Abstract
Quercetin, a ubiquitous dietary flavonoid, has garnered significant scientific interest for its potential as an ergogenic aid in endurance sports. This interest is predicated on robust preclinical evidence demonstrating its potent antioxidant, anti-inflammatory, and mitochondrial biogenesis-stimulating properties. However, a persistent disconnect remains between promising laboratory findings and the equivocal, inconsistent, and often modest results reported in human trials with athletes. This review critically and systematically evaluates the scientific literature concerning quercetin's purported antioxidant and fatigue-resisting properties in the context of endurance training. We dissect the primary molecular mechanisms through which quercetin is proposed to act, including the activation of the nuclear factor erythroid 2–related factor 2 (Nrf2) antioxidant response pathway, modulation of the peroxisome proliferator–activated receptor-gamma coactivator-1α (PGC-1α)/sirtuin-1 (SIRT1) axis for mitochondrial biogenesis, and inhibition of the nuclear factor kappa-light-chain-enhancer of activated B cells inflammatory signaling cascade. The core of this review is a critical analysis of the human performance and recovery data, juxtaposing studies that show benefit with those that report null effects. We synthesize the key controversies that dominate the field, focusing on the critical confounding roles of poor bioavailability, participant training status, supplementation dosage, and timing. The evidence suggests that quercetin's most reliable effects may lie in accelerating recovery from exercise-induced muscle damage (EIMD) and reducing soreness, rather than directly enhancing maximal endurance performance. Its primary value may be as a “training adaptogen” that modulates cellular stress responses, thereby improving fatigue resistance over time. We conclude that quercetin's poor oral bioavailability is the principal barrier that has likely confounded the majority of human research to date. Future research must prioritize the use of high-bioavailability formulations to definitively ascertain whether the impressive preclinical benefits of quercetin can be translated into meaningful, practical applications for endurance athletes.
Keywords
Introduction
Endurance exercise represents a profound physiological challenge that perturbs homeostasis across multiple organ systems. The sustained metabolic demand of prolonged physical activity necessitates a significant increase in oxygen consumption, leading to an acute elevation in the production of reactive oxygen species (ROS) within contracting skeletal muscle, primarily from mitochondrial respiration. 1 For decades, these ROS were viewed almost exclusively as detrimental byproducts responsible for oxidative damage to cellular lipids, proteins, and DNA, contributing to muscle fatigue, damage, and impaired performance. However, this perspective has evolved into a more nuanced understanding. It is now recognized that ROS, at physiological concentrations, function as critical signaling molecules, or “eustress” agents, that are essential for mediating many of the positive adaptations to endurance training, including mitochondrial biogenesis, enhanced endogenous antioxidant defenses, and improved insulin sensitivity. 2 An excessive or chronic imbalance, where ROS production overwhelms the body's antioxidant capacity, results in oxidative stress, which is linked to an amplified inflammatory response, impaired muscle force production, and accelerated fatigue. 3 This dual role of ROS creates a delicate balance; managing excessive oxidative stress to mitigate fatigue and damage without blunting the essential adaptive signaling is a key challenge in sports nutrition. Alongside oxidative stress, strenuous endurance exercise induces micro-trauma to muscle fibers, triggering an inflammatory cascade and leading to delayed onset muscle soreness (DOMS), which can impair subsequent training and performance. 4 Consequently, nutritional strategies aimed at modulating these interconnected processes—oxidative stress, inflammation, and muscle recovery—are of paramount interest to athletes and sports scientists.
Among the vast array of natural compounds investigated for their potential to support endurance athletes, the flavonoid quercetin (3,3′,4′,5,7-pentahydroxyflavone) has emerged as a subject of intense research. Quercetin is one of the most abundant polyphenols in the human diet, found in a wide variety of plant-based foods, including onions, apples, berries, grapes, leafy greens, and tea. In nature, quercetin typically exists not as a free aglycone but as glycosides, where the core quercetin molecule is attached to a sugar moiety, such as in rutin (quercetin-3-O-rutinoside) or isoquercitrin (quercetin-3-O-glucoside). 5 This structural variation is critical, as it profoundly influences the compound's absorption and bioavailability, a central theme of this review. Figure 1 illustrates the chemical structure of the quercetin aglycone and its common glycosidic forms.

Chemical structures of quercetin and its common glycosides.
The scientific rationale for investigating quercetin as an ergogenic aid is compelling and multifaceted. In vitro and animal studies have consistently demonstrated that quercetin possesses potent antioxidant, anti-inflammatory, immunomodulatory, and even “exercise-mimetic” properties. The primary premise is that by leveraging these biological activities, quercetin supplementation could mitigate the negative aspects of strenuous exercise—such as excessive oxidative stress, inflammation, and muscle damage—thereby delaying fatigue, enhancing endurance performance, and accelerating recovery. 6 However, this compelling preclinical promise has collided with a starkly different reality in human clinical trials, creating the central controversy that defines this field of research. Despite hundreds of studies, the evidence for quercetin's efficacy in human athletes remains profoundly equivocal. For nearly every study reporting a modest benefit, another, often with a more rigorous design or in a different population, reports a null effect. This disconnect between the consistent, strong effects observed on the benchtop and in animal models and the inconsistent, often trivial effects seen in human performance studies is the critical issue that this review seeks to dissect.
This review aims to provide a comprehensive and critical evaluation of the scientific literature concerning quercetin's purported antioxidant and fatigue-resisting properties in the context of endurance training. Moving beyond a simple summary of findings, this paper will: 1) dissect the key molecular mechanisms proposed to underlie quercetin's effects, including the Nrf2, PGC-1α/SIRT1, and nuclear factor-κB (NF-κB) pathways; 2) systematically analyze the conflicting body of human clinical trial data on both endurance performance and post-exercise recovery; 3) synthesize the primary controversies that may explain the gap between preclinical promise and clinical reality, with a particular focus on the roles of bioavailability and participant training status; and 4) propose a clear and targeted roadmap for future research designed to resolve these persistent ambiguities. The ultimate goal is to provide the reader with a nuanced understanding of the current state of knowledge and the critical questions that remain to be answered.
Literature Search Strategy and Study Selection
We designed a structured search strategy, informed by PRISMA 2020 reporting guidance, to identify studies on quercetin in the context of endurance exercise, performance, and recovery. Searches were conducted in PubMed/MEDLINE, Scopus, Web of Science Core Collection, SPORTDiscus, Cochrane CENTRAL, ScienceDirect, and Google Scholar (first 200 results by relevance) from database. The core Boolean string combined controlled vocabulary and free-text terms for the exposure (quercetin and major glycosides) with exercise/performance and recovery constructs, and with key mechanistic pathways (Nrf2, PGC-1α/SIRT1, NF-κB). Example PubMed query:
(“quercetin”[MeSH] OR quercetin[tiab] OR “quercetin-3-O-glucoside”[tiab] OR isoquercitrin[tiab] OR rutin[tiab]) AND (exercise[MeSH] OR endurance[tiab] OR “endurance training”[tiab] OR aerobic[tiab] OR VO2max[tiab] OR cycling[tiab] OR runner*[tiab] OR “time trial”[tiab] OR “muscle damage”[tiab] OR DOMS[tiab]) AND (random*[tiab] OR trial[tiab] OR placebo[tiab] OR crossover[tiab] OR “cross-over”[tiab]) NOT (review[Publication Type] OR editorial[Publication Type]).
A mechanistic string was also used to capture pathway studies: (quercetin[tiab]) AND (Nrf2[tiab] OR “nuclear factor erythroid 2-related factor 2”[tiab] OR “PGC-1α”[tiab] OR SIRT1[tiab] OR “NF-κB”[tiab]) AND (skeletal muscle[tiab] OR mitochondria*[tiab] OR oxidative stress[tiab]).
Eligibility criteria were prespecified. For human performance and recovery outcomes, we included randomized or quasi-randomized controlled trials and crossover trials assessing oral quercetin (alone or with clearly separable analyses) in healthy adults (≥18 y), reporting endurance-relevant outcomes or recovery endpoints. For mechanistic insight, we additionally included in vivo animal studies and in vitro skeletal-muscle investigations focused on Nrf2, PGC-1α/SIRT1, or NF-κB signaling under exercise or muscle-stress contexts. We excluded non-original articles, case reports, parenteral administration, multinutrient blends where quercetin effects were not isolable, and studies lacking a control/comparator.
Quercetin's Role in Mitigating Exercise-Induced Oxidative Stress
The foundational hypothesis for quercetin's use in exercise is rooted in its well-documented antioxidant capabilities. Strenuous endurance exercise transiently increases ROS production, and if this production exceeds the cell's buffering capacity, it can lead to oxidative stress, cellular damage, and fatigue. The theory posits that quercetin, as a potent antioxidant, could help neutralize this excess ROS, thereby protecting muscle cells and preserving function. This section explores the molecular basis for this hypothesis, the evidence from preclinical models, and the complex and often contradictory findings from human exercise trials.
The Molecular Basis of Quercetin's Antioxidant Capacity
Quercetin's chemical structure is exceptionally well-suited for antioxidant activity. Its efficacy as a free radical scavenger is attributed to specific structural features, including the catechol group (an o-dihydroxyl group) on the B ring, the 2,3-double bond in conjugation with a 4-oxo group on the C ring, and hydroxyl groups at positions 3 and 5 (Figure 2). These features allow quercetin to readily donate hydrogen atoms or electrons to neutralize highly reactive free radicals, such as superoxide (O2⋅) and hydroxyl radicals (⋅OH), forming a more stable and less reactive quercetin radical in the process. 7 Its direct scavenging capacity is potent; some reports indicate it is over six times more effective than Trolox, a water-soluble vitamin E analog commonly used as an antioxidant reference standard. 8 Beyond direct scavenging, quercetin can also chelate transition metal ions like iron (Fe2+) and copper (Cu2+), preventing them from participating in Fenton reactions that generate highly damaging ⋅OH. 9
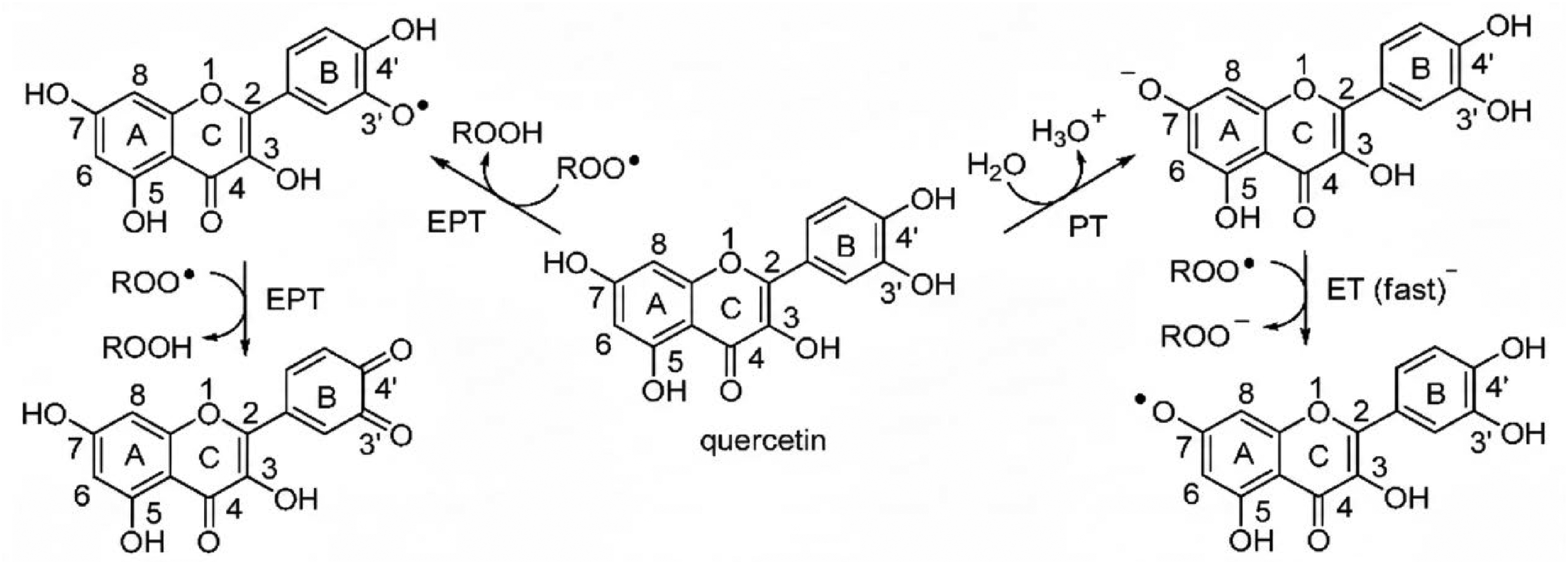
Possible reaction pathways for quercetin.
The Nrf2 Signaling Pathway
While direct radical scavenging is an important property, a more sophisticated mechanism of antioxidant action involves the upregulation of the body's own endogenous defense systems. The primary regulator of this cellular response is the Nrf2 signaling pathway. 10 Under basal, non-stressed conditions, Nrf2 is bound in the cytoplasm by its negative regulator, Kelch-like enzyme complex homologous-associated protein 1 (Keap1), which targets it for ubiquitination and proteasomal degradation, keeping its activity low. 11 However, in the presence of oxidative or electrophilic stress, specific cysteine residues on Keap1 are modified, causing a conformational change that releases Nrf2. The stabilized Nrf2 then translocates to the nucleus, where it heterodimerizes with small musculoaponeurotic fibrosarcoma oncogene homolog proteins and binds to a specific DNA sequence known as the antioxidant response element (ARE) located in the promoter region of numerous target genes. 12 This binding initiates the transcription of a broad suite of cytoprotective proteins, including antioxidant enzymes like heme oxygenase-1 (HO-1), nicotinamide adenine dinucleotide (phosphate) quinone dehydrogenase 1 (NQO1), superoxide dismutase (SOD), and catalase (CAT), as well as enzymes involved in the synthesis and regeneration of glutathione (GSH), the body's most abundant endogenous antioxidant. 13 Activation of the Nrf2-ARE pathway thus provides a coordinated and amplified defense against oxidative stress.
Quercetin as an Nrf2 Activator: The Preclinical Evidence
A substantial body of preclinical evidence strongly supports the role of quercetin as a potent activator of the Nrf2 pathway. In vitro studies have repeatedly shown that pretreating various cell types with quercetin protects them from chemically induced oxidative stress. For example, in mammary epithelial cells challenged with hydrogen peroxide (H2O2), quercetin pretreatment restored cell viability and antioxidant capacity by activating the Nrf2 pathway. 14 Similarly, in broiler chicken intestines challenged with lipopolysaccharide (LPS), quercetin attenuated oxidative stress by preventing the LPS-induced inhibition of Nrf2 activation and translocation. 15
This effect is not limited to cell culture. Animal models of diverse pathologies consistently demonstrate quercetin's ability to leverage the Nrf2 pathway for tissue protection. In a mouse model of traumatic brain injury, quercetin treatment upregulated Nrf2 expression, promoted its translocation to the nucleus, and restored levels of antioxidant enzymes like SOD, suggesting that quercetin's neuroprotective effects are mediated, at least in part, through Nrf2 activation. 16 Likewise, in a rat model of aluminum-induced neurotoxicity, quercetin co-treatment upregulated Nrf2 along with its downstream targets, mitigating oxidative damage in the brain. 17 The interaction appears to be direct; molecular docking analyses have shown that quercetin binds with high affinity to a pocket in the Nrf2 protein, suggesting a direct modulatory interaction that could facilitate its release from Keap1. 18 This robust preclinical evidence has solidified the view of quercetin as a powerful Nrf2-activating compound (Figure 3).

Quercetin's modulation of the Nrf2 antioxidant pathway.
The Human Exercise Conundrum and the “Antioxidant Paradox”
Despite the compelling mechanistic and preclinical data, the translation of quercetin's antioxidant effects to human endurance exercise has been fraught with inconsistency. Several well-controlled studies in trained athletes have failed to demonstrate that quercetin supplementation can attenuate systemic markers of exercise-induced oxidative stress. In a study by McAnulty et al, 19 trained cyclists supplemented with 1000 mg/day of quercetin for three weeks before undergoing three consecutive days of intense 3-h cycling bouts. Despite successfully elevating plasma quercetin levels, the supplement had no effect on the exercise-induced rise in plasma F2-isoprostanes (a marker of lipid peroxidation) or C-reactive protein (a marker of inflammation) compared to placebo. Similar null findings regarding oxidative stress and inflammation markers have been reported in other studies with trained athletes. 20
This inconsistency has fueled debate centered on the ‘antioxidant paradox’. 21 The paradox lies in the dual role of ROS. While excessive ROS are damaging, the transient burst of ROS produced during exercise is a vital physiological signal that triggers beneficial long-term adaptations, such as mitochondrial biogenesis and the upregulation of the body's own antioxidant enzymes. 22 The concern is that indiscriminately blunting this crucial signaling cascade with high doses of exogenous antioxidants like quercetin might actually be counterproductive, potentially hindering the very adaptations that athletes train to achieve. 23 This viewpoint challenges the simplistic notion that all antioxidants are beneficial in an exercise context and suggests that a certain level of oxidative stress is not only tolerable but necessary for improvement.
However, this narrative is complicated by meta-analytic evidence. A recent systematic review and meta-analysis by Rojano-Ortega et al 21 pooled data from studies and found that quercetin supplementation did, in fact, lead to a statistically significant, moderate reduction in post-exercise markers of oxidative stress. This suggests that an effect exists, but its detection may be dependent on the specific markers measured, the study population, and the exercise protocol.
This discrepancy between individual studies and meta-analytic findings points toward a more complex mechanism than simple ROS scavenging. One potential explanation is that quercetin's primary mode of action is not direct scavenging but rather an indirect, hormetic effect. Quercetin itself, particularly at higher concentrations, can exhibit pro-oxidant properties. 24 It is plausible that, in vivo, quercetin acts as a mild cellular stressor, generating a small pro-oxidant signal that is sufficient to activate the powerful Nrf2-mediated endogenous antioxidant system without causing significant damage. 25 This would result in a net protective effect, but one that is driven by an initial, mild stress rather than direct neutralization of exercise-induced ROS. This hormetic model could explain why direct antioxidant effects are difficult to measure systemically, yet downstream protective outcomes are sometimes observed.
Furthermore, the disconnect may stem from methodological limitations. Most human trials measure oxidative stress markers in the blood, which may not accurately reflect the biochemical environment within the skeletal muscle mitochondria, the primary site of action. 26 Additionally, quercetin is rapidly and extensively metabolized in humans, with conjugated metabolites, not the parent aglycone, being the dominant forms in circulation. 27 The antioxidant capacity of these metabolites is not fully characterized and may differ from the aglycone form tested in most in vitro assays. Therefore, the failure to detect a consistent antioxidant signal in human blood may be an artifact of measuring the wrong thing, in the wrong place, at the wrong time, rather than a true failure of the biological mechanism. Collectively, small changes in circulating oxidative stress markers may under-represent intramuscular redox effects because plasma redox does not reliably mirror muscle signaling, and exercise-generated ROS function as compartmentalized signals for adaptation rather than purely as damage mediators. We infer that any benefit of quercetin in endurance contexts is more likely via optimizing “redox eustress” and Nrf2-dependent cytoprotective programs than by broad ROS suppression. Near-term hotspots include in vivo muscle redox readouts and harmonized biomarker panels that pair plasma with serial muscle sampling in athletes receiving higher-bioavailability quercetin, with target-engagement of Nrf2 genes tracked alongside performance endpoints. 28
The Mitochondrial Biogenesis Hypothesis
Perhaps the most tantalizing hypothesis for quercetin's potential as an ergogenic aid is its purported ability to act as an “exercise mimetic” by stimulating mitochondrial biogenesis. Mitochondria are the powerhouses of the cell, and an increase in their density and function is a hallmark adaptation of endurance training, directly correlating with improved aerobic capacity and fatigue resistance. The theory that a dietary supplement could trigger this adaptive process, even in the absence of exercise, has driven a significant portion of the research in this field.
The PGC-1α/SIRT1 Axis in Skeletal Muscle Adaptation
The process of mitochondrial biogenesis is governed by a complex network of signaling pathways, at the center of which lies the transcriptional coactivator, PGC-1α. 29 Often referred to as a “master regulator” of mitochondrial biogenesis, PGC-1α does not bind to DNA directly but co-activates various transcription factors to orchestrate the expression of a broad program of genes required for building new mitochondria. 30 This includes nuclear genes encoding mitochondrial proteins and key transcription factors like nuclear respiratory factors 1 and 2 (NRF-1, NRF-2) and mitochondrial transcription Factor A (TFAM), which is essential for the replication and transcription of mitochondrial DNA (mtDNA). 31
The activity of PGC-1α is tightly controlled by post-translational modifications, most notably deacetylation by SIRT1. 32 SIRT1 is a nicotinamide adenine dinucleotide (NAD+)-dependent protein deacetylase, which positions it as a critical sensor of the cell's energy status. 33 During states of high energy demand, such as exercise or caloric restriction, the ratio of NAD+ to NADH increases, activating SIRT1. Activated SIRT1 then deacetylates PGC-1α, enhancing its transcriptional activity and initiating the mitochondrial biogenesis program. 34 This PGC-1α/SIRT1 axis thus forms a crucial link between physiological stress and the adaptive increase in mitochondrial capacity.
The “Exercise Mimetic” Theory: Evidence from Animal Models
The theory that quercetin could act as an exercise mimetic originated from a series of striking preclinical studies. The landmark investigation by Davis et al 35 provided the foundational evidence. In this study, mice were fed quercetin at doses of 12.5 or 25 mg/kg for just seven days without any exercise training. The results were remarkable: quercetin supplementation led to significant increases in the mRNA expression of both PGC-1α and SIRT1 in skeletal muscle and brain tissue. This was accompanied by downstream increases in markers of mitochondrial mass, including mtDNA and the protein cytochrome c. Most importantly, these molecular changes translated directly to functional improvements. The quercetin-fed mice exhibited a 37% increase in treadmill run time to fatigue and also showed greater voluntary wheel-running activity compared to placebo-fed controls.
These findings, demonstrating an increase in both mitochondrial machinery and endurance capacity without a single training session, established the “exercise mimetic” hypothesis and spurred a wave of further research. Subsequent studies in various animal models have largely corroborated these effects. For instance, in models of cachexia, diabetes, and traumatic brain injury, quercetin has been shown to preserve or improve mitochondrial content and function, often in conjunction with the upregulation of the PGC-1α pathway.36,37 Table 1 summarizes key animal studies that have investigated quercetin's effects on skeletal muscle mitochondrial biogenesis, providing a clear picture of the robust preclinical support for this hypothesis.
Summary of Animal Studies on Quercetin and Skeletal Muscle Mitochondrial Biogenesis.

The Human Enigma
In stark contrast to the compelling animal data, attempts to replicate these exercise-mimetic effects in humans have been largely unsuccessful, particularly in athletic populations. A key study by Dumke et al 20 recruited trained cyclists and supplemented them with 1000 mg/day of quercetin for three weeks. Subsequent analysis of vastus lateralis muscle biopsies revealed no differences between the quercetin and placebo groups in PGC-1α mRNA expression, citrate synthase activity (a marker of mitochondrial content), or muscle oxidative capacity. Another study in rats by Mukai and Sale 39 also directly challenged the original findings, reporting that 7 days of quercetin treatment failed to stimulate mitochondrial biogenesis in skeletal muscle or enhance endurance capacity, suggesting the initial findings might not be robust even in rodents. While a few studies in untrained individuals have reported modest increases in performance that were associated with non-statistically significant trends towards increased mitochondrial markers, the powerful, exercise-free biogenesis seen in the initial mouse studies has not been convincingly demonstrated in humans. 20
Dissecting the Controversy
The profound disconnect between the animal and human data on mitochondrial biogenesis is a central controversy in quercetin research. Several compelling hypotheses have been proposed to explain this gap.
First and foremost is the “ceiling effect” hypothesis. 42 This theory posits that well-trained endurance athletes, through years of consistent training, have already achieved a near-maximal level of skeletal muscle mitochondrial density. Consequently, there is very little physiological headroom for a nutritional supplement to induce further biogenesis. 42 The initial, untrained state of the mice in the Davis et al 43 study provided a low baseline and thus a large window for improvement, a condition that does not exist in elite athletes. This hypothesis is supported by the observation that the most positive human performance results, though modest, have tended to occur in untrained or recreationally active individuals.
Second, there may be fundamental species-specific differences in the absorption, metabolism, and cellular response to quercetin between rodents and humans. 44 The dose equivalency, metabolic pathways, and even the sensitivity of the PGC-1α/SIRT1 axis may differ significantly, making direct translation of the mouse findings problematic. 45
Finally, the issue of dosage and bioavailability is critical. It remains uncertain whether the standard oral dose of 1000 mg/day in humans achieves a sufficient intracellular concentration of active quercetin metabolites within skeletal muscle to effectively activate the SIRT1/PGC-1α pathway. The promising results from a high-bioavailability quercetin nanoemulsion in mice, which succeeded in activating the pathway where ordinary quercetin failed, underscore the potential importance of formulation and delivery. 46 This suggests that previous human studies may have failed not because the mechanism is invalid, but because an effective dose never reached the target tissue. The proposed mechanism is illustrated in Figure 4.

Proposed role of quercetin in the PGC-1α/SIRT1 mitochondrial biogenesis pathway.
The consistent failure to demonstrate mitochondrial biogenesis in human athletes has led to a re-evaluation of quercetin's role. Rather than acting as a generative stimulus to build new mitochondria, its primary benefit may be protective. Intense exercise generates significant oxidative stress that can damage existing mitochondrial membranes and proteins, impairing their function and contributing to fatigue. 47 By leveraging its antioxidant capabilities, particularly through the more robustly supported Nrf2 pathway, quercetin may primarily act to preserve the function and integrity of the existing mitochondrial pool during periods of high stress. 48 This “mitochondrial preservation” hypothesis offers a more plausible explanation for the modest recovery and fatigue-resistance benefits observed in some human trials, shifting the focus from biogenesis to maintenance and protection. Rodent data show that quercetin increases SIRT1, PGC-1α, mitochondrial DNA, and cytochrome-c with improved endurance; however, randomized trials in trained cyclists did not increase muscle PGC-1α/SIRT1 mRNA or performance, suggesting inadequate intramuscular exposure and/or ceiling effects in well-trained athletes. We infer that translation may require higher tissue exposure and metabolic context (eg, “train-low” microcycles). Hotspots include combining high-bioavailability quercetin with low-glycogen training, with biopsies quantifying citrate synthase and TFAM proteins and single-fiber respiration. 35
Quercetin's Impact on Endurance Performance and Recovery: A Critical Evaluation of Human Trials
The ultimate test of any purported ergogenic aid is its ability to translate mechanistic potential into tangible improvements in human performance and recovery. For quercetin, this translation has been the subject of extensive investigation, yielding a complex and often contradictory body of evidence. This section critically evaluates the human clinical trials, separating the data on direct endurance performance from the data on post-exercise recovery, and examines the underlying anti-inflammatory mechanisms.
Endurance Performance
The allure of quercetin as a performance enhancer stems from its potential to increase maximal aerobic capacity (VO2max) and time to fatigue. Some of the earliest human studies provided encouraging results. Davis et al 43 conducted a randomized, double-blind, placebo-controlled crossover study in 12 healthy but untrained participants. After only seven days of supplementation with 1000 mg/day of quercetin, they observed a modest but statistically significant 3.9% increase in VO2max and a substantial 13.2% increase in cycle ride time to fatigue compared to placebo. Another study in untrained men reported a similar small improvement of 2.9% in a 12-min treadmill time trial. 49
However, these positive findings are challenged by a larger volume of research reporting null effects, especially when the subjects are well-trained athletes. For instance, a study on elite male cyclists found no effect of 1000 mg/day of quercetin on a cycling time trial performance after six weeks of supplementation. 49 Similarly, a comprehensive study by Bigelman et al 42 on 58 moderately trained reserve officers’ training corps (ROTC) cadets found that six weeks of quercetin supplementation (1000 mg/day) failed to improve VO2max, performance on the Army Physical Fitness Test, or any other measure of physical performance compared to placebo. Studies on ultra-marathon runners have also failed to show performance benefits. 50
This discrepancy in the literature is reflected in meta-analyses, which have attempted to provide a conclusive summary. An early meta-analysis by Kressler et al, 51 which included 11 studies, found that quercetin provided a statistically significant but very small overall benefit to endurance capacity (combining VO2max and performance measures), with an effect size of 0.15, equating to a practical improvement of about 2% over placebo. However, using magnitude-based inferences, they concluded that these effects were likely to be “trivial” in a practical sense and that quercetin was unlikely to be a meaningful ergogenic aid for either trained or untrained individuals. 52 A more recent meta-analysis focusing on flavonoids more broadly found that quercetin supplementation alone was associated with a lower proportion of studies showing improved performance (25%) compared to other flavonoids like anthocyanins (54%). 53 The collective evidence, summarized in Table 2, points towards a conclusion that if quercetin has any direct effect on endurance performance, it is small, unreliable, and most likely to manifest in untrained populations.
Summary of Human Clinical Trials on Quercetin and Endurance Performance.

Recovery: EIMD and DOMS
While the evidence for direct performance enhancement is weak, the data supporting quercetin's role in post-exercise recovery is considerably more compelling. Strenuous or unaccustomed exercise, particularly with an eccentric component, causes mechanical damage to muscle fibers, leading to a loss of strength, swelling, and DOMS. 54 A growing body of evidence suggests that quercetin can mitigate these effects.
A comprehensive systematic review and meta-analysis by Rojano-Ortega et al 21 provides the strongest evidence to date. After analyzing 13 randomized controlled trials, the authors concluded that quercetin supplementation, typically at 1000 mg/day for at least seven days, significantly accelerated the recovery of muscle function, decreased perceived muscle soreness at 24 h post-exercise, and significantly reduced blood levels of creatine kinase (CK), a key biochemical marker of muscle damage, at 24–48 h post-exercise.
Individual studies support these meta-analytic findings. Bazzucchi et al 54 had participants perform a damaging eccentric exercise protocol for the elbow flexors. They found that 14 days of quercetin supplementation (1000 mg/day) significantly attenuated the loss of isometric strength, reduced the decay in muscle fiber conduction velocity, and resulted in lower levels of CK and lactate dehydrogenase (LDH) compared to placebo. Another study found that quercetin supplementation significantly reduced post-exercise levels of tumor necrosis factor-alpha (TNF-α) and CK after a damaging leg press protocol. 55 These findings suggest that quercetin can protect the structural integrity of muscle fibers and temper the inflammatory response to damage.
However, the evidence is not uniformly positive. A study by O'Fallon et al 56 using a similar eccentric elbow flexor protocol found that 1000 mg/day of quercetin for two weeks had no effect on strength loss, muscle soreness, swelling, or CK elevations compared to placebo. The reasons for such discrepancies are not entirely clear but may relate to differences in the specific exercise protocol, the training status of the participants, or the bioavailability of the quercetin formulation used. Despite these occasional null findings, the overall balance of evidence, as summarized in Table 3, points toward a beneficial effect of quercetin on recovery from EIMD. This suggests that quercetin's primary value for athletes may not be in boosting peak performance in a single event, but rather in facilitating a quicker and more complete recovery, allowing for higher quality and more frequent training sessions over time. This reframes quercetin as less of a direct ergogenic aid and more of a “recovery aid” or “training adaptogen.”
Summary of Human Clinical Trials on Quercetin, EIMD, and DOMS.

Inflammation: The NF-κB Pathway in an Exercise Context
The inflammatory response is intrinsically linked to both muscle damage and oxidative stress. Strenuous exercise activates the transcription factor NF-κB, a master regulator of inflammation. 59 In its inactive state, NF-κB is held in the cytoplasm by an inhibitory protein called IκBα. 60 Inflammatory stimuli, including cytokines and ROS generated during exercise, activate the IκB kinase (IKK) complex. IKK then phosphorylates IκBα, tagging it for ubiquitination and degradation by the proteasome. This frees NF-κB to translocate into the nucleus, where it binds to the promoters of pro-inflammatory genes, driving the production of cytokines like TNF-α and IL-6. 61
Quercetin is proposed to exert its anti-inflammatory effects by directly interfering with this cascade. Preclinical studies suggest that quercetin can inhibit the activity of the IKK complex, thereby preventing the phosphorylation and degradation of IκBα. This action effectively “locks” NF-κB in the cytoplasm, preventing it from activating pro-inflammatory gene transcription. 62 This proposed mechanism is illustrated in Figure 5.

Quercetin's inhibition of the NF-κB inflammatory pathway.
Human studies investigating this effect have yielded results that appear to be highly dependent on the supplementation protocol. Studies using acute ingestion of quercetin just before exercise have generally failed to attenuate the post-exercise inflammatory response. Konrad et al 63 reported that 1000 mg of quercetin ingested 15 min before a 2-h run did not suppress the exercise-induced increase in inflammatory markers like TNF-α. In contrast, studies employing chronic supplementation protocols have been more successful. Similarly, two weeks of supplementation was shown to lower post-exercise TNF-α in swimmers. 64
This discrepancy provides a clear example of how pharmacokinetics likely dictates biological outcomes. The inflammatory response to exercise unfolds over a period of hours to days. An acute dose of quercetin is rapidly metabolized and cleared from the body, with a half-life that may be too short to exert a meaningful effect on the entire inflammatory cascade. Chronic supplementation, on the other hand, allows for the accumulation of quercetin and its metabolites in relevant tissues, such as muscle and immune cells, potentially “priming” them to better manage the inflammatory stimulus of exercise. This provides a mechanistically plausible explanation for why chronic, but not acute, dosing appears to be effective for modulating exercise-induced inflammation. Meta-analyses indicate trivial-to-small average effects on VO2max/endurance performance, with more consistent benefits for post-exercise recovery and attenuation of EIMD (eg, CK, soreness) after chronic intake. We therefore interpret quercetin as a “training-aid” that helps preserve function between demanding sessions rather than an acute race-day ergogenic. Hotspots include mesocycle-length trials in endurance athletes that integrate functional tests and time-loss days with standardized loads while mapping CK/LDH and soreness trajectories under quercetin versus placebo. 51
Synthesis, Controversies, and Future Directions
The body of research on quercetin and endurance training is a case study in the complexities of translating preclinical science to human application. While the foundational mechanisms are compelling, the clinical results are a mosaic of conflicting, modest, and context-dependent findings. This section synthesizes the key themes of this review, focuses on the central controversies that likely explain the observed discrepancies, and proposes a clear roadmap for future research needed to move the field forward.
Synthesis: The Disconnect Between Bench and Bedside
The central narrative of this review is the stark disconnect between the robust, multi-pathway effects of quercetin observed in laboratory settings and its inconsistent performance in human athletes. Preclinical studies paint a picture of a powerful bioactive compound. In vitro and animal models consistently show that quercetin can activate the Nrf2 antioxidant pathway, stimulate mitochondrial biogenesis via the PGC-1α/SIRT1 axis, and suppress inflammation by inhibiting the NF-κB pathway.65,66 These mechanisms provide a strong theoretical basis for its use as an ergogenic and recovery aid. Yet, when tested in humans, these effects often diminish or disappear entirely. The potent “exercise-mimetic” effect on mitochondrial biogenesis seen in mice has not been replicated in human muscle biopsies. 39 The direct antioxidant effects are difficult to detect systemically. 19 The performance benefits are, by most meta-analytic accounts, trivial and practically insignificant for trained athletes. 67 The most promising area appears to be in enhancing recovery from muscle damage, but even these findings are not unanimous. 21 This chasm between bench and bedside suggests that one or more powerful confounding factors are at play in the human context.
The Bioavailability Barrier: The Primary Confounding Factor
The most critical and likely dominant factor confounding the field is quercetin's notoriously poor oral bioavailability. In its common aglycone form used in many supplements, quercetin is a lipophilic, crystalline compound with very low water solubility. This limits its absorption in the gastrointestinal tract. Furthermore, what little is absorbed undergoes extensive first-pass metabolism in the intestinal wall and liver, where it is rapidly conjugated into glucuronide and sulfate metabolites. 68 As a result, the concentration of free, unmetabolized quercetin in systemic circulation is extremely low, with some estimates of absolute bioavailability being less than 2%. The dominant circulating forms are metabolites, whose own biological activity is not fully understood. Some evidence suggests they may be deconjugated back to active quercetin at sites of inflammation, but this remains a point of debate. 69
This pharmacokinetic reality calls into question the validity of much of the existing human literature. It is highly probable that in many trials, the active compound never reached the target tissues (ie, skeletal muscle) in a concentration sufficient to elicit the biological effects observed in cell culture or high-dose animal studies. The inconsistent results seen across human trials may simply reflect the high inter-individual variability in quercetin absorption and metabolism. 70
Recognizing this limitation, researchers have developed novel formulation strategies to enhance quercetin's bioavailability. These advancements represent a potential turning point for the field. Studies have shown that quercetin glycosides, particularly quercetin-3-O-glucoside found in onions, are more bioavailable than the aglycone form. 71 More advanced delivery systems have shown even more dramatic improvements. Phytosomes were highlighted because they form stable molecular complexes of quercetin with dietary phospholipids, which increase membrane partitioning, favor lymphatic transport, and have shown ≈20-fold higher plasma exposure than unformulated quercetin in humans. Hydrogels based on fenugreek galactomannans were included because they are self-emulsifying and mucoadhesive, prolonging gut residence and enhancing dissolution of lipophilic quercetin; a human pharmacokinetic study showed an ≈18.6-fold increase in free quercetin and ≈62-fold in total quercetin compared with aglycone. These two platforms were specifically cited because they provide the strongest human evidence to date of overcoming quercetin's absorption and first-pass metabolism barriers, which is essential to achieve therapeutic tissue levels in athletes. 72 These technologies offer a viable way to overcome the absorption barrier. A comparison of these formulations is presented in Table 4.
Bioavailability of Different Quercetin Formulations in Humans.

Human pharmacokinetic work shows that dietary quercetin form dictates exposure: quercetin glucosides from onions achieve ≈50% absorption, while phospholipid “phytosome” and fenugreek galactomannan hybrid-hydrogel systems markedly increase maximum plasma concentration and area under the curve versus unformulated quercetin. We infer that null athletic trials may reflect subtherapeutic muscle levels rather than inefficacy per se. Hotspots are dose–exposure–response studies in trained athletes randomizing food-based glucosides against optimized delivery (phytosome or hydrogel), with intramuscular conjugates, Nrf2/PGC-1α signaling, and recovery outcomes measured concurrently. 74
Context is Key: Synthesizing the Impact of Training Status, Dosage, and Timing
Beyond bioavailability, the context in which quercetin is studied is paramount. This review has highlighted several key moderating factors. The training status of participants appears to be a critical determinant of outcomes. The “ceiling effect” hypothesis provides a plausible explanation for why well-trained athletes, who have already maximized their physiological adaptations, show little to no response to quercetin, whereas untrained individuals with a lower baseline are more likely to exhibit modest improvements. 42 In terms of dosage, 1000 mg per day has become the de facto standard in human exercise trials, and there is little evidence to suggest that higher doses provide additional benefit. Finally, the timing and duration of supplementation are crucial. For modulating inflammation and supporting recovery, chronic supplementation for at least 7–14 days appears necessary to achieve effective tissue concentrations, whereas acute, pre-exercise dosing is largely ineffective. 21
A Roadmap for Future Research
The existing body of literature, while extensive, has been largely built on the potentially flawed premise of studying a non-bioavailable compound. To resolve the field's central controversies and determine if quercetin has any true, practical value for athletes, future research must be more targeted and methodologically rigorous. The following directions are proposed:
Prioritize high-bioavailability formulations: Future clinical trials should abandon the use of standard quercetin aglycone and instead employ formulations with demonstrated high bioavailability, such as phytosomes or hydrogels. This is the most critical step to test whether overcoming the absorption barrier translates to more robust and consistent physiological effects.
Conduct mechanistic human studies using validated, preferably non-invasive readouts. Where feasible, pair high-bioavailability quercetin trials with in vivo assays of skeletal-muscle oxidative function such as 31P-magnetic resonance spectroscopy and blood-flow–corrected near-infrared spectroscopy, which have been cross-validated against each other and against in vitro oxidative capacity. 75 For redox signaling and Nrf2 target engagement, measure peripheral-blood mononuclear cell (PBMC) expression of canonical Nrf2-regulated genes (eg, NQO1, HO-1, GCLC) alongside robust systemic oxidative-stress indices. 76 For NF-κB–linked inflammation, quantify PBMC nuclear p65 translocation or DNA-binding activity together with cytokine panels. Muscle biopsies should be reserved for focused sub-studies when indispensable, for example to confirm intramuscular target engagement in a small subset. 77 This will allow researchers to finally determine if quercetin, when delivered effectively, can activate the Nrf2, PGC-1α, and NF-κB pathways in human muscle tissue, bridging the gap between preclinical mechanisms and clinical outcomes.
Design recovery-focused protocols: The primary outcome measures should shift away from single-bout VO2max or time-trial performance. Instead, protocols should be designed to tax recovery, such as multi-day training blocks or simulated stage races, with performance on subsequent days being the key variable. This aligns with the evidence suggesting quercetin's strongest effects are on recovery and fatigue resistance.
Directly test the “ceiling effect": Studies should be designed to directly compare the effects of a high-bioavailability quercetin formulation in distinct cohorts of sedentary individuals versus elite endurance athletes under the same protocol. This would provide a definitive test of the training status hypothesis.
Vascular and immune-cell models demonstrate that quercetin-3-O-glucuronide can be deconjugated in situ by β-glucuronidase under inflammatory stress, releasing active aglycone and eliciting measurable vascular effects; this is blocked by β-glucuronidase inhibition and has been observed in vivo. 78 Macrophage studies likewise show inflammation-linked deconjugation of quercetin glucuronides, with β-glucuronidase-dependent release of aglycone during mitochondrial stress. 79 In the context of EIMD, the early inflammatory phase is characterized by neutrophil accumulation followed by pro-inflammatory macrophage infiltration into injured muscle. 80 Importantly, multiple rodent studies using downhill/eccentric exercise demonstrate that intramuscular β-glucuronidase activity rises during recovery (hours–days), supporting a biochemical milieu permissive for local deconjugation of circulating flavonoid glucuronides. 81 Taken together, these data substantiate the mechanistic plausibility that quercetin conjugates act as pro-drugs that are deconjugated within damaged muscle during EIMD recovery.
Conclusion
The investigation into quercetin as an ergogenic aid for endurance athletes is a field characterized by a compelling theoretical framework that has largely failed to translate into consistent, practical benefits in human trials. The preclinical evidence supporting its role as a modulator of the Nrf2, PGC-1α/SIRT1, and NF-κB pathways is robust, positioning it as an ideal candidate to combat exercise-induced oxidative stress, stimulate mitochondrial adaptations, and control inflammation. However, the human clinical evidence is a landscape of contradiction. While there is promising and relatively consistent evidence that chronic quercetin supplementation can accelerate recovery from muscle damage and reduce soreness, its ability to directly enhance maximal endurance performance is, at best, trivial and unreliable, particularly in trained athletes.
This review concludes that the primary reason for this disconnect is quercetin's poor oral bioavailability. The vast majority of human studies have likely failed to deliver a pharmacologically effective dose of the active compound to the target tissues. Therefore, the entire body of existing literature must be interpreted with this critical limitation in mind. Quercetin's true potential remains an open question, one that can only be answered by a new generation of research utilizing novel, high-bioavailability formulations. Future studies must leverage these technologies in well-designed, mechanistically-focused trials to finally determine if the promise of quercetin seen on the bench can become a reality for the athlete. Until then, while quercetin may offer modest benefits for recovery, its status as a potent endurance performance enhancer remains unproven.
Footnotes
Acknowledgements
This work was financially supported by The Henan Province Soft Science Research Program Project “Research on the Practical Path of Special Sports Culture Supporting Rural Revitalization in Henan Province”, Project Number: 252400410353.
Author Contribution Statement
Yan Li: methodology; software; validation; formal analysis; investigation; data curation; writing—original draft preparation,
Funding
This work was financially supported by The Henan Province Soft Science Research Program Project (252400410353).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
