Abstract
Introduction
Antibiotics are losing their effectiveness given the spread of antimicrobial-resistant bacterial pathogens. Natural products are potential alternatives to antibiotics. This paper aims to provide insight into the potential antibacterial effect of AhaEO combined with antibiotics
Materials and methods
AhaEO was hydrodistilled and subsequently analysed using GC–MS. The phytochemical compounds were screened as prospective inhibitors of β-lactamase by using molecular docking, and their antibacterial activities against some disease-causing bacteria were assessed by the antimicrobial disk assay and a serial dilution method in microplates.
Results
The characteristic chemical constituents of this essential oil were: Camphor (30.76%) and Eucalyptol (16.96%). The combinations of AhaEO with antibiotics displayed an antagonism effect, except with AX and TIC. ADMET profiles were favourable, confirming the safe consumption of all tested phytochemicals with no side effects. Among the tested compounds, patchoulane and chrysanthone scored binding energies of (−6.0 kcal/mol) and (−5.1 kcal/mol), respectively.
Conclusion
This work demonstrated that AhaEO can enhance the antibiotic effect. Additional experiments are necessary to validate the obtained results.
Introduction
Nowadays, antibiotic resistance, known as the “silent pandemic“, is a disconcerting worldwide health issue. In 2019, there were around 4.95 million death cases related to bacterial resistance, 1.27 million of them mostly due to bacterial resistance.1–4 Also, it is expected that 10 million incidents of death will be due to the same cause each year. 5 Bacterial pathogens have developed various mechanisms to withstand different types of antibiotics, one of which is the production of β-lactamases. 6 Scientists worldwide are paying more attention to pharmacologically active natural chemicals extracted from plants, such as essential oils, as possible treatments for infections. Studies have demonstrated that various essential oils and their isolated constituents exhibit antibacterial activity.6,7 A pattern of metabolic and biochemical reactions occurring inside the bacterial cell mediates the antimicrobial actions of essential oils, with each component displaying a distinct mechanism of action depending on its specific chemical constituents. 8 The lipophilic constituents of essential oils penetrate cell membranes, increasing permeability and causing leakage of ions, ATP, and intracellular components. This also causes the loss of membrane potential and inhibition of energy metabolism. These effects disrupt vital cellular functions, such as enzyme activity, nutrient transport, and respiration, ultimately resulting in bacterial cell death.8,9 Consequently, improved antibacterial effects could result from a combination of antibiotics and essential oils.10,11
Desert or white warmwood is the common name for
A high chemical variability has been reported in
Changes in the relative proportions of major constituents, including thujone isomers, camphor, 1,8-cineole, and borneol, may influence both antibacterial potency and selectivity, resulting in different responses among Gram-positive and Gram-negative bacteria. 8 Essential oils rich in oxygenated monoterpenes and monoterpene ketones are often reported to be more effective against Gram-positive organisms, whereas Gram-negative bacteria generally exhibit lower or more variable susceptibility.22,23 Several studies have shown that AhaEOs obtained from different regions or representing distinct chemotypes can display markedly different antibacterial activities.17,21,24
Previous studies have investigated the antibacterial efficacy of AhaEO against several bacteria, including
In this context, our study aimed to characterize the qualitative and quantitative chemical profile of AhaEO, and to evaluate its antibacterial efficacy against selected pathogenic bacterial strains. Furthermore, this work explored the effects of AhaEO combined with antibiotics and employed computational approaches to screen its chemical constituents for potential β-lactamase inhibitory activity.
Materials and Methods
Sampling
In April 2024, aerial parts of
Extraction of AhaEO
One hundred grams of dry matter were immersed in distilled water and hydrodistilled for 3 h using a Clevenger-type apparatus. 26 The obtained essential oil was separated from water in the Florentine part of the Clevenger, and then the last drops were separated by freezing the water. The AhaEO is then stored in sealed opaque tubes and preserved at −4 degrees Celsius until use.
GC-MS Analyses of AhaEO
A Shimadzu GC-MS-QP2020 system was used to analyze the chemical composition of AhaEO. The system was fitted with an Rxi-5 ms capillary column measuring 30 m in length, 0.25 mm in internal diameter and 0.25 µm film thicknesses. A high-purity helium (99.99%) gas, flowing at a steady rate of 1 mm/min, was used as the carrier gas. A volume of 0.5 microliters of the sample was injected, in split mode (1:80), into an injector maintained at 250 °C. The detector temperature was set to 310 °C. The column temperature started at a low degree, 50 °C (for 2 min), then gradually rose at a rate of 3 °C/min to reach 310 °C, and was maintained for 2 min. The AhaEO composition was determined by recording the mass spectra for an ionization voltage of 70 eV covering the 45-600 m/z range. The identification of volatile components was based on comparisons of their relative retention times and mass spectra with data from the Wiley, NIST17, and W11N17MAIN1 libraries of essential oil constituents. 27
Bacterial Strains and Antibiotics
-Gram-negative bacteria:
Antibiotics from multiple classes and with different mechanisms of action were used in this study (Table 1).
List of Antibiotics Used.

Antibacterial Activity
The antimicrobial properties of AhaEO were tested using two techniques: the disc diffusion assay to determine the sensitivity of microorganisms to AhaEO and the diameters of growth zones, and serial dilution method in microplates to determine minimal inhibitory concentrations (MIC) and minimal bactericidal concentrations (MBC). For the study, we initially inoculated a standardized bacterial culture (0.5 McFarland turbidity) of the appropriate strain on the surface of Mueller-Hinton agar. Subsequently, in each culture medium containing the respective bacterial culture, we positioned sterile discs (Ø = 6 mm) loaded with 10 µl of AhaEO. All the Petri dishes that had been prepared were placed in an incubator at 37 °C for 18 to 24 h. 28 After measuring the diameter (Ø) of the inhibition zone, the sensitivity of the bacteria toward AhaEO could be classified as not sensitive if Ø < 8 mm, sensitive if 9 mm ≤ Ø ≤ 14 mm, very sensitive if 15 mm ≤ Ø ≤ 19 mm, and highly sensitive if Ø > 20 mm. 29 The process was repeated three times.
The MIC is the lowest concentration of an antibacterial agent, expressed in mg/L (μg/mL), that, under strictly controlled in vitro conditions, completely prevents visible growth of the test strain. 30 Two-fold serial dilutions of the essential oil were prepared in sterile 96-well microplates. In each row, 100 µL of Mueller–Hinton broth was dispensed into wells 1–12. Then, 100 µL of the essential oil working solution was added to well 1, mixed, and 100 µL transferred sequentially from well 1 to well 10 to obtain a 1:2 dilution series; 100 µL from well 10 was discarded. Wells 11 and 12 served as controls: medium + inoculum (growth control) and medium only (sterility control), respectively. A 100 µL aliquot of standardized inoculum was added to each test well. Plates were sealed to prevent evaporation and incubated at 37 ± 2 °C for 24 h.31–33
The MBC is defined as the lowest concentration of the test substance that results in microbial elimination ≥99.9% and no visible colony growth after subculture from wells at a concentration equal to or greater than the MIC. The determination of minimum bactericidal concentrations (MBC) followed the CLSI recommendations (M07-A9; M27-A3). After reading the MIC, 10 µL from wells containing a concentration equal to or greater than the MIC were spread onto Mueller–Hinton agar plates and incubated under appropriate conditions. The lowest concentration at which no colony growth was recorded is the MBC. 34
Screening the Effect of AhaEO and Antibiotic Combinations
The potential interaction between AhaEO and selected antibiotics was evaluated using a modified disk diffusion method. The assay was performed against the reference strains
To determine whether the combination of antibiotics and essential oil produced synergistic or antagonistic effects, 10 µL of AhaEO was injected into antibiotic discs, which were then placed on the agar surface. The zones of inhibition generated by the AhaEO combined with antibiotics indicated synergism if the zone of combination > zone of AhaEO + zone of the relevant antibiotic, additive if the zone of combination = zone of AhaEO + zone of the relevant antibiotic, and antagonism if the zone of combination < zone of AhaEO + zone of the relevant antibiotic. 37
ADMET for Artemisia Herba-alba Bioactive Compounds
The drug-likeness, physicochemical properties, and the absorption, distribution, metabolism, excretion, and toxicity (ADMET) properties for the compounds found in A. herba-alba were assessed using the SwissADME38,39 and pkCSM tool. 40 The canonical SMILES for these compounds were obtained from the PubChem database. 41 These canonical SMILES were then input into the Swiss ADME online tool to evaluate their physicochemical and drug-likeness properties. Furthermore, we evaluated the ADMET properties and toxicity for all the identified compounds using the pkCSM pharmacokinetics web tool. (https://biosig.lab.uq.edu.au/pkcsm/).
Molecular Docking
The molecular docking analysis of the bioactive compounds was performed using AutoDock Vina 1.2.0 software.42,43 The three-dimensional crystal structure of the target CTX-M-15 extended-spectrum β-lactamase in complex with avibactam (PDB ID 4HBU) was downloaded from the Protein Data Bank in PDB file format. 44 , 45 Before docking, the target was prepared by removing water molecules and small molecules, and adding the polar hydrogen and charges to the structure. Additionally, the ligand structures were retrieved from the PubChem database as a single file in a 3D spatial data file (SDF) format. 41 The ligand SDF files were converted to PDB files using the open babel tool. 46 Ligand preparation was done by adding polar hydrogen and Gasteiger charges and defining the rotatable bonds. Subsequently, the prepared protein and ligands were saved in PDBQT file format. The grid box was adjusted to cover the active site residues that include CYS69, SER70, LYS73, ASN104, TYR105, SER130, ASN132, ASN170, THR216, LYS234, THR235, GLY236, SER237, and GLY238 to ensure the accurate docking of the ligands within the inhibitor binding sites. 45 A grid box with size 60 Å × 60 Å × 60 Å3 was created on the protein. The values −6.643882, −2.634176, and 11.573353 were used for the x, y, and z position coordinates. The molecular docking was then performed by setting the exhaustiveness to 8 for more precise and accurate docking scores. Further, the docking score for all the compounds and the reference drug Avibactam was noted. The protein-ligand conformations were analyzed using the PyMOL Molecular Graphics System, Version 3.0, Schrödinger, LLC. Additionally, the ligand interactions were generated using BIOVIA Discovery Studio Visualizer (v21.1.0.20298; Dassault Systèmes, 2020).
Statistical Analysis
All inhibition zones (mm) were measured in triplicate (n = 3). Data are expressed as mean ± standard deviation (SD). Statistical analyses were performed using JASP 0.95.4 software. 47 Differences among treatments were evaluated by one-way ANOVA followed by Tukey's post-hoc test, with p < 0.05 considered significant.
Results
Extraction Result and Chemical Composition of AhaEO
The yield of AhaEO was computed as given in the subsequent expression: ER(%) = (AhaEOM/AhaM) x 100, in which AhaEOM represents AhaEO mass and AhaM is the mass of the dried
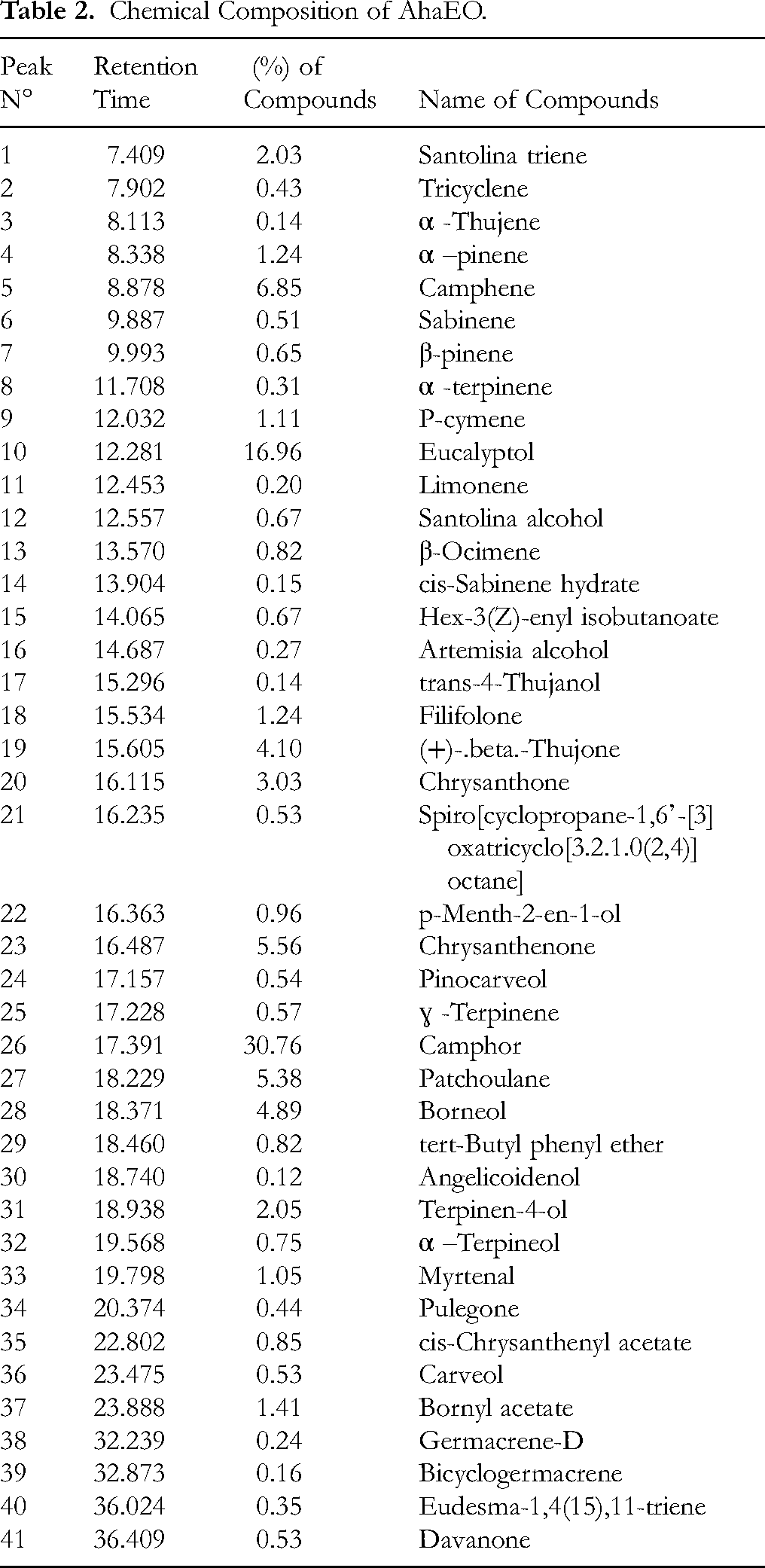
The AhaEO was analyzed by gas chromatography coupled with mass spectrometry (GC-MS). The chromatographic profile revealed the presence of 41 components, collectively representing 100% of the essential oil (Figure 1; Table 2). The composition was dominated by camphor (30.76%) and eucalyptol (16.96%). The other constituents found in significant proportions included camphene (6.85%), chrysanthenone (5.56%), patchoulane (5.38%), borneol (4.89%), β-thujone (4.10%), chrysanthone (3.03%), terpinen-4-ol (2.05%), santolina triene (2.03%), bornyl acetate (1.41%), filifolone (1.24%), alpha-pinene (1.24%), p-cymene (1.11%), and myrtenal (1.05%).

AhaEO chromatogram.
Chemical Composition of AhaEO.
Antibacterial Activity
The evaluation of AhaEO's antibacterial efficiency revealed that it exhibited varying activity against all the pathogens tested (Table 3). A significant effect was noticed against Ec01, Ec02, and Kp, with inhibition zones of 11.0 ± 1.0, 13.33 ± 4.16, and 13.0 ± 1.73 mm, respectively. Sa01 and Sa02 were less sensitive, with inhibition zones of 9 ± 1 and 7.33 ± 1.53, respectively, while Pa01 and Pa02 showed total resistance. The MIC and MBC values revealed a varied response among the tested bacteria. Ec01 was the most susceptible, as shown by the lowest MIC and MBC (3.56 mg/ml). However, all other strains have higher MICs, ranging from 7.12 to 113.91 mg/mL, and MBCs, ranging from 28.48 to 455.65 mg/mL. These results highlighted a strain-dependent sensitivity to AhaEO.
Mean of Inhibition Zone Diameter, MIC, and MBC of the AhaEO Tested Against Different Bacterial Strains.

The Effect of AhaEO and Antibiotics Combinations
Using the disc diffusion method, the antibacterial activity of AhaEO combined with antibiotics TOB, CN, IMP, CAZ, AX, COT, and TIC was tested against multiple bacterial strains. The effects of combinations were classified as synergistic, additive, or antagonistic. Differential results were revealed depending on the antibiotic and the bacterial strain, and antagonism was the most frequent outcome; the results are summarized in Table 4.
Mean of Inhibition Zone Diameter of AhaEO and Antibiotics, Alone and in Combination, Against Different Bacterial Strains.
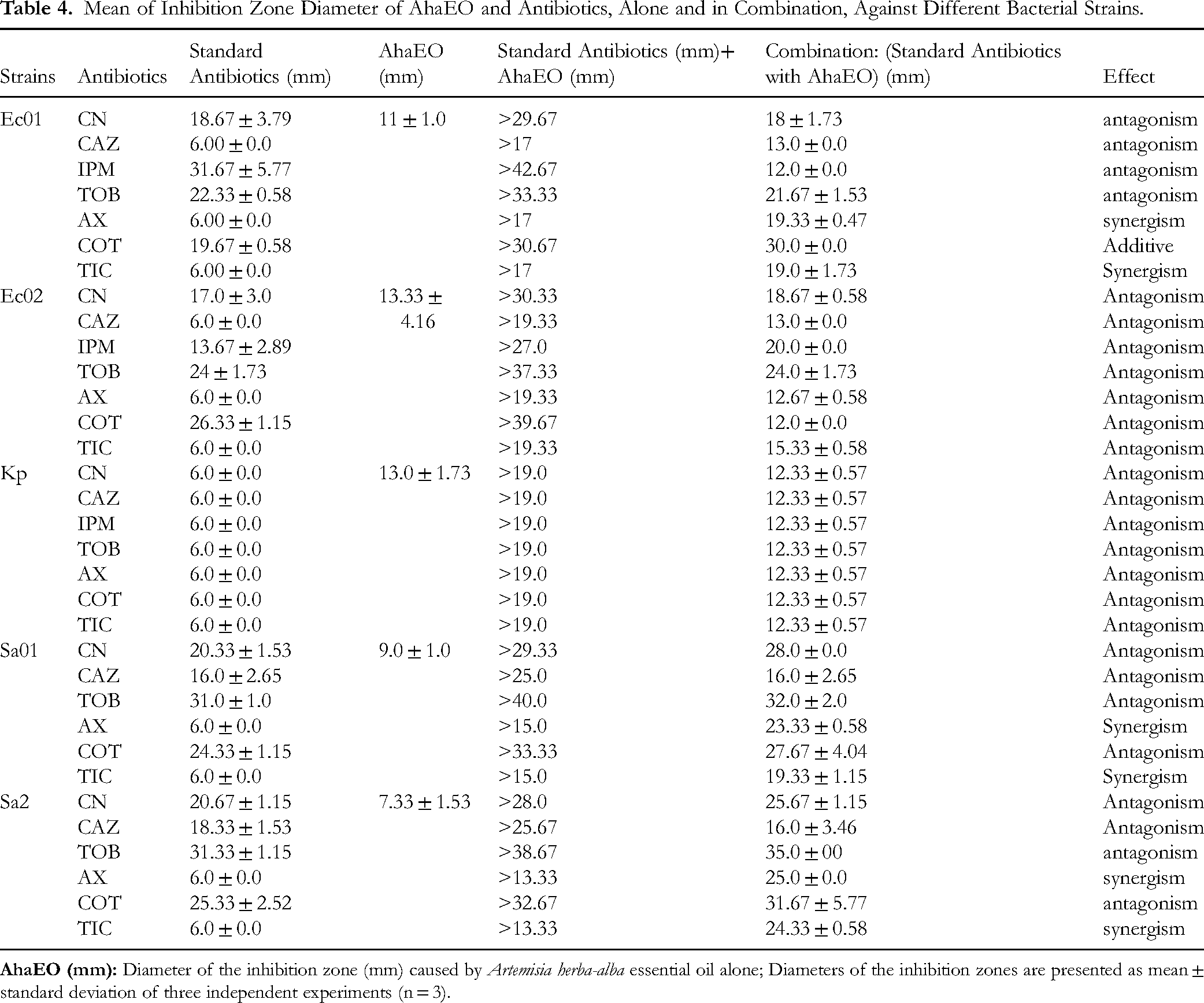
The synergistic effect was mainly observed with antibiotics AX and TIC against Ec01, Sa01 and Sa02, indicating that the presence of AhaEO enhances the antibacterial activity of these antibiotics.
ADMET Analysis
The physicochemical properties, solubility, lipophilicity, and violations of the Lipinski rule of five, as well as human oral bioavailability, were evaluated for all compounds using the SwissADME tool. The assessed parameters included molecular weight (MW), molecular refractivity (MR), the number of hydrogen bond acceptors (HBA), the number of hydrogen bond donors (HBD), and the topological polar surface area (TPSA). All seven compounds were predicted to have a MW between 150-500 g/mol, HBA values of ≤ 10, HBD values of ≤ 5, and MR values within the range of 40-130 Å2. Furthermore, the compounds fell within the acceptable range for bioavailability, with a value of 0.55, and they had a maximum of one violation of the Lipinski rule of five. The chemical structures of compounds identified in

Two-dimensional chemical structures of the phytochemicals identified from the AhaEO.

Bioavailability radar plot for the phytochemicals identified from the AhaEO. The colored zone is the acceptable range for each parameter. LIPO denotes (Liphophilicity), XLOGP3 between −0.7 and +5.0, SIZE (MW) 150 and 500 g/mol, TPSA between 20 and 130 Å2, INSOLU (Insolubility) log S not higher than 6, INSATU (Insaturation) sp3 hybridization not less than 0.25, and FLEX (Flexibility) no more than 9 rotatable bonds.
To evaluate the ADMET profile of all the compounds, the pkCSM pharmacokinetics web tool was used.
The absorption properties include Caco2 permeability, Intestinal absorption (%), Skin Permeability, and P-glycoprotein I inhibitor. All the compounds were predicted to have acceptable Caco-2 and skin permeability and exhibited high intestinal absorption of more than 95%. The volume of distribution (VD) parameter was evaluated to predict how extensively a drug distributes throughout the body and the total dose required to achieve tissue concentrations similar to those in plasma. Compounds with a log VDss value less than −0.15 L/kg are considered to have a low distribution, whereas those with a VDss value greater than 0.45 L/kg are categorized as having a high distribution. The VDss predictions for the seven compounds from
Metabolism predictions provide valuable insights into a compound's interaction with cytochrome P450 (CYP) enzymes, which are crucial for drug metabolism in the body. The results revealed that all compounds do not show any inhibition against the drug metabolizing enzyme. In the case of compounds, Eucalyptol, and Camphene was shown as an inhibitor for the CYP2C19 enzyme. Additionally, the other compounds are predicted to be non-inhibitors for cytochrome P450 enzymes.
Total clearance and Renal OCT2 substrate status provide insights into how compounds are excreted from the body. This excretion rate was evaluated using the total clearance values (log ml/min/kg) for all seven compounds and showed a rate from 0.04 to 1.0, which indicates variability in their elimination rates. Additionally, Camphene showed the lowest rate of 0.04 log ml/min/kg. All compounds present in
The toxicity profiles of the bioactive compounds from the plant were predicted to ensure a safe profile for their potential therapeutic use. These predictions provide significant information on possible adverse effects. The results indicated that none of the compounds studied showed AMES toxicity, and were non-inhibitors of the hERG I and II channels, suggesting their non-mutagenic potential and lower risk of cardiotoxicity. Additionally, they were predicted to be non-hepatotoxic, revealing their safer profile for liver function. The ADMET properties for all compounds in
Molecular Docking Analysis
Based on the antibacterial effect of the AhaEO, we investigated whether the phytochemicals in the plant could interact and inhibit their action, similar to avibactam. To identify these compounds, molecular docking analysis was conducted by targeting β-lactamase.
The molecular docking of the

The diagramshows the surface view of β-lactamase bound to the ligand (A) patchoulane (yellow)and (B) avibactam (control) (violet) shown as sticks. Protein-ligand interaction diagrams show the hydrogen bonds, π-alkyl, alkyl, salt bridge, and van der Waals interactions.
The Docking Scores for the Artemisia Herba alba Compounds and Reference Drug Avibactam with β-Lactamase (PDB ID: 4HBU).

Discussion
In this study, AhaEO was extracted using a Clevenger-type apparatus. The calculated yield, based on the dry plant material weight, was 1.41 ± 0.22%. This yield was higher than that reported for another Algerian species harvested in November 2022 (0.99%).
21
In Morocco, the reported yield was 1.05 ± 0.02%.
13
Meanwhile,
The obtained oil was analyzed for its chemical constituents by GC-MS, using a fused-silica Rxi®-5 ms capillary column, which has a predominantly non-polar stationary phase. The results indicated the predominance of camphor (30.76%) and eucalyptol (16.96%). Our findings were qualitatively in agreement with those reported in the literature.13,15,17,18,24,49–63 However, the composition of our essential oil differed quantitatively. Several studies have reported that camphor.13,15,49–55 was the major component in AhaEO, while other studies on the chemical composition of AhaEO collected from other localities showed that α-Thujone was the predominant component.17,18,56 Notably, chrysanthenone was previously identified in oils from Algeria, Libya, and Morocco by Ouchelli et al, 57 Janaćković et al, 58 and Aljaiyash et al, 59 respectively. A study conducted by Qnais et al in Jordan revealed a different profile, characterized by β-thujone as the main compound. 60 According to Zouari et al, 61 and Houti et al, 63 cis-chrysantenyl acetate was the dominant compound in AhaEO. Zaher et al reported that the primary compound is davana ether. 24 In addition, Abu-Darwish et al reported a different chemotype with 1,8-cineole as the main component. 62
Although all previous studies, including ours, focused on the same species (
This study presents an interdisciplinary evaluation of the antibacterial efficacy of AhaEO, tested independently and in combination with standard antibiotics.
The inhibitory observed for AhaEO is consistent with that documented by Benamar-Aissa et al, who investigated the same essential oil from Laghouat, Algeria. They reported inhibition zones of 10.20 mm against
Moreover, MIC and MBC values are almost identical to those reported by Janackovic et al against
The level of microbial susceptibility to an essential oil depends on its specific chemical profile and on the type of microbe being inhibited. Generally, essential oils demonstrate higher antibacterial effectiveness against gram-positive bacteria compared to gram-negative varieties. This resistance in gram-negative organisms is often attributed to a complex outer membrane that blocks the entry of hydrophobic substances. Moreover, the overall potency of the essential oils is not solely dependent on their major compounds. It results from complex interactions between its various constituents. Ultimately, the minor constituents within an essential oil often play a crucial role in its antibacterial effectiveness.9,33
There is growing interest in the ability of essential oils to enhance the effectiveness of antibiotics, particularly against resistant Gram-negative bacteria.
A synergetic effect was observed for AhaEO in association with cefoxitin against methicillin-resistant
Another study reported a synergistic effect of AhaEO combined with tetracycline, pristinamycin, ofloxacin, and nitrofurantoin antibiotics against the
Amoxicillin and ticarcillin are a beta-lactam antimicrobial drugs belong to the penicillin family of antibiotics and are effective against several kinds of infections. 67
The synergistic interactions identified between AhaEO and AX and TIC in this study may stem from a shared effect of the various modes of action of antibiotics and the bioactive compounds found in the essential oil. This collaborative effort may enhance cell permeability, facilitating greater antibiotic absorption by bacterial cells, or disrupt bacterial resistance mechanisms, such as plasmids and antibiotic-degrading enzymes.68–70
ADMET properties evaluate the pharmacokinetics of a drug by focusing on predicting how a drug behaves in the body. It assesses how effectively the drug is absorbed, distributed, metabolized, and excreted when orally administered. Furthermore, poor ADME can potentially lead to toxicity. 71
ADMET analysis revealed that the tested compounds fall within established drug-likeness criteria, indicating their suitability for further investigation. Predicted toxicity results showed a favorable safety profile for all tested compounds. Nevertheless, experimental pharmacokinetic and toxicity studies are needed to confirm these results.
The present study selected β-lactamase as the target for molecular docking analysis due to its crucial role in bacterial resistance to β-lactam antibiotics. β-lactamases are enzymes that can hydrolyze the β-lactam ring, a significant structural feature of β-lactam antibiotics. This hydrolysis reduces the antibacterial efficacy of these antibiotics and considerably exacerbates the escalating problem of antibiotic resistance. 72 Inhibiting this enzyme is a crucial strategy for addressing resistance and restoring the efficacy of antibiotics. In addition, we have used avibactam (crystallised ligand), a non-β-lactam β-lactamase inhibitor, as a reference compound in this study because of its clinically proven ability to inhibit a wide range of β-lactamases and enhance the activity of β-lactam antibiotics against resistant strains. 73
A previous study on the mechanism of avibactam on class A CTX-M-15 extended-spectrum β-lactamase reported that avibactam forms multiple hydrogen bonds, blocking the hydrolysis of β-lactam antibiotics. It forms hydrogen bonds with LYS73, SER130, GLU166, ASN132, and ASN170, polar and van der Waals contacts with residues,and with TYR105, contributing to its strong inhibitory property. 45
The docking results showed that compounds from AhaEO may interact with β-lactamase. Patchoulane and chrysanthone formed stable interactions with β-lactamase outside the active site. These findings required more experimental confirmation.
Limitations and Future Perspectives
The investigations into the interaction between EOs and antibiotics will provide an understanding of their combined effects. In some instances, the findings may indicate a synergistic relationship that enhances antibacterial activity, which could translate to smaller antibiotic dosages with possibly fewer side effects. It could also point to antagonistic effects by which the treatment may be weakened, drawing attention to a deliberate choice of EO–antibiotic combination. In addition, studies of the mechanism of such interactions can offer valuable input for future in vivo applications. On the other hand, there are also drawbacks associated with variable composition according to the source plant, harvest time, and extraction methodology; poor solubility may require solvents that could interfere with bacterial growth; concentration could be difficult to standardize; and findings may often differ between bacterial strains. Moreover, there is not always a direct translation from results obtained in vitro to the clinical setting, and a lack of a standardized protocol can make comparison between studies difficult.
Conclusion
In this study, the essential oil extracted by hydrodistillation from
Supplemental Material
sj-docx-1-npx-10.1177_1934578X261432720 - Supplemental material for Chemical Profiling, Antibacterial Activity in Combination with Antibiotics and in-Silico Investigation of β-Lactamase Inhibitory Potential of Artemisia Herba alba Essential oil
Supplemental material, sj-docx-1-npx-10.1177_1934578X261432720 for Chemical Profiling, Antibacterial Activity in Combination with Antibiotics and in-Silico Investigation of β-Lactamase Inhibitory Potential of Artemisia Herba alba Essential oil by Turkiya Touhami, Khadidja Houda Benabed, Fatima Zohra Guellouma, Hadjer Boussoussa, Haritha Kalath, Ihcen Khacheba, Mohamed Yousfi, Muhammad Suleman and Abdullah Chaito in Natural Product Communications
Footnotes
Abbreviations
Acknowledgements
We extend our sincere thanks and appreciation to all the staff at the Central Laboratory of Akid Lotfi Hospital in Laghouat for their valuable efforts in collecting reagents and operating various types of equipment.
Ethical Approval
Ethical Approval is not applicable for this article.
Author Contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
All relevant data are included in the manuscript. Additional details or raw data can be provided by the corresponding author upon reasonable request.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
