Abstract
Myrcene, a natural olefinic hydrocarbon, possesses anti-inflammatory, analgesic, antibiotic, and antimutagenic properties, but its anticancer effect has not yet been elucidated. Hence, the present study was framed to investigate the molecular mechanism by which myrcene mediates the anticancer activity of A549 lung adenocarcinoma cells. In vitro, A549 lung cancer cells were cultured either with or without myrcene, and the effects on cellular metabolic activity, levels of reactive oxygen species (ROS), mitochondrial integrity, deoxyribonucleic acid (DNA) damage, and activity of caspases were analyzed. The study demonstrated that compared with control cells, myrcene induces cell death in a dose-dependent manner while inducing ROS levels. Further experiments revealed that the metabolic activity of the A549 lung adenocarcinoma cells was diminished with increased DNA damage and altered cellular integrity. In addition, increased activity of caspase-3 was also evidenced with reduced mitochondrial membrane potential synthesis in the myrcene-treated cells, which demonstrate that lung cancer cells experience signs of toxicity during myrcene treatment through the activation of the apoptosis mechanism via mitochondria-mediated cell death signaling and induction of oxidative stress. The results provide the first report on the evidence of anticancer activity and the possibility of a new drug that could be used for the treatment of lung cancer.
Lung cancer is one of the leading causes of death in elderly patients and people of young age. The incidence of lung cancer in the world population is high and could possibly be caused by smoking, lifestyle, and diet. Although smokers may expect to be out of danger of lung cancer after quitting smoking, the occurrence rate does not decelerate appropriately. Dietary modifications and a healthy lifestyle have resulted in success among traditional smokers, but the availability of a healthy diet and consultation is beyond the reach of many. It has been suggested that the dietary agents acting on various signaling pathways in lung carcinogenesis were not encouraging in clinical trials.
1
The treatments available for lung cancer include surgery, and chemotherapy, and radiotherapy, but the development of resistance by the malignant cells is increasing, and these treatments may not be useful following a relapse.
2
Hence, new drugs must be tested and proved efficacious in the treatment of lung cancer with the elimination of deficiencies in the drug that failed to protect in their mode of action. A surge in the search for new compounds available for the treatment has led researchers to opt for readily available plants and their isolated compounds.
3
Among these, thymoquinone, monoterpenes,
Myrcene is a volatile aromatic hydrocarbon that is abundantly found in plants such as hops, lemongrass, cannabis, and bay leaves. This volatile compound is used as a fragrance in the perfumery, food, and beverage industries. The antioxidant, antibacterial, anti-inflammatory, analgesic,
4
and antimutagenic properties of myrcene have also been well studied.
5
-7
The cytotoxic effects of
Several events related to lung cancer, such as the proliferation of cells, apoptosis, and deoxyribonucleic acid (DNA) damage are well associated with the maintenance of equilibria in mitochondria. 11 When this is lost, then alterations in the mitochondrial integrity will occur, 12 resulting in apoptosis, which is one of the cell regulatory events initiated by the release of cytochrome C from mitochondria and helped in cell death with the action of caspases and the control of the malignant growth of dysregulated cells. Hence, many of the known anticancer drugs are based on targeting the mitochondria of cancerous cells. 13 In our present study, we attempted to prove the efficacy of myrcene in its use as an effective anticancer compound in lung cancer using human lung adenocarcinoma A549 cells. The results obtained showed that the drug had its impact through activation of the mitochondria-mediated mechanism, thus showing that myrcene could be a potential drug candidate in the future for lung adenocarcinoma.
Results
The results obtained in the current investigations demonstrated the antiproliferative effect of myrcene on A549 lung carcinoma cells. Initial experiments on cell toxicity using a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) dye-based assay demonstrated that myrcene at concentrations of 0.25, 0.50, and 1.0 µg/mL exhibited a dose-dependent and time-dependent response (24 hours and 48 hours, respectively) for cell death compared with control cells with no treatment (Figure 1).
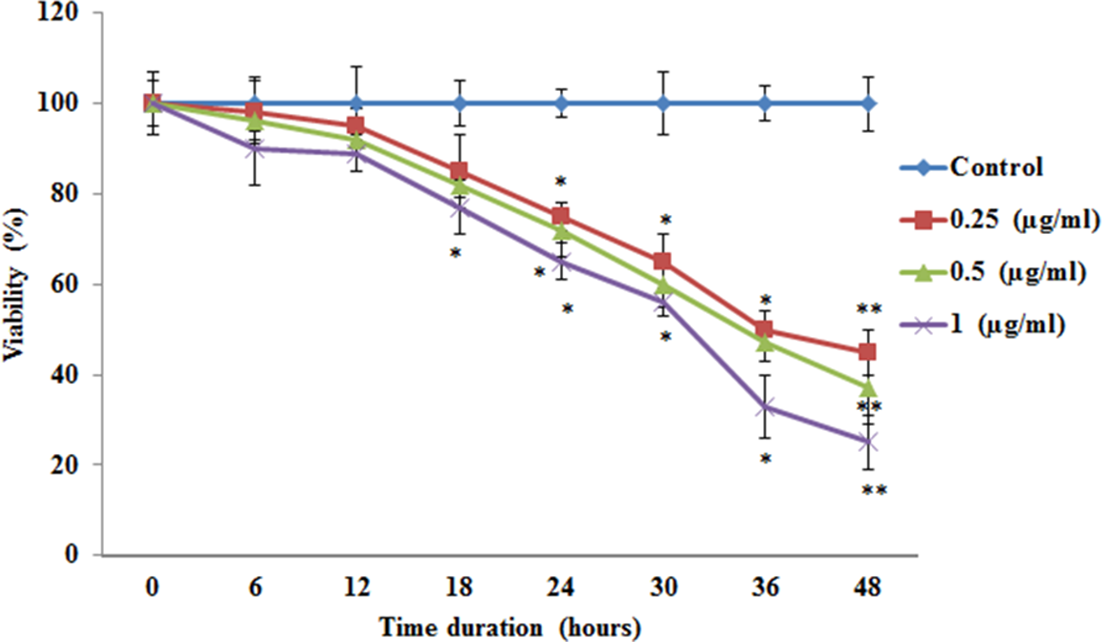
The cytotoxic effects of various concentrations of myrcene in A549 lung carcinoma cells for 48 hours. Details of the assay are given in the Materials and Methods section. The data are expressed as mean ± SD of 3 concurrent similar experiments. Statistical significance expressed as *
Figure 2 shows the results for cell cycle analysis (G0/G1, S, G2/M) in the control and experimental group of cells stained with propidium iodide (PI) for the DNA content using flow cytometry. The results documented that the normal progression of cell growth was altered after 6 hours of drug treatment. More marked effects were observed in cells incubated for more than 24 hours, with a significant difference in the cell cycle histogram compared with control cells. The alteration in cell growth favored a reduction in cell division in the myrcene-incubated cells (Figure 2(A) and (B)). Further, the effect of myrcene on clonal sphere formation in A549 cells was elucidated. After 24 and 48 hours of drug treatment, in comparison with the control group, the number of colony-forming units in the 0.25 µg/mL, 0.50 µg/mL, and 1.0 µg/mL drug-treated groups had undergone 93.4%, 89.1%, and 83.9% reduction and 84.6%, 75.2%, and 66.5% reduction, respectively, compared with the control cells. The results showed that the drug had had a high impact on the proliferation of lung carcinoma cells in a time-dependent and dose-dependent manner (Figure 3). In order to elucidate the mechanism of cell death induced by myrcene, the mitochondrial integrity was tested using rhodamine, and the results are presented in Figure 4. It is well documented that alteration in mitochondrial membrane potential (Δψm) is a major event leading to apoptosis. In the present study, cancer cells exposed to myrcene demonstrated a reduction in Δψm in a dose-dependent manner, whereas, the control cells, with no drug treatment, maintained structural integrity (Figure 4).

(A and B) The cell cycle analysis in control and experimental groups of cells. The total deoxyribonucleic acid (DNA) content is represented, analyzed by propidium iodide staining followed by flow cytometry using different concentrations of myrcene for 24 and 48 hours. The data are expressed as mean ± SD of 3 concurrent similar experiments. Statistical significance expressed as *

The effect of myrcene on clonal sphere formation in A549 cells after 24 and 48 hours of drug exposure, respectively. The relative number of colonies after treatment with various concentrations of myrcene is represented. The data are expressed as mean ± SD of 3 concurrent similar experiments. Statistical significance expressed as *
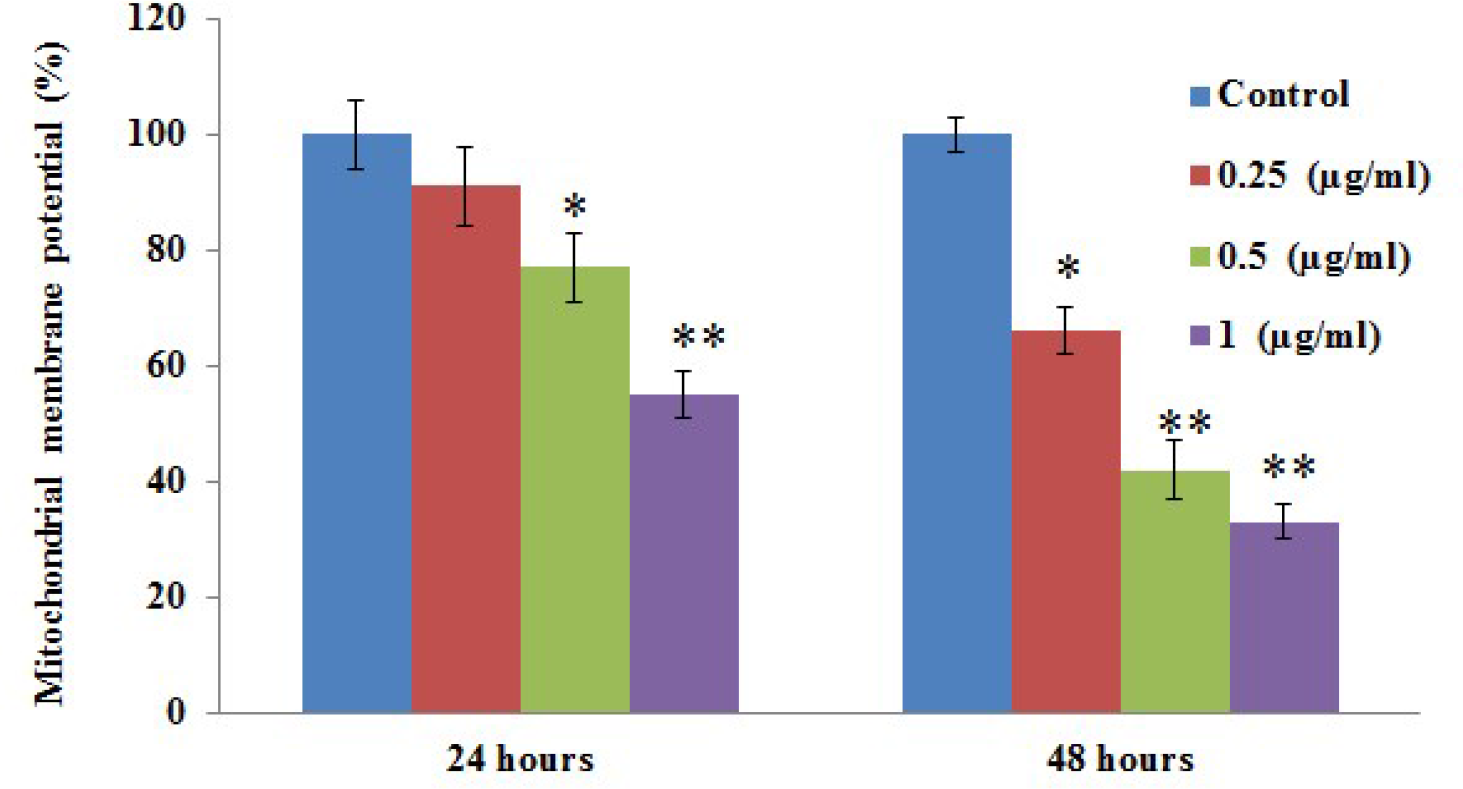
The effect of myrcene on the mitochondrial membrane potential (Δψm) in control and experimental groups of A549 cells. The results of 24 and 48 hours of drug exposure are shown. The data are expressed as mean ± SD of 3 concurrent similar experiments. Statistical significance expressed as *
Following the cellular organelle integrity assay, the role of enzymes involved in the activation of cell death was elucidated using the release of caspases, a well-known marker of the apoptosis execution phase. Using commercial assay kits, the activity of caspase-3 and caspase-9 was elucidated. The release of caspases was found to be significantly increased in the myrcene-treated cells in a dose-dependent manner compared with the control cells (Figure 5(A) and (B)). On the other hand, the observed loss of ΔΨm is related to the involvement of cytochrome C release in cells. As expected, the drug-treated cells indicated a dose-dependent increase in cytochrome C release in cancer cells, which illuminates the valid mechanism for the cell death induced by the drug (Figure 5(C)).

(A and B) The effect of myrcene on the activity of caspases and (C) the release of cytochrome c in A549 cells of control and experimental group, respectively. The data are expressed as mean ± SD of 3 concurrent similar experiments. Statistical significance expressed as *
Figure 6 displays the results attained from the analysis of phosphorylation of the histone H2AX (γH2AX form), a potent marker of cell death that is related to DNA damage. In the present study, cells exposed to myrcene exhibited a significant surge in γH2AX phosphorylation in a dose-dependent manner, which indicates that DNA damage is activated by myrcene (Figure 6).
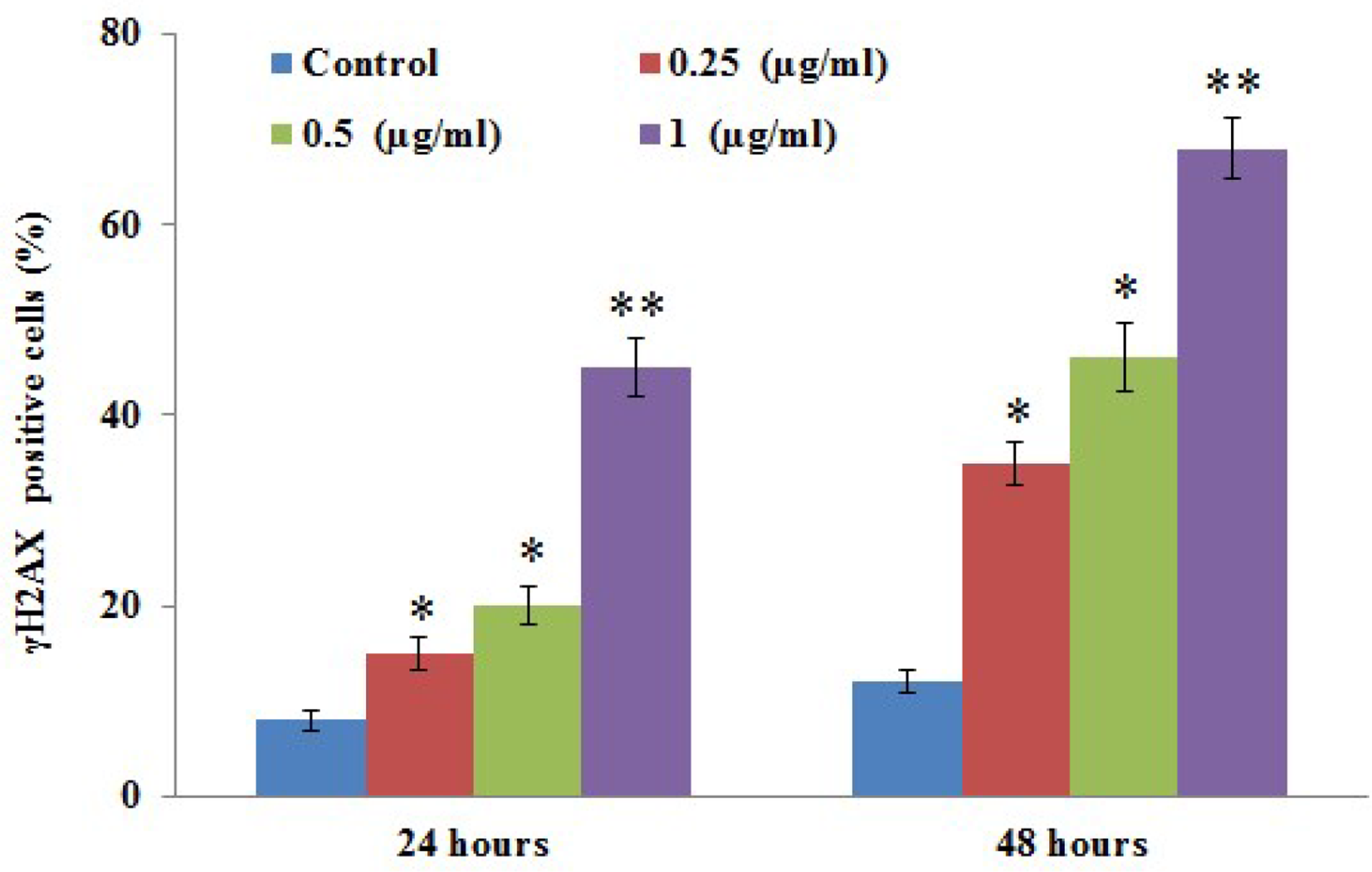
Phosphorylation of histone H2AX (γH2AX) in control and experimental groups of A549 cells exposed to myrcene is presented in the figure. The data are expressed as mean ± SD of 3 concurrent similar experiments. Statistical significance expressed as *
Discussion
The importance of myrcene for its antinociceptive, antimutagenic, and antiulcer effects has already been reported. 14 We have tried to exploit its antioxidant properties in testing its effectiveness against A549 lung carcinoma cells. Various physiological and functional aspects of cancer cells that give them their properties were examined in the presence of various concentrations of myrcene; the effectiveness of myrcene on A549 cells was dose-dependent. The maximum number of nonviable A549 cells was observed when the concentration of myrcene used was 1 µg/mL (Figure 1), with a half-maximal inhibitory concentration of 0.5 ug/mL.
The cell proliferative capacity of the A549 cell line in the presence of increasing dosages of myrcene indicated that the compound is effective in inhibiting cell proliferation and reducing the viability of cells, which is typical of an anticancer compound. This property of inhibiting the viability of cancerous cells is in line with the development of growth inhibition to cisplatin, to which the cancer cells are susceptible. This significant reduction in the growth of A549 cells over 48 hours highlights the probable therapeutic capability of this natural product.
From our results, we infer that the cell proliferation inhibition is mediated through the activation of various cellular pathways that would arrest the cells in various phases of the cell cycle. Our treatment of the cells with increased myrcene concentrations and flow cytometry results have shown that the maximal number of cells were arrested in the G0/G1 phases of the cell cycle. In our present study, we have proven the ability of myrcene’s antiproliferative effects by demonstrating on A549 cells a decrease in colony formation, activation of the classical apoptotic pathway, decrease in the mitochondria membrane potential, increase in caspase activation, and induction of DNA damage. The effect is dose-dependent and time-dependent with its inhibitory effect deduced from the colony formation assay (Figure 3(A) and (B)). In order to investigate this, we grew the cells in the presence of myrcene at different concentrations and observed the formation of colonies. These data on the formation of colonies would provide a measurable parameter in estimating the toxicity of myrcene on the number of colonies that are formed after the inhibition of cell proliferation. The number of colonies formed by A549 cells under control conditions was higher but decreased with increasing concentrations of myrcene. The effects were more pronounced and significant when the same cells were observed after 48 hours. Thus, myrcene inhibited the colony formation in A549 cells in a dose-dependent manner and inhibited the anchorage-independent growth of A549 cells.
Programmed cell death or apoptosis is a physiological event that occurs due to induction by various apoptotic stimuli on dysregulated cells, causing cytotoxicity. Among the major apoptotic pathways that are known, we have shown that myrcene-treated A549 cells have undergone the intrinsic pathway (mitochondrial) and exhibited significant cytotoxicity. In this intrinsic pathway, the permeability of the mitochondrial membrane, cytochrome C release, activation of caspase-9, and the effector caspase-3 activation are the prime factors that are considered. Myrcene inhibited the proliferation of A549 cells and can induce apoptosis in such cells, at a concentration that is significant at 1 µg/mL, with a reduction in mitochondrial membrane potential.
The reduced membrane potential makes the mitochondrial pores more permeable to the release of cytochrome C from mitochondria to cytosol. Cytochrome C then activates caspase-9 to produce a cascade effect on caspase-3 to activate the downstream apoptotic signaling pathway, leading to cell death. As shown in Figure 5, the increase in activity of caspase-9 and caspase-3 coordinated with the increase in cytochrome C by the weakened mitochondrial membrane potential of A549 cells treated with myrcene, at a concentration of 1 µg/mL over 48 hours, is significant. Apoptosis of the cells is accompanied by DNA damage, which is evident with the increase in the percentage of cells that are positive for γH2AX found in the cells treated with myrcene. This is characteristic of the apoptotic property of DNA fragmentation and is accompanied by the formation of γH2AX in cells. 15 Such cells show an increase in fragmented DNA, packaged into apoptotic bodies, which would be eliminated by cell scavenging.
γH2AX is important in the maintenance of genome stability 16 -18 and is an indicator of DNA damage and double-stranded breaks. 19,20 It is involved in the induction of apoptosis in cells treated with an anticancer drug. 21 It is known that the increase in soluble γH2AX is directly related to the inability of DNA replication exhibited in cells that are under cell cycle arrest and the stress associated with it. 22 This downstream effect would lead to the sensitization of cells to undergo apoptosis and disruption of gene transcription. A549 cells that are arrested in the G0/G1 phase of the cell cycle would be stressed by the effect of myrcene and would express soluble γH2AX 23 to undergo apoptosis and eventually be eliminated.
Materials and Methods
Cell Lines and Experimentation
Human lung cancer cell line (A549) was purchased from American Type Culture Collection (ATCC), USA. The cells were cultured in Roswell Park Memorial Institute 1640 medium supplemented with 10% heat-inactivated fetal bovine serum (Life Technologies, Thermo Fisher Scientific Inc. Waltham, MA, USA) with 2 mM/L glutamine, 50 U/mL penicillin, and 50 µg/mL streptomycin, and these cells were incubated in an incubator with 5% carbon dioxide at 37 °C. The cells were grown in culture plates and flasks until confluence. Control untreated A549 cells were grown without any treatment; other A549 cells were exposed to myrcene 0.25 µg/mL, 0.50 µg/mL, and 1.0 µg/mL for 24 and 48 hours to elucidate its antitumor effects.
Cytotoxicity Assessment
At the end of the treatment, cell toxicity was tested using the MTT cell proliferation assay (ATCC 30‐1010K) (Abcam Inc., Cambridge, MA, USA). Briefly, to control and experimental cells, 10 µL of MTT solution was added into each well and after incubation, 100 µL detergent was added to each well, and the plates were kept in the dark with shaking for 30 minutes to facilitate dissolution of the crystals; the color developed in each well was measured at 570 nm in a plate reader. Absorbance values of each well were repeated 3 times for an average measurement reading.
Cell Cycle Analysis
To determine the cell cycle status in the cultured cell lines, the DNA content was measured using a flow cytometer; a commercial PI flow cytometry kit for cell cycle analysis from Abcam was used (Ab139418, Abcam Inc., Cambridge, MA, USA). Briefly, control and myrcene exposed cells were harvested and fixed using 70% ethanol and stored at 4 °C for 2 hours, and the cells were rehydrated in phosphate-buffered saline (PBS). The cells were then stained with PI containing RNase for 30 minutes, and the fluorescence intensity of PI was read on an FL2 flow cytometer with 488 nM laser excitation (493ex; 636em).
Colony Formation Assay
The cells were tested for their ability to form colonies (clonal sphere formation) on myrcene exposure using a commercial Clonogenic Assay Kit (BioPioneer Inc. San Diego, CA, USA). Briefly, an equal number of cells grown in regular media up to 70% confluence were treated with different concentrations of myrcene, while the control cells were maintained with no treatment. At the end of the treatment, the medium was removed, and the cells were washed with PBS buffer 2‐3 times and fixed with the fixing solution provided in the kit. After that, the cells were stained using the staining solution and left at room temperature for 45 minutes. Finally, the cells were washed twice with PBS, and the blue colonies counted. The colony formation rate was calculated according to the following formula: Colony formation rate = Number of colonies/Number of inoculated cells × 100%.
Mitochondrial Membrane Potential
In order to determine the mechanism of cell death, a tetramethylrhodamine ethyl ester (TMRE)-mitochondrial membrane potential (Δψm) Assay Kit (ab113852; Abcam Inc., Cambridge, MA, USA) was utilized to spotlight the vitality of mitochondria. Briefly, cells grown in well plates with or without myrcene treatment for 24 and 48 hours and the media were aspirated and replaced with 100 µL of 0.2% bovine serum albumin in PBS. Later, the TMRE stain provided in the kit was added to the cells and incubated for 20 minutes at 37 °C. Then the Δψm in the cells was calculated using a fluorescence plate reader with a setting suitable for TMRE: Ex/Em = 549/575 nm. The results were expressed as fluorescence intensity as a percentage compared with that of the untreated control.
Caspases Activation and Cytochrome Release
For the estimation of caspase-3 (b39401) and caspase-9 activity, commercial assay kits (ab65608; Abcam Inc., Cambridge, MA, USA) were used. In addition, a Cytochrome c Releasing Apoptosis Assay (KHO1051) was carried out using a commercial kit (Thermo Fisher Scientific Inc. Waltham, MA, USA). The assays were performed according to the manufacturer’s instructions. Briefly, for the caspases assay, control and drug-treated cells were lysed, incubated with a reaction buffer containing the labeled specific substrates, incubated at 37 °C for 1 hour, and the samples read at 400 nm using the microplate reader to detect the release of chromophore p-nitroaniline after cleavage from the labeled substrate. The results are expressed as fold increase compared with untreated controls. For cytochrome C activity, the results are expressed as ng/mL.
γH2AX Assay
An H2AX phosphorylation assay kit (Catalog # 17‐344) for flow cytometry (Millipore, Temecula, CA, USA) was used. For the analysis, control and drug-treated cells were collected after trypsinization and resuspended in 1× Fixation solution at a cell density of 2 × 106/mL and incubated for 20 minutes on ice to fix cells and washed using PBS before permeabilizing. Later, anti-phospho-Histone H2A.X (Ser139), fluorescein isothiocyanate (FITC) conjugate, or the negative control mouse immunoglobulin G-FITC conjugate and PI were added to each well and incubated on ice for 20 minutes. Then, the cells were washed to remove excess FITC labeled antibody, and the cell pellets in 150 µL flow cytometry staining (FACS) buffer per well were run in a FACS machine. The results were calculated from the percentage of gated cells positive for γH2AX, and PI were calculated regarding the total cells gated.
Statistical analysis
Data obtained were expressed as mean ± standard error (SE) of the mean. GraphPad Prism (Version 8) was used for statistical analysis (GraphPad Software, San Diego, CA, USA). To compare two groups, Student’s test was performed. Differences in the values were considered to be statistically significant when
Conclusion
We have satisfactorily proven that myrcene has a positive effect on the control of lung cancer through its actions on A549 cells, and the compound was confirmed to be involved in the antiproliferation and apoptosis of the cells. These findings suggest that myrcene may be an effective chemopreventive agent in cancer, and further experiments to explore the possibilities are required.
Footnotes
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
All the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care guidelines and approved by the Administration Committee of Experimental Animals of Shanxi Bethune Hospital, Shanxi Academy of Medical Sciences, Taiyuan, Shanxi, China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
