Abstract
Objectives
The current study evaluated the protective mechanism of Danshensu and Notoginsenoside R1 combination (DR1) on obesity from the perspective of gut microbiota and short-chain fatty acids (SCFAs).
Methods
Male C57BL/6J mice were fed either a normal diet or a high-fat diet (HFD). DR1 (60 mg/kg) was administered to both normal and HFD-induced obese mice for 12 weeks. The obese phenotype and pharmacodynamics effects were assessed through hematoxylin-eosin staining of epididymal fat, liver, small intestine, and colon tissues, as well as serum biochemical detection. Moreover, 16S ribosomal ribonucleic acid (16S rRNA) sequencing was employed to profile the taxonomy and function of gut microbiota, while SCFAs were quantified using ultra-performance liquid chromatography coupled with mass spectrometry methods.
Results
Compared to HFD group, the tissue lesions were significantly alleviated by DR1 treatment in obese mice, accompanied by the significant reduction of average body, epididymal fat and liver weights, as well as the normalization of insulin sensitivity and glucose homeostasis (P < .05, P < .01 or P < .001). 16S rRNA sequencing analysis showed that DR1 treatment potently ameliorated HFD-induced dysbiosis of gut microbial composition and function. Notably, DR1 reversed the ratio of Firmicutes/Bacteroidetes to normality. Moreover, DR1 improved intestinal barrier structures and enhanced SCFAs concentrations in the caecal contents (especially propionate and butyrate).
Conclusion
These results suggested that DR1 could reverse the gut microbial dysbiosis and increased SCFAs levels in HFD-induced obese mice, thereby ultimately supporting the anti-obesity value of DR1.
Introduction
The rapid advancement of contemporary society has exerted a profound impact on our dietary patterns and lifestyles. The exponential surge in the prevalence of obesity and other cardiometabolic disorders have emerged as the most critical public health concern.1,2 Intense research into the development of more effective and safer pharmaceutical interventions holds the potential to significantly benefit the obesity, thereby improving the overall health and well-being of society.
Chinese medicine, a form of Asian traditional medicine, has drawn worldwide interest in the life sciences and drug discovery. 3 Massive evidence has demonstrated the significant anti-obesity activities of active ingredients derived from Chinese herbal medicines (CHMs). 4 The herb pair of Salvia miltiorrhiza and Panax notoginseng can effectively promote blood circulation and alleviate blood stasis. A study revealed that this herb pair exhibited remarkable insulin resistance efficacy through the regulation of glucolipid metabolism. 5 Moreover, we previously demonstrated that this herb pair could treat acute myocardial ischemia via regulating lipid and amino acids metabolisms. 6 These evidences strongly suggest that Salvia miltiorrhiza and Panax notoginseng herb pair have the potential for anti-obesity. The active components compatibility in herb pairs possess both the characteristic of complex formulae and the feature of simplicity to facilitate new drug research. 7 Danshensu and Notoginsenoside R1 combination (DR1) have been investigated in Han et al's patent for preparing medicaments to prevent and treat diseases caused by microcirculation disorder. 8 Danshensu is the main hydrophilic active ingredient of S. miltiorrhiza with various biological functions including lipidemia control, anti-inflammatory effects, inhibition of platelet aggregation, antioxidant and antithrombotic activities.9–11 Notoginsenoside R1, an unique saponin found solely in P. notoginseng, possesses cardioprotective activity through regulation of lipid metabolism/deposition,12,13 inflammation 14 and/or oxidative stress.15,16 However, no studies have reported on the anti-obesity efficiency of DR1 or its underlying mechanism.
In recent years, groundbreaking research has emerged elucidating the pivotal role of the gut microbiota in human health. This microbial community, inhabiting the intestinal tract, is acknowledged as the largest and most intricate “organ” within the body. 17 Moreover, the gut microbiota serves as an indispensable barrier against pathogenic bacteria by maintaining a harmonious balance within the gut ecosystem. This equilibrium is vital for preserving intestinal integrity and preventing the onset of leaky gut syndrome along with subsequent immune activation and inflammation. The gut microbiota plays a crucial role in nutrient digestion and absorption, while also contributing to essential vitamins and short-chain fatty acids (SCFAs) production. These metabolites subsequently exert a profound impact on host metabolism, influencing body weight regulation, energy homeostasis, and even cognitive function. A large body of evidence suggests that dysregulation of gut microbiota is associated with obesity,4,18 particularly involving two main phyla (Firmicutes and Bacteroidetes). Notably, the gut microbiota is emerging as a crucial mediator of the efficacy of CHMs. CHMs have the potential to modulate the composition of gut microbiota, leading to an increase in the production of representative metabolites such as SCFAs. These metabolites subsequently inhibit pathogen proliferation and reduce lipopolysaccharide and other harmful metabolites levels, thereby establishing a healthy ecosystem. SCFAs also play critical role in regulating glucolipid levels and inflammatory responses.1,4,19 Therefore, it is imperative to investigate the anti-obesity effect of DR1 by manipulating gut microbiota and SCFAs.
In this study, we investigated the underlying mechanism of DR1 on the taxonomic composition and functional capacity of gut microbiota via 16S ribosomal ribonucleic acid (16S rRNA) sequencing analysis and employed an ultra-performance liquid chromatography coupled with mass spectrometry (UPLC-MS) method for quantifying SCFAs concentrations.
Results
Effects of DR1 on Body Weight and serum Biochemical Indicators in Obese Mice
In Figure 1A, the average body weight of obese mice was significantly increased compared to control mice. Following DR1 treatment, the average body weight in obese mice showed a significant decrease (P < .001) along with a significant reduction in epididymal fat weight and fat area (P < .001) (Figure 1B and H). The liver weight of obese mice treated with DR1 exhibited a significant decrease compared to that of obese mice alone (P < .001). Hematoxylin-eosin (H&E) staining revealed that liver lipid droplet size was decreased after DR1 therapy, indicating its potential for reducing high-fat diet (HFD)-induced hepatic fat accumulation (Figure 1C and I). Additionally, DR1 significantly decreased fasting serum glucose (FSG), fasting serum insulin (FINS), and homeostatic model assessment of insulin resistance (HOMA-IR) levels in obese mice as compared to the Mod group, suggestive of its ability to ameliorate insulin resistance (P < .05) (Figure 1D-F). Moreover, the area under the curve (AUC) of oral glucose tolerance test (OGTT) showed a significant difference between the Mod-DR1 group and Mod group (P < .001), highlighting DR1's potential for improving impaired glucose tolerance in obese mice (Figure S1 and Figure 1G). However, no significant effects were observed on these aforementioned indexes in normal chow diet (NCD)-fed mice following DR1 treatment.
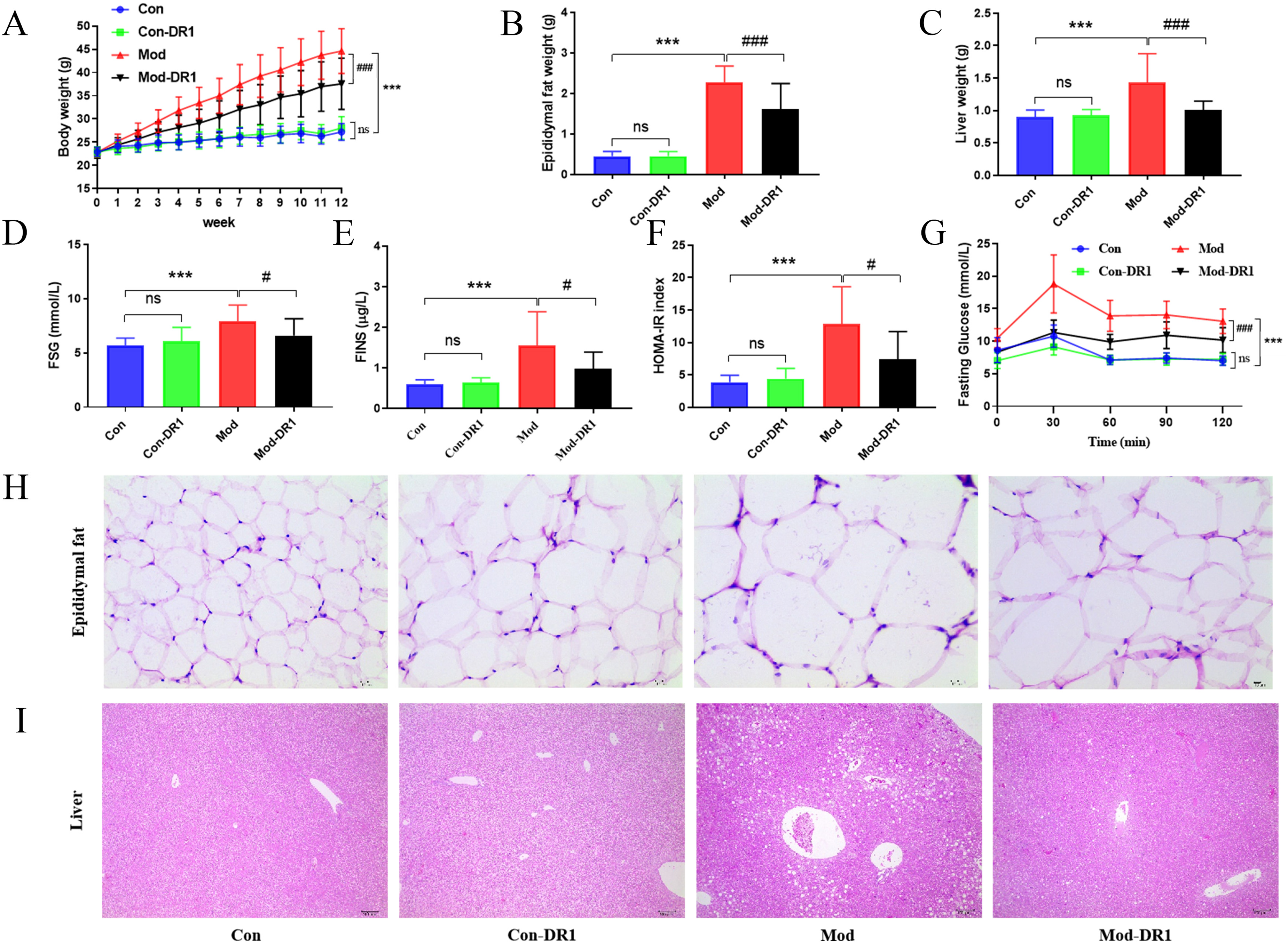
Effects of oral DR1 on body weight, insulin resistance and glucose homeostasis in obese mice. A: body weight; B: epididymal fat weight; C: liver weight; D: fasting serum glucose (FSG) concentration; E: fasting serum insulin (FINS); F: homeostatic model assessment for insulin resistance (HOMA-IR); G: oral glucose tolerance test (OGTT) shown by curves of fasting glucose concentrations. H, epididymal fat hematoxylin-eosin (H&E) staining (Magnification = 400×, scale bar, 10 μm); I, liver H&E staining (Magnification = 100×, scale bar, 100 μm). Data were expressed as mean ± standard deviation (SD) (n = 8∼10). Differences were assessed by using one-way ANOVA followed by Dunnett’s multiple comparisons test or Student's t-test or Mann-Whitney tests. *P < .05, ***P < .01, ***P < .001 compared with NCD group, #P < .05, ##P < .01, ###P < .001 compared with HFD group.
The Impact of DR1 on Serum Lipids and Inflammatory Response in Obese Mice
Obese mice may exhibit disruptions in the levels of serum lipids and pro-inflammatory cytokines compared to normal mice. We assessed the relative concentrations of these serum lipids and inflammatory cytokines in obese mice following treatment with DR1 (Figure 2). The Mod group exhibited significantly elevated levels of low-density lipoprotein (LDL), total cholesterol (TC), total triglyceride (TG) and interleukin (IL)-1β when compared to the Con group. Importantly, DR1 effectively reversed the concentrations of LDL, TC, TG and IL-1β in the Mod-DR1 group but not in the Con-DR1 group (P < .05). However, there was no significant difference in high-density lipoprotein (HDL) level between the Mod and Mod-DR1 groups. These findings suggest that DR1 has potential for regulating lipid metabolism and mitigating inflammatory response in obese mice.

DR1 influenced serum lipids and inflammatory response in obese mice. Relative expressions of LDL, TC, TG, HDL and IL-1β in serum. Data were expressed as mean ± SD (n = 8∼10). Differences were assessed by using one-way ANOVA followed by Dunnett’s multiple comparisons test or Student's t-test or Mann-Whitney tests. *P < .05, **P < .01, ***P < .001 compared with NCD group, #P < .05, ##P < .01, ###P < .001 compared with HFD group.
The Impact of DR1 on SCFAs Production and Intestinal Barrier Integrity in Obese Mice
Obesity is associated with impaired intestinal barrier integrity and gut microbiota, which may influence the production of SCFAs. 18 Concentrations of acetate, isobutyrate and hepatanoic acid were significantly lower in the Mod group compared to the Con group (P < .05), while propionate, butyrate, isovalerate and caproate showed a slight decrease between obese and normal mice. Treatment with DR1 significantly reversed the decrease in propionate, isobutyate, butyrate, isovalerate, caproate and hepatanoic acid levels in obese mice compared to the Mod group (P < .05, P < .01), DR1 also increased the level of valerate in obese mice (Figure 3A and Table S1). Additionally, H&E staining was employed to observe the morphology of the small intestine and colon. The results revealed more severe damage to the intestinal barrier structure in mice fed with HFD than those on a NCD, which was partially restored by treatment with DR1 (Figure 3B). Intervention with DR1 resulted in a more compact and orderly morphology of intestinal villi in the colons of obese mice compared to those in the Mod group. Villi height decreased while crypt depth slightly increased in the Mod group compared to the Con group, but these changes were restored by DR1 intervention. These findings suggest that DR1 can enhance gut microbial SCFA production, and improve intestinal barrier integrity in obese mice.

DR1 affected short-chain fatty acids (SCFAs) and intestinal barrier integrity in obese mice. A: SCFAs including acetate, propionate, isobutyrate, butyrate, isovalerate, caproate, hepatanoic acid and valerate in caecal content (n = 8∼10); B: small intestine and colon (Magnification = 100×, scale bar, 100 μm) (n = 6). Data were expressed as mean ± SD. Differences were assessed by using one-way ANOVA followed by Dunnett’s multiple comparisons test or Student's t-test or Mann-Whitney tests. *P < .05, **P < .01 compared with NCD group, #P < .05, ##P < .01 compared with HFD group, and ns represents no significant difference.
Effects of DR1 on Gut Microbiota in Obese Mice
DR1 can exert a broader regulatory role on gut microbial SCFA production, then we investigated the effects of DR1 on the gut microbial structure and composition. The intervention with DR1 did not significantly affect the α-diversity indices (Chao1, Simpson, etc) compared to the Con group (Figure S2), indicating that the different interventions did not result in significant changes in taxa richness.
20
However, as depicted in Figure 4A, the principal coordinate analysis (PCoA) analysis of β-diversity revealed significant differences between the Con and Mod groups. Notably, the distribution range of gut microbiota in the Mod-DR1 group exhibited a more regulated and concentrated pattern compared to other groups. The Veen diagram was employed for community analysis, revealing that the unique gut microbiotas in the Con group, Mod group, and Mod-DR1 group accounted for 22.7%, 20.52%, and 15.9%, respectively. These findings indicated alterations in gut microbial composition following HFD modeling, with similar changes observed after DR1 treatment (Figure 4B). Unweighted pair group method of arithmetic means (UPGMA) Clustering analysis focuses on characterizing the clustering of samples based on similarity. The hierarchical branch length of the unweighted UniFrac-based Clustering tree revealed significant differences in clustering between the Con and Mod groups, as well as between the Mod and Mod-DR1 groups, indicating that DR1 had a discernible impact on altering the gut microbial structure in obese mice (Figure S3). To further investigate the specific changes in the bacterial communities, the relative abundance of the predominant genus was compared across four groups, especially taxa responding to DR1 treatment. The representative bacteria from normal mice belonged to Bacteroidetes and Firmicutes, while the Mod and Mod-DR1 groups exhibited an expansion to include TM7, Proteobacteria, and Actinobacteria. At both phylum and genus levels, there was a significant increase in Firmicutes/Bacteroidetes (F/B) ratio in obese mice compared to normal mice; however, DR1 treatment significantly reversed the change in obese mice (P < .01) (Figure 4C-E

Effects of DR1 on the gut microbial structure and composition in obese mice. A: principal coordinate analysis (PCoA) of β-diversity; B: Venn diagram; C: the abundance of Firmicutes/Bacteroidetes; D and E: taxonomic composition of gut microbiota on the phylum and genus levels. *P < .05, **P < .01, ***P < .001 compared with NCD group, #P < .05, ##P < .01, ###P < .001 compared with HFD group, and ns represents no significant difference.
A two-dimensional ordering diagram based on PCoA using Bray-Curtis distance matrix was employed to explore functional unit differences among gut microbiota across each group. The Figure S5 demonstrated a partial overlap in the sample range among groups, with significant non-overlapping areas indicating distinct variations in the functional units of KEGG orthology. 22
The linear discriminant analysis effect size (LEfSe) analysis revealed the evolutionary relationship of different gut microbiota in each group (Figure 5A). At the genus level, a heat map was used to further elucidate the impact of DR1 treatment on the structure of gut microbiota. Figure 5B demonstrated a significant increase in relative abundances of Mucispirillum, Lactobacillus, and Bilophila in the Mod group. However, there was a marked decrease in relative abundances of Clostridium, Mucispirillum, and Alistipes in the Mod-DR1 group. Additionally, random forest analysis enabled effective and accurate classification of gut microbiota samples from each group by identifying complex nonlinear correlations between variables. 23 As shown in Figure 5C, multiple bacteria were significantly affected by HFD and DR1 treatment. For instance, Lactobacillus and Desulfovibrio exhibited substantial increases in obese mice while Blautia, Streptococcus and Helicobacter showed significant increases in the Mod-DR1 group. After examining the structural characteristics, Picrust2 software was employed to investigate the influence of gut microbiota on metabolic pathways. Figure 5D-E illustrated notable differences in abundance for Proteasome as well as Synthesis and degradation of ketone bodies among Con and Mod groups along with an alteration related to Toxoplasmosis pathway activation levels among these groups. The metabolic pathways observed within each group primarily focused on metabolism and genetic information processing, with metabolism related pathways accounting for the highest proportion.
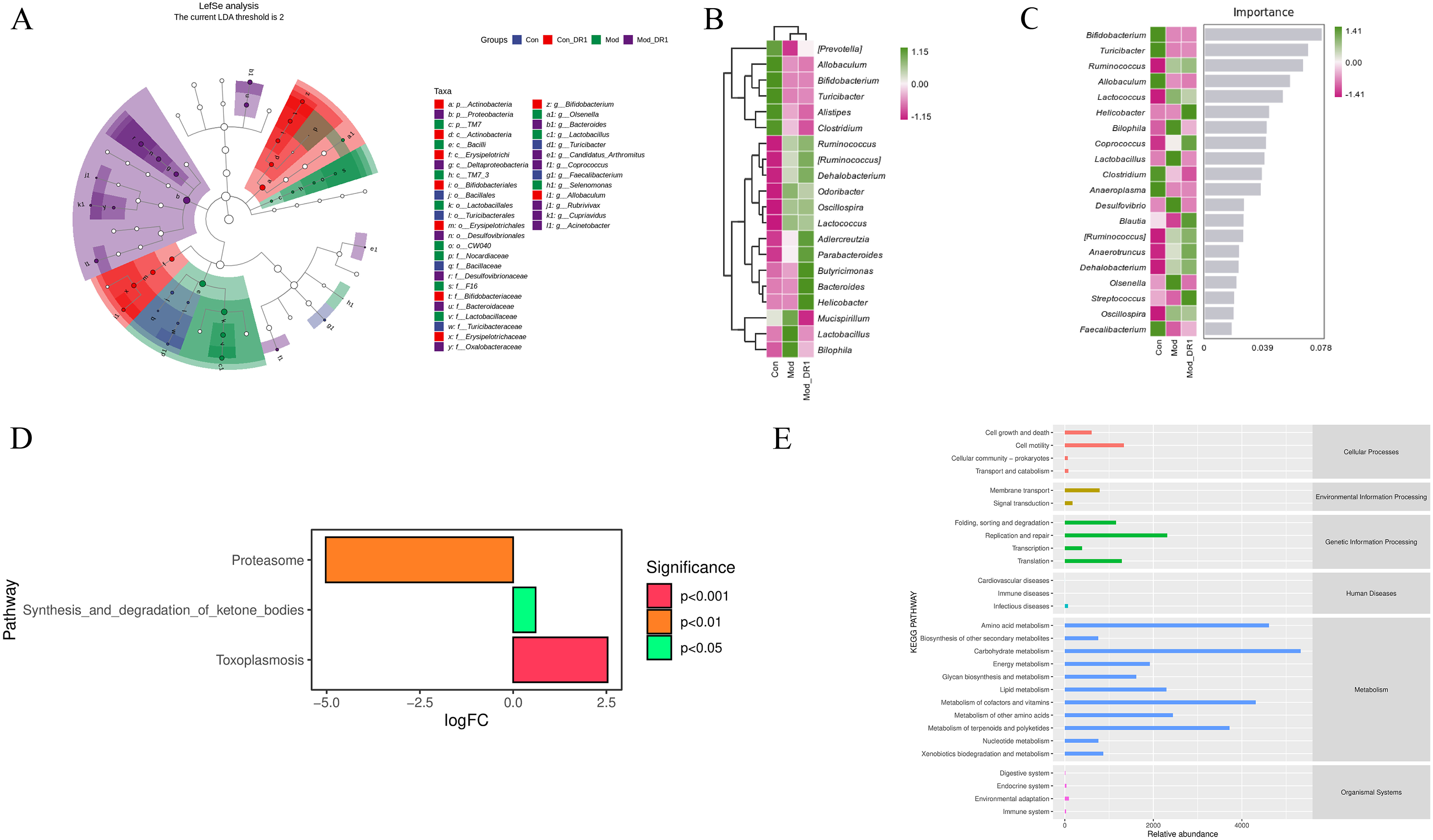
Effects of DR1 on the gut microbial characteristics and metabolic pathway in obese mice. A: linear discriminant analysis effects size (LEfSe) analysis of biomarker among groups (LDA threshold is 2); B: heat map of genus composition; C: random forest model of genus levels; D: the difference analysis of KEGG metabolic pathway between Con and Mod groups; E: the average abundance of metabolic pathways analysis (KEGG database) across all samples.
The Correlations Between Gut Microbiota, Phenotypes and SCFAs
The correlations between the relative abundances of several important bacteria, phenotypic indicators, and SCFAs were constructed to further elucidate the effects and mechanism of DR1 on obesity. Figure 6 illustrated that the abundance of Bacteroidetes was negatively correlated with FSG, TC, isovalerate. The abundance of Firmicutes was positively correlated with IL-1β, and negatively correlated with acetate, and heptanoic acid levels. Proteobacteria and Desulfovibrionaceae abundances showed positive correlations with butyrate, caproate, isobutyrate, isovalerate and valerate. Erysipelotrichaceae and Allobaculum abundances exhibited negative correlations with FINS and positive correlations with heptanoic acid. Lactobacillus abundance was negatively correlated with acetate, butyrate, heptanoic acid, isobutyrate, and propionate. Additionally, heptanoic acid and isobutyrate exhibited a negative correlation with FSG and TC, while caproate showed a negative correlation with TG and IL-1β (Figure S6). These findings suggested that DR1 may modulate gut microbiota as well as SCFAs levels to improve glucolipid metabolism and attenuate inflammatory response.

Pearson correlation integrating the gut microbiota, phenotypes and SCFAs (SPSS 22.0 software). * means P < .05, ** means P < .01. FSG: Fast serum glucose; FINS: fast serum insulin.
Discussion
The study aimed to investigate the effects of DR1 on glucolipid metabolism, inflammatory response, SCFAs, and gut microbiota to elucidate its potential mechanisms underlying weight loss. We observed that treatment with DR1 significantly reduced serum levels of inflammatory markers, glucose, lipids, and HOMA-IR in obese mice. Furthermore, DR1 improved colon morphology, regulated SCFA levels, and increased the Firmicutes/Bacteroidetes ratio. Gut microbiota play a crucial role in shaping SCFA production which modulates various metabolic pathways associated with obesity, insulin resistance, and type 2 diabetes. 24 Accumulating evidence suggests that metabolites generated by the gut microbiota are key signaling molecules in microbial communities, as well as in host-microbial cross-talk. 25 Previous research has demonstrated that SCFA-induced activation of the peroxisome proliferator-activated receptor (PPAR) pathway can regulate lipid metabolism by enhancing energy expenditure and reducing body weight. 26 SCFAs play critical roles in regulating host physiological function and intestinal homeostasis, particularly enhancing the gut barrier integrity. 20 Additionally, SCFAs have been demonstrated to stimulate intestinal gluconeogenesis (IGN) in enterocytes. 27 DR1 might impact the host's production of mucus, glycans, or other nutrients that serve as substrates for microbial fermentation, thereby influencing the gut microbiota composition and SCFA production. Our study found that feeding DR1 to HFD-induced obese mice significantly attenuated the reduction in fecal SCFAs in these mice, especially acetate, propionate and butyrate. Acetate functions as a signaling molecule in IGN metabolism and adipogenesis by inhibiting key basic and β-adrenergic receptors involved in adipocyte breakdown, thereby promoting insulin sensitivity. 28 Oral administration of acetate has been shown to confer beneficial effects on weight gain, TG levels, and postprandial glucose levels. 29 Furthermore, propionate and butyrate can regulate body weight, improve glucose tolerance, and enhance insulin sensitivity. The propionate compound demonstrates the ability to inhibit cholesterol synthesis. 30 Butyrate has the potential to mitigate metabolic disorders by activating the expression of AMP-activated protein kinase and glucose transporter 4 in adipose tissue, thereby reversing the gut microbiota imbalance caused by a HFD and stimulating lipoprotein biosynthesis.1,31 Additionally, butyrate reduces gut inflammation and ectopic lipid deposition while promoting mitochondrial adaptations that enhance fatty acid oxidation. 32 Also, we observed that DR1 treatment could improve the impaired barrier function of obese mice. Together, the mechanism of the anti-obesity action of DR1 was likely via stimulating the SCFAs-producing bacteria and subsequently increasing the SCFAs contents, leading to gut barrier restoration and insulin resistance reversal.
Furthermore, our results demonstrate that treatment with DR1 could optimize both the structure and function of gut microbiota in obese mice. This optimization is characterized by a decrease in Firmicutes abundance and an increase in Bacteroides abundance. The ratio of Firmicutes/Bacteroidetes is widely recognized as a hallmark of “obese gut microbiota”. 33 Many species within the Bacteroides genus possess polysaccharide-utilizing capabilities and produce fermentative end products that provide nutrients and other beneficial properties for hosts. 4 By influencing metabolic activities such as carbohydrate metabolism, gut microbiota create a favorable environment within the intestine. 34 Previous studies have shown that Bacteroides species harbor genes involved in carbohydrate metabolism, 35 which aligns with our finding indicating higher levels of carbohydrate metabolism within the gut microbiome (Figure 5E). Additionally, it has been reported that dominant phylum Firmicutes promotes energy absorption in obese and diabetic rats. 36 DR1 might directly interact with the cell walls or metabolic pathways of certain gut bacteria, either promoting the growth of beneficial species or inhibiting the growth of harmful ones. DR1 Supplementation also enriched the abundance of Ruminococcus, which was reported to be negatively correlated with metabolic disorders and diabetes. 37 Ruminococcus is capable of degrading resistant starches, thereby enhancing intestinal energy absorption, which contributes to weight gain in individuals. It also influences gut health by facilitating oxidative stress and inflammatory responses, and is regarded as being associated with adverse human health outcomes. Additionally, Ruminococcus produce succinate, an intermediate product of propionate, through the succinate pathway. 38 Furthermore, a recent study indicated that Oscillospira abundance is influenced by diet in obese individuals and is positively correlated with gut permeability, obesity, and systemic inflammation. 39 Remarkably, DR1 treatment led to a substantial reduction in Oscillospira abundance in obese mice. Correlation analysis revealed significant associations between important bacteria, phenotypic indicators and SCFAs, DR1 could significantly regulate bacteria, SCFAs and phenotypic indicators, suggesting a correlation between the regulatory effects of DR1 on gut microbiota, SCFAs, glucolipid metabolism and inflammatory response. Our findings also unveiled an inverse relationship between most bacteria and levels of glucolipid induced by HFD while demonstrating a positive correlation with SCFAs, suggesting that DR1 may modulate gut microbial composition to enhance SCFAs production and regulate glucolipid metabolism. In addition, the anti-obesity mechanisms associated with DR1 warrant further investigation. It is crucial to recognize that the gut microbiome constitutes a complex ecosystem, with its dynamics shaped by numerous factors beyond the scope of our current study. For example, environmental conditions, psychological stress, dietary habits, host genetics, individual variation in gut microbiota composition, and other microbial metabolites are known to impact research outcomes and may interact with the effects of DR1.
However, there are certain limitations of our study. It is essential to set different doses of DR1 and the positive drug in further pharmacodynamic studies. Furthermore, multiomics (eg, metabolomics and transcriptomics) and fecal microbiome transplantation are of great significance for exploring the mechanisms by which DR1 exerts the anti-obesity effects.
In conclusion, the present findings validate the efficacy of DR1 intervention in mitigating HFD-induced obesity in mice, thereby offering a crucial experimental avenue for weight management by targeting gut microbiota and SCFAs.
Methods
Materials
The Danshensu and Notoginsenoside R1 (purity ≥ 98%) were procured from Shanghai Yuanye Biotechnology Co., Ltd (Shanghai, China). The HFD (D12492) and NCD (D12450J) were obtained from Research Diets Inc. (New Brunswick, NJ, USA). All other chemicals used in this study were of MS grade and sourced from Thermo Fisher Scientific Inc. (Waltham, MA, USA).
Animal Experiment
Six-week male C57BL/6J mice (SPF grade, certificate NO. SCXK (Jing) 2016-0006) were procured from Beijing Vital River Laboratory Animal Technology Co., Ltd (Beijing, China). The mice were placed in a standard SPF environment (23 °C and 12-h light/dark cycle). All animals adhered to the ethics standards set by the animal care and welfare committee of Nanjing University of Chinese Medicine (certificate NO. 201909A016).
Forty mice were subjected to one-week acclimatization period during which they were provided with NCD and water at libitum. Subsequently, they were randomly assigned into four groups (n = 10) as follows: (1) Con group, receiving NCD; (2) ConDR1 group, receiving NCD along with 60 mg/kg DR1 (4:1, according to related literature 40 and our pre-experiments); (3) Mod group, fed with HFD; and (4) ModDR1 group, fed with HFD along with 60 mg/kg DR1. The DR1 was administered orally on a daily basis for a duration of 12 weeks, and the body weight of mice was measured once per week. OGTT was conducted, and fresh feces were collected at the last week. The specific operating methods of collecting feces could be found in our previous paper. 4 Once mice underwent an overnight (∼12 h) fasting period, retro-orbital bleeding was performed, and the collected blood samples were then centrifuged at 1520 g and 4 °C for 10 min. The resulting upper serum was collected and stored at −80 °C. Additionally, liver, epididymal fat, colon, small intestine and caecal contents were collected. After measuring their weights, these tissues were fixed in 4% paraformaldehyde, while the caecal contents were frozen at −80 °C.
Oral Glucose Tolerance Test (OGTT) and Serum Biochemical Analyses
The operation of OGTT was conducted according to our previous study. 41 The levels of serum TC, TG, LDL, and HDL were determined using kits from Nanjing Jiancheng Bioengineering Research Institute (Jiangsu, China), while FSG was measured with a glucose oxidase kit from Applygen Technologies Inc. (Beijing, China), while FINS was quantified using the ELISA Kit from Mercodia Inc. (Uppsala, Sweden). Serum IL-1β was detected using ELISA kits obtained from Shanghai Mlbio Biotechnology Co., Ltd The HOMA-IR was calculated as [FSG (mmol/L) multiply FINS (mU/L)] divided by 22.5. 42
Histopathological Analysis
Those fixed tissues (epididymal fat, liver, small intestine, and colon) were embedded and sectioned at a thickness of 4 μm, thereafter undergoing standard H&E staining and were examined and photographed with light microscopy (Nikon, Japan). 43
Short-chain Fatty Acids (SCFAs) Analysis
The levels of SCFAs in the caecal contents were quantified using UPLC-MS. Chromatographic separation was performed on a Waters UPLC I-Class system (Waters, Corp., Milford, MA, USA), coupled with a Waters XEVO TQ-XS mass spectrometer for detection. The column temperature was 45 °C, the injection volume was set to 5 µL, the spray voltage was 3.0 kv, the turbospray temperature was 150 °C, the cone was 21 V, the desolvation temperature of 500 °C, the aux gas flow rate was 10, and the desolvation gas flow was1000 L/Hr. Data were collected and processed using MassLynx software (V.4.2). The mobile phase consisted of 0.01% formic acid in water (A) and methanol/isopropanol (B) at a ratio of 4:1 (v/v). A gradient elution method was employed as follows: starting from 95%-85% A at 0∼2 min, followed by a linear decrease to 45% A at 2∼9 min, further decreasing to 0% A at 9∼10 min, maintaining at 0% A until 11 min, then increasing back to the initial condition of 95% A (11∼11.1 min), and finally holding steady at this composition until the end of the run (13 min).
For sample preparation, each sample weighing approximately 50 mg was mixed with 1 mL an additional volume of methanol-water solution containing derivatization reagent in a ratio of approximately half-and-half. After vigorous vortexing for extraction over a period of 30 min and subsequent centrifugation for 5 min at a speed corresponding to approximately 1520 g under refrigerated conditions (4 °C), 50 µL of the upper layer was carefully transferred into an injection vial equipped with an integrated liner tube having 200 μL capacity for subsequent analysis. 41
Microbiome Analysis Based on the 16S rRNA Sequencing
Fecal samples were shipped on dry ice to Personal Biotechnology (Shanghai, China) for the analysis of gut microbiota using 16S rRNA sequencing, following previously described methods. 1 For data analysis, PCoA based on weighted unifrac distance was conducted, and arithmetic mean clustering using the unweighted pair group method was applied. Visual representations such as Veen diagrams and heat maps displayed differences between various groups while t-test and LEfSe determined significant variations among groups. UPGMA clustering was used as a hierarchical clustering method to interpret the distance matrix using the average linkage.
Data Processing and Statistical Analysis
Our data were presented as mean ± standard deviation (SD) and analyzed using GraphPad Prism version 8.0 (La Jolla, CA, USA). For comparisons between only two groups, unpaired two tailed Student’s t test were used for parametrically distributed data and Mann-Whitney tests for nonparametrically distributed data when applicable. Datasets that involved more than two groups were assessed by one-way analysis of variance (ANOVA) followed by Dunnett’s multiple comparisons test. A P value less than 0.05 was considered to indicate statistical significance. The correlations were identified by Pearson correlation, and the correlations analysis was conducted using SPSS software (version 22.0, Chicago, Illinois, USA) and GraphPad Prism.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241293024 - Supplemental material for Danshensu and Notoginsenoside R1 Combination Alleviates Obesity in Mice Via Modification of Gut Microbiota and Short-Chain Fatty Acids
Supplemental material, sj-docx-1-npx-10.1177_1934578X241293024 for Danshensu and Notoginsenoside R1 Combination Alleviates Obesity in Mice Via Modification of Gut Microbiota and Short-Chain Fatty Acids by Huijuan Tao, Wenxiao Wang, Hui Cao, Ning Tong, Li Ma, Jie Shen, Qiao Zhang, Yuping Tang and Shijun Yue in Natural Product Communications
Footnotes
Acknowledgements
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Shaanxi Province (grant number 2022SF-221) and School-level Project of Shaanxi University of Chinese Medicine (grant number 2023GP24).
Ethical Approval
This study was approved by the animal care and welfare committee of Nanjing University of Chinese Medicine (certificate NO. 201909A016).
Statement of Informed Consent
These are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
All animal experiments were approved by the animal and welfare committee of Nanjing University of Chinese Medicine. All procedures were conducted in accordance with the guidelines for the care and use of laboratory animals (china).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
