soil used for cereal cultivation. 73
The metabolomic changes observed in this study correspond to shifts in the gut microbiota. An increased abundance of Akkermansia likely promotes mucin turnover and SCFA production, which can improve host lipid and glucose metabolism. Meanwhile, higher Lactobacillus levels may influence amino acid and lipid metabolites through the fermentation of millet fibers and polyphenols. Together, these microbial changes help to explain the differences in serum metabolites—particularly highlighting the gut–metabolome connection, which has been underexplored in previous research on millet. 75
Previous studies have shown that millet and its components increase the abundance of beneficial bacteria such as Lactobacillus, Bifidobacterium, and Akkermansia, improving the gut microbiota composition and metabolic health in models of diabetes and obesity. Our study adds novel data by quantifying these changes and linking them to metabolomic shifts. These findings suggest that millet could help to reduce the risk of metabolic syndrome by modulating the gut microbiota and metabolic factors. In this regard, further human trials are needed to confirm effective doses for and the health benefits of millet. 76
In addition, the results of the present study indicate that serum GC–MS-based metabolomics is a useful approach for metabolite screening and distribution in response to grain millet intervention diets and, in turn, to changes in gut bacterial composition, enabling the selection of metabolites as potential biomarkers reflecting the status of the gut microbiota.
Conclusions
This study demonstrated that pearl millet (MPG) and its ethanolic extract (MPGethanolE) improve the composition of the gut microbiota, significantly increasing the abundances of Akkermansia muciniphila and Lactobacillus spp., in addition to altering serum metabolomic profiles in high-fat-diet-induced obese rats. These changes are accompanied by improved intestinal architecture, suggesting a prebiotic-like effect. The study's limitations include the use of an animal model and the lack of direct measurement of microbial metabolites such as SCFAs. Future studies should explore the underlying mechanisms, dose–response relationships, and relevant effects in human clinical trials to evaluate the potential of millet for preventing or managing metabolic syndrome.
Study Limitation
Despite its promising findings, this study has several limitations that warrant consideration. First, the experiment was conducted in a rat model which, although widely used for metabolic and gut microbiome research, may not fully replicate human physiological responses. Therefore, the results should be cautiously extrapolated to human populations. Second, the study duration may not have been sufficient to observe long-term metabolic adaptations and stable shifts in the gut microbiota. Third, the analysis focused on selected bacterial taxa using qPCR rather than comprehensive high-throughput sequencing approaches, which would have enabled a more complete characterization of microbial diversity and functional capacity. Additionally, serum metabolomics was performed using GC-MS only, and the absence of targeted SCFA quantification limits interpretation of the results regarding fermentation-derived metabolites. Inflammatory markers, gut barrier integrity, and metabolic hormones were also not assessed, which can provide further mechanistic insight regarding obesity and gut health. Finally, only male rats were used, preventing the evaluation of potential sex-specific responses. Future research should incorporate long-term interventions, advanced multi-omics approaches, SCFA profiling, inflammatory markers, and human clinical studies to validate and expand the presented findings.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X261431451 - Supplemental material for Impact of Millet Grain Diets on High-Fat-Diet-Induced Obese Rats: Gut Microbiota and Metabolomic Analysis
Supplemental material, sj-docx-1-npx-10.1177_1934578X261431451 for Impact of Millet Grain Diets on High-Fat-Diet-Induced Obese Rats: Gut Microbiota and Metabolomic Analysis by Nadiah S. Alzahrani, Ghedeir M. Alshammari, Abu ElGasim A. Yagoub, Rajapandiyan Krishnamoorthy, Mohammad A. Alshuniaber, Nadine M. S. Moubayed and Mohammed Abdo Yahya in Natural Product Communications
Footnotes
Acknowledgments
The authors extend thanks to the Ongoing Research Funding Program, (ORF-2026-84), King Saud University, Riyadh, Saudi Arabia for support this work.
Ethical Considerations
The Research Ethics Committee at King Saud University, Riyadh, Saudi Arabia, approved the experimental protocol (Ref. No.: KSU-SE-21-49).
Statement of Informed Consent
There are no human subjects in this article, and informed consent is not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Ongoing Research Funding Program, (ORF-2026-84), King Saud University, Riyadh, Saudi Arabia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
