Abstract
Background
Exercise-induced muscle damage (EIMD) was characterized by microstructural disruption, sterile inflammation, oxidative stress, and delayed-onset soreness, transiently reducing force production and training capacity. Conventional analgesics alleviated pain yet carried safety concerns and did not consistently accelerate biological recovery.
Knowledge Gap/Problem Statement
Evidence for curcumin, a pleiotropic polyphenol, had grown but remained fragmented by heterogeneous dosing, formulations, timing, and outcome measures; the magnitude of benefit and key moderators were not clearly framed for practice.
Objectives
This review aimed to synthesize mechanistic, preclinical, and clinical evidence on curcumin for EIMD; to summarize effect sizes on soreness, function, and biomarkers; to identify moderators (formulation, dose, timing, training status); and to outline safety and translational implications.
Methodology
We conducted a structured narrative review with a systematic search of PubMed, Scopus, and Web of Science from inception to April 2025 using predefined terms for curcumin/turmeric, EIMD/DOMS, inflammation, and oxidative stress; English-language in-vitro, animal, and human studies were included; conference abstracts without full text and studies lacking direct relevance were excluded.
Results
Mechanistic and preclinical data showed that curcumin inhibited NF-κB, JAK/STAT, and MAPK signaling; reduced COX-2/5-LOX eicosanoids; activated Nrf2-driven antioxidant defenses; and preserved mitochondrial function, collectively limiting secondary muscle damage and facilitating regeneration. In human trials, curcumin supplementation—especially with bioavailability-enhanced preparations—consistently attenuated post-exercise increases in muscle-damage and inflammatory markers and improved recovery metrics. Across randomized studies and meta-analytic syntheses, curcumin reduced perceived soreness, lowered CK and IL-6 elevations, and modestly preserved ROM; dosing/timing strategies (pre- and early post-exercise, multi-day courses) and enhanced formulations strengthened effects.
Conclusions
The totality of evidence positioned curcumin as an NSAID-sparing adjunct that safely mitigated EIMD-related inflammation and oxidative stress and supported faster functional recovery. Standardizing PK-informed dosing and reporting, and testing long-term use across training cycles, were warranted to refine guidance for athletes and clinicians.
Keywords
Introduction
Exercise-induced muscle injury (EIMD) referred to the microtrauma and physiological strain that occurred in muscle tissue after intense or unaccustomed exercise. Such injury was common after eccentric muscle contractions (lengthening under load) and was characterized by structural damage to muscle fibers, increased muscle cell membrane permeability (leading to leakage of enzymes like creatine kinase), and impaired excitation-contraction coupling. 1 Clinically, EIMD manifested as delayed onset muscle soreness (DOMS), swelling, stiffness, reduced range of motion (ROM), and temporary loss of muscle strength in the affected area.1,2 Symptoms typically began 12-24 h post-exercise, peaked around 24-72 h, and persisted for several days, impacting athletic performance in subsequent workouts. 2 Underlying these symptoms were physiological processes including acute inflammation and oxidative stress in the muscle tissue. Effective recovery from EIMD was crucial for athletes and active individuals to maintain training consistency and performance. 3 However, muscle recovery was often challenging, and conventional interventions had limitations. 4 Common practices for managing DOMS and muscle injury included rest, massage, stretching, and RICE, as well as nonsteroidal anti-inflammatory drugs (NSAIDs) for pain relief. 5 While these approaches alleviated symptoms, they often did not significantly accelerate the intrinsic healing process of muscle fibers.6,7 In the case of NSAIDs, although they reduced pain and inflammation, their repeated use had been associated with potential side effects and concerns about blunting muscle adaptation to training. 8 There was thus interest in adjunct therapies that could safely reduce inflammation and oxidative damage without impairing muscle regeneration. In recent years, nutritional and nutraceutical interventions had gained popularity for aiding muscle recovery. 9 Among these, curcumin had emerged as a prominent natural compound of interest. 10
Curcumin was a polyphenolic compound derived from the rhizomes of Curcuma longa (turmeric), a plant in the ginger family. Turmeric had a long history of use in traditional medicine; in Ayurvedic practices, turmeric was applied to treat sprains, swelling, and other inflammatory ailments. 11 The bright yellow-orange spice contained curcuminoids (approximately 77% curcumin, along with demethoxycurcumin and bisdemethoxycurcumin) as its active pigments. Chemically, curcumin was classified as a diarylheptanoid; it was hydrophobic and exhibited tautomeric forms (enol and keto) that contributed to its reactivity. Curcumin was renowned for its broad pharmacological profile, notably its potent anti-inflammatory and antioxidant properties. 12 It had been studied in various disease contexts such as arthritis, metabolic syndrome, and neurodegenerative diseases for its ability to modulate inflammatory pathways and neutralize reactive oxygen species. These same properties suggested curcumin could mitigate the inflammation and oxidative stress associated with EIMD. Research into curcumin as a sports supplement had accelerated, examining whether curcumin ingestion before and/or after exercise could reduce DOMS, limit muscle damage markers, and improve recovery. Curcumin's mechanisms – including inhibition of key inflammatory signaling pathways and direct antioxidant action – aligned with the pathophysiological factors of EIMD, providing a strong rationale for its use in this context. Notably, curcumin in encapsulated or formulated forms had shown high bioactivity in reducing exercise-induced muscle inflammation and damage. 13 Early evidence from human trials and animal studies had been promising, but results were sometimes mixed due to differences in study design and curcumin formulations. Nonetheless, the therapeutic potential of curcumin for muscle recovery was significant enough to warrant a comprehensive review.
This article provided an in-depth review of curcumin as a natural therapeutic agent in exercise-induced muscle injury and recovery. First, it outlined the pathophysiology of EIMD, including the roles of inflammation and oxidative stress in muscle damage and DOMS. Next, it described the pharmacological actions of curcumin relevant to these pathways (anti-inflammatory and antioxidant effects, molecular targets, and bioavailability challenges). It then examined the experimental evidence from in vitro studies, animal models, and human clinical trials on curcumin's efficacy in muscle recovery, including outcomes on soreness, muscle function, and biochemical markers. Finally, it discussed practical considerations, current limitations, and future directions for research and application of curcumin in exercise recovery. By synthesizing findings across these areas, the aim of the review was to investigate the therapeutic potential of curcumin in enhancing recovery in active people from muscle injury and soreness.
Pathophysiology of Exercise-Induced Muscle Injury
During strenuous or unfamiliar exercise, especially with eccentric contractions, muscle fibers experienced mechanical strain that exceeded their structural capacity. 14 The immediate result was microscopic damage within the muscle: disruption of myofibrils, damage to the cytoskeleton, and micro-ruptures of the sarcolemma. Calcium homeostasis was disturbed as extracellular calcium leaked into the damaged fibers and intracellular stores released excess calcium. 15 The elevated intracellular Ca2+ activated proteases and phospholipases that further degraded structural proteins and membrane phospholipids, exacerbating the initial injury. 16 The mechanical and chemical damage impaired excitation-contraction coupling, meaning the muscle had a reduced ability to generate force until repairs occurred. Functionally, this corresponded to an immediate loss of strength in the affected muscle and manifested as acute muscle weakness or instability post-exercise. 17
The structural damage and calcium overload also triggered a localized inflammatory response that unfolded over the hours and days following exercise. The body treated the microdamage similarly to a mild injury or infection, initiating an innate immune response. 18 The process began with resident immune cells and stress signals from the damaged muscle fibers. 19 Injured muscle fibers released danger signals that activated endothelial cells and resident macrophages. Within a few hours, neutrophils were recruited to the muscle through the bloodstream. 20 Neutrophils migrated into the interstitial spaces of the muscle, facilitated by upregulation of adhesion molecules on blood vessels. 21 These neutrophils began phagocytosing debris and released reactive oxygen species (ROS) via the respiratory burst, as well as proteolytic enzymes. 22 While neutrophils helped clear damaged tissue, the ROS they produced also caused secondary damage to surrounding healthy muscle cells. The oxidative burst from neutrophils and the initial injury together created an environment of oxidative stress in the muscle, where the production of ROS outpaced the muscle's antioxidant defenses. 23 This oxidative stress oxidized lipids in cell membranes (lipid peroxidation), damaged proteins, and impaired mitochondria, further reducing the muscle cell's energy production and function. 24
Activated neutrophils also secreted chemokines that attracted monocytes, which differentiated into macrophages in the muscle tissue. The infiltration of macrophages marked the next phase of inflammation. 25 In the first 1-2 days after injury, many macrophages exhibited a pro-inflammatory phenotype (often termed M1 macrophages). 26 These M1 macrophages released pro-inflammatory cytokines such as tumor necrosis factor-alpha (TNF-α), interleukin-1 beta (IL-1β), and interleukin-6 (IL-6). 27 TNF-α was particularly influential: it activated the ubiquitin-proteasome system in muscle cells, increasing expression of muscle-specific E3 ubiquitin ligases (MuRF1 and MAFbx) that tagged proteins for degradation. 28 Through this mechanism, excessive or prolonged TNF-α signaling contributed to muscle protein breakdown and atrophy, although in the short term after acute exercise this breakdown was part of removing damaged proteins. 29 TNF-α and IL-1β also activated the nuclear factor kappa B (NF-κB) pathway, a pivotal transcription factor complex that amplified inflammation. 30 NF-κB activation in muscle and immune cells led to further production of cytokines, chemokines, and cell adhesion molecules, creating an inflammatory cascade. 31 This cascade was necessary for calling in more immune cells and initiating repair, but it also intensified the inflammatory damage if unchecked. 32 For example, NF-κB-driven pathways increased cyclooxygenase-2 (COX-2) expression, raising local prostaglandin levels that contributed to pain and swelling. 33
By around 48 h after injury, the inflammatory response began to shift. 34 The initial wave of neutrophils and M1 macrophages peaked and then declined, and macrophages started to adopt an anti-inflammatory, pro-recovery phenotype (M2 macrophages). Under the influence of anti-inflammatory signals (like IL-10, IL-4, and IL-13 from immune and muscle cells), macrophages transitioned to M2 status. 35 M2 macrophages released growth factors such as insulin-like growth factor-1 (IGF-1) and transforming growth factor-beta (TGF-β) that stimulated muscle regeneration. They also cleared away apoptotic cells and secreted anti-inflammatory cytokines (IL-10) to resolve inflammation. 36 This switch from a pro-inflammatory to a pro-regenerative environment was crucial for muscle recovery and adaptation. 37 The M2 macrophages activated muscle satellite cells, which proliferated and then differentiated into myoblasts to repair and form new muscle fibers, ultimately replacing or reinforcing the damaged tissue. It was noteworthy that skeletal muscle itself produced cytokines, termed myokines, during exercise. 38 For instance, IL-6 was released in large amounts from working muscle fibers, but interestingly, muscle-derived IL-6 acted in an anti-inflammatory manner, stimulating IL-1 receptor antagonist and IL-10, unlike macrophage-derived IL-6 which could be pro-inflammatory. 39 This meant that not all IL-6 measured after exercise was harmful; some was actually part of a signaling mechanism to improve metabolic and immune responses. 40 Nonetheless, excessive IL-6 and other cytokines from immune cells contributed to the symptomatic inflammation following muscle damage. 41
Another consequence of the inflammatory process was pain and soreness – ie, DOMS. 42 The exact cause of DOMS was multifactorial, but a major contributor was the sensitization of nerve endings in the muscle by inflammatory mediators. 43 Prostaglandins and bradykinin, as well as cytokines like TNF-α and IL-1β, lowered the activation threshold of pain receptors in the muscle. 44 Microscopic damage and swelling increased intramuscular pressure, stimulating mechanoreceptors and nociceptors. As a result, movements or pressure on the muscle post-exercise triggered pain signals more readily. 45 DOMS typically peaked when the inflammatory process was in full swing (24-48 h post-exercise) and subsided as inflammation resolved and tissues healed. In addition to pain, muscle function was affected: swelling and damage reduced the range of motion and increased stiffness, while the loss of structural integrity and excitation-contraction coupling led to decreased strength and power output until repair occurred. 1 Mechanical strain initiated sarcomere micro-tears, an early neutrophil ROS surge (0-24 h), and a cytokine-driven shift from M1- to M2-macrophage dominance (48-72 h). These temporal events underpinned DOMS and ultimately satellite-cell-mediated regeneration, as illustrated in Table 1 and Figure 1.

Schematic Illustration of the Pathophysiological Progression and Recovery Phases in EIMD. The Process Begins with Mechanical Disruption of Muscle Fibers, Followed by an Inflammatory Response and Oxidative Stress, Leading to Delayed DOMS. Recovery Involves Activation of Satellite Cells and Adaptive Remodeling, Resulting in Increased Muscle Resilience.
Pathophysiological Factors in EIMD.

Pharmacological Properties of Curcumin Relevant to Muscle Injury
Curcumin's therapeutic effects were attributed to its multi-modal action on molecular pathways involved in inflammation and oxidative stress. In the context of exercise-induced muscle injury, curcumin modulated the very processes that caused secondary damage and delayed recovery. Below, we elaborated on curcumin's key pharmacological properties—particularly its anti-inflammatory and antioxidant effects—and how they related to mitigating muscle injury. This review also addressed curcumin's metabolism and bioavailability, which limited clinical translation because oral curcumin was absorbed poorly and underwent rapid first-pass metabolism and elimination. Co-administration with the alkaloid piperine, a potent UDP-glucuronyl-transferase inhibitor, increased the area-under-the-curve of curcumin by ∼2000% in humans, thereby overcoming these barriers and widening its therapeutic window. Contemporary delivery systems (micellar, phytosomal, fenugreek-fiber, and solid-lipid particles) further enhanced systemic exposure by 27- to 185-fold, allowing effective intakes as low as 90 mg day−1. 46
Anti-Inflammatory Effects: Curcumin was widely recognized as a natural anti-inflammatory agent. One of its central actions was the inhibition of the NF-κB signaling pathway, often termed a “master switch” of inflammation (Figure 2). 47 NF-κB was normally activated in muscle cells and infiltrating immune cells in response to stress signals (like TNF-α and ROS) during muscle injury. 48 Upon activation, NF-κB translocated to the nucleus and induced genes encoding pro-inflammatory cytokines (TNF-α, IL-1β, IL-6, IL-8), chemokines, and enzymes such as inducible nitric oxide synthase (iNOS) and COX-2. 49 Curcumin blocked the activation of NF-κB, even when triggered by upstream signals like TNF-α or oxidative stress. 50 By inhibiting NF-κB activation, curcumin led to a broad downregulation of pro-inflammatory mediators. 51 Studies showed that curcumin supplementation resulted in lowered levels of cytokines including TNF-α, IL-1β, IL-6, IL-8, and IL-12 in various inflammatory contexts. 52 In muscle inflammation specifically, curcumin's NF-κB inhibition was significant because it both diminished the acute inflammatory response and reduced muscle protein breakdown; with NF-κB held in check, the expression of muscle-wasting factors (like MuRF1 driven by TNF-α signaling) was also reduced. 53 This implied that curcumin could help preserve muscle proteins after injury while still allowing necessary repair.

Curcumin Inhibits NF-κB Signaling in Muscle Inflammation. Muscle Injury and Oxidative Stress (eg, ROS, TNF-α) Activate the IκB Kinase (IKK) Complex, Leading to IκB Degradation and Nuclear Translocation of NF-κB (p65/p50). NF-κB Promotes Transcription of Pro-inflammatory Cytokines (TNF-α, IL-1β, IL-6, IL-8, IL-12) and Enzymes (COX-2, iNOS), Contributing to Inflammation and Muscle Protein Breakdown (eg, MuRF1 Expression). Curcumin Inhibits IKK Activation and NF-κB Nuclear Translocation, Thereby Reducing Inflammatory Mediator Expression and Preserving Muscle Tissue.
Beyond NF-κB, curcumin influenced other inflammatory signaling cascades (Figure 3). 54 It suppressed the JAK/STAT pathway, which was activated by certain cytokines and growth factors.47,55 By dampening JAK/STAT activation, curcumin reduced the downstream production of inflammatory cytokines that were controlled by this pathway. 56 Curcumin also interfered with the mitogen-activated protein kinase (MAPK) pathways, including the stress-activated kinases JNK and p38, as well as ERK1/2. 57 These kinases were typically activated in muscle cells under mechanical stress and in immune cells during inflammation, contributing to the expression of inflammation-related genes.58,59 Curcumin's ability to inhibit JNK, p38, and ERK activity added another layer of anti-inflammatory effect, potentially reducing muscle fiber apoptosis and fibrosis that resulted from MAPK overactivation. 60

Curcumin Suppresses Multiple Inflammatory Signaling Pathways Involved in Muscle Injury. Curcumin Exerts Broad Anti-inflammatory Effects by Modulating Three Key Signaling Cascades: (1) the NF-κB pathway, Where it Inhibits IKK Activation and Prevents NF-κB Nuclear Translocation; (2) the JAK/STAT Pathway, Where it Blocks JAK Activation and Reduces Downstream Cytokine Expression; and (3) the MAPK Pathway, Where it Suppresses the Phosphorylation of JNK, p38, and ERK1/2. These Actions Collectively Downregulate Pro-inflammatory Mediators Such as IL-1β, IL-6, and COX-2, and Reduce Muscle Apoptosis and Fibrosis.
At the enzymatic level, curcumin directly downregulated pro-inflammatory enzymes. 61 It inhibited COX-2, thereby reducing the synthesis of pro-inflammatory prostaglandins (like PGE2), which were major contributors to pain and edema in DOMS. Simultaneously, curcumin inhibited 5-lipoxygenase (5-LOX), an enzyme in the leukotriene pathway, thus lowering leukotriene production that would otherwise have attracted more neutrophils and caused bronchoconstriction or vascular permeability. 62 By attenuating both COX-2 and LOX pathways, curcumin broadly decreased the eicosanoid mediators of inflammation. 63 Additionally, curcumin reduced the expression of adhesion molecules such as ICAM-1 and VCAM-1 on endothelial cells. 64 These molecules were critical for leukocyte adhesion and migration into tissues; curcumin's reduction of ICAM-1/VCAM-1 meant fewer immune cells extravasated into the muscle, potentially limiting the extent of inflammation and tissue disruption. 65 Curcumin also downregulated iNOS, which curtailed the production of high levels of nitric oxide that could form reactive nitrogen species and contribute to inflammation-associated tissue damage. 66 Through these combined actions, curcumin created a more moderate inflammatory milieu: sufficient to clear damage and signal repair but milder than the unchecked response, thereby reducing pain and secondary muscle damage. 67
Interestingly, curcumin not only suppressed the “bad” aspects of inflammation but also promoted resolution and healing. 68 Research indicated that curcumin upregulated anti-inflammatory cytokines like IL-10 in certain contexts, acting as an immunomodulator. 69 Moreover, curcumin was associated with increased expression of the anti-apoptotic proteins Bcl-2 and Bcl-xL in cells under stress. 70 In muscle cells, higher Bcl-2/Bcl-xL protected against cell death induced by exercise stress or inflammatory cytokines, preserving muscle fiber integrity. 71 Curcumin also influenced heat shock proteins and other stress responses that aided in cellular protection. 72 For example, there was evidence that curcumin enhanced the heat shock response, which helped refold damaged proteins and protect cells from thermal or oxidative stress. 73 All these effects suggested curcumin not only tempered the initial inflammatory damage but also set the stage for quicker recovery and regeneration. One striking demonstration of curcumin's anti-inflammatory potency in muscle came from preclinical trauma studies. In a classic study by Thaloor et al, 13 systemic administration of curcumin (as an NF-κB inhibitor) in mice with muscle injuries led to significantly faster and greater muscle regeneration compared to controls. The curcumin-treated mice had reduced inflammation and enhanced muscle fiber repair, highlighting that curcumin's modulation of NF-κB and related pathways translated into tangible improvements in muscle healing. While exercise-induced damage was less severe than traumatic injury, the principle remained that controlling excessive inflammation facilitated better muscle recovery. 74
Antioxidant Effects: Curcumin was a well-established antioxidant, capable of combating the oxidative stress that accompanied intense exercise. 75 Its antioxidant activity was twofold: direct scavenging of free radicals and upregulation of the body's own antioxidant defenses. 76 Curcumin's molecular structure allowed it to directly quench ROS like superoxide anions, hydroxyl radicals, and peroxyl radicals. 77 In chemical assays, curcumin neutralized free radicals by donating hydrogen atoms or electrons, thereby terminating radical chain reactions. 78 For instance, curcumin exhibited strong DPPH radical scavenging activity in vitro. 79 In muscle cell experiments, curcumin dose-dependently increased radical scavenging and protected cells from peroxide-induced death. This direct antioxidant effect limited the oxidative damage to muscle cell membranes and organelles in the aftermath of strenuous exercise.
In addition to directly scavenging ROS, curcumin bolstered endogenous antioxidant systems mainly through activation of the transcription factor Nrf2 (nuclear factor erythroid 2–related factor 2). 80 Under oxidative stress, Nrf2 dissociated from its inhibitor and moved to the nucleus to induce expression of antioxidant and phase II detoxification enzymes. 81 Curcumin was described as an Nrf2 activator, meaning it helped trigger this protective pathway. 82 In cultured muscle cells, curcumin strongly activated an Nrf2-dependent antioxidant response element, leading to increased levels of heme oxygenase-1 (HO-1), glutathione cysteine ligase (the rate-limiting enzyme in glutathione synthesis), and NAD(P)H quinone oxidoreductase-1 (NQO1). These enzymes worked to neutralize peroxides, maintain glutathione stores, and detoxify reactive molecules. 83 As a result, curcumin pretreatment of muscle cells significantly reduced ROS accumulation and lipid peroxidation after an oxidative challenge, while also preserving intracellular glutathione levels. 84 This indicated that curcumin “pre-armed” cells with greater antioxidant capacity to face stress. 85 In vivo, similar effects were observed; animals given curcumin showed higher activity of antioxidant enzymes like superoxide dismutase (SOD), catalase, and glutathione peroxidase in tissues after exercise or toxic insult. 86 One human study on exercise-induced oxidative stress found that curcumin supplementation maintained total antioxidant capacity (TAC) significantly higher post-exercise compared to placebo, suggesting curcumin supported the body's antioxidant network during recovery. 87
The antioxidant impact of curcumin directly addressed the oxidative stress aspect of EIMD. 75 By neutralizing ROS and boosting antioxidant defenses, curcumin reduced oxidative damage to muscle cell membranes, 88 protect mitochondria, 89 and decrease the activation of redox-sensitive inflammatory pathways. 90 Consequently, muscles under the influence of curcumin experienced less secondary damage from free radicals. 91 This also had implications for fatigue; reducing oxidative damage helped muscle contractile function recover faster. 92 It was worth noting that curcumin's anti-inflammatory and antioxidant roles were interconnected. 93 Oxidative stress could activate inflammation, and inflammation could generate ROS. Curcumin broke this vicious cycle from both ends. 94 For example, by scavenging ROS, curcumin indirectly prevented ROS-mediated activation of NF-κB and MAPKs. 95 Conversely, by inhibiting inflammatory enzyme systems, curcumin reduced the production of new ROS. This dual action made it especially effective in conditions like EIMD, where both inflammation and oxidative stress were present concurrently. 96
Beyond anti-inflammatory and antioxidant actions, curcumin had other pharmacological effects that benefitted muscle recovery. It influenced muscle metabolism and cell signaling pathways that governed energy use and recovery. Some studies showed that curcumin activated AMP-activated protein kinase (AMPK) in skeletal muscle. 97 AMPK was an energy sensor; when activated, it enhanced glucose uptake and fatty acid oxidation, which helped muscle cells restore energy balance after exhaustive exercise. Activation of AMPK also had anti-inflammatory effects. Additionally, curcumin's effect on PGC-1α was noted in animal models. 98 In a study where rats were given curcumin during an exercise training program, their muscle showed higher PGC-1α levels, which was a key regulator of mitochondrial biogenesis and endurance adaptation. This suggested curcumin aided the muscle's adaptive remodeling by promoting mitochondrial improvements, thereby not just recovering from damage but potentially coming back stronger. Curcumin also showed anti-catabolic effects in other models of muscle wasting by reducing proteolytic pathways and inflammation. 99 This again tied to its NF-κB inhibition, as NF-κB was implicated in muscle atrophy; curcumin's suppression of NF-κB helped maintain muscle mass in recovery periods.
A practical challenge with curcumin was its metabolism and bioavailability. 100 Orally administered curcumin had notoriously poor bioavailability, which meant the amount that actually got absorbed into the bloodstream and reached target tissues like muscle was quite low. This was due to several factors: curcumin's low water solubility, its tendency to be rapidly metabolized in the intestinal mucosa and liver, and its quick elimination. 101 After ingestion, curcumin was subject to extensive phase II metabolism—mainly glucuronidation and sulfation—which converted it into more water-soluble metabolites that had less biological activity.102,103 As a result, very little free curcumin appeared in plasma even with gram-level doses. 104 Human clinical trials indicated that curcumin's oral bioavailability was extremely low; one summary noted that curcumin was “poorly absorbed and rapidly metabolized and eliminated” in humans. Plasma curcumin levels in many studies were in the nanomolar range or below detection after standard doses. This pharmacokinetic profile limited the effectiveness of plain curcumin—essentially, one might have needed impractically high doses to get sufficient curcumin to muscle tissues if no measures were taken to improve its delivery.
To overcome this, several strategies were developed and were pertinent when considering curcumin for EIMD. One common approach was co-administration of piperine, an alkaloid from black pepper. 105 Piperine inhibited certain enzymes in the liver and gut that metabolized curcumin, thereby reducing curcumin's conjugation and excretion. 106 The addition of piperine dramatically increased curcumin bioavailability—by up to 2000% in one study. 13 For instance, a curcumin supplement taken with piperine led to much higher plasma curcumin levels than curcumin alone. 107 Many studies on curcumin and exercise utilized this combination or similar absorption enhancers for this reason. Another strategy was to use advanced curcumin formulations. 68 Liposomal curcumin, nano-curcumin, and curcumin phospholipid complexes, such as the commercially known phytosome formulation Meriva® (a patented complex of curcumin with phosphatidylcholine designed to enhance intestinal absorption), were examples. 108 Similarly, Longvida® (a solid lipid curcumin particle formulation developed to enhance curcumin stability and enable targeted absorption) demonstrated markedly improved bioavailability in human studies. 109 These delivery systems increased systemic curcumin exposure by up to 27-185-fold, facilitating therapeutic effects at lower oral doses.
Curcumin's metabolism yielded some metabolites that were also biologically active, albeit often weaker than curcumin itself. There was ongoing investigation into whether these metabolites contributed meaningfully to curcumin's effects. 110 Nonetheless, to maximize efficacy in muscle recovery, maintaining higher levels of the parent compound was considered important, which was why bioavailability enhancements were emphasized. Indeed, differences in outcomes among studies could sometimes be traced to whether a bioavailability-enhanced form of curcumin was used or not. 111 Studies using formulations that achieved even modest plasma levels of 100 ng/mL curcumin tended to show more significant effects on muscle inflammation markers than those with lower levels. 13 Thus, any practical application of curcumin for EIMD needed to consider formulation and dosing carefully.
In summary, curcumin's pharmacodynamic profile (inhibiting NF-κB, COX-2, and inflammatory cytokines; scavenging ROS and activating Nrf2; modulating signaling pathways like JAK/STAT and MAPK; and supporting muscle cell survival and metabolism) directly addressed the pathological mechanisms of exercise-induced muscle damage. Table 2 showed that curcumin simultaneously dampened NF-κB, COX-2, and 5-LOX while activating Nrf2-regulated antioxidant enzymes; these concerted actions explained the 25%-50% decrements in post-exercise TNF-α and CK. The major caveat in translating these effects to real-world muscle recovery was ensuring adequate bioavailability of curcumin. However, with modern supplement formulations and dosing strategies, it was feasible to attain effective concentrations. The following sections examined how these properties of curcumin translated into experimental and clinical outcomes in the context of muscle injury and recovery.
Curcumin's key Molecular Actions Relevant to Muscle Injury.

Applications of Curcumin in Treating EIMD
Given curcumin's promising pharmacological profile, numerous studies investigated its actual effects on muscle cells, animal models of muscle damage, and human participants undergoing exercise-induced muscle injury. In this section, we summarized the body of evidence from laboratory experiments to clinical trials, highlighting how curcumin supplementation influenced markers of muscle damage, inflammation, oxidative stress, muscle soreness, and functional recovery.
In Vitro Studies: Controlled experiments on muscle cells provided proof-of-concept for curcumin's protective effects. For instance, C2C12 myoblasts (a mouse muscle cell line) were used to model muscle oxidative stress and inflammation. 112 In one study, curcumin (delivered as a curcumin-rich spray-dried powder) significantly fortified muscle cells against hydrogen peroxide-induced damage. 79 Cells pretreated with curcumin showed higher viability after H2O2 exposure compared to untreated cells, correlating with curcumin's activation of the Nrf2 pathway and increased antioxidant enzyme levels. The curcumin-treated cells exhibited reduced ROS accumulation and less lipid peroxidation of their membranes under stress, supporting the idea that curcumin could directly prevent oxidative injury to muscle fibers. Additionally, the same study showed that curcumin reduced the production of IL-6 in muscle cells challenged with bacterial endotoxin (LPS), demonstrating an anti-inflammatory effect at the cellular level. Another in vitro investigation found that curcumin prevented heat-induced damage in muscle cells by inhibiting the excessive production of ROS via NADPH oxidase and helped maintain mitochondrial function during heat stress. 113 These cellular studies collectively suggested that curcumin entered muscle cells and modulated their response to stressors, making them more resilient to the kind of oxidative and inflammatory insults that occurred in EIMD. Curcumin was also shown to influence muscle cell differentiation and regeneration in laboratory settings. A notable early study observed that curcumin promoted the differentiation of myoblasts into myotubes, partly by inhibiting NF-κB signaling, which otherwise suppressed muscle formation. This finding tied to the idea that curcumin might have assisted not only in protecting existing muscle fibers but also in the formation of new muscle tissue during recovery. 114 While in vitro results had to be interpreted with caution for real-world implications, they provided mechanistic insight: curcumin at the cellular level reduced oxidative damage, lowered inflammatory signaling, and even enhanced regenerative processes—all desirable effects for muscle recovery.
Animal models of muscle injury or intense exercise allowed researchers to observe curcumin's effects in a whole-body system, where bioavailability and immune responses were factors. One of the seminal animal studies on curcumin and muscle was by Thaloor et al 13 using a traumatic muscle injury model in mice. Systemic curcumin administration in those mice led to a marked acceleration of muscle regeneration. The curcumin-treated mice had larger regenerating myofibers and earlier functional recovery of the injured muscle. 115 This was attributed to curcumin's blockade of NF-κB, which in turn allowed muscle precursor cells to differentiate more efficiently and reduced the inflammatory impediments to regeneration. Essentially, this study provided a dramatic visual demonstration of curcumin's capacity to improve muscle healing after severe damage. 116
In the context of exercise-induced damage, several rodent studies examined curcumin. One study on rats supplemented with curcumin (using a CurcuWin® formulation for better absorption) during a 6-week exercise training regimen found significant benefits. 98 The curcumin-supplemented rats ran to exhaustion longer than controls, indicating improved endurance performance. 117 Biochemical analysis revealed the reasons: curcumin reduced markers of muscle damage and fatigue such as muscle malondialdehyde and blood lactate levels. Meanwhile, it increased antioxidant enzyme activities in muscle and elevated muscle glycogen content post-exercise. 117 Importantly, curcumin prevented the exercise-induced increase in muscle NF-κB and heat shock protein, both of which were typically elevated under stress. It also upregulated muscle proteins associated with mitochondrial function and recovery: PGC-1α, sirtuin 1, thioredoxin, and GLUT4 were higher in curcumin-treated exercising rats than in exercise alone. 118 These changes aligned with curcumin reducing oxidative/inflammatory stress and promoting a more oxidative, fatigue-resistant muscle phenotype. The results suggested that curcumin not only attenuated muscle damage in these rats but also enhanced their adaptive responses to training.
Some animal studies mimicked human DOMS by using downhill running or electrical stimulation. In one such mouse study, curcumin supplementation attenuated the rise of inflammatory cytokines and oxidative markers after downhill running, and muscles from curcumin-fed mice showed less histological damage and edema than controls. 119 Curcumin-treated mice also retained more muscle strength in the days following the exercise compared to untreated mice, indicating faster recovery of function. These results echoed that curcumin's anti-inflammatory action (such as reducing IL-1β, IL-6, and TNF-α in muscle tissue) protected the muscle from excessive damage. Although not the primary focus, it was worth noting that curcumin showed beneficial effects in animal models of chronic muscle injury or degeneration, such as the mdx mouse model of Duchenne muscular dystrophy. Curcumin-treated mdx mice exhibited reduced inflammation in muscle and improved muscle integrity and strength relative to placebo. 120 This highlighted curcumin's potential to stabilize muscle membranes and reduce chronic inflammation in muscle tissue, which was encouraging for its use in recurrent or long-term muscle stress situations. Overall, animal studies consistently supported that curcumin reduced indicators of muscle damage, quelled inflammatory responses, diminished oxidative stress, and sometimes even improved performance or recovery of strength. These positive findings in animals set the stage for human trials, with the caveat that dosing and bioavailability required adjustment when moving from animals to humans.
Human Clinical Trials: In recent years, multiple randomized controlled trials (RCTs) and other clinical studies examined curcumin supplementation in the context of exercise-induced muscle damage and recovery. 121 These studies often involved healthy volunteers (sometimes trained athletes, other times untrained individuals) performing a muscle-damaging exercise protocol (such as downhill running, eccentric single-leg presses, or heavy resistance exercise) and then taking either curcumin or a placebo around the time of the exercise. The outcomes assessed typically included muscle soreness ratings, muscle function tests (strength, power, jump performance, etc), and blood biomarkers of muscle damage and inflammation (like creatine kinase, lactate dehydrogenase, IL-6, TNF-α, CRP, etc). 122 Analyzing the collective evidence from these trials revealed a generally positive trend in favor of curcumin, though not every study found benefits on every measure. A narrative synthesis of 15 human studies reported that curcumin dosages from 90 to 5000 mg day−1, taken 1-2 h before and/or within the first 24 h after strenuous exercise, consistently reduced creatine kinase, TNF-α, IL-6, and IL-8 while improving perceived soreness and functional recovery. 46 Benefits appeared formulation-dependent, with high-bioavailability preparations producing larger effects at lower absolute doses.
To date, at least three systematic reviews and meta-analyses were conducted on curcumin for exercise recovery, indicating the substantial interest and data in this area.13,97,123,124 A 2024 meta-analysis by Liu et al 97 compiled 14 RCTs with a total of 349 subjects, evaluating curcumin's effects on muscle soreness, creatine kinase (CK), IL-6, and range of motion. The meta-analysis found that curcumin supplementation significantly mitigated muscle damage and promoted recovery across these outcomes. On average, curcumin-treated groups reported lower DOMS ratings (pooled mean difference about −0.6 on a standard scale), meaning subjects felt less sore post-exercise. Blood CK levels, a marker of muscle fiber damage, were markedly reduced in curcumin groups (mean difference roughly −137 U/L) compared to placebo. In practical terms, curcumin users experienced a smaller spike in CK after exercise and a faster return toward baseline, indicating less muscle membrane disruption or quicker healing. 125 Inflammatory IL-6 levels were also modestly lower with curcumin (mean difference −0.33 pg/mL, in whatever units reported). Additionally, curcumin helped preserve range of motion in the affected muscles, with an average improvement of ∼4 degrees in ROM in curcumin versus placebo. This aligned with reduced swelling and stiffness when inflammation was moderated. 126 The meta-analysis also probed effect modifiers: it suggested that timing and dosage mattered. Prolonged curcumin supplementation (several days of dosing) yielded the best results, especially for individuals not habitually accustomed to muscle-damaging exercise. It also found that taking curcumin before exercise in a preventative, low-dose manner was beneficial for reducing subsequent soreness and maintaining ROM, whereas taking curcumin immediately after exercise was most effective for lowering CK and IL-6 levels. These nuanced findings hinted that an optimal strategy might have involved a combination of pre- and post-exercise dosing.
Individual clinical trials gave more detailed insights. For example, Nicol et al 127 conducted a double-blind crossover RCT in which 17 men performed heavy eccentric exercise and received either curcumin or placebo around the exercise. The curcumin dose was quite high (5 g/day, given as 2.5 g twice daily) and started 2 days before exercise, continuing until 3 days after. They found that curcumin significantly reduced DOMS pain. At 24 and 48 h post-exercise, curcumin users reported notably less pain during exercises like a squat or stretch compared to placebo (reductions of about 1.0-1.9 cm on a 10 cm visual analog scale). This was a meaningful decrease, often representing mild versus moderate soreness. The curcumin group also performed better in functional tests: they had a smaller decline in single-leg jump height post-exercise and actually showed a faster recovery of jump performance (by ∼15% improvement relative to placebo). Biologically, Nicol's trial observed that curcumin led to a smaller increase in CK (22-29% lower than placebo at 24-48 h), consistent with less muscle damage. One interesting finding was that curcumin modulated IL-6 in a time-dependent way: the curcumin group had a higher IL-6 response immediately after exercise but a lower level at 24 h into recovery. This observation was particularly interesting in light of earlier discussion on the dual role of IL-6. The initial, higher IL-6 spike might have represented a beneficial myokine-driven signal for metabolic and immune responses, while the subsequent reduction in IL-6 at 24 h indicated a blunting of the pro-inflammatory macrophage-derived cytokine response, thereby facilitating the resolution of inflammation without hindering the initial adaptive signaling. This suggested that curcumin acted as a modulator, preserving the beneficial, acute IL-6 response while suppressing the prolonged, damaging inflammatory cascade.
Another pivotal study was by McFarlin et al, 128 who examined a commercially enhanced curcumin formulation (400 mg/day of Longvida® curcumin, known for good absorption) in 28 active adults undergoing a muscle-damaging leg exercise. Participants took curcumin or placebo for 2 days before and 4 days after performing 50 eccentric leg presses. The results showed clear anti-inflammatory and muscle-protective effects: the curcumin group had significantly smaller increases in serum CK (48% lower peak CK than placebo), TNF-α (25% lower), and IL-8 (21% lower) over the 4 days of recovery. 128 This meant that curcumin curtailed both the muscle damage indicator (CK) and key inflammatory cytokines compared to no supplementation. Notably, IL-6 and IL-10 did not differ between groups, and—corroborating some other findings—the curcumin group did not report significantly less muscle soreness in this study. 128 Both groups had similar DOMS ratings in the quadriceps despite the biochemical differences. The authors posited that while curcumin clearly reduced biological inflammation, the sample size might not have been large enough to detect a difference in pain perception, or that pain in this context could have been influenced by factors other than just the measured cytokines. Nonetheless, they concluded that curcumin's reduction of muscle inflammation might have translated to faster recovery and better capacity in subsequent exercise, even if pain was unchanged. This study was often cited as evidence that curcumin could “work in the background” to aid recovery, which could have been beneficial for long-term training, regardless of immediate soreness.
Tanabe and colleagues 129 in Japan conducted multiple studies on curcumin timing and dosing. In one trial, they tested curcumin ingestion either before or after an eccentric exercise bout of the elbow flexors. They found that taking curcumin after exercise was more effective in improving recovery than taking it only before. The group that received curcumin post-exercise experienced significantly less muscle soreness and greater recovery of elbow joint range of motion over the next few days, whereas the pre-exercise curcumin group did not differ much from placebo in that study. In a related study, Tanabe et al 129 reported that curcumin led to improvements in certain blood biomarkers: the curcumin groups had lower plasma IL-8 12 h after exercise and a blunted peak of CK activity 4-5 days later. Specifically, the curcumin-supplemented participants showed a significantly lower CK peak and faster decline compared to controls, indicating an accelerated recovery from muscle fiber damage. Tanabe's work reinforced that curcumin reduced inflammatory chemokines like IL-8 and limited muscle damage, and also suggested timing nuances.
There were also studies focusing on different muscle groups and functional outcomes. For example, some RCTs looked at downhill running or plyometric exercise and measured soreness in leg muscles. A study by Hillman et al 130 found that curcumin supplementation (likely in a similar dosing scheme) improved recovery of single-leg hop performance and reduced thigh pain after downhill running. Another by Drobnic et al 131 used a curcumin phytosome (Meriva®) in soccer players and found a reduction in DOMS intensity after simulated match play. However, Drobnic's study noted no significant difference in CK levels despite reported pain reduction, illustrating that not all markers moved in unison. To illustrate the range of evidence, Table 3 summarized a selection of human studies on curcumin for muscle recovery, including their design and key findings.
Selected Studies of Curcumin Supplementation in Exercise-Induced Muscle Damage.
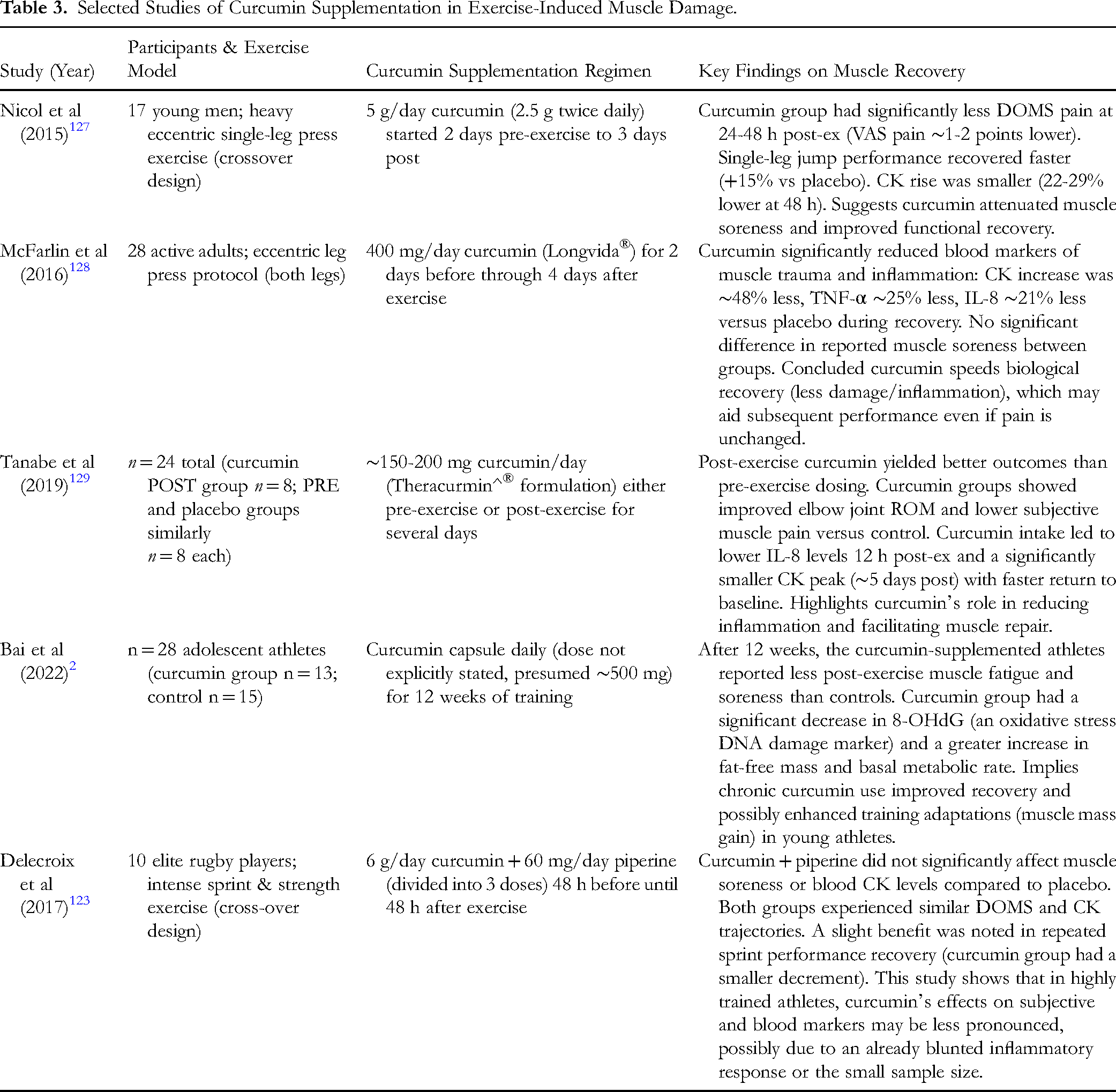
As shown in Table 3, most human studies reported at least one beneficial outcome with curcumin: whether it was reduced soreness, lower CK/inflammatory markers, or improved muscle function. The evidence was strongest for curcumin's effect on subjective DOMS and inflammation markers. Several trials consistently found that curcumin users felt less sore in the days after intense exercise.13,127 This represented a meaningful quality-of-life improvement for recreational exercisers and allowed athletes to train again sooner or at a higher intensity due to less discomfort. Mechanistically, the reduction in soreness was likely due to curcumin's suppression of prostaglandins and inflammatory stimuli that drove pain. The reduction in CK and other muscle damage markers like LDH or myoglobin with curcumin indicated that muscles suffered less structural damage or that curcumin enhanced membrane repair. For instance, multiple studies saw a significant curcumin effect on CK. 127 This not only reflected muscle fiber integrity but also could have meant better preservation of performance, since extreme CK elevations often correlated with strength loss. However, not all studies saw CK differences, which likely depended on exercise type and timing of measurements. The meta-analysis overall confirmed a CK-lowering effect of curcumin. 97
For inflammatory cytokines, curcumin's effect seemed to depend on sufficient dosing and frequency. McFarlin's study clearly showed reductions in TNF-α and IL-8. 128 Others showed lower IL-6, but results were variable because exercise itself elevated IL-6 from muscle in a beneficial way. Some trials found no difference in IL-6 or IL-10 between curcumin and placebo, likely because their exercise protocol caused only a mild inflammatory response. On the other hand, a study by Walters et al 97 observed curcumin lowering IL-6 and CRP after downhill running in untrained men, aligning with an anti-inflammatory effect when the response was robust. The meta-analysis suggested an overall IL-6 reduction, albeit modest. Additionally, curcumin was found to reduce IL-8 quite consistently, 47 which could have meant less neutrophil accumulation and therefore reduced secondary muscle damage.
A particularly noteworthy aspect was curcumin's safety and the minimal side effects reported in these trials. 132 Virtually none of the exercise studies reported adverse events from curcumin; at most, some participants might have had mild gastrointestinal upset with very high doses. This safety profile held even at high intakes (6 g/day in the Delecroix study was well tolerated). Athletes generally completed the studies without knowing whether they were on curcumin or placebo, implying curcumin did not produce noticeable negative symptoms that could have broken blinding. This was important because it suggested curcumin could be used without the concerns that accompanied NSAID use. 133 For inflammatory cytokines, curcumin's effect seemed to depend on sufficient dosing and frequency. McFarlin's study clearly showed reductions in TNF-α and IL-8. In contrast, results for IL-6 were variable, and this variability could be explained by the dual nature of IL-6. As discussed in the pathophysiology section, IL-6 released from contracting muscle could play a beneficial, anti-inflammatory role, while IL-6 from immune cells contributed to inflammation. Curcumin's action might not have been a simple suppression of all IL-6, but rather a modulation of its source or timing. By inhibiting NF-κB and macrophage infiltration, curcumin likely suppressed the pro-inflammatory IL-6 from immune cells. However, it might not have interfered with the myokine-driven IL-6 surge that was part of the muscle's immediate adaptive response to exercise. A study by Walters et al 97 observed curcumin lowering IL-6 and CRP after downhill running in untrained men, aligning with an anti-inflammatory effect when the response was robust. The overall IL-6 reduction suggested by the meta-analysis, albeit modest, likely reflected a net effect of blunting the harmful inflammatory component while sparing the beneficial signaling. This suggested that curcumin's benefit lay in helping the body transition from the pro-inflammatory phase to the regenerative phase of muscle recovery more efficiently, rather than simply reducing all inflammation universally.
It should have been acknowledged that not all studies were uniformly positive—some saw neutral effects on certain outcomes, and a few small studies failed to find significant benefits at all. Factors such as curcumin formulation, dose, exercise model, and subject training status influenced outcomes. For example, studies using plain curcumin powder at lower doses (like 150 mg) sometimes showed marginal or no effects unless the exercise bout was extremely strenuous. In contrast, studies using either a high dose or an enhanced form often found clearer results. 134 Trained athletes might have had a lower inflammatory response to a given exercise (the repeated-bout effect), thus the window for curcumin to show an improvement was smaller. This could have explained why a study in elite rugby players (who regularly experienced muscle stress) did not see much difference in soreness or CK with curcumin 123 – their bodies might already have handled EIMD efficiently. The timing of supplementation was another consideration, as highlighted earlier. Some protocols gave curcumin only after exercise, some only before, and some both before and after. The most logical approach appeared to be a combination: a little beforehand to pre-load tissues and primarily in the acute post-exercise period when inflammation peaked. 97 Tanabe's work supported post-exercise dosing for DOMS relief. 13 Meanwhile, if one anticipated a particularly damaging workout, starting curcumin a day or two prior (to saturate tissues and possibly pre-activate antioxidant defenses) could have been beneficial as well. 47
In addition to acute studies, the longer-term use of curcumin during training emerged as an area of interest. The 12-week study on adolescent athletes by Bai et al 2 was illustrative. Though not randomized, it suggested that daily curcumin over weeks might cumulatively reduce muscle fatigue and improve recovery to the point of allowing better training adaptations (like greater gains in lean mass). Athletes taking curcumin had improved antioxidant status (lower oxidative DNA damage) and felt less fatigued after their training sessions. This raised the possibility that curcumin could be used not just for isolated bouts but as a recovery aid throughout a competitive season or training cycle. However, the balance between reducing harmful inflammation and not interfering with beneficial training adaptation needed further study. Bai et al 2 demonstrated that curcumin supplementation (400 mg/day for 8 weeks) significantly reduced CK and IL-6 levels after eccentric exercise, indicating both anti-inflammatory and recovery-promoting effects. While these findings aligned with curcumin's proposed benefits, a theoretical concern existed regarding the potential for prolonged attenuation of inflammatory or oxidative signals to blunt the repeated-bout effect (RBE), an adaptive response that enhanced muscle resilience to subsequent exercise. Although studies in other antioxidant interventions raised such concerns, 135 c current evidence did not support this effect for curcumin. In fact, both Tanabe et al 129 and Bai et al 2 reported improved recovery and performance outcomes without compromising adaptive responses, suggesting that curcumin's modulatory effects were sufficiently mild to avoid interference with beneficial training adaptations. Future long-term studies were warranted to fully elucidate these interactions.
Another interesting potential benefit was curcumin's effect on connective tissue and joints, which could also have been stressed by exercise. 136 While the focus here was muscle, curcumin's anti-inflammatory action may have helped with exercise-induced joint or tendon inflammation. 121 This holistic recovery improvement was hard to quantify but could have contributed to an athlete's overall feeling of readiness.
Comparatively, curcumin was evaluated head-to-head against NSAIDs in clinical settings, most notably in knee osteoarthritis, which—while chronic—offered insights applicable to pain modulation and inflammatory relief after muscle injury. In a double-blind non-inferiority RCT involving 367 patients, administration of Curcuma domestica extract at 1500 mg/day for four weeks produced reductions in pain, stiffness, and functional impairment that were statistically non-inferior to ibuprofen 1200 mg/day. Importantly, gastrointestinal adverse events were significantly less frequent in the curcumin group than in the ibuprofen group. 137 Similarly, multiple reviews indicated that curcuminoid supplementation (1000-1500 mg/day) achieved moderate analgesic effect sizes comparable to standard NSAIDs such as ibuprofen or diclofenac for musculoskeletal pain, while maintaining a markedly superior safety profile, including fewer gastrointestinal and cardiovascular risks.138,139
Although specific comparisons in exercise-induced DOMS were lacking, the underlying mechanism—COX-2 inhibition and modulation of inflammatory cytokines—was shared between NSAIDs and curcumin. Given that curcumin attenuated COX-2 activity and lowered prostaglandin levels in preclinical muscle injury models, it was plausible that it approached the magnitude of pain reduction seen with ibuprofen in EIMD, albeit with a more gradual onset. In human RCTs of exercise-induced muscle soreness, curcumin typically reduced DOMS ratings by approximately 0.6 cm on a 10 cm VAS and lowered CK elevations by ∼137 U/L compared to placebo in meta-analysis, reflecting clinically meaningful effects (albeit not as instantaneous as a single NSAID dose).138,140 Taken together, while direct RCT comparisons in the EIMD context were limited, current evidence across musculoskeletal pain and recovery settings suggested that curcumin's analgesic effect was broadly in the same range as that of ibuprofen, but with a distinct advantage in safety when taken over multiple days—making it a promising alternative adjunct for post-exercise pain management.
In summary, clinical evidence strongly suggested that curcumin supplementation aided recovery from exercise-induced muscle injury. The most commonly observed benefits were: reduction in DOMS intensity, blunting of exercise-induced increases in CK and inflammatory cytokines, and, in some cases, faster restoration of strength or performance. These benefits were seen with various forms of curcumin at doses roughly ranging from ∼150 mg up to 5 g per day, often enhanced with piperine or advanced formulations. The data supported curcumin as an effective nutraceutical for managing post-exercise muscle soreness and damage, especially in situations of high mechanical stress. It was important to manage expectations: curcumin was not magic and typically yielded moderate improvements, but those improvements could be quite meaningful in practice.
Challenges, Future Directions, and Practical Applications
Despite the promising evidence that supported curcumin's role in exercise recovery, several challenges and considerations still needed to be addressed. This section discussed the limitations of existing research, safety and tolerability of curcumin for regular use, potential synergies with other interventions, and outlined future directions for both research and practical application.
A notable limitation in many curcumin exercise studies was the relatively small sample size. Most RCTs in this area included on the order of 10-30 participants per group, which limited statistical power and the generalizability of the findings. While meta-analyses helped aggregate these data, larger trials would have provided more confidence and allowed examination of how results varied across different populations. Another issue was the heterogeneity of study protocols. The studies differed in the type of exercise used to induce muscle damage, the timing and duration of curcumin supplementation, the curcumin dose (ranging from <200 mg to several grams), and the form of curcumin.13,47 This heterogeneity made direct comparisons difficult and might have explained some conflicting results. For example, a study using a highly bioavailable curcumin could have found an effect where another using plain curcumin at the same dose did not. Or an untrained population might have shown bigger improvements with curcumin than a trained population, due to higher baseline inflammatory responses in the former. The variety of outcome measures also introduced variability—since not all studies examined the same endpoints, and some outcomes were more prone to placebo effects.
Additionally, the timing of measurements post-exercise varied. Some studies caught certain peaks or windows of inflammation, while others missed them. If a study measured IL-6 only at 24 h post-exercise but IL-6 differences occurred mainly earlier at 8 h, that effect was missed. Similarly, if muscle soreness was measured at 48 h when curcumin mostly helped at 24 h, one might have incorrectly concluded no effect. Future studies ideally should have included multiple time-point measurements to fully capture the time course of recovery in both placebo and curcumin conditions. Another limitation was that many studies did not measure plasma curcumin levels or otherwise verify how much curcumin entered the system. Curcumin's bioavailability issues meant that a lack of effect in some trials could simply have been because not enough curcumin was absorbed to exert any effect. Only a few studies measured plasma curcumin, and they reported the concentrations achieved. 13 It would have been useful if future trials had included pharmacokinetic analyses or at least used standardized high-bioavailability preparations to reduce this uncertainty. Despite these limitations, the trend in the data was positive. The inconsistent results in a minority of studies were likely due, in part, to the factors above. 47 Improving study design homogeneity and including more subjects (perhaps multicenter trials) would have helped confirm curcumin's efficacy and refine understanding of when it worked best.
Curcumin was generally regarded as safe; regulatory agencies assigned an ADI of 0-3 mg kg−1 bw. Nevertheless, dose-response trials (500-12 000 mg) recorded transient diarrhea, headache, rash, and yellow stools in a minority of participants. 141 These events were self-limiting, and no hepatotoxicity was observed. Athletes using piperine-enhanced formulations needed to monitor for possible drug–nutrient interactions. Studies reported that curcumin at doses ranging from ∼150 mg to 5 g per day for weeks or months was well tolerated. For example, in clinical trials for other conditions, cancer patients took 3.6 g/day for 4 months with no serious adverse effects. The most common side effects at high doses included gastrointestinal discomfort, such as mild nausea, diarrhea, or bloating, but these were relatively infrequent. In exercise studies specifically, side effect reports were rare; participants typically could not distinguish curcumin from placebo based on how they felt. One reason curcumin was safe was ironically its low bioavailability—very little entered circulation to cause systemic toxicity. However, when taken with bioavailability enhancers like piperine, caution was warranted regarding drug-supplement interactions. Piperine not only boosted curcumin but also affected the metabolism of various drugs by inhibiting cytochrome P450 enzymes and drug transporters. This meant that if an athlete or individual was on medications, curcumin + piperine could have potentially increased those drug levels. Therefore, while curcumin itself was safe, the combinations used to enhance it needed to be considered in context.
For athletic use, curcumin was not on any doping ban lists; it was a legal supplement, and in fact some professional teams and athletes already incorporated it as part of their recovery nutrition. One safety consideration specific to intense training was to ensure that curcumin's anti-inflammatory effect was not counterproductive if used chronically—as mentioned, some inflammation was needed for adaptation. However, a critical caveat was that chronic anti-inflammatory or antioxidant supplementation could in some cases attenuate desired training adaptations. For example, long-term use of high-dose vitamin C and E was shown to blunt mitochondrial biogenesis and hypertrophy in response to endurance and resistance training. 142 Data on chronic curcumin were more limited, but Davis et al 143 observed improved recovery without reporting any ergolytic effects in subsequent training adaptation contexts. One preclinical caution stemmed from the acute Theracurmin study by Tanabe et al, 129 which attenuated strength loss and CK rise acutely—but the authors specifically noted that chronic consumption of curcumin warranted examination for potential interference with muscle adaptations induced by resistance training. This suggested that at least moderate doses of curcumin did not blunt adaptations in a noticeable way for young healthy individuals. It might have been because curcumin's effects were mild and targeted enough. Nevertheless, it remained an area to monitor in future research—perhaps using muscle biopsies to see if muscle protein synthesis or anabolic signaling after exercise was altered by curcumin. Curcumin in high amounts could cause stool to be yellowish and possibly mild liver enzyme elevations in extremely rare cases, but these had not been a significant concern in the literature. Overall, for healthy individuals, including athletes, curcumin supplementation was very safe when used responsibly. It offered a natural alternative to repeated NSAID use, avoiding NSAIDs’ gastrointestinal and cardiovascular risks.
Based on the evidence from both meta-analyses and human RCTs, Table 4 summarized practical dosing, timing, and formulation strategies for curcumin supplementation for exercise-induced muscle injury and recovery. These recommendations were grounded in controlled trials and consensus reviews and aimed to guide athletes and clinicians in designing evidence-based protocols. A common approach was to start supplementation 1-2 days before a known strenuous event and continue for 2-3 days after. This covered the period of muscle injury and the peak of inflammation/DOMS. The dosage varied depending on the product: for standard curcumin extract with piperine, a dose in the range of 500 mg to 1 g twice daily appeared effective, as that roughly emulated the high end of dosing in studies like Nicol's. For specialized high-bioavailability curcumin, the effective doses were lower. Many commercial products for sports recovery provided around 200-400 mg curcumin in a bioenhanced form per day—which appeared to fall within the effective range seen in literature. It was advisable to take curcumin with a meal that contained some fat, since curcumin was fat-soluble and this improved absorption if not using a pre-formulated enhanced version. Including black pepper in the meal helped as well. For athletes, timing could have been: one dose the night before the hard workout, one dose a few hours before, then doses in the evening after and on the next day morning and evening.
Evidence-Based Practical Recommendations for Curcumin Supplementation in Athletes and Clinicians.

Curcumin might have worked synergistically with other recovery strategies. Nutritionally, combining curcumin with protein could have been beneficial—protein provided amino acids for muscle repair, while curcumin reduced the inflammatory drag on recovery. There was no evidence that curcumin interfered with protein absorption or muscle protein synthesis. Thus, a post-exercise shake that included whey protein plus some curcumin (as attempted in certain recovery supplements) could have addressed both rebuilding muscle and controlling inflammation. Curcumin could also have complemented omega-3 fatty acids, which had their own anti-inflammatory effects on muscle soreness and joint health. Omega-3 s acted via different pathways, so using both might have yielded additive benefits. Similarly, antioxidants like vitamin C and E had been used for DOMS—though high doses of those antioxidants raised concerns about blunting adaptation, curcumin had not shown such effects at moderate doses. Still, an athlete might have preferred curcumin over high-dose pure antioxidants because curcumin's polyphenolic action appeared more balanced. I In terms of synergy with physical modalities, curcumin's systemic anti-inflammatory action could have worked in concert with local strategies like ice baths or compression garments. For example, an athlete might have taken curcumin to manage whole-body inflammation while also icing a particularly sore muscle group to reduce localized swelling. There had not been formal research on combining curcumin with other recovery modalities, but in practice it was likely that many small effects summed to a larger benefit. One interesting synergy mentioned in traditional medicine was curcumin with bromelain. 144 Bromelain itself possessed anti-inflammatory properties and might have improved curcumin absorption. Some supplements combined these, claiming better outcomes for joint and muscle inflammation. Though not studied in the EIMD literature, this remained an area worth exploring.
While the available evidence was quite supportive of curcumin's efficacy, several avenues remained for future investigation. More research was needed to determine the minimal effective dose and to compare head-to-head different curcumin formulations. To address the challenge of dosing heterogeneity in a meaningful way, future research needed to move beyond standardizing oral dosage (in mg) and progress towards standardizing based on pharmacokinetic outcomes. A key recommendation was for studies to preferentially use and compare bio-enhanced curcumin formulations (eg, micellar, phytosomal, solid-lipid particles) that had demonstrated the capacity to achieve meaningful systemic exposure. For example, an RCT could have been designed not just to compare Formulation A versus Formulation B, but to determine the oral dose of each required to achieve a target plasma curcumin concentration. As noted in this review, formulations that achieved plasma levels exceeding 100 ng/mL appeared to be more consistently associated with significant effects on inflammatory biomarkers. Therefore, a crucial step in standardizing the field would have been to design trials aiming for a minimum peak plasma concentration (Cmax) within a therapeutic window, such as >100 ng/mL, and to systematically report the achieved pharmacokinetic profiles (including Cmax, Tmax, and AUC). This concentration-centric approach would have allowed more meaningful cross-study comparisons, helped determine a true minimal effective systemic exposure, and clarified whether the observed benefits were dose-dependent in a systemic manner. Such a strategy would have directly addressed the limitations of prior work, refined practical, evidence-based recommendations for athletes, and helped lower costs by identifying the most efficient formulations.
Most trials enrolled predominantly young men; a 2024 meta-analysis involving 349 participants reported that only 18% were female, underscoring the need for sex-specific investigations. 13 Older adults could also have benefited greatly from curcumin, as they experienced more severe DOMS and slower recovery. Studying curcumin in masters athletes or in rehabilitation scenarios would have extended its applicability. Additionally, people with inflammatory conditions or at risk of muscle damage could have been studied. While one study covered 12 weeks, it would have been useful to conduct a longer-term RCT in which athletes were supplemented throughout a training season to evaluate effects on performance, incidence of injury, and markers of chronic inflammation. Endpoints could have included not just immediate recovery but also whether curcumin users handled higher training loads or experienced fewer overtraining symptoms. As suggested, using transcriptomics or proteomics could have yielded insights into how curcumin affected muscle recovery at the molecular level. For example, taking muscle biopsies from exercise + curcumin versus exercise + placebo groups at early and late recovery times and performing RNA sequencing might have shown which genes curcumin altered. Researchers might have observed upregulation of genes related to antioxidant defense or downregulation of genes in inflammatory pathways. Metabolomics on blood could have revealed changes in inflammatory lipid mediators with curcumin.
Ultimately, the question for competitive athletics was: did curcumin meaningfully improve subsequent performance or reduce time to full recovery in a way that gave an advantage? Small improvements in soreness were good, but did athletes return to baseline performance faster? Some studies hinted yes, but more direct measures—such as time trial performance 2 days after a muscle-damaging run, or the number of repeats an athlete could perform—would have been valuable. If evidence showed that curcumin allowed athletes to train at a higher intensity or frequency, that would have solidified its role. In planning future studies, researchers also needed to consider blinding and placebo effects, as the color of curcumin could tint urine or stools, which might have unblinded a very observant participant. Using a placebo with a similar appearance might have been necessary to keep subjects blind if high doses of curcumin were used.
Conclusion
In conclusion, curcumin had transitioned from a traditional remedy and laboratory curiosity to a compound with substantial scientific backing for use in exercise-induced muscle damage and recovery. It addressed the core issues of EIMD—namely inflammation and oxidative stress—and demonstrated efficacy in reducing soreness and biological markers of muscle damage in many studies. Its safety profile and natural origin made it especially attractive as a recovery aid for athletes and exercisers who sought alternatives to pharmaceuticals. As research continued to refine understanding, curcumin supplementation was increasingly optimized for enhancing muscle recovery. In the near future, curcumin was expected to become a staple recommendation in sports nutrition for recovery, much like protein or electrolytes, especially if ongoing studies continued to confirm its benefits.
Methodology
This review was conducted using a structured narrative approach to ensure comprehensive coverage of the literature while allowing for critical synthesis of mechanistic, preclinical, and clinical findings related to curcumin in exercise-induced muscle injury and recovery. The author systematically searched PubMed, Scopus, and Web of Science from database inception to April 2025 using the following keywords: “curcumin,” “turmeric,” “exercise-induced muscle damage,” “delayed-onset muscle soreness,” “oxidative stress,” and “inflammation.” Boolean operators and Medical Subject Headings (MeSH) were applied where appropriate. Secondary data were obtained from PubMed, Scopus, and Web of Science databases. Data were based on in vitro, animal-based in vivo and human-observation (clinical) studies. Non-English articles, conference abstracts without full text, and studies lacking direct relevance to curcumin and muscle recovery were excluded.
Although formal risk-of-bias tools were not applied to all included studies (given the narrative nature of the review), preference was given to well-designed randomized trials, studies that used bioavailability-enhanced curcumin formulations, and meta-analyses. This structured approach ensured that the conclusions presented in this review were derived from the best available evidence and aligned with current recommendations for narrative reviews.
Footnotes
Abbreviations
Acknowledgements
This work has been supported by The Henan Province Soft Science Research Program Project “Research on the Practical Path of Special Sports Culture Supporting Rural Revitalization in Henan Province”, Project Number: 252400410353.
Author Contribution Statement
Yan Li: methodology; software; validation; formal analysis; investigation; data curation; writing—original draft preparation,
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
