Abstract
Kaempferide is an O-methylated flavonol that has received much attention due to its various biological activities. In this study, we explored the underlying mechanisms of kaempferide in human lung cancer A549 cells. The Cell Counting Kit-8 (CCK-8) assay, Hoechst 33342/propidium iodide double staining, flow cytometry, scratch wound healing assay, and Western blot analysis were used to measure cell apoptosis, the cell cycle, reactive oxygen species (ROS) levels, and cell migration of human lung cancer cells. Kaempferide significantly inhibited human lung cancer cell proliferation, and its toxic effects on normal cells were significantly lower than those of 5-fluorouracil. Kaempferide induced A549 cell apoptosis by decreasing the mitochondrial membrane potential and the expression level of B-cell lymphoma 2, and by increasing the expression levels of Bcl-2-associated X protein and caspase-3. It also regulated mitogen-activated protein kinase (MAPK), signal transducer and activator of transcription 3 (STAT3), and nuclear factor kappa B (NF-κB) signaling pathways by increasing the expression levels of phosphorylated c-Jun N-terminal kinase, p-p38, I kappa B, and by decreasing the expression levels of phosphorylated extracellular signal-regulated kinase, p-STAT3, and NF-κB. Kaempferide induced cell cycle arrest in the G0/G1 phase in A549 cells by downregulating the expression levels of p-AKT, cyclin D1, and cyclin-dependent kinase 2. Furthermore, kaempferide blocked A549 cell migration by downregulating the expression levels of transforming growth factor beta 1 (TGF-β1), p-β-catenin, p-glycogen synthase kinase 3 beta, N-cadherin, and vimentin, and by upregulating the expression level of E-cadherin. Kaempferide enhanced the accumulation of ROS, and N-acetyl-
Lung cancer, a common lung malignant disease, is recognized as the deadliest cancer by the American Cancer Society. There are about 2 million new lung cancer patients and 1.5 million deaths reported worldwide every year. 1 At present, the main treatment methods for lung cancer are surgery, radiotherapy, and chemotherapy. However, there are risks in the course of treatment and side effects after treatment. 2,3 Therefore, it is urgent to find efficient therapeutic agents.
Apoptosis, the process of programmed cell death, is an important link in the process of cell growth. 4 Several studies have reported that apoptosis is regulated by reactive oxygen species (ROS). 5,6 Meantime, ROS regulates mitogen-activated protein kinase (MAPK), signal transducer and activator of transcription 3 (STAT3), nuclear factor-kappa B (NF-κB), and protein kinase B (AKT) signaling pathways to induce apoptosis and regulate transforming growth factor beta (TGF-β) to limit cell migration. 7 -9 The MAPK signaling pathway plays a key role in the regulation of gene expression and cytoplasmic function. The transmission of important signals is mainly through 4 pathways, namely, c-Jun N-terminal kinase (JNK), extracellular signal-regulated kinase (ERK), ERK5, and p38. 10 Meanwhile, the STAT3 signaling pathway is involved in the regulation of cell growth and apoptosis through the regulation of cytokines and growth factors that change the state of cell growth. 11 The NF-κB signaling pathway has a strong role in promoting cell proliferation, and controlling its expression can increase the rate of cell apoptosis. 12 Furthermore, AKT plays an important role in cell survival and apoptosis and is involved in regulating the rate of cell division. 13 TGF-β is a transforming growth factor; when activated in cancer cells, cells become more active, causing lesions in surrounding cells and promoting cancer cell spread and metastasis. 14,15 The characteristic proteins of each pathway can regulate cell growth and apoptosis. Kaempferide is a kind of methyl flavonoid that is widely found in Sophora japonica, tea, and ginger plants. 16 It has good anti-inflammatory and hypolipidemic effects, and can regulate the transmission of malaria and osteoclastic nerve signals, boosting the human immune system. 17 -20 It has been shown that kaempferide can prevent myocardial ischemia, protect the myocardium, and reduce myocardial injury through phosphoinositide 3-kinases, glycogen synthase kinase-3β (GSK-3β), and other pathways. 21 It also has the ability to reverse cognition and has certain preventive effects against Alzheimer’s disease. 22 Furthermore, kaempferide has strong inhibitory effects on colon cancer cells. 23
In this study, we elucidated the mechanism of action of kaempferide in A549 human lung cancer cells to provide a basis for the clinical application of drugs and the development of novel anticancer drugs.
Results
Kaempferide Inhibits the Proliferation of Human Lung Cancer Cells
As shown in Figure 1(A), kaempferide had obvious killing effects on human lung cancer cells, which were significantly higher than those in control cells treated with 5-fluorouracil (5-FU). The IC50 values of A549, NCI-H460, and NCI-H23 cell lines were 22.5 ± 1.35, 29.1 ± 1.5, and 26.2 ± 1.4 µM, respectively. Kaempferide had no obvious toxic effects in 3 human normal cell lines (Figure 1(B)). Meantime, as shown in Figure 1(C), the survival rate of human lung cancer cells treated with kaempferide for 0, 3, 6, 12, 24, and 36 hours was determined, and the cell survival rate decreased with increased incubation times. There was no significant decrease in the survival rate of normal cells (Figure 1(D)). In contrast, A549 cells were more sensitive to kaempferide, and thus, were chosen for subsequent experiments.

Killing effects of kaempferide on human lung cancer cells. (A) A549, NCI-H460, and NCI-H23 lung cancer cells were treated with different concentrations of kaempferide (1, 3, 10, 30, and 100 µM) for 24 hours. The cell viabilities were determined by CCK-8 assay. (B) Human normal liver L-02 cells, normal gastric GES-1 cells, and normal lung IMR-90 cells were treated with different concentrations of kaempferide (1, 3, 10, 30, and 100 µM) for 24 hours. The cell viabilities were determined by CCK-8 assay. (C) Human lung cancer A549, NCI-H460, and NCI-H23 cells were treated with 20 µM kaempferide for 0, 3, 6, 12, 24, and 36 hours. Cell viabilities were determined by CCK-8 assay. (D) Human normal liver L-02 cells, normal gastric GES-1 cells, and normal lung IMR-90 cells were treated with 20 µM kaempferide for 0, 3, 6, 12, 24, and 36 hours. Cell viabilities were determined by CCK-8 assay. *P < .05, **P < .01, or ***P < .001 vs 5-fluorouracil group.
Kaempferide Induces Mitochondrial-Dependent Apoptosis in A549 Cells
As shown in Figure 2(A), with an increase in kaempferide treatment time, the amount of apoptosis significantly increased and was higher than that in the 5-FU control group. As shown in Figure 2(B), the flow cytometry results showed that the total number of early apoptotic and late apoptotic cells increased by 45.1%, and the number of apoptotic cells treated with kaempferide was significantly higher than that of the control group (P < .001). Kaempferide had a regulatory effect on mitochondrial membrane potential (MMP; Figure 2(C)). It also induced apoptosis by upregulating the expression levels of Bcl-2-associated X protein (Bax), caspase-3, and poly (ADP ribose) polymerase (PARP) and by downregulating the expression level of B-cell lymphoma 2 (Bcl-2; Figure 2(D)). These results showed that kaempferide induced apoptosis in A549 cells via a mitochondrial-dependent pathway.

Apoptotic effects of kaempferide on A549 cells. (A) A549 cells were treated with 20 µM kaempferide for 0, 3, 6, 12, and 24 hours. Cell morphology and fluorescence intensity were analyzed by fluorescence microscopy. (B) A549 cells were treated with 20 µM kaempferide for 0, 3, 6, 12, and 24 hours. The distribution of apoptosis was analyzed by flow cytometry. (C) A549 cells were treated with 20 µM kaempferide for 0, 3, 6, 12, and 24 hours. Changes in mitochondrial membrane potential were analyzed by flow cytometry. (D) A549 cells were treated with 20 µM kaempferide for 0, 3, 6, 12, and 24 hours. The expression of Bax, Bcl-2, caspase-3, and PARP were analyzed by Western blotting. α-Tubulin was used as the internal control. *P < .05, **P < .01, or ***P < .001 vs control group.
Kaempferide Induces Apoptosis in A549 Cells via the MAPK, STAT3, and NF-κB Signaling Pathways
The expression levels of p-JNK, I-κB, and p-p38 in A549 cells were significantly increased with increasing kaempferide treatment time, but the expression levels of p-ERK, p-STAT3, and NF-κB were decreased (Figure 3(A)). To evaluate the relationship among MAPK, STAT3, and NF-κB signaling pathways, we pretreated the cells with the following MAPK inhibitors: JNK inhibitor (SP600125), p38 inhibitor (SB203580), and ERK inhibitor (FR180204). As shown in Figure 3(B), when SP600125 and SB203580 were added, the inhibitory effect of kaempferide was alleviated and the expression levels of p-STAT3 and NF-κB were increased; the activation effect of kaempferide was limited, and the expression levels of p-JNK, p-p38, I-κB, and caspase-3 were decreased. FR180204 enhanced the inhibitory ability of kaempferide and decreased the expression levels of p-ERK, p-STAT3, and NF-κB, but limited kaempferide activation to the expression level of I-κB. It also increased the activation ability of kaempferide and increased the expression level of caspase-3. These results showed that kaempferide induced the apoptosis of A549 human lung cancer cells through MAPK, STAT3, and NF-κB pathways.

Kaempferide induces apoptosis of A549 cells via the MAPK/STAT3/NF-κB signaling pathways. (A) The expression of MAPK/STAT3/NF-κB signaling pathway-related proteins was analyzed by Western blotting. (B) A549 cells were treated with MAPK inhibitors (SP600125, SB203580, and FR180204) and kaempferide. The expression of p-JNK, p-p38, p-ERK, p-STAT3, I-κB, NF-κB, and caspase-3 was analyzed by Western blotting. α-Tubulin was used as the internal control. *P < .05, **P < .01, or ***P < .001 vs control group and inhibitor + kaempferide group.
Kaempferide Induces Apoptosis by Increasing Intracellular Reactive Oxygen Species Levels in A549 Cells
As shown in Figure 4(A), kaempferide significantly increased ROS levels in a time-dependent manner. N-acetyl-

Kaempferide stimulates reactive oxygen species to induce apoptosis of A549 cells. (A) A549 cells were treated with 20 µM kaempferide for 0, 3, 6, 12, and 24 hours. Intracellular reactive oxygen species levels were analyzed by flow cytometry. (B) A549 cells were treated with N-acetyl-
Kaempferide Arrests A549 Cells in the G0/G1 Cell Cycle
The percentage of cells in the G0/G1 phase significantly increased, and the percentage of cells in the G2/M phase gradually decreased with increasing treatment time, indicating that kaempferide arrested A549 cells in the G0/G1 phase (Figure 5(A)). The expression levels of p-AKT, cyclin-dependent kinase 2 (CDK2), CDK4, CDK6, cyclin D1, and cyclin E were decreased; the expression levels of p21 and p27 were increased with the prolongation of treatment time (Figure 5(B)). Flow cytometry analysis showed that after pretreatment with NAC, G0/G1 cell cycle arrest was reversed (Figure 5(C)). Meanwhile, after adding NAC, kaempferide inhibited p-AKT, CDK2, CDK4, CDK6, cyclin D1, and cyclin E were decreased; active p21 and p27 were restricted (Figure 5(D)). These results showed that kaempferide induced G0/G1 cell cycle arrest through the ROS-mediated AKT signaling pathway.

Kaempferide arrests A549 cells in the G0/G1 phase. (A) A549 cells were treated with 20 µM kaempferide for 0, 3, 6, 12, and 24 hours. Periodic distribution was analyzed by flow cytometry. (B) A549 cells were treated with 20 µM kaempferide for 0, 3, 6, 12, and 24 hours. The expression levels of p-AKT, p21, p27, CDK2, CDK4, CDK6, cyclin D1, and cyclin E were analyzed by Western blot. (C) A549 cells were treated with N-acetyl-
Kaempferide Inhibits A549 Cell Migration via the Transforming Growth Factor Beta Pathway
As shown in Figure 6(A), the control group had a significant healing trend with the extension of culture time. In the kaempferide group, there was a tendency to heal in a shorter time, but there was no obvious sign of healing at 24 hours, and some cells had shrinkage and rounding, and there were signs of apoptosis. As shown in Figure 6(B), the expression levels of TGF-β1, p-GSK-3β, SNAI 1, p-β-catenin, N-cadherin, and vimentin had a significant downward trend, whereas the expression levels of E-cadherin had a significant upward trend. As shown in Figure 6(C), after NAC and kaempferide treatment, Western blot analysis showed that NAC decreased the expression level of E-cadherin increased by kaempferide. N-Acetyl-

Kaempferide inhibits A549 cell migration. (A) A549 cells were treated with 20 µM kaempferide for 0, 3, 6, 12, and 24 hours. Cell morphology was analyzed by fluorescence microscopy. (B) A549 cells were treated with 20 µM kaempferide for 0, 3, 6, 12, and 24 hours. The expression of TGF-β1, SANI1, p-β-catenin, p-GSK-3β, E-cadherin, N-cadherin, and vimentin was analyzed by Western blotting. (C) A549 cells were treated with N-acetyl-
Discussion
Kaempferide, a flavonoid extracted from herbal medicine, has a variety of biological activities. Some studies have shown that it has strong inhibitory effects on cervical cancer, as determined by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. 24 In this study, kaempferide had significant killing effects on lung cancer cells and no obvious side effects on normal cells. The killing effects were via mitochondrial-dependent apoptosis. Mitochondrial-dependent apoptosis is mainly regulated by the Bcl-2 family of proteins, which transfer signals from the cytoplasm to the outer membrane of mitochondria. The signals interact with voltage-dependent anion channels on the membrane to release apoptotic factors from the mitochondria into the cytoplasmic matrix, to induce apoptosis. 25,26 In this study, we found that kaempferide upregulated expression of the pro-apoptotic protein Bax and downregulated expression of the anti-apoptotic protein Bcl-2, which led to decreases in the MMP, destruction of mitochondrial homeostasis, and initiation of the caspase cascade, ultimately leading to cell apoptosis. Some research has shown that MAPK, STAT3, and NF-κB pathways mediate cell apoptosis. The MAPK pathway is one of the most important pathways that regulate cell survival; disruption of this pathway leads to the inhibition of A549 cell growth and reproduction. 27 STAT3 acts as a transmitter of cell differentiation, which affects the proliferation of cells during the process of conduction. When the expression of p-STAT3 is inhibited, the cells move toward apoptosis. 28,29 At the same time, the STAT3 signaling pathway can be regulated by MAPKs in cell apoptosis. 30 The expression of NF-κB protein plays an important role in cell proliferation and apoptosis. 31,32 Reddy et al have also found that the drug can promote cell apoptosis through the p38, MAPK, and NF-κB signaling pathways, and this has been confirmed from the gene aspect. 33 In this study, kaempferide activated the p38 and JNK signaling pathways through protein phosphorylation, and inhibited the ERK signaling pathway, further inhibiting STAT3 and NF-κB activity, resulting in cell apoptosis.
The AKT pathway plays an important role in cell cycle expression, which can inhibit cell proliferation by restricting AKT signal transduction. 34 Kaempferide has been shown to arrest colon cancer cells in the G0/G1 phase and inhibit their proliferation. 23 Similarly, our results showed that A549 cells treated with kaempferide were blocked in the G0/G1 phase. Some studies have indicated that the cell cycle is regulated by CDK and cyclins. Cyclin-dependent kinases can form a complex with the corresponding cyclin to promote the cell cycle. 35,36 Here, we found that kaempferide promoted the expression of p21 and p27 by inhibiting the AKT signaling pathway, and p21 and p27 could bind to CDK2, CDK4, CDK6, cyclin D1, and cyclin E, preventing the cells from passing the cell cycle checkpoint. Eventually, the cells were arrested in the G0/G1 phase, and normal cell division could not be completed, which limited cell proliferation.
The proliferation rate of cancer cells is limited, which is of great significance for the treatment of cancer. The expression of TGF-β proteins in cells is closely related to their proliferation and metastasis. 37 Some studies have shown that the TGF-β/β-catenin pathway plays an important role in limiting cell metastasis and can also either promote or inhibit the expression of downstream proteins. 38,39 In the present study, we found that the expression level of E-cadherin was upregulated, and those of TGF-β1, SANI1, p-β-catenin, p-GSK-3β, N-cadherin, and vimentin were decreased to different extents. These results showed that kaempferide regulated the signal transduction pathways in A549 cells, so that the cells could not proliferate normally and limiting their range of activities, thereby inhibiting the migration of lung cancer cells.
It is well known that cell apoptosis can be regulated by ROS, and increasing the accumulation of ROS causes strong killing effects on cells. 40 Meanwhile, ROS can regulate the MAPK, STAT3, NF-κB, and AKT signaling pathways, which are involved in apoptosis, proliferation, and other life processes. 41,42 In this study, kaempferide increased ROS levels, which led to the regulation of downstream signaling pathways and promoted cell apoptosis. However, when the cells were treated with NAC and kaempferide, NAC had no effect on the control group, but had a strong inhibitory effect on the kaempferide group. Thus, kaempferide stimulated ROS after entering the cells. Then, it mediated the pathways of MAPK, STAT3, NF-κB, AKT, and TGF-β to inhibit A549 cell proliferation, to control the growth and metastasis of cells, thereby achieving anticancer effects.
There are still some shortcomings. Reddy et al also carried out gene expression analysis while determining the changes of protein expression in cells, which could give greater clarification about the drug on cancer cells targets. 43 In summary, kaempferide induced apoptosis, caused cell cycle arrest, and limited cell migration in lung cancer cells though ROS-mediated MAPK, STAT3, AKT, and TGF-β pathways (Figure 7). The results of this study lay the foundation for the development of novel drugs for the treatment of lung cancer, promoting the development of therapies for cancer prevention and control.
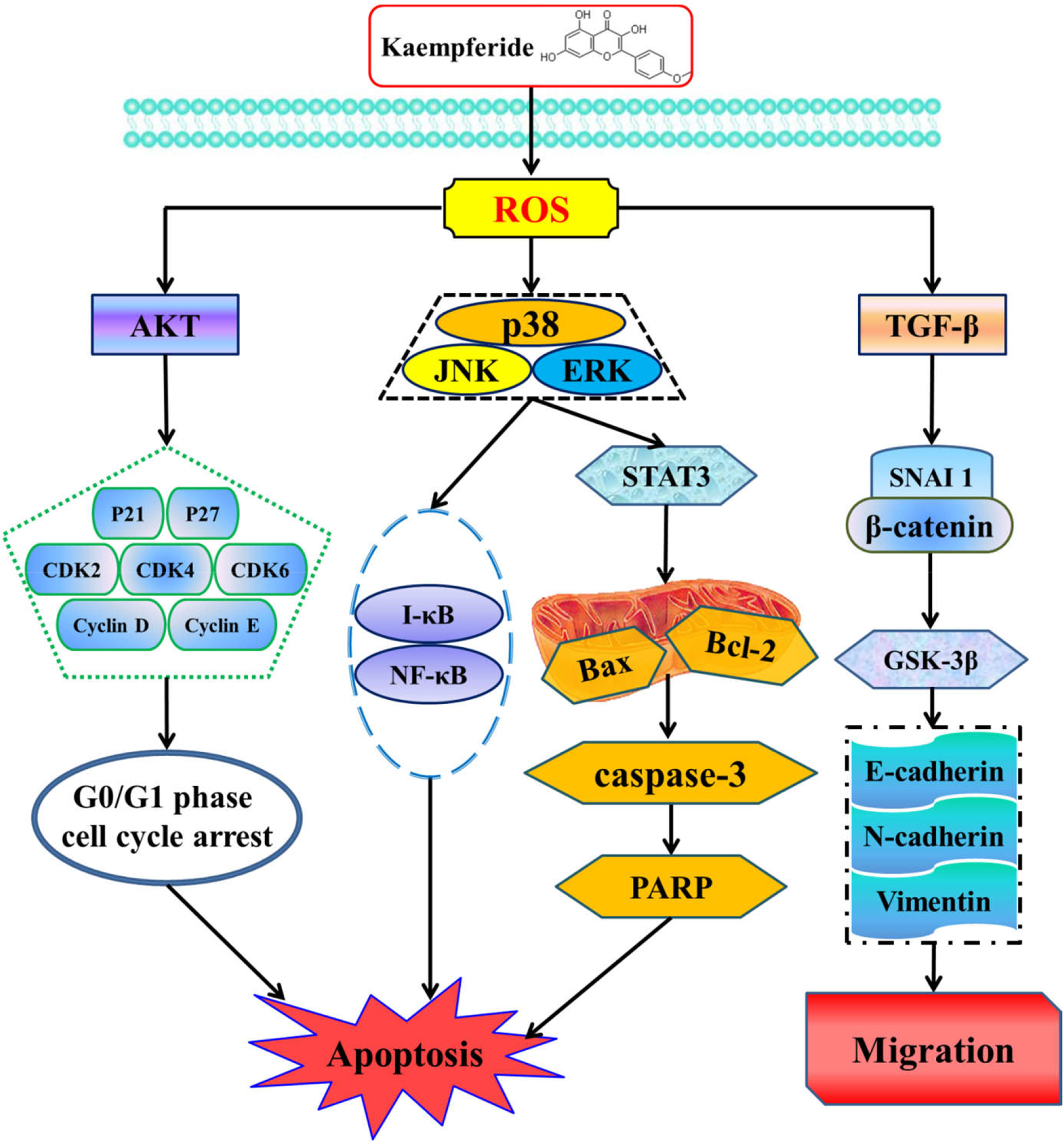
Mechanism of kaempferide in human lung cancer A549 cells.
Experimental
Cell Activity Analysis
The cells were inoculated in 96-well plates (5 × 104 cells/well) and cultured for 24 hours. Different concentrations of kaempferide (1, 3, 10, 30, and 100 µM) were added to the cells and incubated for 24 hours. Then, the cells were incubated with 10 µL CCK-8 solution for 2 hours, and the absorbance was measured at 450 nm using an Automatic Microplate Reader (BioTek Instruments, Inc., Winooski, VT, United States). The half-maximal inhibitory (IC50) values were calculated with GraphPad Prism software.
Double Staining With Hoechet 33342/Propidium Iodide
A549 cells were seeded in a 6-well plate (1 × 105 cells/well) and cultured for 24 hours. Cells were incubated with kaempferide (20 µM) for different times (0, 3, 6, 12, and 24 hours). After removing the culture medium, 1 mL cell staining buffer, 5 µL Hoechst staining solution, and 5 µL propidium iodide (PI) staining solution were added to the cells, followed by shaking and incubation at 4 °C for 30 minutes. The cell morphology was observed using the EVOS FL Auto Cell Imaging System (Thermo Fisher Scientific, Waltham, MA, United States) at a magnification of 400×.
Apoptosis Analysis
A549 cells were seeded in a 6-well plate (1 × 105 cells/well) and cultured for 24 hours. Cells were incubated with kaempferide (20 µM) for different times (0, 3, 6, 12, and 24 hours). Then, the cells were removed and rinsed 2 to 3 times with PBS. The A549 cells were resuspended in 1× binding buffer and adjusted to 1 × 106 cells/mL. Then, 100 µL cell suspension was mixed with 5 µL Annexin V-FITC and incubated at room temperature without light for 5 minutes, followed by the addition of 10 µL PI and 400 µL PBS. The apoptotic rate was detected by flow cytometry (BD Biosciences, San Diego, CA, United States).
Detection of the Mitochondrial Membrane Potential
JC-1 was used as a fluorescent probe. When the MMP was high, JC-1 emitted red fluorescence. A549 cells were cultured in a 6-well plate (1 × 105 cell/well) and incubated with 20 µM kaempferide for different times (0, 3, 6, 12, and 24 hours), after which the cells were removed and incubated with JC-1 for 20 minutes at room temperature. The data were analyzed by flow cytometry.
Analysis of Reactive Oxygen Species Generation
A549 cells were seeded in a 6-well plate (1 × 105 cells/well) and cultured for 24 hours. Cells were treated with 20 µM kaempferide for different times (0, 3, 6, 12, and 24 hours), followed by incubation with a 2′,7′-dichlorofluorescein diacetate probe at 37 °C for 20 minutes. Then, flow cytometry was conducted to measure ROS levels.
Scratch Wound Healing Assay
The A549 cells were inoculated in a 6-well plate at a density of 5 × 105 cells/well. When the cells were completely fused, the lineation treatment was conducted and the exfoliated cells were washed with PBS. Then, A549 cells were treated with kaempferide (20 µM) for different times (0, 3, 6, 12, and 24 hours). Images were analyzed using the EVOS FL Auto Cell Imaging System at a magnification of 100×.
Cell Cycle Analysis
A549 cells were seeded in a 6-well plate (1 × 105 cells/well) and cultured for 24 hours. Kaempferide (20 µM) were incubated for different times (0, 3, 6, 12, and 24 hours). The cells were collected and fixed in 70% ethanol for 2 hours. Briefly, the supernatant was removed after washing with PBS. The cells were resuspended with RNase A and incubated at 37 °C in the dark for 30 minutes. Then, the cells were incubated with PI solution and incubated at 4 °C in the dark for 30 minutes. Flow cytometry was used to detect the cell cycle.
Western Blot Analysis
Collected cells were washed with PBS and then lysed using protein lysis solution. The protein was resolved on 8% to 12% SDS-PAGE gels and transferred to a nitrocellulose membrane using the semidry transfer method. The membrane was blocked in 5% skimmed milk at room temperature for 2 hours, and then incubated with the following primary antibodies for 12 hours at 4 °C: Bcl-2, Bax, cleaved caspase-3, and cleaved PARP, as characteristic proteins of apoptosis; JNK, p38, ERK, STAT3, I kappa B (I-κB), and NF-κB to infer the relationship between signaling pathways; AKT, p21, p27, CDK2, CDK4, CDK6, cyclin D1, and cyclin E to verify cell cycle arrest; and TGF-β1, phosphorylated GSK-3β (p-GSK-3β), SANI1, p-β-catenin, E-cadherin, N-cadherin, and vimentin to determine the authenticity of cell migration. The membranes were probed with either goat antimouse IgG or goat antirabbit IgG secondary antibodies for 2 hours at room temperature. Then, membranes were detected using the Amersham Imager 600 (GE Healthcare, Fairfield, CT, United States). Image J software was used for evaluation and analysis, and α-tubulin was used as the internal control.
Statistical Analysis
All of the experiments were repeated 3 times. The data were analyzed using SPSS 21.0 and expressed as mean
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Postdoctoral Scientific Research Foundation of Heilongjiang Province of China under Grant (LBH-Q13132), the Heilongjiang Farms & Land Reclamation Administration Support Project for Key Scientific Research (HKKYZD190705), the Heilongjiang Bayi Agricultural University Support Program for “San Zong” (TDJH201905), and Heilongjiang Touyan Innovation Team Program (2019HTY078). In addition, we thank LetPub (www.letpub.com) for their linguistic assistance during the preparation of this manuscript.
