Abstract
Introduction
It is recognizable that
Results and Discussion
Chemical Analysis
Chemical compounds identified in the rhizome oil and their percentages are tabulated in Table 1. In general, 36 compounds were identified, which accounted for 98.0%. The main chemical classes were monoterpene hydrocarbons (62.5%) and their oxygenated derivatives (29.0%). Sesquiterpene hydrocarbons, oxygenated sesquiterpenes, and nonterpenic compounds were present in this essential oil at 1.5%, 1.6%, and 3.4%, respectively. 1,8-Cineole (29.0%), geraniol (15.2%), β-pinene (12.8%), α-pinene (7.5%), and myrcene (6.6%) could be seen as the major compounds in
Chemical Compositions in

Bold: major compounds; compounds were identified based on RI and MS comparisons.
Retention time (Rt).
Retention indices (RIE) relative to
Retention indices (RIL) from Adams’ book 23 and NIST standard database. 24
A total of 45 compounds were identified in the leaf oil, which represented 95.3% (Table 1). Monoterpene hydrocarbons (60.4%) and their oxygenated derivatives (18.1%) were still the two main chemical classes. In the meantime, sesquiterpene hydrocarbons, nonterpenic compounds, and oxygenated sesquiterpenes were found to achieve 8.7%, 4.7%, and 3.2%, respectively. The principal compounds consisted of 1,8-cineole (25.7%), β-pinene (21.3%), geraniol (9.0%), and α-pinene (6.1%). Obviously, in contrast to β-pinene, the percentages of 1,8-cineole, α-pinene, and geraniol in the leaf oil were lower than those in the rhizome oil. Some other compounds also reached a significant amount in the leaf oil, comprising linalool (4.4%), myrcene (3.7%), α-terpineol (2.8%), limonene (2.7%), β-caryophyllene (2.0%), and β-sesquiphellandrene and δ-cadinene (1.0%, each).
Various compounds, such as nonanal, δ-terpineol, methyl chavicol, (2
Cytotoxicity and α-Glucosidase Inhibition
In the cytotoxic assay, both tested oil samples showed activity against four cancer cell lines KB, Hep-G2, Lu-1, and MCF-7 with the IC50 values ranging from 82.39 to 216.46 µg/mL (Table 2), when ellipticine was used as a positive control (IC50 0.29-0.40 µg/mL). At the highest concentration of 500 µg/mL, the inhibitory percentages for the two samples were more than 80% (Table 2). Among the obtained results, the leaf oil exhibited the best effect against Lu-1 and MCF-7 with the IC50 value of 82.39 and 84.05 µg/mL, respectively.
Cytotoxicity of Tested Essential Oil Samples.

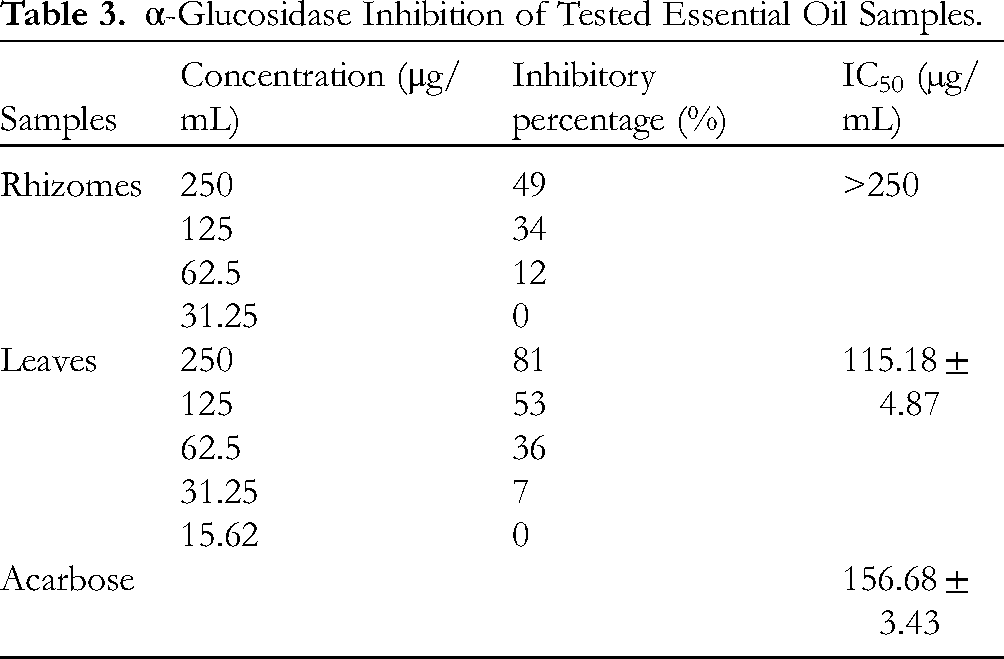
Two essential oil samples were further submitted to α-glucosidase inhibitory assay. As shown in Table 3, the leaf oil with the IC50 value of 115.18 ± 4.87 µg/mL is better than the standard acarbose (IC50 156.68 ± 3.43 µg/mL). However, the rhizome oil was inactive (IC50 > 250 µg/mL). At the highest concentration of 250 μg/mL, the inhibitory percentage of the leaf oil is two times higher than that of the rhizome oil. This matches well with previous reports, eg,
α-Glucosidase Inhibition of Tested Essential Oil Samples.
Molecular Docking Study
It suggests that the major compounds 1,8-cineole, geraniol, α-pinene, and β-pinene were mainly responsible for the α-glucosidase inhibitory activity of

The major compounds in essential oils of
AutoDock Vina v1.2.3 was utilized to conduct the molecular docking study.18,19 The protein’s active site was identified by selecting residues of the catalytic triad. Then, the validated docking method was used to analyze the binding ability of the chosen compounds over the enzyme active site pocket, as compared with that of the standard compound acarbose.18,19 We used redocking of (3

Superimposition of the native (cyan) and docked (magentas) of ligand (3
1,8-Cineole exhibited highly favorable adaptability of the docked molecule within the active site of the α-glucosidase enzyme (PDB ID: 3WY1) with a binding affinity of −5.429 kcal/mol (Table 4). Numerous noncovalent interactions between the docked molecule and active site residues can be observed upon visual analysis. Hydrophobic interactions with Phe166, Ile146, Tyr389, Phe147, and Phe206 were identified (Figure 3). Geraniol induced a stable receptor–ligand complex with a binding affinity of −5.752 kcal/mol. This complex was subjected to a more in-depth analysis to assess the molecular occupancy potential within the enzyme’s active site. Among the eight studied conformations, the best docking pose revealed various interactions with four different amino acid residues of the target enzyme. The docked ligand can be observed to engage in pi–sigma interactions with Tyr65 and pi–alkyl interactions with His332, Phe206, and Ile146 (Figure 4). Also, geraniol established hydrogen bonding with His105 and Asp62, similar to the positively controlled acarbose compound (Figures 4 and 7). α-Pinene occupied the active site pocket of the receptor protein with a binding affinity of −5.768 kcal/mol. Analysis of the best docking pose for α-pinene revealed interactions with several amino acid residues, including pi–alkyl and alkyl interactions with Tyr389, Phe297, Phe147, Ile146, and Phe166 (Figure 5). In the final complex of β-pinene and α-glucosidase enzyme (Figure 6), multiple intermolecular interactions were formed between the docked molecule and the active amino acid residues of α-glucosidase. These interactions included pi–alkyl interactions with four amino acids: Phe297, Phe206, Ile146, and Phe166 (Figure 7).

Predicted interaction mode for 1,8-cineole with α-glucosidase enzyme. (A) 2D diagram of 1,8-cineole interacting with amino acid residues. (B) 3D view of ligand–receptor interactions. (C) The 3D hydrophobicity surface map comparing substrates and α-glucosidase. (D) View of the active site gorge from the top, displaying 1,8-cineole bound to the observed amino acid residues of α-glucosidase as cyan sticks.

Predicted interaction mode for geraniol with α-glucosidase enzyme. (A) 2D diagram of geraniol interacting with amino acid residues. (B) 3D view of ligand–receptor interactions. (C) The 3D hydrophobicity surface map comparing substrates and α-glucosidase. (D) View of the active site gorge from the top, displaying geraniol bound to the observed amino acid residues of α-glucosidase as cyan sticks.

Predicted interaction mode for α-pinene with α-glucosidase enzyme. (A) 2D diagram of α-pinene interacting with amino acid residues. (B) 3D view of ligand–receptor interactions. (C) The 3D hydrophobicity surface map comparing substrates and α-glucosidase. (D) View of the active site gorge from the top, displaying α-pinene bound to the observed amino acid residues of α-glucosidase as cyan sticks.

Predicted interaction mode for β-pinene with α-glucosidase enzyme. (A) 2D diagram of β-pinene interacting with amino acid residues. (B) 3D view of ligand–receptor interactions. (C) The 3D hydrophobicity surface map comparing substrates and α-glucosidase. (D) View of the active site gorge from the top, displaying β-pinene bound to the observed amino acid residues of α-glucosidase as cyan sticks.

Predicted interaction mode for acarbose with α-glucosidase enzyme. (A) 2D diagram of acarbose interacting with amino acid residues. (B) 3D view of ligand–receptor interactions. (C) The 3D hydrophobicity surface map comparing substrates and α-glucosidase. (D) View of the active site gorge from the top, displaying acarbose bound to the observed amino acid residues of α-glucosidase as yellow sticks.
The Binding Affinity of Major Compounds and Their Potential Molecular Interactions With Amino Acid Residues.

It is also found that β-pinene exerted the best binding affinity of −5.778 kcal/mol, whereas the binding affinities for α-pinene, geraniol, and 1,8-cineole were −5.768, −5.752, and −5.429 kcal/mol, respectively (Table 4). It is worth noting that these values were comparable with that of acarbose (−5.767 kcal/mol).
Materials and Methods
Plant Materials
Hydrodistillation
The fresh rhizome powder (1.0 kg) was hydrodistilled using the Clevenger apparatus for 3.0 h in three replicates. Three distillates were combined, to afford a yellow essential oil. This product was dried over anhydrous Na2SO4 and kept in small vials at −5 °C for further analyses. The same procedure was assigned to the fresh leaf powder. The extraction yields for rhizomes and leaves, which were estimated from the fresh materials, achieved 0.24% and 0.19%, respectively.
The Gas Chromatography–Flame Ionization Detection/Mass Spectrometry Analysis
The gas chromatography–flame ionization detection (GC–FID) analytical procedure was performed as described in our previous reports6,21,22: including HP-5MS column (30 m × 0.25 mm, film thickness 0.25 m, Agilent Technologies), the gaseous carrier He (1.0 mL/min), injector's temperature 270 °C, detector's temperature 280 °C, column temperature establishment: 60 °C (4 min hold), increase to 240 °C (5 °C/min), 240 °C (9 min hold), inlet pressure of 5.0 kPa, split ratio of 9:1, and 1.1 µL injection volume.
The same setup was used for the GC–mass spectrometry (MS) analysis: HP-5MS column (30 m × 0.25 mm, film thickness 0.25 µm, Agilent Technologies HP 7890A Plus Chromatograph), HP 5973 MSD mass detector, the gaseous carrier He (1.0 mL/min), the MS ionization voltage of 70 eV, emission current of 40 mA, acquisition range of 50 to 500 amu, and sampling rate of 1.0 scan/s. The retention index (RI) values were obtained by coinjecting a reference homologous series of
Cytotoxic and α-Glucosidase Inhibitory Assays
Experimental assays were carried out following our previous reports.18,19,25‐27
Molecular Docking Study
To conduct docking research, the AutoDock Vina v1.2.3 program was utilized, which is one of the rapid and accurate computational programs used.18,19 These chemical structures of the studied compounds were optimized using the MMFF94s force field. 28 The structure of α-glucosidase enzyme was obtained from the RCSB Protein Data Bank and prepared according to previous research. 17
For the docking simulation, the parameters included the grid center coordinates based on the active site region of the α-glucosidase enzyme (
Conclusions
For the first time, the present study reports chemical compositions in the rhizome and leaf oils of
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231206280 - Supplemental material for Essential Oils of Alpinia vietnamica Rhizomes and Leaves: Chemical Composition, Cytotoxicity, α-Glucosidase Inhibition, and Molecular Docking Approach
Supplemental material, sj-docx-1-npx-10.1177_1934578X231206280 for Essential Oils of
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financially supported by a grant from Vietnam Academy Science and Technology (VAST04.04/22-23).
Ethical Approval
Not applicable.
Statement of Human and Animal Rights
Not applicable. This article does not contain any guidelines followed for performing experimental procedures.
Statement of Informed Consent
Not applicable. This article does not contain any studies with human or animal subjects.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
