Abstract
Introduction
Lung cancer remains a major global health challenge and is the second most common malignancy worldwide, often leading to fatal outcomes if not diagnosed and treated promptly. Herba Houttuyniae exhibits diverse pharmacological properties, including antioxidant, antiviral, antitumor, and anti-inflammatory activities. However, its apoptotic effects on lung cancer cells have not been fully elucidated. In this study, we investigated the impact of Herba Houttuyniae on the viability of A549 and H1299 lung cancer cells.
Methods
This study investigated the effects of aqueous extract of Herba Houttuyniae (aqueous extract of Herba Houttuyniae (AEHH)) on A549 and H1299 lung cancer cells. A range of aqueous extract of Herba Houttuyniae (AEHH) concentrations was applied, and assays were conducted to assess cell viability, mitochondrial membrane potential, and the expression of apoptosis-related proteins, including Bcl-2-associated X (Bax), caspases, cytochrome c, binding immunoglobulin protein (binding immunoglobulin protein (BiP)), and phosphorylated endoplasmic reticulum (ER)K (p-endoplasmic reticulum (ER)K). Cell migration and wound healing were evaluated using Transwell and scratch assays, respectively. Statistical analyses were performed using analysis of variance (ANOVA) followed by Tukey's post hoc test.
Results
AEHH inhibited lung cancer cell proliferation by decreasing the mitochondrial membrane potential. Western blot analysis demonstrated increased expression of cytochrome c, binding immunoglobulin protein (BiP), phosphorylated extracellular signal–regulated kinase (ERK) (p-extracellular signal–regulated kinase (ERK)), Bcl-2-associated X protein (Bax), and caspases 3 and 8. Furthermore, AEHH suppressed both the migratory capacity and invasive potential of the lung cancer cells examined.
Conclusion
In conclusion, AEHH exhibited concentration-dependent anticancer effects on the A549 and H1299 cell lines and shows potential for use in lung cancer treatments.
This is a visual representation of the abstract.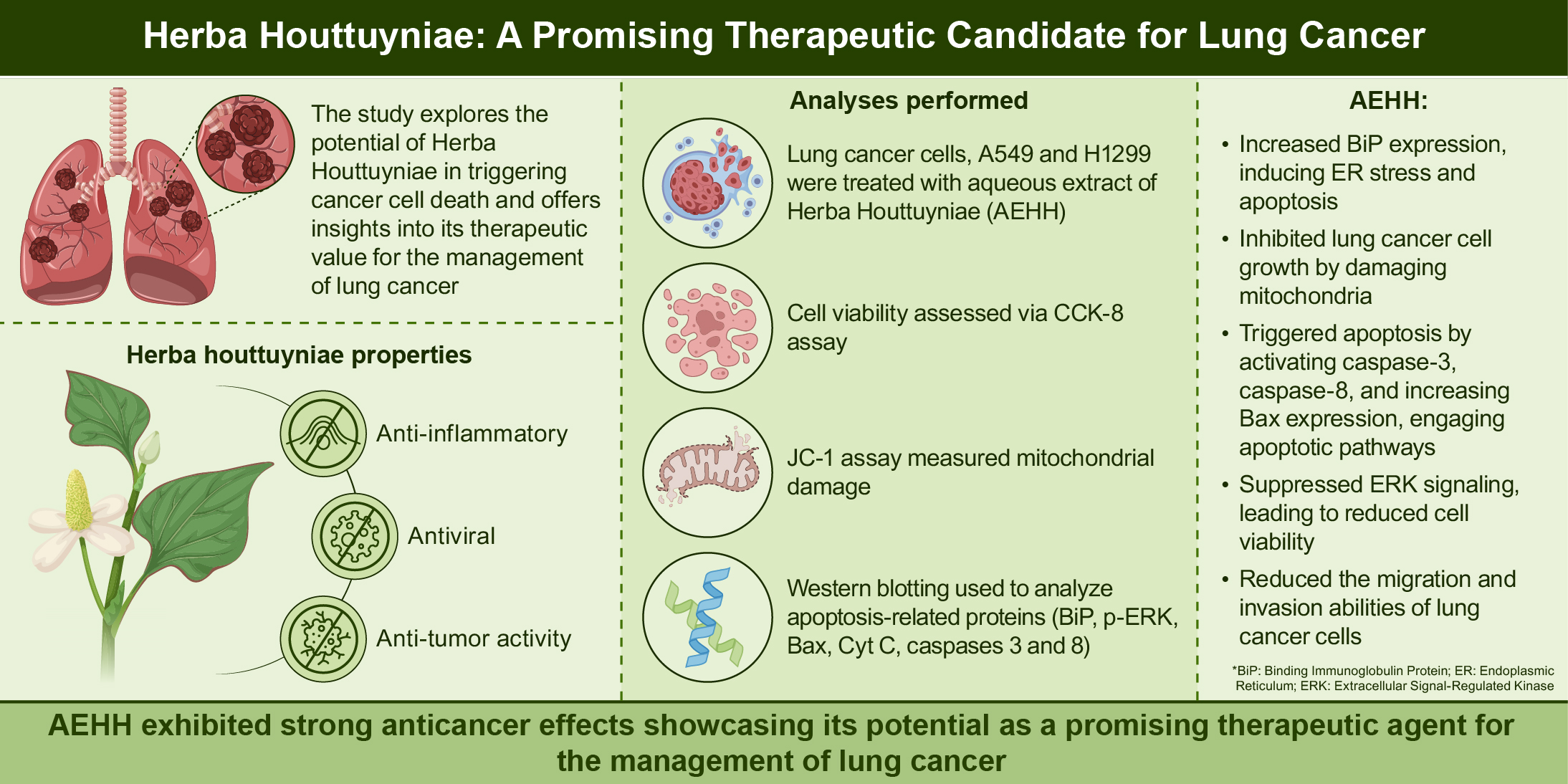
Introduction
In 2024, lung cancer was the cancer most frequently diagnosed—with there being approximately 2.5 million new cases of lung cancer (representing 12.4% of all cancers globally). Additionally, lung cancer poses the greatest risk of mortality due to cancer; approximately 1.8 million deaths were caused by lung cancer in 2024 (representing 18.7% of all cancer-related mortality). 1 Non-small-cell lung carcinoma (non-small-cell lung carcinoma (NSCLC) is the most prevalent lung cancer. Approximately 85% of all cases of lung cancer involve NSCLC. 2 NSCLC is caused by smoking, exposure to toxic substances, including secondhand smoke or radon, and a family history of the disease. 3 Surgery can facilitate complete recovery when the cancer is treated at an early stage. Nevertheless, approximately half of patients who undergo surgery and recover completely go on to experience relapse, and early diagnosis of NSCLC is challenging. Over half of patients with NSCLC die within a year, leading to a low 5-year survival rate. 4 Chemotherapy, which is employed to treat lung cancer, can have several adverse effects. Patients who receive chemotherapy may experience chemoresistance or recurrence, and chemotherapy can damage normal cells. 5 Studies are urgently needed to investigate how chemopreventive agents used to treat patients with advanced and recurrent lung cancer influence survival rates.
The ER has a critical role in maintaining cellular function. ER function can be impaired by factors such as infection and hypoxia. 15 Impairment of ER function can lead to the excessive accumulation of unfolded proteins. 16 Inositol-requiring enzyme 1 alpha (IRE1a) has an important role in restoring ER function in response to stress. IRE1a is a protein located in the ER membrane that acts as a sensor for stress. Binding immunoglobulin protein (BiP) is a chaperone protein that normally binds to IRE1a, keeping it inactive. During periods of ER stress, BiP releases from IRE1a. Once BiP dissociates, IRE1a undergoes autophosphorylation and dimerization and triggers the unfolded protein response, 17 which is also crucial for restoring ER function and maintaining protein homeostasis in response to stress. 17
During ER stress, IRE1a plays a dual role, regulating whether a cell adapts or undergoes apoptosis. To restore ER function, IRE1a triggers the unfolded protein response. If the ER stress is excessively severe, IRE1a signals cells to undergo apoptosis. 18 Furthermore, the ER–mitochondria interface is critical for many physiological functions, such as signaling to and from the ER, lipid transport, energy metabolism, and cell survival.19,20
In mammals, mitochondria contribute to apoptosis regulation. During apoptosis, pro-apoptotic proteins are released into the cytosol from mitochondrial intermembrane spaces. 21 Mitochondrial calcium regulates the mitochondrial outer membrane's permeability and subsequent apoptosis. An excessive accumulation of calcium in the mitochondria causes mitochondrial swelling and rupture, leading to the activation of caspases. A major mitochondrial calcium source is the ER. Mitochondria-associated membranes facilitate the direct transfer of calcium ions between mitochondria and the ER. These membranes contribute to the complex interplay between these organelles to regulate apoptosis.22,23 Calcium levels are associated with various types of cell death, including autophagy and necrosis. 24
In one study, Herba Houttuyniae reduced the viability of human lung cancer cells by inducing mitochondrial dysfunction and subsequent reactive oxygen species production. 25 Whether and how Herba Houttuyniae regulates organelle function in lung cancer is unknown. This paper presents an exploration of how Herba Houttuyniae influences the viability, mitochondrial function, signaling pathways, and invasive properties of lung cancer cells of the types A549 and H1299.
Materials and Methods
Reagents
Aqueous extract of Herba Houttuyniae (AEHH; Yu Xing Cao, #421118004) was purchased from Ko Da Pharmaceutical (Taoyuan, Taiwan).
Antibodies
ABclonal (Woburn, MA, USA) was our source of anti-mouse and anti-rabbit IgG-conjugated horseradish peroxidase and rabbit polyclonal antibodies specific for Bcl-2-associated X (Bax), cytochrome c (Cyt C), cleaved caspase-3, caspase-8, glyceraldehyde 3-phosphate dehydrogenase, BiP, CHOP, activating transcription factor 4, ERK, and p-ERK.
Chemicals
Kits for the assay of JC1 mitochondrial membrane potential (MtMP) were purchased from Abcam (Cambridge, UK). Gibco-BRL Life Technologies (Grand Island, NY, USA) was our source of fetal bovine serum, Dulbecco's modified Eagle's medium (DMEM), and other cell culture reagents.
Cell Culture
Human lung epithelial cells of types A549 (CRM-CCL-185) and H1299 (CRL-5803) were purchased from the American Type Culture Collection (Manassas, VA, USA). DMEM supplemented with 100 U/mL penicillin/streptomycin and 10% fetal bovine serum was employed to culture the cells at 37 °C under 5% CO2.
Experimental Protocol
At a density of 1 × 106 cells/well, A549 or H1299 cells were seeded into a six-well plate, in which they were incubated for 24 h. Subsequently, they were divided into a control group and two experimental groups. The control group was cultured in DMEM with 1% fetal bovine serum (FBS). The first and second experimental groups were cultured in basal medium containing 250 and 500 μg/mL AEHH, respectively. The cells were then harvested and extracted.
Cell Viability Assay
Cell viability was evaluated utilizing the CCK-8 assay in the method stipulated in the manufacturer's specified protocol. At a density of 2 × 104 cells/well, A549 or H1299 cells were seeded in a 96-well plate and subjected to overnight incubation. The subsequent day, each well received 10 µL of CCK-8 solution, and 2-h incubation was conducted at 37 °C with 5% CO2 and with different concentrations of chrysophanol. An Infinite F50 Plus microplate reader (Tecan, Männedorf, Switzerland) was employed to measure absorbance at 450 nm. To determine whether AEHH interferes with CCK-8 reagent, a control experiment was performed in which no cells were present. AEHH at the indicated concentrations was added to cell culture medium containing CCK-8 reagent and incubated under the same conditions as in the cell viability assay (37 °C, 2 h). The absorbance measurement process was also the same. The results were compared with those of a reagent-only control (medium + CCK-8 without AEHH). No significant difference in absorbance was observed, indicating that AEHH does not interfere with CCK-8 reagent and does not produce nonspecific absorbance. Therefore, all measured cell viability changes reflect the biological effect of AEHH.
Western Blotting Analysis
Cold RIPA buffer was employed to lyse the collected cells with protein inhibitors. Sodium dodecyl sulfide–polyacrylamide gel electrophoresis was performed, with the cells transferred to polyvinylidene difluoride membranes. Specific primary antibodies and horseradish peroxidase–conjugated secondary antibodies were used to conduct Western blotting. Enhanced chemiluminescence was employed to quantify peroxidase activity. The ChemiDoc MP (Bio-Rad, Hercules, CA, USA) was used for analysis of reactive band intensities.
MtMP Assay
At a density of 2 × 104 cells/well, A549 or H1299 cells were seeded into 96-well plates for 24 h. After culturing of the cells, a JC-1 MtMP assay kit was employed to measure MtMP.
Migration Assay
A 24-well dish (Transwell; Costar, Corning, NY, USA) with pores measuring 8 μm was employed to perform the migration assay. A suspension of A549 or H1299 cells (approximately 1 × 104) in 200 μL of serum-free DMEM was placed in the Transwell dish's upper chamber. The lower chamber contained 300 µL of DMEM containing AEHH at one of several concentrations and 10% fetal bovine serum. Twenty-four-hour incubation was conducted at 37 °C in 5% CO2. Subsequently, methanol was employed to fix the cells for 15 min, and 0.05% crystal violet in phosphate-buffered saline was used to stain them for 15 min. Cotton-tipped swabs were employed to remove cells on the upper side of each filter, and phosphate-buffered saline was then used to rinse the filters. Finally, we used a microscope to count the number of cells on the underside of each filter.
Wound Healing Assay
At a density of 1 × 106 cells/well, A549 or H1299 cells were seeded in six-well plates, and 100% confluency was achieved. A straight line was created in each well by using a sterilized 10-mL pipette tip. AEHH treatment was applied to the cells for 24 h, and relative gap distances were measured using photographs.
Statistical Analysis
GraphPad Prism 10 for MacOS (version 10.2.3; GraphPad, San Diego, CA, USA) was used to analyze the collected data, which are expressed in terms of the mean ± standard deviation. We made intergroup comparisons through one-way analysis of variance and for post hoc analysis used Tukey's multiple comparison test. Statistical significance was indicated by
Results
AEHH Inhibited the Proliferation of A549 and H1299 Cells
The viability of the investigated lung cancer cells after AEHH treatment was assessed through CCK-8 assays (Figure 1). The results indicated that AEHH dose-dependently lowered the percentage of proliferating cells. Treatment with 250 and 500 μg/mL AEHH had reduced cell proliferation by 22.38% and 44.33%, respectively, in A549 cells and by 35.75% and 47.49%, respectively, in H1299 cells after 24 h. After 48 h, 125, 250, and 500 μg/mL AEHH had reduced cell proliferation by 20.49%, 34.25%, and 48.41%, respectively, in A549 cells and by 23.36%, 35.76%, and 53.57%, respectively, in H1299 cells.

Aqueous Extract of Herba Houttuyniae (AEHH) Affected the Percentage of Viable Cells in (A and B) A549 and (C and D) H1299 Human Lung Cancer Cells, Which Were Cultured with Different AEHH Concentrations for 24 and 48 h. Results are Represented as the Mean ± Standard Deviation (SD).
To further characterize the dose-dependent cytotoxic effects of AEHH, we assessed cell viability at even lower (31.25 and 62.5 μg/mL) and higher (750 and 1000 μg/mL) concentrations. AEHH exerted minimal cytotoxic effects for the two lowest concentrations, with cell viability remaining higher than 85%. At 750 and 1000 μg/mL, only modestly higher cytotoxicity compared with that at 500 μg/mL was observed, suggesting a saturation effect. These findings confirm a dose-dependent response within the 125-500 μg/mL range and support the selection of this window as the biologically active concentration range for subsequent experiments (Supplemental Figure 1).
AEHH Increased ER Stress in Lung Cancer Cells
To determine whether AEHH inhibits cell growth by regulating ER stress in A549 and H1299 cells, Western blot analyses were performed using 250 and 500 μg/mL AEHH. Western blot analysis revealed that BiP expression was significantly increased following 24-h AEHH treatment at 500 μg/mL (Figure 2A, B). To further investigate how AEHH-induced cytotoxicity involved ER stress, we examined the signaling of key markers of ER stress in A549 and H1299 cells treated with 250 or 500 μg/mL AEHH. The results indicated significant upregulation of CHOP expression compared with the untreated control group in the 500 μg/mL AEHH–treated A549 cell line, suggesting activation of proapoptotic ER stress signaling. By contrast, the expression levels of BiP, activating transcription factor 4, and p-ERK were largely unchanged following AEHH treatment. These findings imply that AEHH may selectively trigger CHOP-mediated ER stress responses without broadly activating the canonical unfolded protein response pathways.

AEHH Regulated Endoplasmic reticulum (ER) Stress–Related Proteins in A549 and H1299 Cells. (A and C) Western Blotting Results. (B and D) Quantitative Western Blotting Results. Results are Represented as the Mean ± SD.
AEHH Decreased MtMP in A549 and H1299 Cells
JC-1 staining was used to assess the effects of AEHH (250 and 500 μg/mL) on MtMP levels in A549 and H1299 cells (Figure 3). Treatment with 250 μg/mL AEHH reduced MtMP levels by 33.19% in A549 cells and 25.16% in H1299 cells relative to the levels in dimethyl sulfoxide–treated controls. At 500 μg/mL, AEHH reduced MtMP levels by 49.08% in A549 cells and 35.28% in H1299 cells, indicating that AEHH disrupts MtMP in lung cancer cells.
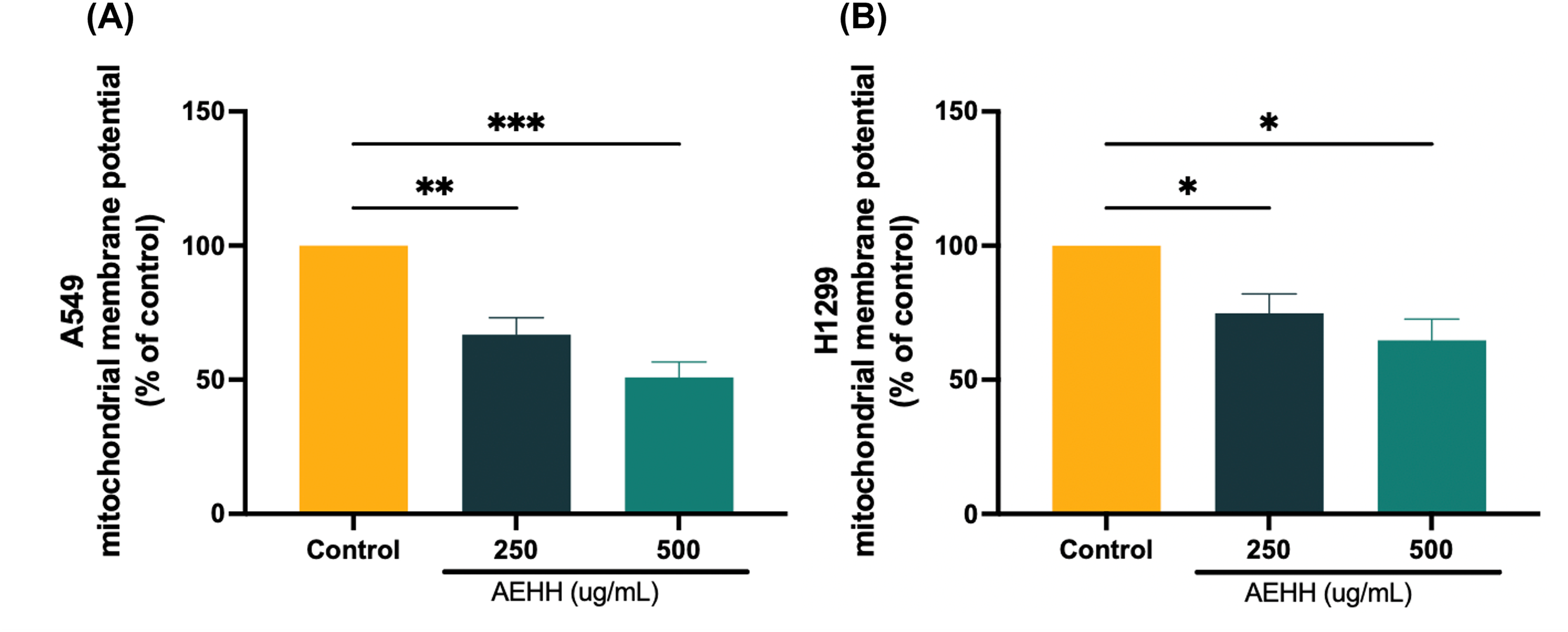
AEHH Reduced Mitochondrial Function in (A) A549 and (B) H1299 Cells. Treatment with AEHH Lasted 24 h. A JC-1 Mitochondrial Membrane Potential Assay kit was Used to Measure Mitochondrial Membrane Potential. Results are Represented as the Mean ± SD.
AEHH Regulated Signal Transduction in Lung Cancer Cells
We made use of Western blot analysis for the purpose of investigating the effects of AEHH on apoptosis-related signaling molecules. Results from three experimental groups for Bax, p-ERK, cleaved caspase-3, Cyt C, and caspase-8 are illustrated in Figure 4A, B. Twenty-four-hour treatment with 500 μg/mL AEHH significantly decreased p-ERK expression and increased levels of cleaved caspase-3, Cyt C, cleaved caspase-8, and Bax relative to the levels in the control cells (Figure 4C–F). At 250 μg/mL, AEHH significantly upregulated cleaved caspase-3, Cyt C, and caspase-8 expression in H1299 and A549 cells (Figure 4D–F), suggesting that AEHH-regulated signaling proteins affect lung cancer cell proliferation.

In A549 and H1299 Cell Lines, Cell Death was Induced by AEHH-Mediated Changes in Apoptosis-Related Proteins. Treatment with AEHH Lasted for 24 h; Total Protein was Prepared and Evaluated. (A) Western Blotting Results. (B–F) Quantitative Western Blotting Results. Results are Represented as Mean ± SD.
AEHH Suppressed Lung Cancer Cell Invasiveness
We performed an invasion assay by using culture dishes and Transwell assays to explore how AEHH inhibits cell invasion. Treatment was performed at AEHH concentrations of 250 or 500 μg/mL for 24 h. AEHH disrupted the interactions between cells and impaired wound healing. AEHH at 500 μg/mL reduced the migration distance by 25.32% in A549 cells and 20.76% in H1299 cells. At 250 μg/mL, the reduction was 20.72% in A549 cells relative to that in vehicle-treated controls (Figure 5A, B).

AEHH Inhibited the Invasiveness of A549 and H1299 Cell Lines. Treatment with AEHH Lasted for 24 h. (A and B) Images of Invasiveness, Analyzed Using Wound Healing Assays. (C and D) A Transwell Assay was Used to Assess AEHH-Induced Inhibition of Cell Invasion. Results are Represented as the Mean ± SD.
The invasiveness of the cells treated with AEHH at 500 μg/mL was reduced by approximately 36.20% in the A549 cells and by 21.54% in the H1299 cells relative to the nontreated cells (Figure 5C, D), suggesting that AEHH significantly reduces the invasive potential of lung cancer cells.
Discussion
This study examined Herba Houttuyniae's chemopreventive effects and the underlying mechanisms. Herba Houttuyniae is popular as a herbal medicine thanks to its favorable anti-inflammatory and antioxidant properties. The herbal extract is also popular for its ability to inhibit the development of lung cancer. Our results improve our understanding of Herba Houttuyniae's protective effects and ability to inhibit lung tumorigenesis.
For many years, Herba Houttuyniae has been a herbal extract widely used among those practicing traditional Chinese medicine. 26 The death receptor apoptotic pathway has long been considered a target of traditional Chinese medicines used to treat human lung cancer. 27 Besides having immunoregulatory and anti-inflammatory properties, AEHH is an antibacterial, antiviral, and anticancer agent. 28 The effectiveness of AEHH in treating cancer has been reported from several studies, with the extract showing promise against colorectal cancer, leukemia, and breast cancer.6,29 Our results demonstrate that the anti–lung cancer activity of AEHH is concentration and time dependent. AEHH has substantial promise in lung cancer as a therapeutic agent.
Signal transduction is strongly reliant on the ER. Upon its sensing of a homeostatic change, the ER forwards feedback to other components of a cell. The ER's protein-folding ability is inhibited by excessive Ca2+ concentration, cellular adenosine triphosphate level perturbations, and fluctuations in the redox state. Unfolded proteins accumulate when the ER's function is impaired, and excessive accumulation of unfolded proteins induces ER stress. 30 The cancer microenvironment, characterized by hypoxia and limited nutrients, further disrupts the ER's function. Disruption of ER function triggers the unfolded protein response and ER stress. 31 BiP, a key chaperone protein, supports antiapoptotic activity and helps transport newly synthesized polypeptides across the ER membrane. 32 As well as regulating the ER stress response, BiP participates in the unfolded protein response signaling pathway. 33 Our findings revealed that BiP expression was upregulated in AEHH-treated A549 and H1299 cells, consistent with recent studies that have highlighted ER stress as a druggable target in oncology.16,30 Thus, AEHH sensitized the lung cancer cells to ER stress–mediated apoptosis.
Mitochondria play vital roles in biosynthetic processes, including adenosine triphosphate generation and intrinsic apoptotic pathway regulation. Mitochondria are the primary energy source for cells. Acting as signaling organelles, mitochondria participate in numerous processes (physiological and pathological), including apoptosis, pyroptosis, and ferroptosis. 34 Regarding apoptosis, the primary pathway through which mitochondria are involved is related to increased permeability of the mitochondrial outer membrane. Such high permeability results in mitochondrial intermembrane spaces releasing Cyt C, which triggers signaling pathways related to apoptosis and leading to the death of cells. Mitochondria-dependent apoptosis is associated with not only caspase cleavage, proapoptotic protein activation (including activation of Bax and Bak), and Cyt C release but also with Ca2+ overload and membrane permeabilization in the mitochondria.35,36 Upon treatment with Herba Houttuyniae, reactive oxygen species production in colorectal cancer and leukemia cells increases, and MtMP in these cells decreases.25,37 These alterations are associated with increased Cyt C and Bax levels, which ultimately induce apoptosis. Accordingly, we investigated how AEHH affects mitochondria-dependent apoptosis. Recent mechanistic studies further confirm the pivotal role of BCL-2 family proteins and mitochondrial dysfunction in cancer cell apoptosis.30,35 The results we obtained are consistent with those reported in other studies; AEHH significantly inhibits MtMP function, activates proteins that have proapoptotic effects, increases Cyt C levels, and upregulates caspase cleavage. These results indicate that ER stress disruption is central to AEHH-induced cell death in A549 and H1299 cells.
In some cases, signaling pathway hyperactivation leads to more rampant cell proliferation and higher antiapoptotic activity. The ERK/MAPK axis is associated with various types of cancers, including ovarian, colon, breast, and lung cancer. 38 Activation of the ERK/MAPK signaling pathway triggers the transformation of healthy cells into tumor cells. Simultaneously, ERK/MAPK pathway inhibition restores the original state of tumor cells. 39 Colon cancer cells can be prevented from moving to the S phase (from the G1 phase) if ERK1/2 activity is inhibited. 40 In addition, PI3K signaling is associated with chemotherapy resistance through the ERK pathway. 41 Our study demonstrates the potential of AEHH for cancer treatment. We observed a significant decrease in p-ERK expression in AEHH-treated A549 and H1299 cells, suggesting a promising avenue for further research and potential clinical applications. This is in line with recent translational studies demonstrating the clinical relevance of MAPK/ERK pathway inhibition in oncology. 39
Effector caspases—such as caspase-3, −6, and −7—and initiator caspases, such as caspase-8 and −9, mediate caspase-induced apoptosis. 42 Caspase-3, −6, and −7 activation causes cellular morphology and biochemical changes associated with apoptosis. 43 We demonstrated that initiator caspase (ie, caspase-3 and −8) activities were triggered in a manner that was dose dependent. Caspases are proteases that play an essential role in the signaling cascade that leads to apoptosis. They are activated by extracellular signals or intracellular stresses and by membrane death receptors (in the case of caspase-8), which are further activated by downstream effector caspases in the intrinsic pathway. Apoptosis mediated by mitochondria is induced by Bax, Bcl-2, and Bcl-2 family members. These proteins regulate mitochondria's release of Cyt C into the cytoplasm and simultaneously induce apoptosis.11,44 A study demonstrated that Bcl-2 family proteins are regulated by AEHH and that AEHH activates caspase-8 and −3, engaging both death receptor and mitochondrial pathways. 12 Similarly, we found that AEHH induced apoptosis in A549 and H1299 cells because of how it promoted Cyt C release into the cytoplasm. This release activates caspase-8 and −3 and regulates expression of Bax. Evidently, extrinsic pathways and pathways mediated by mitochondria are mechanisms through which AEHH induces apoptosis. This study demonstrated that AEHH is involved in promoting cancer cell apoptosis; nevertheless the specific apoptotic pathway and its intrinsic or extrinsic nature have not been conclusively determined. Although we observed that cleaved caspase-3 and total caspase-8 levels were increased, additional pathway-specific markers were not assessed. For example, we did not assess involvement of cleaved caspase-8, caspase-9, Cyt C release, or the Bax to Bcl-2 ratio. These markers are essential to distinguish apoptosis mediated by mitochondria and death receptors (intrinsic and extrinsic, respectively). Future studies will more comprehensively investigate the aforementioned signaling components to identify which precise apoptotic mechanisms are triggered by AEHH. This study demonstrated that AEHH induces apoptosis in lung cancer cells by discovering increased expression of apoptotic markers, such as cleaved caspase-3 and CHOP. Although these findings support the involvement of apoptotic mechanisms, we acknowledge that the use of Annexin V/PI double staining would provide more definitive evidence by directly quantifying apoptotic cell populations, including those corresponding to early and late apoptosis. Annexin V binding to externalized phosphatidylserine and PI uptake are classic indicators of apoptosis and necrosis, respectively, and their combined use in flow cytometry would allow for a more comprehensive assessment of AEHH-induced cell death. Incorporating this assay in future studies will enhance the accuracy and reliability of apoptosis characterization.
Mitochondrial dysfunction is a hallmark of intrinsic apoptosis, often marked by a decrease in MtMP and release of proapoptotic proteins (including Cyt C). In this study, JC-1 staining confirmed that, in A549 and H1299 cells, AEHH treatment significantly disrupted MtMP. The study also demonstrated that AEHH increased Cyt C levels, evidence of activation of the mitochondrial-mediated apoptotic pathway. The experiments conducted in this study used whole-cell lysates—mitochondrial fractionation was not performed. Fractionation would have enabled us to confirm Cyt C's translocation into the cytosol from mitochondria. Although the obtained results suggest involvement of the intrinsic apoptotic pathway, future studies using subcellular fractionation will help yield definitive evidence of permeabilization of the outer layer of the mitochondria and release of Cyt C.
This research focused on evaluating the cytotoxic and mechanistic effects of AEHH on lung cancer cell lines. A limitation is the absence of comparative analysis with nontumorigenic epithelial cell lines, such as BEAS-2B. Such comparisons are important for determining the selectivity and potential therapeutic window of AEHH. Future studies will include these normal cell models to more accurately assess the safety profile and differential sensitivity of AEHH for cancerous versus noncancerous cells. Additionally, although our findings provide mechanistic insight at the cellular level, further in vivo studies and clinical validation are warranted to support the translational potential of AEHH in lung cancer therapy. This study did not assess the effect of AEHH on matrix metalloproteinases (ie, MMP-2 and MMP-9) or on other markers of cell motility and invasion. These factors are critical for evaluating the antimetastatic properties of AEHH. We plan to explore these aspects in future work using both molecular analyses and functional assays to determine whether AEHH inhibits lung cancer cells’ invasion and metastatic potentials. Our study demonstrated that AEHH significantly inhibits A549 and H1299 lung cancer cell migration and invasion, as was demonstrated in wound healing and migration assays. These results suggest that AEHH possesses antimigratory properties in addition to its cytotoxic effects. We acknowledge that the observed reduction in migration could, in part, be influenced by cytotoxicity, particularly for the higher AEHH concentrations. To minimize this confounding factor, we assessed cell motility by using 250 μg/mL AEHH, a sublethal dose that resulted in approximately 80% cell viability after 24 h. Although this provides preliminary support for an antimigratory effect, future studies using strictly nonlethal concentrations, such as ≤125 μg/mL, are warranted to confirm whether AEHH directly impairs migratory behavior independently of its cytotoxic action.
The observed downregulation of p-ERK following AEHH treatment suggests that the ERK/MAPK signaling pathway may be suppressed by AEHH. Cell proliferation and survival are heavily regulated by the ERK/MAPK signaling pathway. Nevertheless, whether this reduction represents a direct effect of AEHH on the ERK pathway or a secondary consequence of broader cellular stress responses induced by AEHH—such as oxidative stress, mitochondrial dysfunction, or ER stress—remains unclear. Given the interconnected nature of these signaling networks, we believe that ERK deactivation might occur downstream of primary stress-related events. Further mechanistic studies will be necessary to clarify the precise role of ERK signaling in AEHH-induced cytotoxicity.
In addition to having cytotoxic and proapoptotic effects, AEHH may also influence the progression of tumors through its modulatory effects on cell motility and invasion. MMP-2 and MMP-9, among other matrix metalloproteinases, are key enzymes involved in extracellular matrix degradation and are commonly associated with tumor invasiveness and metastasis. Although our current study did not evaluate matrix metalloproteinase expression or perform functional migration/invasion assays, we acknowledge that these aspects are highly relevant for determining the broader therapeutic potential of AEHH. Future studies will investigate whether regulation of matrix metalloproteinases or related signaling pathways are responsible for AEHH's suppression of the migration and invasion of lung cancer cells.
We observed significantly elevated expression of CHOP following AEHH treatment in A549 and H1299 cells, suggesting that AEHH-induced apoptosis is mediated by ER stress. CHOP is a key proapoptotic transcription factor activated during prolonged or unresolved ER stress, and its upregulation is often associated with the initiation of apoptosis. Nevertheless, apoptosis is multifactorial and is regulated by a network of signaling pathways. Some of the mechanisms involved in apoptosis are mitochondrial dysfunction, death receptor–mediated signaling, and oxidative stress. Given that only a limited panel of ER stress markers was examined in this study, we cannot definitively conclude that AEHH-induced apoptosis occurs primarily through ER stress. Other pathways not explored in the current study may also have played key roles in the apoptosis we observed. Mechanistic research is required to confirm the relative involvement and interplay of ER stress with other apoptosis-regulating pathways. This study suggests that ER stress is likely involved in AEHH-induced apoptosis because of the observed upregulation of CHOP. Nevertheless, only a subset of ER stress markers was evaluated, and other key apoptotic pathways such as mitochondrial dysfunction, autophagy, and death receptor signaling were not assessed. Therefore, we cannot state with certainty that ER stress is the direct or sole driver of apoptosis in AEHH-treated A549 and H1299 cells. Future studies will include pathway-specific inhibition and rescue experiments to delineate the exact mechanisms underlying AEHH-induced apoptosis.
This study has some limitations. First, we did not confirm the effects of AEHH in humans further clinical studies are warranted. Second, we did not assess the long-term effects of AEHH. Third, our research involved only 1 (AEHH) of the 28 compounds that have been identified in
Conclusion
AEHH increased BiP protein expression, suggesting a cell death mechanism that is mediated by ER stress. AEHH induced apoptosis (mediated by an intrinsic pathway) in A549 and H1299 cells by inhibiting MtMP and Cyt C activity. AEHH also increased proapoptotic protein levels, including that of Bax. Caspase-3 and caspase-8 activities were elevated. AEHH blocked the prosurvival ERK signaling pathway. AEHH triggered apoptosis in A549 and H1299 cells via several pathways, namely intrinsic, extrinsic, and ER stress pathways. AEHH also had the effect of attenuating ERK signaling, as illustrated in Figure 6. AEHH may serve as an effective therapeutic drug in the treatment of lung cancer and warrants further investigation, particularly in vivo studies. In summary, this study demonstrated that AEHH exerts cytotoxic, proapoptotic, and antimigratory effects in lung cancer cells, potentially through modulation of mitochondrial dysfunction, ER stress, and MAPK signaling pathways. These mechanistic findings suggest that AEHH may serve as a promising phytochemical candidate for lung cancer therapy. This aligns with recent evidence highlighting the anticancer potential of phytochemicals, including flavonoids and polysaccharides.13,46 However, the current work is limited to in vitro models. Future research should focus on in vivo validation to assess pharmacokinetics, toxicity, and therapeutic efficacy, as well as its effects on tumor growth and metastasis in animal models. Studies exploring AEHH in combination with established chemotherapeutic or targeted therapies may reveal synergistic benefits and help overcome resistance mechanisms. In parallel, investigations into immune modulation and tumor microenvironment interactions could further clarify its translational potential.

AEHH Inhibited Lung Cancer Cell Proliferation by Reducing MtMP, Sensitizing Cells to ER Stress, Activating Antiapoptotic Protein Bax, Increasing Cytochrome c Expression, and Increasing Caspase Activity.
In conclusion, AEHH exerts cytotoxic, proapoptotic, and antimigratory effects through modulation of mitochondrial dysfunction, ER stress, and MAPK signaling pathways. With comprehensive preclinical evaluation, AEHH holds promise as a novel complementary or standalone therapeutic strategy for lung cancer.
Supplemental Material
sj-pdf-1-npx-10.1177_1934578X251385781 - Supplemental material for Herba Houttuyniae Induces Cell Apoptosis through Caspases, Endoplasmic Reticulum Stress, and Mitochondrial Membrane Potential
Supplemental material, sj-pdf-1-npx-10.1177_1934578X251385781 for Herba Houttuyniae Induces Cell Apoptosis through Caspases, Endoplasmic Reticulum Stress, and Mitochondrial Membrane Potential by Hsien-Yueh Liu, Chan-Yen Kuo, Jen-Wei Lin and Wei-Li Lin in Natural Product Communications
Supplemental Material
sj-pdf-2-npx-10.1177_1934578X251385781 - Supplemental material for Herba Houttuyniae Induces Cell Apoptosis through Caspases, Endoplasmic Reticulum Stress, and Mitochondrial Membrane Potential
Supplemental material, sj-pdf-2-npx-10.1177_1934578X251385781 for Herba Houttuyniae Induces Cell Apoptosis through Caspases, Endoplasmic Reticulum Stress, and Mitochondrial Membrane Potential by Hsien-Yueh Liu, Chan-Yen Kuo, Jen-Wei Lin and Wei-Li Lin in Natural Product Communications
Footnotes
Acknowledgments
The authors would like to express their sincere gratitude to the Ministry of Science and Technology of Taiwan for supporting this research (grant number 109-2320-B-303-004-MY3). We also thank Ko Da Pharmaceutical for providing the aqueous extract of Herba Houttuyniae used in this study. The authors acknowledge the technical assistance provided by the core laboratory staff of Hungkuang University. We are grateful to our colleagues at Tzu Chi University and Chaoyang University of Technology for their constructive input and academic support throughout this project.
Consent to Participate Statement
Not applicable.
Authors’ Contributions
Hsien-Yueh Liu: Writing – review & editing, Writing – original draft, Visualization, Validation, Project administration, Funding acquisition, Formal analysis, Data curation, Conceptualization. Chan-Yen Kuo: Writing – review & editing, Writing – original draft, Visualization, Validation, Supervision, Software, Resources, Project administration, Methodology, Investigation, Formal analysis, Data curation, Conceptualization. Jen-Wei Lin: Visualization, Validation, Investigation. Wei-Li Lin: Project administration, Methodology, Funding acquisition, Conceptualization.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a grant from the Ministry of Science and Technology of Taiwan (grant number: 109-2320-B-303-004-MY3). The sponsors were not involved in the study design; collection, analysis, and interpretation of data; writing of the report; or decision to submit the article for publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Ethics
Please address the following aspects in your Statement of Ethics.
Study Approval Statement
This experiment is basic research and does not involve ethical statements, clinical trial registration numbers, or informed consent.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
