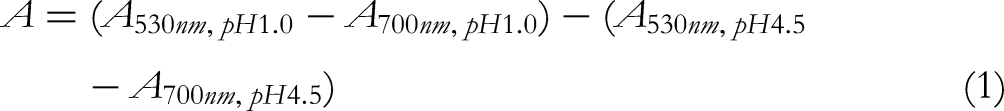Abstract
Objectives
Female obesity may be improved by the consumption of a high-polyphenolic-rich diet. Lycium ruthenicum Murray is a renowned edible plant, the fruit of which is abundant in anthocyanins and exhibits many biological activities. This study aimed to investigate the potential impact of anthocyanins derived from Lycium ruthenicum Murray (ACN) intervention on improving obesity by regulating the gut microbiota and bile acids (BAs) metabolism in high-fat diet (HFD)-induced female mice.
Methods
A total of 32 C57BL/6J female mice were divided into four distinct groups: the Ctrl group (fed a normal diet), Ctrl + ACN group (fed a normal diet plus 8 mg/mL extract of ACN), HFD group (fed a high-fat diet), and HFD + ACN group (fed a HFD plus 8 mg/mL extract of ACN).
Results
The findings showed that ACN significantly reduced the body weight, periovarian adipose mass, and adipocyte diameter, ameliorated lipid accumulation in the liver, and lowered the serum total cholesterol and low-density lipoprotein (LDL-C) levels in HFD-induced female mice. In addition, ACN exhibited a reversal of gut microbial dysbiosis in HFD-fed female mice, such as by enhancing the quantity of Lactobacillus and Allobaculum and reducing the abundance of Blautia and Faecalibaculum. Moreover, the results of fecal BAs showed that ACN led to a decrease in the ratio of primary to secondary BAs, mainly attributed to decreased levels of primary BAs, including CA, CDCA, αMCA, and HCA in HFD-induced female mice. Further analysis revealed that ACN may exert its anti-obesity effect by increasing the relative abundance of Lactobacillus_ jonsonii and Lactobacillus_reuteri within the gut and subsequently affecting the metabolism of fecal HDCA and GUDCA.
Conclusion
These results indicated that ACN effectively inhibits HFD-induced obesity in female mice by regulating gut microbiota-related BA metabolism.
Introduction
The escalating prevalence of overweight and obesity has become an increasing public health problem worldwide. 1 The prevalence of overweight and obesity in women was significantly higher compared to men, as reported by the World Health Organization (WHO). 2 Overweight and obesity can lead to other diseases such as insulin resistance, type 2 diabetes mellitus, hepatic steatosis, and cardiovascular diseases. 3 In addition, overweight or obese females have a significantly heightened risk of adverse pregnancy outcomes such as gestational diabetes mellitus and preeclampsia. 4 Overweight and obesity are multifactorial diseases, with a focus on genetic and environmental determinants caused by the lack of physical exercise, excess energy intake, and a sedentary lifestyle. 5 Therefore, it is crucial to conduct further investigations to intervene and prevent the occurrence of overweight and obesity, particularly among females.
In recent years, the gut microbiota has been considered a crucial factor in the occurrence and development of obesity and related metabolic diseases. 6 The physiological functions of the gut microbiota encompass a wide spectrum, including metabolic regulation, digestion of food ingredients, synthesis of new elements, and gut immune system regulation, thus affecting the host metabolism at multiple levels. 7 Therefore, high-fat diet (HFD) may alter gut microbiota composition, resulting in obesity and related diseases. Bile acids (BAs) constitute a class of metabolites produced by the liver and transformed by the gut microbiota. These have attracted extensive attention due to their important roles in the occurrence and development of obesity and related metabolic diseases.8,9 Diet and the gut microbiota interact with BAs pools through biotransformation reactions, thereby influencing the hydrophobicity, toxicity, and regulatory effects of BAs. 10
There are numerous treatment options available for overweight and obesity, including dietary interventions, prescription weight-loss drugs, and bariatric surgery. However, compared to weight-loss drugs and bariatric surgery, dietary intervention is often considered more convenient, safe, and sustainable. 11 Recently, polyphenolic compounds such as anthocyanins have shown considerable potential anti-obesity effects through different mechanisms. Anthocyanins are widely present in various plants, including sour cherry, cranberry, black beans, blueberry, sweet cherry, strawberry, and pomegranate.12,13 Anthocyanins have been reported to exert anti-obesity effects by improving gut microbiota and different mechanisms.
Lycium ruthenicum Murray, a member of the Lycium genus within the family, is a well-known edible plant containing many anthocyanins. 14 The bioactivities of anthocyanins from Lycium ruthenicum Murray (ACN), especially antioxidant and anti-inflammatory activities, have been widely reported, and they can change the composition of gut microbiota and reduce the risk of chronic diseases, such as obesity. 15 In previous studies, the intervention of ACN was found to enhance intestinal barrier integrity and decrease systemic inflammation in male HFD-induced mice. 16 Meanwhile, ACN attenuated HFD-induced gut microbial metabolic dysbiosis and confirmed inhibition of pancreatic lipase activity in male mice.17,18 However, the role of ACN in the prevention of overweight and obesity in females is not fully understood. Therefore, this experiment aimed to investigate the preventive effects of ACN induced by HFD in female obese mice. In this study, we investigated whether ACN from Lycium ruthenicum Murray exerts its anti-obesity effect in female mice by regulating gut microbiota and fecal BA metabolism.
Results
ACN Reduced the Body Weight and Fat Accumulation in HFD-Fed Female C57BL/6J Mice
To determine the preventive effect of ACN on HFD-induced female overweight and obesity in females, female mice were fed with the normal diet or HFD and with or without the adjunct of an ACN solution. At the endpoint of the experiment, it was observed that HFD significantly increased the body weight, liver index, and the weights of mesenteric, perirenal, back, and periovarian fats in female mice compared to those on a normal diet. Following the intervention of ACN for 32 weeks, compared with the Ctrl group, the body weight in the Ctrl + ACN group remained constant. However, the female mice in the HFD + ACN group exhibited a significant decrease in body weight and periovarian fat compared to those in the HFD group (Figure 1a, c, d). In addition, there was no difference in food intake between the Ctrl and Ctrl + ACN group, nor between HFD and HFD + ACN group, suggesting that the preventive effect of ACN on female obese mice induced by HFD was not due to reduced food intake (Figure 1b). These results suggested that the ACN reduced body weight in female mice could be attributed to a reduction in adipose tissue weights.

ACN reduced HFD-induced body weight in female C57BL/6J mice. (a) Body weight, (b) food intake, (c) liver index, and (d) fat weight. Data were expressed as mean ± SEM, n = 7-8 mice per group. * indicates significant differences between Ctrl and HFD groups, # indicates significant differences between HFD and HFD + ACN groups, #P < 0.05, **P < 0.01. HFD, high-fat diet.
ACN Reduced the Levels of Serum Lipid in HFD-Fed Female C57BL/6J Mice
The biochemical parameters of the serum obtained from mice at the end of the experiment are presented in Figure 2. HFD-fed female mice exhibited a significant increase in the concentrations of total cholesterol (TC) and low-density lipoprotein (LDL) compared to the Ctrl group. The intervention of ACN decreased serum lipids in HFD-fed female mice, as indicated by significant reductions in TC and LDL concentrations. Interestingly, there were no significant differences in the levels of TC and LDL between the Ctrl and Ctrl + ACN groups. In addition, serum triglyceride (TG) and high-density lipoprotein (HDL) levels showed no significant differences among the four groups. These results indicated that ACN can lower serum TC and LDL levels in HFD-fed female mice.

ACN reduced HFD-induced serum lipid in female C57BL/6J mice. (a) Serum T-CHO, (b) serum TG, (c) serum LDL-C, and (d) serum HDL-C. Data were expressed as mean ± SEM, n = 6 mice per group. *P < 0.05, **P < 0.01. TG, triglyceride; HDL-C, high-density lipoprotein; LDL-C, low-density lipoprotein; HFD, high-fat diet.
ACN Ameliorated the Fat Accumulation in HFD-Fed Female C57BL/6J Mice
In comparison to normal diet-fed mice, the liver of HFD-fed female mice exhibited a greasy texture, round shape, and blunt edge, and these features were improved in the HFD + ACN group after ACN intervention (Figure 3a). Furthermore, histological analysis of the liver revealed that ACN intervention resulted in a reduction of hepatic steatosis and lipid droplet accumulation in the liver of HFD-induced female mice (Figure 3b). In addition, histological sections of the periovarian adipose showed a larger adipocyte diameter in the HFD group than that of the Ctrl group. In comparison with the HFD group, the adipocyte size was smaller in the HFD + ACN group (Figure 3c). In the same field of vision, the number of adipocytes in the HFD group was significantly reduced. At the same time, it increased considerably in the HFD + ACN group (Figure 3d). These results showed that ACN has a preventive effect on HFD-induced hyperlipidemia and obesity in female mice.

ACN ameliorated HFD-induced fat accumulation in female C57BL/6J mice. Photo images of H&E staining of liver sections were photographed at ×200 magnification. (a) Liver tissues, (b, c) H&E staining of liver and periovarian fat, and (d) adipocyte numbers of periovarian fat. Data were expressed as mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001. H&E, hematoxylin and eosin; HFD, high-fat diet.
ACN Regulated the Gut Microbiota in HFD-Fed Female C57BL/6J Mice
To evaluate the intervention of ACN on HFD-induced female mice, the 16S rRNA gene sequences of microbial samples isolated from feces were determined. Alpha diversity analysis provides insights into microbial communities’ richness, evenness, and diversity. Chao1 and ACE indices indicate community richness, while the Shannon and Simpson indices show evenness and diversity. No significant differences were observed between the ACE, Chao1, and Simpson values between the four groups. However, the HFD group significantly lowered the Shannon index compared to the Ctrl group. At the same time, there was no significant difference between Ctrl + ACN and HFD + ACN groups, suggesting that the HFD led to a decrease in the evenness and diversity of the bacterial community (Figure 4a-d). The beta-diversity analyses, including the nonmetric multidimensional scaling (NMDS), revealed a distinct clustering of intestinal microbe communities for each experimental group. This suggested that ACN intervention significantly ameliorated the HFD-induced gut microbiota dysbiosis in female mice (Figure 4e).

Effects of ACN intervention on gut microbiota in α and β diversities indices. (a-d) Indices of Shannon, Simpson, ACE, and Chao 1 and (e) NMDS plot of unweighted UniFrac data. Data were expressed as mean ± SEM, n = 7 mice per group. *P < 0.05, **P < 0.01. NMDS, nonmetric multidimensional scaling.
To further investigate the specific changes in bacterial communities, the relative abundance of the predominant phylum, genus, and species, especially taxa responding to ACN intervention, were compared among the four groups. At the phylum level, the majority of the relative abundance belonged to Firmicutes and Bacteroidota, followed by Verrucomicrobiota (Figure 5a). Moreover, compared to the Ctrl group, the HFD group had significantly reduced the abundance of Bacteroidota (Figure 5b) and significantly increased the abundance of Verrucomicrobiota (Figure 5c) in the feces of female mice. The relative abundance of Bacteroidota was decreased, while the relative abundance of Firmicutes was increased in the HFD + ACN group when compared with that of the HFD group (Figure 5d).

Effects of ACN intervention on gut microbiota structure at the phylum level. (a) Phylum-level taxonomic distributions of the microbial communities in feces, (b) the relative abundance of Bacteroidota, (c) the relative abundance of Verrucomicrobiota, and (d) the relative abundance of Firmicutes. Data were expressed as mean ± SEM, n = 7 mice per group. *P < 0.05, **P < 0.01.
At the genus level (Figure 6a), the relative abundances of Blautia, Faecalibaculum, Bacteroides, Romboutsia, and Akkermansia were significantly increased in the HFD group compared with the Ctrl group (Figure 6b-f). Interestingly, higher abundances of Lactobacillus (Figure 6g) and Allobaculum (Figure 6h) in the HFD + ACN group were observed. Meanwhile, ACN dramatically inhibited the relative abundances of Blautia and Faecalibaculum in HFD-induced female mice.

Effects of ACN intervention on gut microbiota structure at the genus level. (a) Genus-level taxonomic distributions of the microbial communities in feces and (b-h) the relative abundance of Blautia, Faecalibaculum, Bacteroides, Romboutsia, Akkermansia, Lactobacillus, and Allobaculum. Data were expressed as mean ± SEM, n = 7 mice per group. *P < 0.05, **P < 0.01.
At the species level (Figure 7a), the relative abundances of Lachnospiraceae_bacterium_28-4, Faecalibaculum_rodentium, Bacteroides_sartorii, Romboutsia_ilealis, and Akkermansia_muciniphila were more abundant in HFD group than in Ctrl group (Figure 7b-f). However, the HFD + ACN group was found to considerably decrease the abundances of Lachnospiraceae_bacterium_28-4, Faecalibaculum_rodentium (Figure 7b,c) and increase the abundances of Lactobacillus_johnsonii and Lactobacillus_reuteri (Figure 7g-h) compared with the HFD group. These results indicated that ACN significantly improves HFD-induced gut microbiota disturbances in female mice. In particular, ACN increased the abundance of beneficial bacteria such as Allobaculum, Lactobacillus_johnsonii, and Lactobacillus_reuteri in Firmicutes.
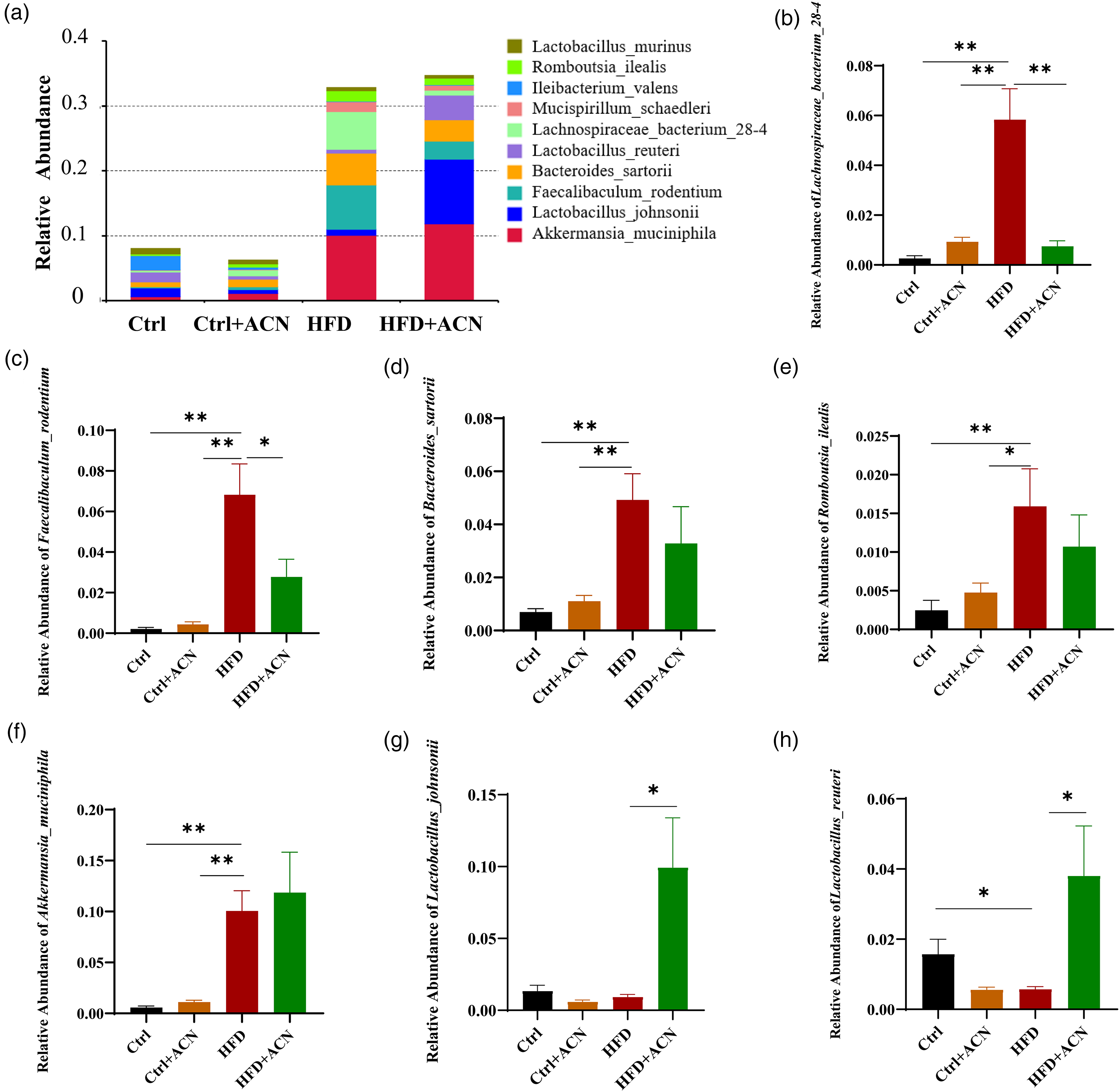
Effects of ACN intervention on gut microbiota structure at the species level. (a) Species-level taxonomic distributions of the microbial communities in feces and (b-h) the relative abundance of Lachnospiraceae_bacterium_28-4, Faecalibaculum_rodentium, Bacteroides_sartorii, Romboutsia_ilealis, Akkermansia_muciniphila, Lactobacillus_johnsonii and Lactobacillus_reuteri. Data were expressed as mean ± SEM, n = 7 mice per group. *P < 0.05, **P < 0.01.
ACN Altered the Levels of Fecal BAs Composition in HFD-Fed Female C57BL/6J Mice
BAs are important in obesity as signaling molecules. To investigate the BAs profile in four groups of mice, UHPLC-MS/MS was applied to acquire their fecal BAs profiles. PCoA results showed a clear separation between the Ctrl group and the HFD group. Simultaneously, an obvious result showed that the samples from HFD and HFD + ACN groups were remarkably separated, suggesting that ACN had a significant effect on fecal BAs metabolism of female mice induced by HFD (Figure 8a). The levels of fecal total BAs, primary BAs, and secondary BAs were all significantly increased in the HFD group compared to the Ctrl group. Notably, the levels of fecal primary BAs in the HFD + ACN group decreased significantly compared to the HFD group. Interestingly, there were no significant differences in the level of fecal total BAs, primary BAs, and secondary BAs between Ctrl + ACN and Ctrl groups (Figure 8b). These results revealed that ACN could decrease the levels of primary BAs induced by HFD in female mice (Figure 8c). Moreover, the ratio of primary to secondary BAs was significantly elevated in the HFD group compared with the Ctrl group, which was mainly manifested by increasing the levels of fecal primary BAs such as CA, CDCA, αMCA, βMCA, UDCA, and HCA (Figure 8d). However, the ratio of primary to secondary BAs in the HFD + ACN group was significantly lower than that in the HFD group, which was mainly due to reduction in the levels of CA, CDCA, αMCA, and HCA, and increase in the level of HDCA (Figure 8e). These results revealed that ACN from Lycium ruthenicum Murray altered the fecal BAs profiles, specifically reversing the increase in the ratio of primary to secondary BAs induced by HFD.

Effects of ACN intervention on fecal bile acids levels. (a) PCoA plot of unweighted UniFrac, (b) total BA, primary BA, and secondary BA, (c) the ratio of primary to secondary, (d) primary BAs, and (e) secondary BAs. Data were expressed as mean ± SEM, n = 7 mice per group. *P < 0.05, **P < 0.01. BA, bile acid.
Consistency and Correlation Between Fecal Abundance of Gut Microbiotas and Bile Acids
As shown in Figure 9a, Procrustes’ analysis suggested a strong correlation between microbiota and BAs. To clarify the relationship between gut microbiota and BAs, a Spearman correlation was conducted between the levels of fecal BAs and the relative abundances of differential bacteria species identified above. HFD significantly increased the relative abundances of Lachnospiraceae_bacterium_28_4, Faecalibaculum_rodentium, Bacteroides_sartorii, and Romboutsia_ilealis, which were negatively correlated with the levels of fecal HDCA and GUDCA in female obese mice (Figure 9b). However, the intervention of ACN significantly increased the relative abundances of Lactobacillus_johnsonii and Lactobacillus_reuteri in HFD-fed female mice, and correlation analysis showed that Lactobacillus_johnsonii and Lactobacillus_reuteri were positively correlated with the levels of fecal HDCA and GUDCA (Figure 9b). Therefore, our data suggested that ACN may exert its anti-obesity effect by increasing the relative abundances of Lactobacillus_johnsonii and Lactobacillus_reuteri in the gut and subsequently affecting the metabolism of fecal HDCA and GUDCA.

Consistency and correlation between fecal abundance of gut microbiotas and bile acids. (a) Procrustes analysis and (b) a heat map generated by Spearman's correlation analysis reveals the relationship between the abundance of gut microbiota at the species level and BA profile. Data were expressed as mean ± SEM, n = 7 mice per group. *P < 0.05, **P < 0.01. BA, bile acid.
Discussion
The increasing prevalence of overweight and obesity poses a significant threat to public health globally, notably higher rates observed among adult women than men.2,19 Accumulating evidence suggests that sex differences exist in obesity and related diseases. The primary contributors to these differences are gonadal hormones, which play a central role in distinguishing between males and females.20,21 As a natural bioactive substance, anthocyanins exhibit antioxidative and anti-inflammatory properties, contributing to their role in preventing various chronic diseases such as metabolic disorders.22,23 Previous studies confirmed that ACN derived from Lycium ruthenicum Murray effectively reduces body weight in HFD-induced male C57BL/6J mice mainly by enhancing bacterial communities’ diversity, enhancing intestinal barrier integrity, inhibiting pancreatic lipase activity, and lowering the Firmicutes to Bacteroidota ratio. 18 In this study, our investigation revealed notable distinctions in the gut microbiota composition between male and female mice following ACN intervention at the taxonomic level. Specifically, we observed a significant increase in the abundance of Lactobacillus_jonsonii and Lactobacillus_reuteri in the fecal samples of female mice post-ACN intervention. These findings suggest that sex differences may be crucial in influencing the positive alterations in gut microbiota composition associated with ACN intervention.
The present study demonstrated that ACN effectively reduced body weight in HFD-induced female mice by suppressing the weight of visceral periovarian adipose tissue but without affecting their food intake. Furthermore, the histopathological analysis showed that the intervention of ACN reduced hepatic steatosis and accumulation of lipid droplets and reduced the periovarian adipocyte size. The results agreed with the report of Ding et al, 24 who reported a similar reduction in body weight and adipose tissue associated with the consumption of Honokiol in female C57BL/6J mice. In addition, the most significant was that intervention of ACN could decrease the serum lipids of TC and LDL levels in female HFD mice. These results suggested that ACN from Lycium ruthenicum Murray has a beneficial effect on the overweight and obesity of HFD-induced female mice, which is achieved by lowering food intake and reducing fat accumulation in mice.
The gut microbiota is closely related to obesity, and the interaction between dietary components and the gut microbiota determines the composition of gut microbiota, which has an important impact on host metabolism.6,25 Recent studies have presented evidence indicating variations in gut microbiota composition between the sexes and the effects of diet and sex hormones on gut microbiota composition.26,27 A study showed that bacterial diversity was significantly reduced in HFD-induced obese mice, accompanied by insulin resistance and dyslipidemia. 28 In this study, we also observed that HFD significantly decreased the fecal bacterial community evenness and diversity in female mice. At the phylum level, the intervention of ACN significantly increased the relative abundance of Firmicutes and decreased the relative abundance of Bacteroidota in HFD-induced female mice. This result deviates from prior findings in male mice, which suggested Firmicutes are energy collectors and Bacteroidota are energy consumers. 26 However, recent reports have suggested that changes in other components at the genus or species level may be more relevant to obesity than variations in Firmicutes and Bacteroidota. 29 At the genus level, ACN intervention significantly increased the relative abundances of Lactobacillus, Allobaculum, while decreased the abundances of Blautia, Faecalibaculum in HFD-induced female mice. Studies have shown that Lactobacillus and Allobaculum may exhibit a negative correlation with inflammation and obesity; these bacterial genera are believed to contribute to improving obesity and related diseases through their anti-inflammatory properties and alleviating intestinal dysbiosis.30,31 In contrast, Blautia and Faecalibaculum have been documented to show a positive association with obesity and impaired glucose tolerance. 32 The results at the species level were consistent with those observed at the corresponding genus level. These findings suggested that ACN from Lycium ruthenicum Murray exerts its anti-obesity effect on female mice through the regulation of the gut microbiota, specifically by increasing the abundances of beneficial bacteria such as Allobaculum, Lactobacillus_johnsonii and Lactobacillus_reuteri within Firmicutes.
BAs have been recognized as crucial for the emulsification and absorption of fat and cholesterol. 33 The composition of BAs is also affected by microbial community structure and function.34,35 Thus, gut bacteria-BA interaction significantly affects cholesterol metabolism, lipid homeostasis, and energy harvest of the host. 36 Our results showed that in HFD-induced female mice, the concentration of fecal total BAs, primary BAs, and secondary BAs was significantly increased, while the increase in primary BAs expression was substantially reversed following ACN intervention. One of the important findings in the current investigation was that the intervention of ACN also reversed the increased ratio of primary to secondary BAs in fecal induced by the HFD. Previously, an elevated primary/secondary BA ratio has been reported to have an adverse effect on health. 37 In the report by Mouzaki et al, 38 it was also found that NASH patients had higher levels of total fecal BAs levels and a higher ratio of primary to secondary BAs. This is consistent with what we have observed in female mice. Considering the interaction between BAs and gut microbiota, we examined their correlation. The correlation analysis between fecal profiles and gut microbiota showed that ACN intervention increased the abundance of Lactobacillus_reuteri and Lactobacillus_ johnsonii in female obese mice induced by HFD, further increasing the levels of HDCA and GUDCA. Lactobacillus_reuteri and Lactobacillus_ johnsonii have been identified as probiotics with the potential to positively impact obesity by promoting stability within the microbiota. A study has reported that Lactobacillus_johnsonii JNU3402 plays a key role in inhibiting hepatic lipid accumulation by attenuating hepatic lipid accumulation and reducing liver and plasma TG and free fatty acid levels in HFD-induced mice.39,40 In a recent study, Kuang et al 41 found significant improvements in liver TG, histological evaluation, and serum ALT following the treatment of NAFLD mice using HDCA. Additionally, HDCA significantly increased the abundance of probiotic species, which enhances lipid catabolism through fatty acid-hepatic peroxisome proliferator-activated receptor alpha signaling. These results suggest that ACN could alter BAs metabolism and suppress fat accumulation by improving the gut microbiota environment.
There are several limitations to this study. First, the study only measured fecal BAs levels. This does not fully reflect liver and serum BAs levels. Second, our available data only suggests an association between gut microbiota and fecal BA profile after ACN intervention. Therefore, further experiments will be designed to provide evidence of causality in the future.
Our study revealed that ACN from Lycium ruthenicum Murray significantly regulates gut microbiota dysbiosis and BAs metabolism in female C57BL/6J mice induced by HFD. These results showed that ACN significantly increased the abundances of Lactobacillus, Allobaculum and decreased the abundances of Blautia and Faecalibaculum. Alterations in the gut microbial composition further alter the composition of the BAs profile, particularly increasing the levels of fecal HDCA and GUDCA. Therefore, our results indicated that gut microbiota and BA metabolism play an important role in obesity and being overweight. ACN from Lycium ruthenicum Murray attenuated body weight induced by HFD by regulating the gut microbiota and BA metabolism in female mice.
Conclusion
In conclusion, our present results demonstrate that ACN from Lycium ruthenicum Murray reduces body weight in HFD-induced obese female mice. This is evidenced by reduced fat accumulation, decreased serum lipid levels, and improved hepatic steatosis. In addition, studies on gut microbiome and BAs metabolomics studies have shown that ACN exerts anti-obesity effects by regulating the composition of gut microbiome and fecal BAs. Therefore, ACN, as an extract of Lycium ruthenicum Murray, could be a potent candidate in functional foods used for anti-obesity among females.
Materials and Methods
Chemicals and Reagents
Fruits of Lycium ruthenicum Murray were obtained from the National Engineering Technology Research Center (Yinchuan, China) and stored at −20 °C until use. The normal diet (containing 11.3% lipids, 25.1% proteins, and 63.6% carbohydrates) was acquired from (Jiangsu Xietong Pharmaceutical Bio-engineering Co., Ltd). The HFD of MD12015 (containing 21% lipids, 20% proteins, 50% carbohydrates, and 0.15% cholesterol) was purchased from Medicience Company (Yangzhou, China). TC, TG, high-density lipoprotein (HDL-C), and low-density lipoprotein (LDL-C) kits were provided by Nanjing Jiancheng Biological Institute (Nanjing, China). The hematoxylin and eosin (H&E) staining solutions were supplied by Solarbio Science & Technology Co., Ltd (Beijing, China). All 33 BA standards and 6 stable isotope-labeled standards were obtained from ZZ Standards Co., LTD. (Shanghai, China). Methanol, acetonitrile, and formic acid were purchased from Thermo-Fisher Scientific (Fairlawn, NJ, USA). Ammonium acetate was of analytical grade and acquired from Sigma-Aldrich (St. Louis, MO, USA).
Preparation of the Anthocyanins from Lycium ruthenicum Murray
Total anthocyanins were extracted from the fruits of Lycium ruthenicum Murray according to the method reported by Yan et al.
42
Anthocyanins were extracted using a 0.5% TFA methanol solution. The extracts were concentrated after vacuum suction and filtration, and then the crude anthocyanin extracts were further purified by XAD-7 microporous resin column. The recovery of XAD-7 microporous resin column was then freeze-dried for later use. The total concentration of anthocyanins in the ACN extract was 15.1 ± 1.2%, using the pH differential method reported by Mi et al.
43
In brief, the obtained anthocyanin extracts were diluted with pH 1.0 and pH 4.5 buffers, respectively, and the absorbance was measured at 530 nm and 700 nm. The total anthocyanin concentration in the ACN extract was calculated according to equations (1-3).
The compounds identified in the extract of Lycium ruthenicum Murray have been reported in our previous research. 44 In addition to anthocyanins, other polyphenolic compounds had been identified in ACN extract, such as naringenin, naringenin-O-rutinose-7-O-hexoside, naringenin-O-rutinoside, naringenin-O-hexoside, chrysin-p-ferulic acid-hexoside, and chrysin-p-ferulic acid.
Animal Study
All animals and experimental procedures were approved by the Animal Ethics and Welfare Committee of Ningxia Medical University (Approval number: IACUC-NYLAC-2022-019). Thirty-two 6-week-old female C57BL/6J mice were purchased from Laboratory Animal Centre (Ningxia Medical University, Yinchuan, China). They were maintained in a specific-pathogen-free laboratory animal room with a controlled environment (22 ± 2 °C temperature, 50 ± 10% humidity, 12-h light/dark cycle), and were allowed free access to diet and water. After 1 week acclimatization, the mice were randomly divided into four groups, eight mice per group: (1) control group (Ctrl) that received normal diet and autoclaved, sterilized water; (2) ACN plus normal diet group (Ctrl + ACN) that received normal diet with 8 mg/ml ACN solution; (3) HFD group that received HFD and autoclaved, sterilized water; and (4) ACN plus HFD group (HFD + ACN) that received HFD with 8 mg/ml ACN solution. During the experiment protocol, their body weights, food intakes, and ACN/water consumption were recorded once a week for 32 weeks. Fecal samples were collected from mice at week 31, which were carried out in individual cages in the morning to avoid contamination, kept in liquid nitrogen, and then stored at −80 °C until further analysis.
At the end of the experiment, mice were fasted overnight (10 h) before being euthanized. Blood samples were collected and centrifuged at 4 °C, 4000 × g for 10 min to obtain serum samples. Tissues including liver, mesenteric, perirenal, back, and periovarian fats were weighted immediately, some of which were fixed in 4% paraformaldehyde for histopathological analysis, and the remainder were flash-frozen in liquid nitrogen and stored at −80 °C for further analysis.
Measurement of serum Biochemical Indicators
The serum TC, TG, LDL-C, and HDL-C were measured using the commercial assay kits, according to the manufacturer's protocol.
Histopathological Analysis
Liver and periovarian fats were fixed with 4% paraformaldehyde solution, dehydrated in an automatic tissue dehydrator, embedded in paraffin blocks, and then cut into 5-μm sections. Tissue sections were deparaffinized and rehydrated using a graded ethanol series and distilled water, then treated with a hematoxylin solution for 4 min and washed in running tap water for 10 min. Then, it was counterstained with an eosin solution for 2 min, washed in pure water for 5 min, dehydrated in a series of ethanol, and cleared in xylene. The H&E histopathological sample images were acquired using optical microscopy from Nikon (Tokyo, Japan).
16S rRNA Gene Sequencing and Data Analysis
Fecal samples from each group were selected for 16S rRNA analysis. Total genome DNA from fecal samples was extracted using Cetyltrimethylammonium Bromide method reported by Bansal et al. 45 The concentration and purity of DNA were determined via 1.0% agarose gel electrophoresis and diluted to 1 ng/μL as a template using sterile water. The DNA template was amplified using barcoded universal bacterial primers of the 16S rRNA gene: 341F (5′-CCTAYGGGRBGCASCAG-3′) and 806R (5′-GGACTACNNGGGTATCTAAT-3′). All PCR reactions were carried out with 15 µL of Phusion®High-Fidelity PCR Master Mix (New England Biolabs). The PCR reaction was carried out as follows: initial denaturation at 98 °C for 1 min, followed by 30 cycles of denaturation at 98 °C for 10 s, annealing at 50 °C for 30 s, and elongation at 72 °C for 30 s, with a final extension of 5 min at 72 °C. The PCR product was extracted from 2% agarose gel and purified, then sequenced on an Illumina Miseq platform with the paired-end method.
The raw sequencing tags of 16S rDNA sequencing were spliced using FLASH (V1.2.7, http://ccb.jhu.edu/software/FLASH/), filtered interfering data using QIIME (V1.9.1, http://qiime.org/scripts/split_libraries_fastq.html) to obtain the clean tags. In addition, the clean tags through the UCHIME algorithm (http://www.drive5.com/usearch/manual/uchime_algo.html) to detect chimera sequences, and then the chimera sequences were removed to obtain the effective tags. The effective tags from each sample were clustered into operational taxonomic units (OTUs) based on the 97% sequence identity by UPARS (Uparse v7.0.1001, http://www.drive5.com/uparse/), and then representative sequence for each OTU was screened for further annotation. The α-diversity of the fecal microbial communities was applied in analyzing the complexity of species diversity for a sample through 4 indices, including Chao1, Shannon, Simpson, and ACE. All these indices were calculated with QIIME (Version 1.7.0) and displayed with R software (Version 2.15.3). The β-diversity of the fecal microbial communities was explored with NMDS plots based on the unweighted UniFrac distance using R software (Version 2.15.3).
BA Analysis
Fecal samples (50 mg) were pulverized with liquid nitrogen and extracted with 300 µL of acetonitrile/methanol (v/v = 8 : 2). Then internal standard mixed solution (consisting of GCA-d4, UDCA-d4, CA-d4, GCDCA-d4, LCA-d4, and CDCA-d4)was added into the extract. Following a precipitation procedure at −20 °C for 10 min, samples were centrifuged at 4 °C at 12 000 rpm for 10 min. Supernatants were collected for further UHPLC-MS/MS analysis. The stock solution of individual BA was mixed and prepared in a BA-free matrix to obtain a series of BA calibrators at a concentration of 25,000, 15,000, 5000, 2500, 500, 250, 50, 25, 15, 5, 2.5 or 1.5 ng/mL. LC-MS was used to detect the concentration series of standard solution. The ratio of the concentration of standard to internal standard as abscissa and the ratio of peak area of the standard to internal standard is ordinated to investigate the linearity of standard solution. BA contents of the sample were detected by an ultra-high-performance liquid chromatography coupled to tandem mass spectrometry (UHPLC-MS/MS) system (ExionLC™ AD UHPLC-QTRAP® 6500+, AB SCIEX Corp., Boston, MA, USA) in Novogene Co., Ltd (Beijing, China).
Separation was performed on a Waters ACQUITY UPLC BEH C18 column (2.1 × 100 mm, 1.7 μm), which was maintained at 50 °C. The mobile phase, consisting of 0.1% formic acid in water (solvent A) and acetonitrile (solvent B), was delivered at a 0.30 mL/min flow rate. The solvent gradient changed according to the following conditions: initial 20% solvent B in 0.5 min; 20–35% solvent B in 1 min; 35–37% solvent B in 2.5 min; 37–38% solvent B in 4.1 min; 38–39% solvent B in 6 min; 39–40% solvent B in 6.5 min; 40–44% solvent B in 8.5 min; 44–45% solvent B in 9 min; 45–52% solvent B in 9.5 min; 52–65% solvent B in 12.5 min; 65–100% solvent B in 13 min; 100–20% solvent B in 15.1 min; 20% solvent B in 17 min. BAs were analyzed using scheduled multiple reaction monitoring. Parameters were as follows: IonSpray Voltage (−4500 V), Curtain Gas (35 psi), Ion Source Temp (550 °C), Ion Source Gas of 1 and 2 (60 psi). Finally, the data obtained were analyzed using SCIEX OS.
Statistical Analysis
All results were presented as mean ± standard error. Comparisons between groups were performed using one-way analysis of variance and the Mann-Whitney U test. All the bar plots in this study were generated with GraphPad Prism 8.0. Spearman's correlation coefficients were computed for the relationships between BA monomers and the relative abundances of gut microbiota, and a heatmap was designed for the correlation matrix using the Omicshare tools. Statistically significant differences are indicated with asterisks: * P < 0.05, **P < 0.01, *** P < 0.001.
Footnotes
Acknowledgments
The authors would like to thank Abdul Rehman from the School of Clinical Medicine, Ningxia Medical University, for revising and correcting the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Committee of Animal Ethics and Welfare of Ningxia Medical University (IACUC-NYLAC-2022-019).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (NSFC, 81960588), Natural Science Foundation of Ningxia (2023AAC03168), and Ningxia Key Research and Development Program (2020BBF02006, 2021BEF02008).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
All animal experiments were approved by the Ethical Committee of Ningxia Medical University. All procedures were conducted in accordance with the guidelines for the care and use of laboratory animals (China).