Abstract
Introduction
Lipid metabolism is crucial for maintaining the energy balance, cellular structure, hormone production, molecular signaling, and overall metabolic health.1,2 Dysfunction in lipid metabolism can lead to various metabolic diseases, including metabolic dysfunction-associated fatty liver disease (MAFLD), obesity, and hyperlipidemia.3,4 In modern society, unhealthy habits like sedentary lifestyles and a high-fat, high-sugar diet have led to excessive lipid accumulation, fueling the global rise in metabolic diseases. 5 Emerging evidence highlights the influence of the gut microbiota and peripheral serotonin on host metabolism through both correlation and causation.6,7 Despite this, safe and effective therapeutics to combat this pandemic remain limited. 8 Therefore, it is imperative to explore diverse strategies and techniques for controlling excessive lipid accumulation.
Fermentation is an extraordinary biological process utilized globally for thousands of years, allowing microorganisms to produce beneficial compounds that enhance preservation, flavor, and health. 9 In Asia, fermented foods such as kimchi in Korea and pu-erh tea in China are renowned for their positive effects on lipid metabolism.10,11 Additionally, fermentation effectively reduces toxic components in herbal medicines, such as aconite root. 12 Consequently, researchers are increasingly demonstrating that fermentation is an effective approach for enhancing bioactivity associated with the amelioration of various metabolic disorders. 13
Bacillus subtilis is a well-known probiotic bacterium commonly found in the soil, fermented soy products, and the mammalian gastrointestinal tracts. 14 Numerous studies have verified that it can serve as a fermentation starter for herbs such as Amomum xanthioides, Cornus officinalis, Tetragonia tetragonioides, and Radix astragali, which exhibit anti-hyperlipidemic, anti-inflammatory, antioxidant, and anti-aging properties.15–18 Cynanchum atratum, also known as “Bai-Wei” or “Baeg Mi,” is a traditional herbal medicine commonly used for its antipyretic, analgesic, and diuretic effects. 19 Moreover, it is frequently used for the treatment of nephritis, urinary tract infections, bronchitis, and rheumatic lumbocrural pain. 20 In addition, our previous study demonstrated that C. atratum exerts an anti-hepatic steatosis effect by reducing lipogenesis and enhancing fatty acid oxidation in a mouse model. 21 Nonetheless, our previous studies also revealed potential risks of cardiac toxicity and alopecia at high doses of C. atratum, raising significant concerns that limit its clinical use.21,22
Therefore, the present study is the first to attempt the fermentation of C. atratum using identified and quantified B. subtilis. The ameliorative effect of B. subtilis-fermented C. atratum (BSCA) on lipid accumulation was investigated using a mouse model fed a high-fat, high-fructose diet. Moreover, potential mechanisms related to the gut microbiota and peripheral serotonin levels were evaluated to provide a basis for further research into the development of promising therapies for metabolic diseases.
Materials and Methods
Herbal Fermentation and Extraction
Cynanchum atratum, compliant with the standards of the Korean Pharmacopeia, was sourced from Jeong-Seong Pharmaceutical Company (Daejeon, Korea). Soil-derived B. subtilis was isolated and cultured for 24 h in a shaking incubator at 37°C and 150 rpm prior to fermentation. The strain type was confirmed through 16S ribosomal DNA sequencing using the Basic Local Alignment Search Tool (https://blast.ncbi.nlm.nih.gov/Blast.cgi; accessed on March 17, 2022). In detail, the BLAST results identified the bacteria used in the present study as a Bacillus subtilis strain with high sequence similarity (>99%) to several well-characterized reference strains, providing strong evidence for species-level identification (Table S1). For fermentation, 500 g of C. atratum powder was mixed with 2500 mL of MRS medium (BD Difco, Marshfield, WI, USA). To eliminate interference from other bacteria and enhance the fermentation efficiency, the mixture was autoclaved at 115°C for 15 min. After cooling, B. subtilis was inoculated at 2% (2 × 108 CFU), and the mixture was incubated at 30°C for 1 week without shaking (Figure 1A).
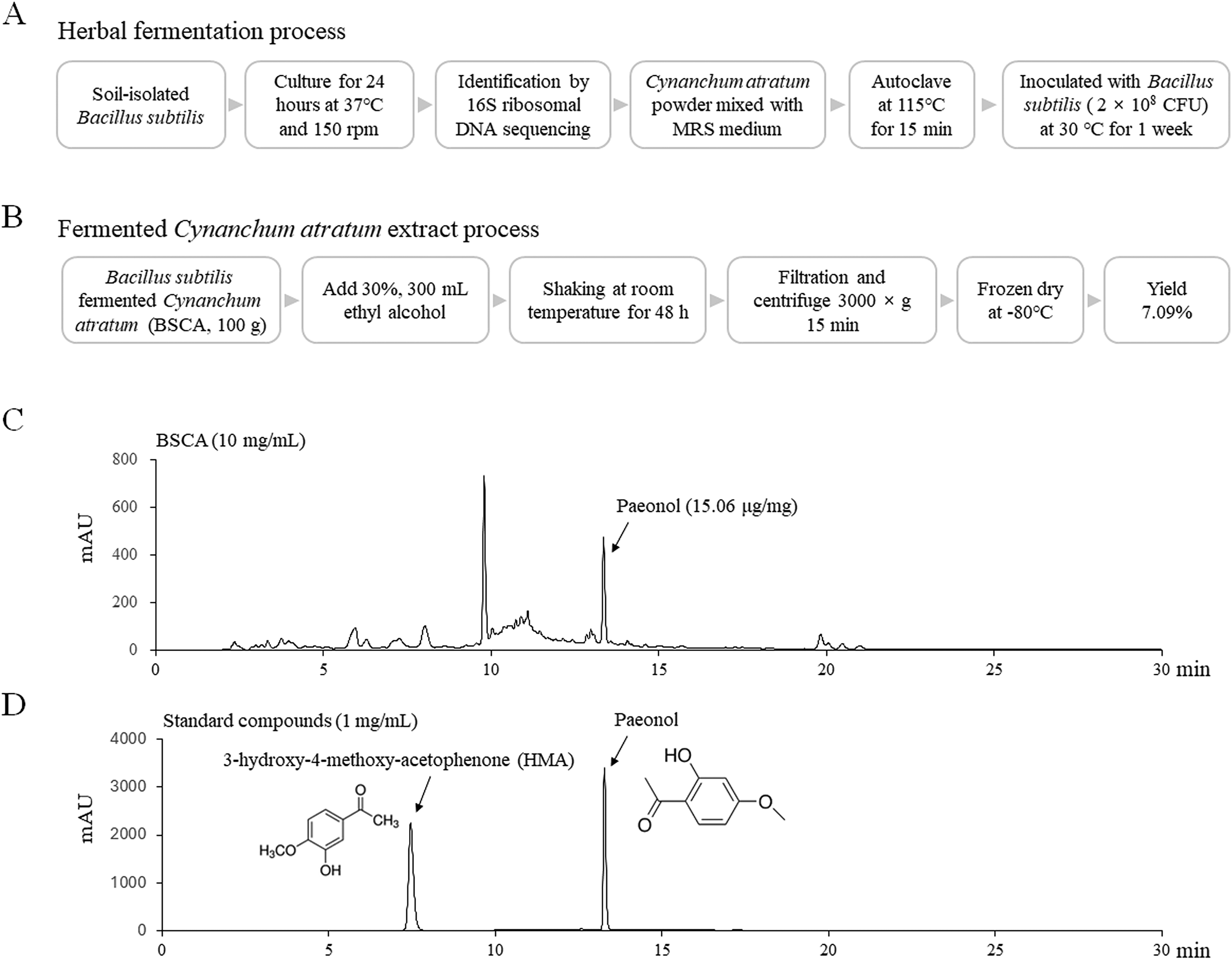
Herbal Fermentation and Extraction Procedures. (A) Schematic Representation of the Fermentation Process of Cynanchum atratum. (B) The Extraction Process of Fermented Cynanchum atratum Using 30% Ethanol. (C) High-Performance Liquid Chromatography (HPLC) Fingerprint Analysis of the Extracted Fermented Cynanchum atratum, Illustrating the Chemical Profile of the Potential Bioactive Compounds.
To maximize the extraction of both lipophilic and hydrophilic components, 30% ethanol was used based on Official restrictions. In detail, the ethanol extract of BSCA was prepared as follows: 100 g of BSCA was mixed with 300 mL of 30% ethanol, followed by shaking at 150 rpm for 48 h at room temperature (RT). The mixture was then centrifuged at 3000 × g for 15 min, and the supernatant was concentrated to 80 mL using a rotary vacuum evaporator. The concentrated extract was freeze-dried at −80°C for 72 h, yielding 7.09% of the original weight (Figure 1B). The final powder was stored at −20°C for future in vitro and in vivo studies and was labeled with the voucher specimen number BSCA-202105. Metformin was purchased from Sigma-Aldrich (St. Louis, MO, USA) and used as the positive control.
High-Performance Liquid Chromatography (HPLC) Fingerprinting
A 50% methanol solution of BSCA was analyzed using an Agilent 1260 HPLC system (Agilent Technologies, Wilmington, DE, USA), equipped with a binary pump, degasser, autosampler, column oven, and diode array detector set to 300 nm. Separation was performed on an Eclipse XDB-C18 column (Agilent, 4.6 × 250 mm, 5 μm) at RT. Gradient elution with H2O and acetonitrile (CH3CN) was employed, starting at 20% CH3CN and increasing it to 100% over 45 min at a flow rate of 1 mL/min. Paeonol (Glentham Life Sciences, Wiltshire, UK), a known constituent of C. atratum, was used as an internal standard. Chromatographic data were analyzed using Agilent ChemStation software (Figure 1C and D).
Animal Experiment Design
The animal experiments adhered to the ARRIVE 2.0 guidelines (Animal Research: Reporting of In Vivo Experiments 2.0) 23 and were approved by the Institutional Animal Care and Use Committee of Daejeon University (approval number: DJUARB-2021016). To minimize variation caused by hormonal fluctuations, 6-week-old male C57BL/6J mice (SPF grade) were purchased from Daehan BioLink (Gyeonggi-do, Korea). Throughout the experiment, the mice were housed in standard polycarbonate cages with aspen bedding under controlled conditions (22 ± 2°C, 50 ± 20% humidity) and a 12-h light/dark cycle, with ad libitum access to food. After a 7-day acclimation period, 30 mice were randomly assigned to five groups (n = 6 per group). Mice in all treatment groups were fed a high-fat diet (HFD, D12492, 60% kcal from fat; Research Diet, New Brunswick, NJ, USA) along with liquid fructose (dissolved in distilled water [DW], 20% w/v; Sigma-Aldrich, Darmstadt, Germany), whereas the control group received a chow diet (Rodent NIH-41 K, 11.88% kcal from fat; Zeigler Bros., Inc. Gardner, PA, USA) and DW. The mice were fed a high-fat, high-fructose diet (HFHFD) for 10 weeks, and from the fifth week, they were treated with BSCA (100 or 200 mg/kg/day) or metformin (100 mg/kg/day) via oral gavage for 6 weeks (Figure 2A). The BSCA dosage was calculated based on the clinical dose and adjusted for the animal's body surface area. 24 The control and HFHFD groups received DW following the same administration procedure as the treatment groups.

Experimental Schedule and Food Consumption. (A) Animal Experimental Timeline, (B-C) Average Food and Fructose Intake, (D-E) Average Caloric Intake from Food and Total Caloric Intake. # p < .05 and ## p < .01, as Compared to Normal Group.
Food and water intake were monitored weekly, and body weights were recorded at the same intervals. Fecal samples were collected the day before sacrifice and stored at −80°C. Following an 18 h fast, a drop of blood was collected from the tail vein to measure fasting blood glucose levels using an ACCU-CHEK glucometer (Roche Diabetes Care GmbH, Mannheim, Germany). On the final day of the experiment, approximately 500 μL blood was drawn from the abdominal vein under anesthesia using a combination of Zoletil (20 mg/kg, tiletamine-zolazepam, Virbac, Carros, France) and Rompun (10 mg/kg, xylazine-hydrochloride, Bayer, Leverkusen, Germany) to minimize suffering. Complete blood counts (CBCs) were performed using an automated hematology analyzer (Exigo Eos, Boule Medical AB, Spanga, Sweden). Liver and adipose tissues were dissected and weighed, and liver samples were preserved in 10% neutral formalin or frozen at −80°C for further analyses. Mesenteric, epididymal, and perirenal adipose tissues were excised and weighed individually.
Biochemical Analysis of Serum and Hepatic Tissue
After allowing the blood to clot for 1 h, the serum was separated via centrifugation at 3000 × g for 15 min. Serum levels of aspartate transaminase (AST), alanine transaminase (ALT), total cholesterol (TC) and triglyceride (TG) determined using an Auto Chemistry Analyzer (Chiron, Emeryville, CA, USA). Serum insulin levels were measured using a mouse insulin ELISA kit following the manufacturer's instructions (Invitrogen, Carlsbad, CA, USA). Hepatic TG and total cholesterol (TC) levels were quantified using commercial assay kits (ASAN Pharm, Anseong-si, Korea).
Histopathological Analysis
Paraffin-embedded liver tissues were sectioned into 5 μm thick slices using a microtome (Leica RM225, Nussloch, Germany). The sections were subsequently stained with hematoxylin and eosin (H&E) and mounted using a commercial mounting medium (Biomeda, Foster City, CA, USA). Frozen liver tissues, embedded in FSC22 clear frozen section medium (Leica, Germany), were sectioned into 5 μm-thick slices and mounted on silicone-coated slides. Slides were stained with either H&E or Oil Red O solution. After staining and drying, liver sections were imaged at 200× magnification using an Olympus IX71 microscope and a DP70 camera (Tokyo, Japan). The extent of Oil Red O staining was quantified using ImageJ software (National Institutes of Health, USA).
ELISA for Inflammatory Cytokines and Peripheral Serotonin
Serum samples were diluted five-fold, whereas tissue homogenate supernatants were diluted two-fold with phosphate-buffered saline at pH 7.2. Tumor necrosis factor-alpha (TNF-α) levels in hepatic tissue and serotonin levels in serum and duodenal tissue were determined using commercial ELISA kits, following the manufacturers’ protocols: R&D Systems® (Minneapolis, MN, USA) for mouse TNF-α (DY410) and LDN (Nordhorn, Germany) for mouse serotonin (BAE-5900).
Fecal Microbiota Analysis by 16S rRNA Sequencing
Fecal metagenomic DNA was extracted using the QIAamp DNA Stool Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer's protocol. The quality of the extracted DNA was evaluated using 1% agarose gel electrophoresis. Bacterial DNA was amplified using primers targeting the V3–V4 region of the 16S rRNA gene (341F and 805R). PCR-amplified DNA was sequenced by ChunLab, Inc. (Seoul, Republic of Korea) using the MiSeq sequencing system (Illumina, San Diego, CA, USA). Low-quality reads, defined as those with average quality scores of <25 or read lengths <300 base pairs, were removed. The remaining sequences were denoised using DUD-Seq software. Quality-controlled sequences were used for taxonomic classification. Alpha and beta diversity analyses were conducted using CLcommunityTM software (Version 3.46, Chunlab Inc., Seoul, Korea).
Statistical Analysis
All statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS, version 19.0, Chicago, NY, USA). Data are presented as means ± standard deviations. One-way analysis of variance, followed by the least significant difference post-hoc test was used to determine significant differences between groups. Statistical significance was set at a p-value < .05.
Results
BSCA Contains Paeonol but Lacks HMA
Both paeonol and HMA are naturally present in C. atratum. However, HPLC profiling revealed that only paeonol was detected in BSCA, with a concentration of 15.06 μg/mg (Figure 1C and D). In contrast, HMA was not detected in BSCA, indicating its absence or presence at levels below the detection limit.
BSCA Did not Change Food and Calorie Intake
Mice fed an HFHFD for 10 weeks showed a significant reduction in food and fructose intake compared with those in the control group (Figure 2B and C, p < .01). However, their food and total calorie intake was significantly increased (Figure 2D and E, p < .05 or p < .01). Interestingly, treatment with BSCA and metformin did not affect either food or calorie intake compared with that in the group administered an HFHFD alone. In the current study, no apparent signs of cardiac toxicity or alopecia were observed throughout the study period. In additional assessment, C57BL/6J mice treated with a single oral gavage dose of 1 g/kg or 2 g/kg BSCA showed no acute mortality, and serum AST and ALT levels remained within normal ranges (data not shown).
BSCA Attenuated Excessive Body, Liver, and Fat Weights
The long-term administration of an HFHFD notably enhanced body, liver, and fat weights compared to those in the control group (Figure 3A to F, p < .05 or p < .01). However, BSCA, but not metformin, significantly reduced body weight in a dose-dependent manner (Figure 3B, p < .05 or p < .01). Only the high dose of BSCA treatment significantly decreased liver weight compared to that in the HFHFD control group (Figure 3C, p < .05). Additionally, BSCA noticeably reduced mesenteric and perirenal fat weights compared to that in the HFHFD control group (Figure 3D to F, p < .05), but it did not affect epididymal fat weight.

BSCA Inhibited Excessive Body and Organ Mass in HFHFD-fed Mice. At the Beginning (A) and End (B) of the Experiment, the Average Body Weight of Mice in Each Group was Measured and Compared. After Sacrifice, the Mean Weights of the Liver (C), Mesenteric Fat (D), Perirenal Fat (E), and Epididymal Fat (F) Were Calculated and Compared as Well. # p < .05 and ## p < .01, as Compared to Normal Group; * p < .05 and ** p < .01, as Compared to the HFHFD Group.
BSCA Inhibited Lipid Accumulation and Inflammation in Hepatic Tissue
The administration of HFHFD resulted in a marked increase in vacuolation and Oil Red O-positive areas in the liver tissue compared to those in the control group (Figure 4A to C). Furthermore, HFHFD treatment significantly increased hepatic TG levels. In contrast, BSCA administration led to a dose-dependent reduction in vacuole size in H&E-stained liver tissues and a decrease in Oil Red O-stained areas (Figure 4A to C, p < .01). Consistently, BSCA significantly reduced hepatic TG levels in a dose-dependent manner (Figure 4D, p < .05 or p < .01). Additionally, HFHFD treatment significantly elevated levels of the inflammatory cytokine TNF-α in liver tissue compared to those in the control group (Figure 4E, p < .01). As expected, BSCA notably inhibited the elevated TNF-α levels induced by HFHFD treatment (Figure 4E, p < .05 or p < .01).
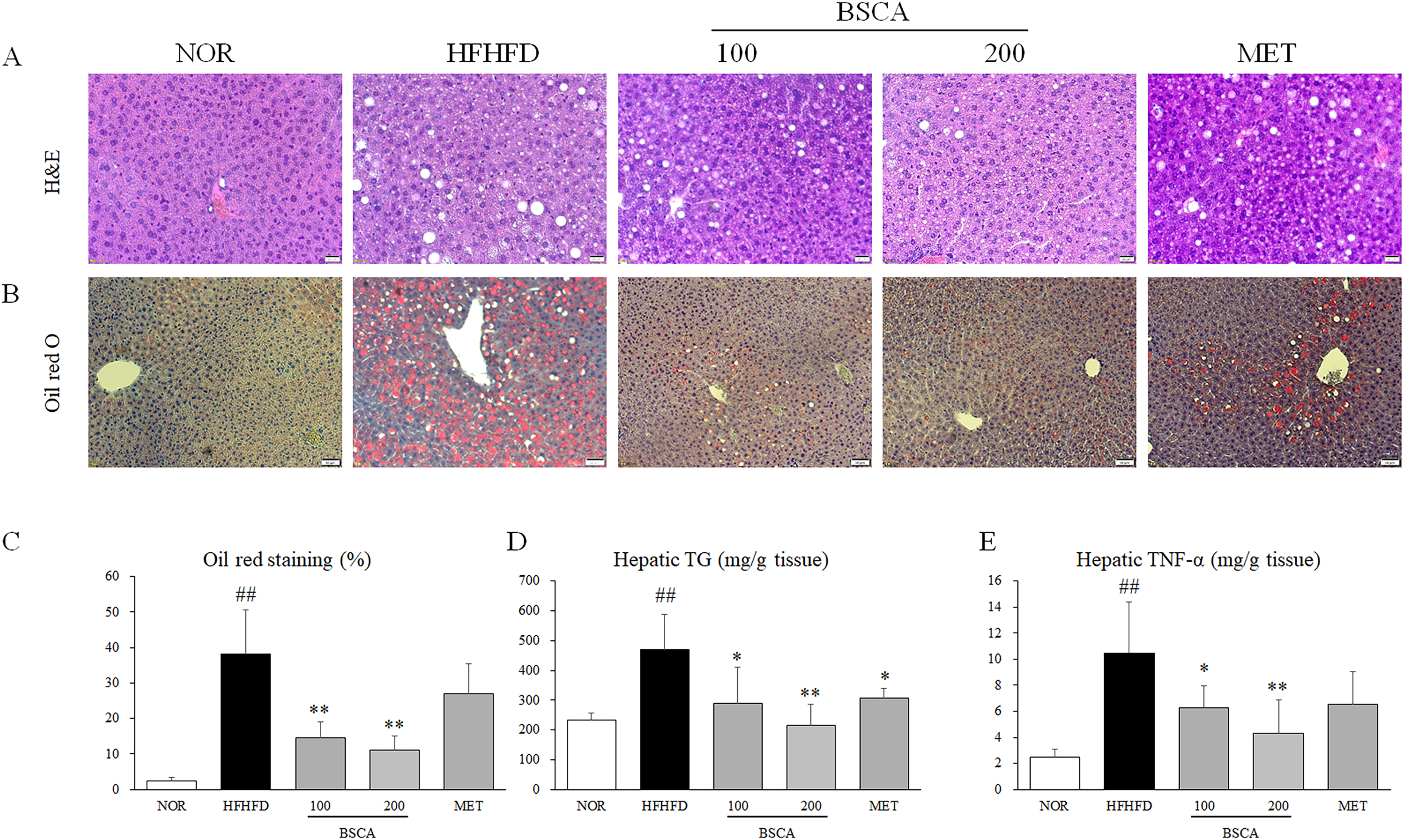
BSCA Inhibited Lipid Accumulation in Hepatic Tissue. (A) H&E Staining and (B) Oil Red O Staining were Performed on Liver Tissue (200× Magnification). (C) The Percentage of the Oil Red O Staining Area was Calculated Using ImageJ. The levels of TG (D) and TNF-α (E) were Analyzed in Liver Tissue. ## p < .01 Compared to the Normal Group; * p < .05 and ** p < .01 Compared to the HFHFD Group.
BSCA Reduced Superfluous Aminotransferases and Lipid Profiling in Serum
HFHFD treatment significantly increased the serum levels of AST, ALT, TC, TG, LDL, and HDL compared to those in the control group (Figure 5A to F, p < .05 or p < .01). However, BSCA treatment significantly normalized serum levels of AST, ALT, TC, TG, and LDL (Figure 5A to E, p < .05 or p < .01). Interestingly, BSCA did not affect serum HDL levels compared to those in the HFHFD control group (Figure 5F).
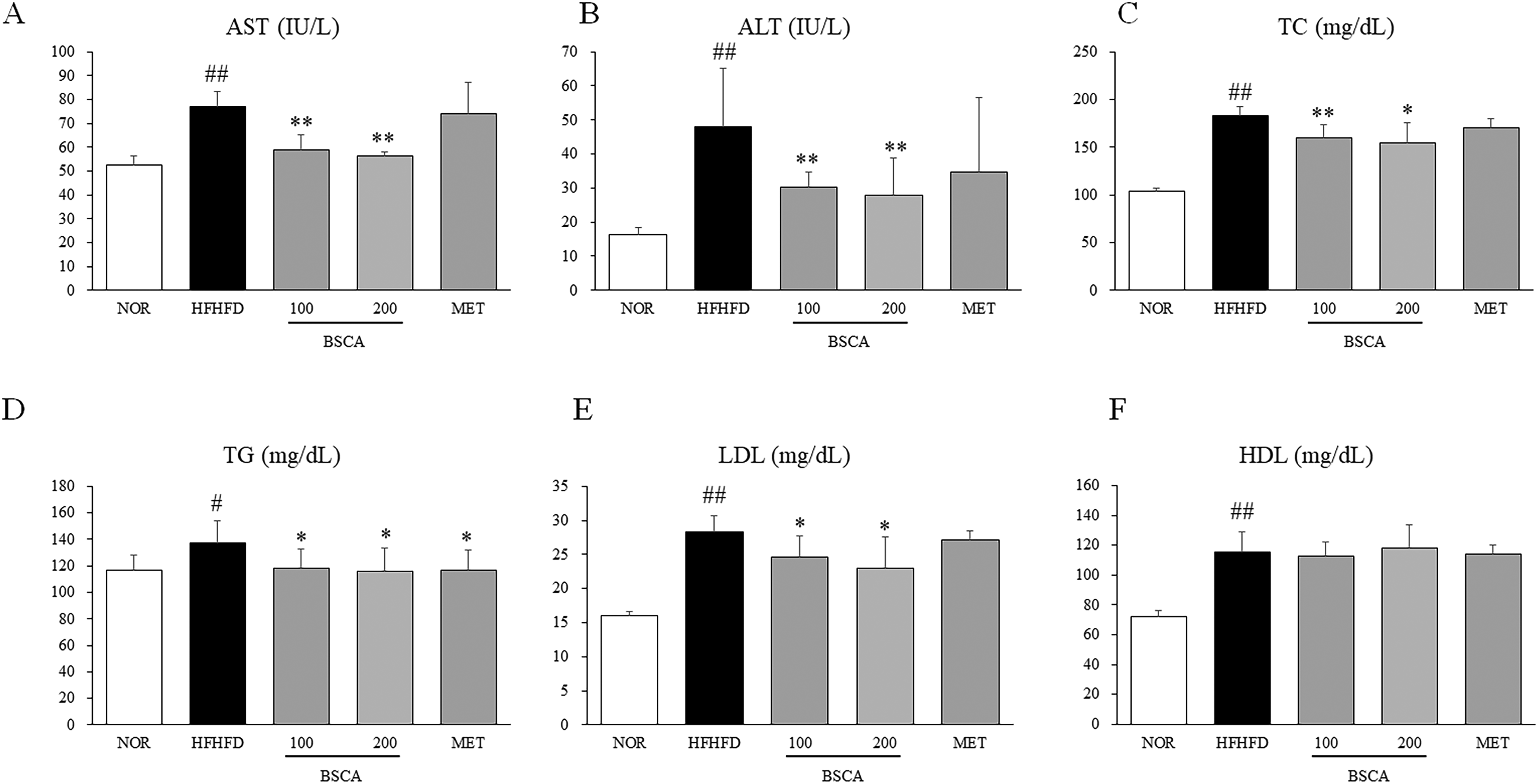
Serum Biochemistry Measurement. After Sacrifice, the Blood was Collected from the Abdominal Vein Under Anesthesia. After Allowing the Blood to Clot for 1 h, the Serum was Separated via Centrifugation at 3000 × g for 15 min. Serum Levels of Aspartate Transaminase (AST), Alanine Transaminase (ALT), Total Cholesterol (TC) and Triglyceride (TG) were Determined using an Auto Chemistry Analyzer (Chiron, Emeryville, CA, USA). # p < .05 and ## p < .01, as Compared to Normal Group; * p < .05 and ** p < .01, as Compared to the HFHFD Group.
BSCA Ameliorated Long-Term HFHFD-Induced Thrombocytosis and Leukopenia
Neither HFHFD nor BSCA treatment significantly affected RBC or HGB levels compared to those in the control group. However, HFHFD treatment resulted in a marked increase in platelet count and a significant decrease in WBC counts relative to those in the control group (F, p < .05 or p < .01). In contrast, BSCA treatment significantly decreased platelet count and normalized the WBC count (Table 1, p < .05 or p < .01). Notably, the reduction in lymphocyte and monocyte counts induced by the HFHFD was significantly mitigated by BSCA in a dose-dependent manner (Table 1, p < .05 or p < .01).
Complete Blood Count Analysis.

Abbreviations: NOR, normal; HFHFD, high-fat and high-fructose diet; BSCA, bacillus subtilis-fermented cynanchum atratum; MET, metformin; RBC, red blood cell; PLT, platelet; HGB, hemoglobin; WBC, white blood cell; LY, lymphocyte; MO, monocyte; GR, granulocyte; PLR, platelet-lymphocyte ratio. # P < .05, ## P < .01, as compared to NOR; * P < .05, ** P < .01, as compared to HFHFD.
BSCA Improved Fecal Microbiota Diversity
According to the rarefaction curve, we confirmed the sequencing depth is sufficient to capture microbial diversity (Figure S2). Fecal 16S rDNA sequencing results demonstrated that HFHFD treatment significantly reduced the α-diversity of the fecal microbiota, as indicated by the observed OTUs, Chao1 index, and Shannon index (Figure 6A to C, p < .01). Notably, only BSCA, but not metformin, significantly restored the α-diversity across all assessed metrics (Figure 6A to C, p < .01). Beta diversity analysis revealed substantial differences in the microbial communities between the control and drug-treated groups (Figure 6D). Furthermore, the microbial communities associated with BSCA treatment were more similar to those in the control group than to those in the HFHFD control group (Figure 6D, Figure S1).
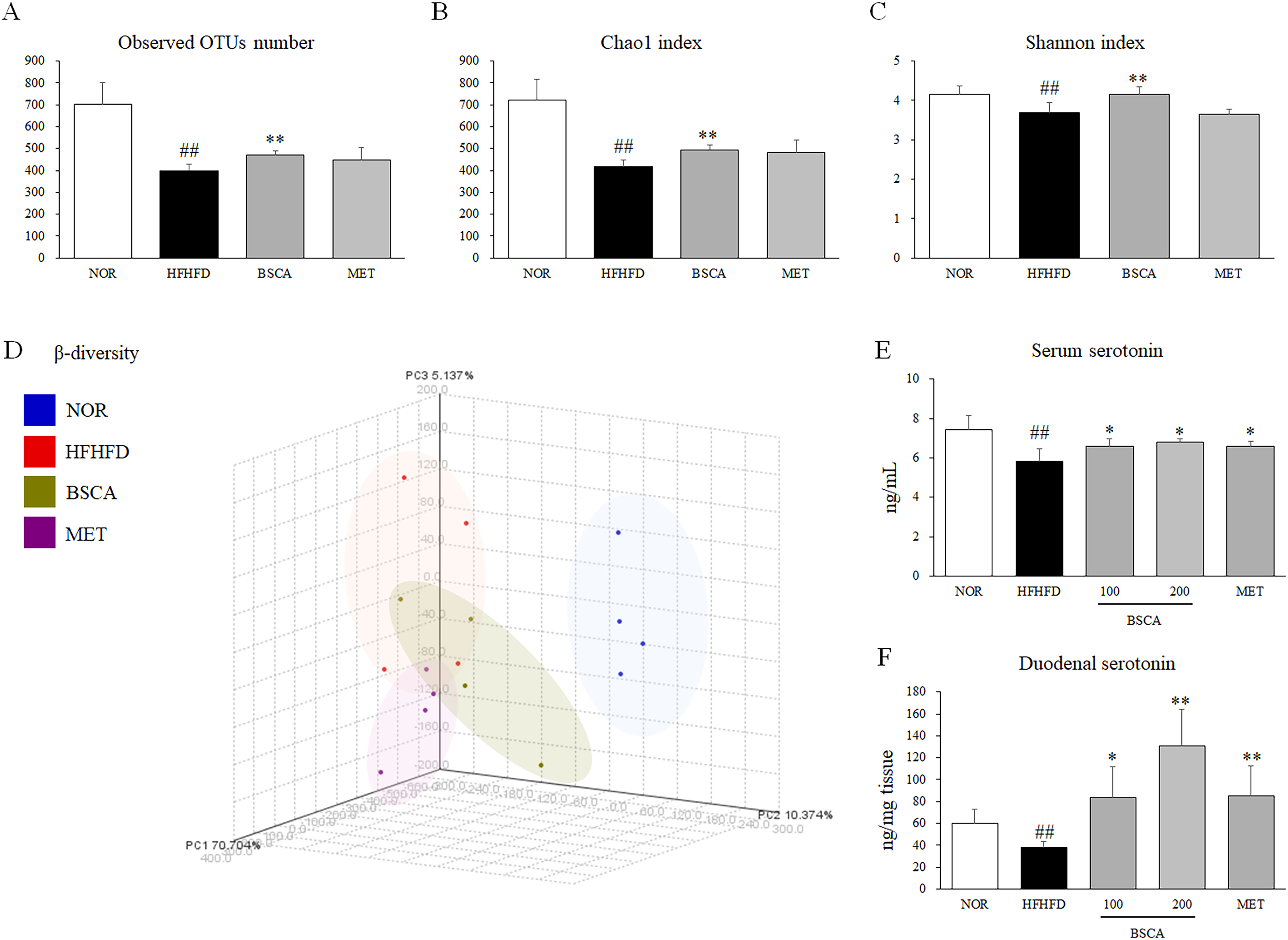
BSCA Restored the Diversity of Gut Microbiota and Peripheral Serotonin in Mice. On the Final Experimental Day, Fecal Samples were Collected, and Bacterial DNA was Isolated for 16S rDNA Sequencing Using the MiSeq Sequencing System. Then, α and β Diversity Analyses were Conducted Using CLcommunityTM Software. (A) Observed OTU Number, (B) Chao1 Index, and (C) Shannon Index was Used to Assess the α-Diversity of Gut Microbiota. (D) PCoA Analysis was Performed to Evaluate the β-Diversity of Gut Microbiota. (E) Serum and Duodenal Serotonin Levels were Measured Using an ELISA Kit (LDN, BAE-5900, Nordhorn, Germany). ## p < .01, as Compared to Normal Group; * p < .05 and ** p < .01, as Compared to the HFHFD Group.
BSCA Elevated Peripheral Serotonin Levels in the Serum and Duodenum
Serotonin levels in the serum and duodenal tissue were significantly reduced following HFHFD treatment (Figure 6E and F, p < .01). In contrast, BSCA treatment notably restored serum serotonin levels (Figure 6E, p < .05) and significantly elevated serotonin levels in the duodenum when compared with those in the HFHFD control group (Figure 6F, p < .05 or p < .01). As a positive control, metformin treatment demonstrated a comparable pattern of change in peripheral serotonin levels to that observed with BSCA.
Discussion
In modern society, long-term consumption of an unhealthy diet often leads to extensive lipid accumulation and various metabolic disorders by creating a positive energy balance, disrupting hormonal and metabolic pathways, inducing inflammation, and altering the gut microbiota. 4 Compared to a HFD alone, a combined HFHFD more closely reflects the modern Western diet, characterized by the excessive intake of both fats and sugars. 25 Therefore, the present study employed a 10-week HFHFD administration to induce lipid metabolism dysfunction in mice. This model of lipid dysmetabolism was successfully established, as evidenced by increased white adipose tissue weight in the mesenteric and perirenal regions, elevated hepatic TG levels, enlarged areas of Oil Red O staining in the liver tissue, and excessive serum TG and LDL levels. These findings demonstrate significant lipid accumulation in the adipose tissue, liver, and blood of the current mouse model.
To evaluate the potential of BSCA to mitigate lipid accumulation and associated metabolic disorders, BSCA treatment was initiated during the fifth week of HFHFD administration and continued for 6 weeks. Appetite regulation is a critical target in developing anti-obesity medications. 26 In the present study, although BSCA significantly reduced body and liver weights, it did not affect food or caloric intake. This suggests that the observed effects of BSCA on obesity are not associated with reduced food consumption. This finding has important implications for the development of therapeutic strategies, as it indicates that BSCA might enhance metabolic health without requiring dietary restrictions.
Furthermore, BSCA treatment noticeably reduced surplus lipid accumulation in the fat and blood; however, it did not affect epididymal fat weight or serum HDL levels. Mesenteric and perirenal adipose tissues are generally more responsive to hormonal and metabolic fluctuations than epididymal adipose tissue. 27 Consequently, the epididymal fat depot is often regarded as a more stable form of adipose tissue, exhibiting lower metabolic activity than mesenteric and perirenal fats. 28 This difference may explain its relative resistance to the lipid-reducing effects of BSCA. Furthermore, the anti-hyperlipidemic properties of BSCA were confirmed, suggesting that it primarily inhibits TG and cholesterol synthesis in the liver, leading to reduced serum LDL and total cholesterol levels, without significantly affecting HDL levels.
BSCA treatment also markedly decreased lipid accumulation in hepatic tissues, as evidenced by reduced Oil Red O staining and lower hepatic TG levels. Lipotoxicity can lead to hepatocyte injury, resulting in elevated aminotransferase levels. 29 However, BSCA mitigated excessive secretion of serum aminotransferases, indicating that it not only inhibits lipogenesis but also alleviates inflammation, as evidenced by significantly reduced levels of the inflammatory cytokine TNF-α and histopathological amelioration in the liver.
Recent research increasingly shows that a healthy gut microbiota is essential for maintaining lipid homeostasis and preventing metabolic disorders such as obesity and fatty liver disease.30,31 The gut microbiota plays a key role in regulating host lipid metabolism through mechanisms such as fermentation, bile acid modification, serotonin production, and modulation of inflammation.31,32 An HFHFD can disrupt the gut microbiota, leading to dysbiosis, increased intestinal permeability, and systemic inflammation. 33 Moreover, the gut microbiota significantly affects serotonin production, with approximately 90% synthesized in the gastrointestinal tract by enterochromaffin cells. 34 In our study, BSCA markedly restored the gut microbiota and serotonin levels, as demonstrated by changes in both alpha and beta diversity. Thus, we propose that the capacity of BSCA to restore gut microbiota and peripheral serotonin levels may be a key mechanism for improving host lipid metabolism.
Platelets are involved in inflammation and tissue repair, whereas a decrease in lymphocytes is often associated with chronic inflammation.35,36 Consequently, recent clinical studies have increasingly recognized the platelet-to-lymphocyte ratio as a marker of inflammation. 37 In this study, we found that BSCA alleviated the hematological disturbances caused by the HFHFD, including thrombocytosis and leukopenia. The restoration of WBCs, particularly lymphocytes rather than monocytes and granulocytes, induced by BSCA suggests that it may have broader systemic effects beyond lipid metabolism, potentially enhancing overall health and metabolic functions. These findings support the notion that fermented products, such as BSCA, could serve as promising herbal medicines for chronic metabolic disorders, including MAFLD and obesity. This study highlights the need for further research to investigate the mechanisms underlying these effects and assess the long-term safety and efficacy of BSCA in human populations.
Nevertheless, the current study has several limitations. For instance, we did not directly compare the effects of fermented and non-fermented CA, and the specific bioactive components responsible for the observed effects have not been identified. Therefore, future studies should include a direct comparison to elucidate the compositional changes induced by fermentation and their impact on efficacy. Moreover, we did not evaluate the probiotic properties of this B. subtilis strain itself. Besides, we selected only male animals to eliminate hormonal variability in this study. Therefore, it remains unclear whether BSCA has the same effect in females.
Conclusion
The fermentation of C. atratum demonstrates significant promise in alleviating chronic inflammation, highlighting its potential as a novel herbal medicine to enhance metabolic health. Chronic inflammation is a key contributor to various metabolic disorders, making effective interventions crucial for overall health. Moreover, BSCA exhibits noteworthy anti-fatty liver and anti-hyperlipidemic effects by inhibiting lipogenesis, a process intricately influenced by alterations in the gut microbiota composition and elevated serotonin levels. The relationship between gut health and metabolic function underscores the importance of a balanced microbiome for the prevention and management of metabolic disorders. These findings suggest that fermented C. atratum could serve as a valuable herbal medicine for various chronic metabolic conditions, including MAFLD, metabolic syndrome, and obesity. As research continues to unveil the mechanisms underlying these effects, C. atratum might offer a promising complementary approach to conventional therapies, ultimately contributing to improved metabolic health and the prevention of related diseases. Future studies should focus on the long-term safety and efficacy of this herbal remedy in diverse populations.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251331114 - Supplemental material for Bacillus subtilis-Fermented Cynanchum atratum Ameliorates Lipid Accumulation in Mice with Obesity Induced by High-Fat, High-Fructose Diet: Implications for Gut Microbiota and Peripheral Serotonin
Supplemental material, sj-docx-1-npx-10.1177_1934578X251331114 for Bacillus subtilis-Fermented Cynanchum atratum Ameliorates Lipid Accumulation in Mice with Obesity Induced by High-Fat, High-Fructose Diet: Implications for Gut Microbiota and Peripheral Serotonin by Mukesh Kumar Yadav, Hojun Kim, Song-Yi Han, Chang-Gue Son and Jing-Hua Wang in Natural Product Communications
Footnotes
Ethical Considerations
This study was approved by the Institutional Animal Care and Use Committee of Daejeon University (approval number: DJUARB-2021016).
Author Contributions/CRediT
Mukesh Kumar Yadav: writing-original draft, methodology, data curation, and formal analysis. Hojun Kim: resources, validation, and project administration and funding acquisition. Song-Yi Han: data curation, formal analysis, software, validation, investigation, and visualization. Chang-Gue Son: methodology, writing-review and editing. Jing-Hua Wang: conceptualization, methodology, writing-review and editing, and supervision.
Funding
The author(s) disclose receipt of the following financial support for the research, authorship, and/or publication of this article: this research was funded by the National Research Foundation of Korea (NRF) under grant number 2020R1F1A1074155 and by the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI) funded by the Ministry of Health & Welfare, Republic of Korea (HF20C0020)
National Research Foundation of Korea, Korea Health Industry Development Institute, (grant number 2020R1F1A1074155, HF20C0020).
Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
