Abstract
Six compounds previously isolated and reported from Macropidia fuliginosa were evaluated for antimicrobial activity against 11 different microbial strains, with all compounds displaying broad spectrum activity. In addition, the compounds were also assessed for their antiproliferative activity and cytotoxicity. Further investigation of M. fuliginosa has led to the discovery of a previously described acenaphthylenediol from the bulbs of the plant.
In recent years, the Marine and Terrestrial Natural Product (MATNAP) research group has been extensively investigating the chemistry of plants that belong to the Haemodoraceae family.
1
-4
As part of our continuing efforts to chemically profile Australian plants derived from this family, Macropidia fuliginosa (Hook.) Druce, commonly known as the Black Kangaroo paw which is an endemic species of Western Australia, was recently investigated, with 16 compounds being reported from the bulbs and flowers of the plant.
5
Antibiotic assessment of the isolated compounds showed that some were active against both Gram-positive and Gram-negative bacteria.
5
Among the compounds isolated in the original study of the plant were anigorufone (

Compounds isolated from Macropidia fuliginosa.
These 6 compounds were selected for further evaluation of their antibiotic and antifungal against a selection of microorganisms, including Gram-positive and Gram-negative bacteria and against various fungi. The activity of the isolated compounds was compared to standard antibiotic and antifungal compounds. The antiproliferative effect and cytotoxicity of the compounds were also evaluated.
Further investigation of the plant has subsequently identified the presence of a previously described acenaphthylenediol derivative from an enriched fraction of the bulbs of the plant.
Results and Discussion
Antimicrobial evaluation of the compounds was carried out against 6 bacterial strains (3 Gram-positive and 3 Gram-negative), and 5 fungal strains, at a concentration of 1 mg/mL.
All 6 natural products evaluated showed a range of activity across all microorganisms tested, particularly across the bacteria. The results of the antimicrobial assays are provided in Table 1, whereby the zones of inhibition are quoted as the mean diameter in mm of the clear inhibition zone after 24 hours of incubation. The positive controls were ampicillin and ciprofloxacin (antibiotic positive controls), as well as amphotericin B and carbendazim (antifungal positive control).
Antimicrobial Activity of Compounds (1-6) Isolated From Macropidia fuliginosa.
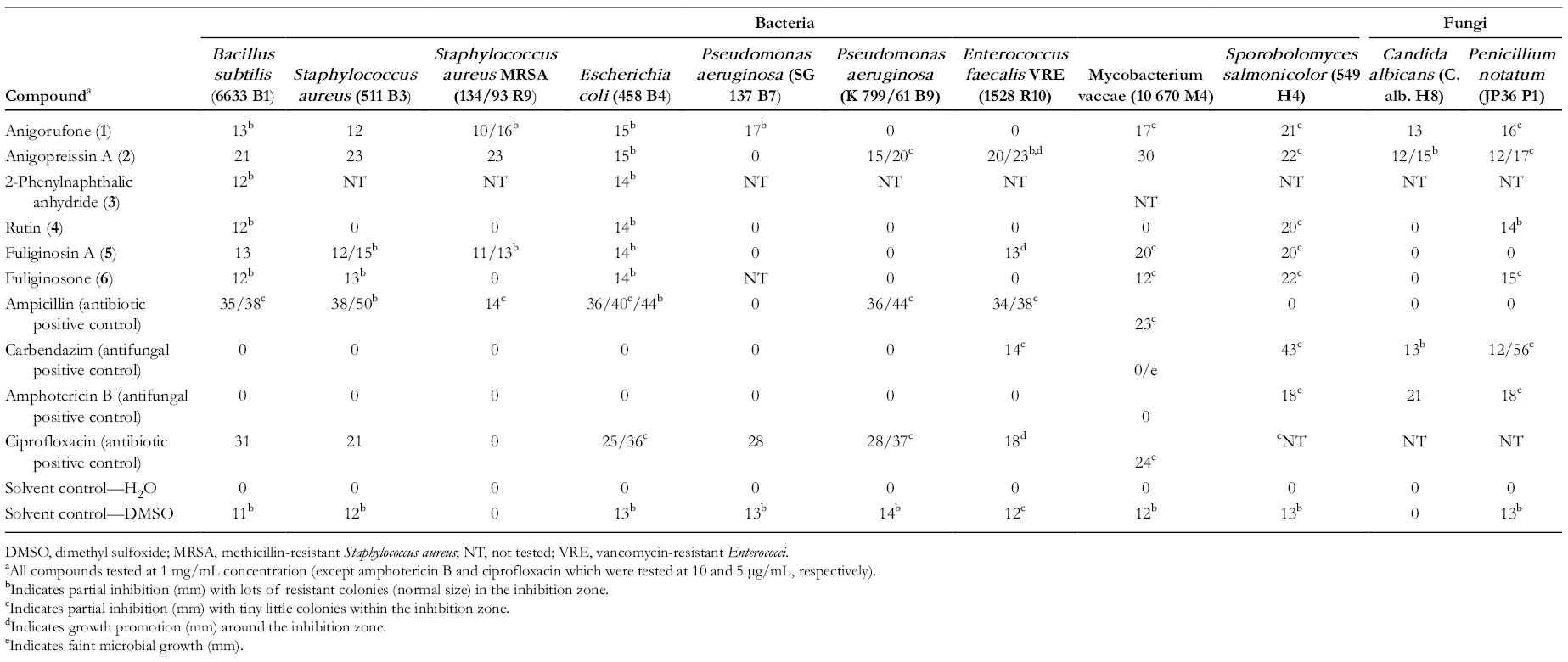
DMSO, dimethyl sulfoxide; MRSA, methicillin-resistant Staphylococcus aureus; NT, not tested; VRE, vancomycin-resistant Enterococci.
aAll compounds tested at 1 mg/mL concentration (except amphotericin B and ciprofloxacin which were tested at 10 and 5 µg/mL, respectively).
bIndicates partial inhibition (mm) with lots of resistant colonies (normal size) in the inhibition zone.
cIndicates partial inhibition (mm) with tiny little colonies within the inhibition zone.
dIndicates growth promotion (mm) around the inhibition zone.
eIndicates faint microbial growth (mm).
Interestingly, 2 strains of Pseudomonas aeruginosa tested showed vastly different results for 2 of the compounds (
Anigopreissin A (
2-Phenylnaphthalic anhydride (
Compared to the positive controls, some compounds (
The antiproliferative activity and cytotoxicity of the compounds is given in Table 2. The antiproliferative activity is the ability of a compound to reduce the proliferation of cells by not allowing the cells to multiply rapidly. Cytotoxicity refers to causing harm to cells and thereby killing them. These results indicated that the strongest cytotoxic compound was anigopreissin A (
Antiproliferative Activity and Cytotoxicity of Compounds Isolated From Macropidia fuliginosa.

Further investigation of an enriched nonpolar fraction derived from the bulbs of M. fuliginosa (see the “Experimental” section) yielded a previously described acenaphthylenediol derivative (
In the original assignment of the Nuclear Magnetic Resonance (NMR) data of
Conclusion
Compounds isolated from M. fuliginosa showed broad spectrum activity against both the bacteria and fungi tested. Four of the compounds showed equal or better inhibition against various microorganisms, including resistant strains of bacteria. The compounds also showed promising antiproliferative activity.
Experimental
General Experimental Conditions
1H (500 MHz) and 13C (125 MHz) NMR spectra were acquired in CDCl3 on a 500 MHz Agilent DD2 NMR spectrometer with reference to solvent signals (δH 7.26; δC 77.0). Two-dimensional NMR experiments recorded included gCOSY, HSQCAD, and gHMBCAD. Silica gel flash chromatography was carried out using Davisil LC35Å silica gel (40-60 mesh) with a 20% stepwise solvent elution from 100% petroleum spirits (60°C-80°C) to 100% CH2Cl2 to 100% EtOAc and finally to 100% MeOH. All analytical High Pressure Liquid Chromatography (HPLC) analyses and method development were performed on a Dionex P680 solvent delivery system equipped with a PDA100 UV detector (operated using “Chromeleon” software). Analytical HPLC analyses were carried out using either a gradient method (0-2 minutes 10% CH3CN/H2O; 14-24 minutes 75% CH3CN/H2O; 26-30 minutes 100% CH3CN; and 32-40 minutes 10% CH3CN/H2O or an isocratic method (70% CH3CN/H2O) on an Alltech Alltima HP C18 (250 × 4.6) 5 µm column at a flow rate of 1.0 mL/min. Semipreparative HPLC was carried out on a Varian Prostar 210 solvent delivery system equipped with a Prostar 335 PDA detector (operated using “Star Workstation” software) using an isocratic method (70% CH3CN/H2O) using an Alltech Alltima C18 (250 × 10) 5 µm column at a flow rate of 3.5 mL/min. Mass Spectrometry (MS) for pure compounds was carried out on a Perkin Elmer AxION 2 TOF.
Previously Isolated Natural Products for Biological Evaluation
Compounds
Current Natural Products Isolated
The crude extract was decanted and concentrated under reduced pressure and sequentially solvent partitioned (triturated) into CH2Cl2 and MeOH soluble extracts, respectively. The CH2Cl2 extract was subjected to flash silica gel column chromatography (20% stepwise elution from petroleum ether (60°C-80°C) to CH2Cl2 to EtOAc and, finally, to MeOH). The 100% CH2Cl2 fraction was subjected to Sephadex LH-20 column chromatography (100% MeOH) to yield 4 fractions. The first fraction was subjected to Reversed Phase - High Pressure Liquid Chromatography (RP-HPLC) (70% CH3CN/H2O) to yield compound (
Antimicrobial Assays
To determine their biological activities, anigorufone (
Antiproliferative Assays
Cell proliferation and cytotoxicity assays are applied to assess the efficacy and potency of natural products. The following cell lines and cell culture mediums were used: HUVEC (ATCC CRL-1730) and Dulbecco′s Modified Eagle′s Medium (DMEM) (CAMBREX 12-614F); K-562 (DSM ACC 10) and RPMI 1640 (CAMBREX 12-167F); and HeLa (DSM ACC 57) and RPMI 1640 (CAMBREX 12-167F).
Cells were grown in the appropriate cell culture medium supplemented with 10 mL/L ultraglutamine 1 (CAMBREX 17-605E/U1), 550 µL/L (50 mg/mL) gentamicin sulfate (CAMBREX 17-518Z), and 10% heat-inactivated fetal bovine serum (SIGMA F7524) at 37°C in 5% CO2 in high-density polyethylene flasks (NUNC 156340).
To decide if the compounds have an antiproliferative and/or a cytotoxic effect on human cells, the cytotoxicity (cell death) and the antiproliferative activity (retardation of cell proliferation) were measured. For this, different colorimetric assays for determining the number of viable cells were used. Under these experimental conditions, the optical density measured from the CellTiter-Blue reagent and methylene blue assay is proportional to the number of viable cells.
The test substances were dissolved in DMSO before being diluted in Dulbecco′s Modified Eagle′s Medium (DMEM). The adherent cells were harvested at the logarithmic growth phase after soft trypsinization using 0.25% trypsin in Phosphate Buffer Solution (PBS) containing 0.02% Ethylenediaminetetraacetic acid (EDTA) (Biochrom KG L2163). For each experiment, approximately 10 000 cells were seeded with 0.1 mL culture medium per well of the 96-well microplates (HUVEC: flat-bottomed NUNC 167008, K-562: round-bottomed NUNC 163320). To test the antiproliferative effect of the natural products on HUVEC and K-562, the cells were incubated for 72 hours in plates prepared with control and different dilutions of test substances. The GI50 values were defined as being where the inhibition of proliferation is 50% compared to the untreated control.
For the cytotoxicity assay, HeLa cells were preincubated for 48 hours without the test substances. To test the cytotoxic effect of the natural products on HeLa, the dilutions of the compounds were carried out carefully on the subconfluent monolayers of HeLa cells after the preincubation time. After incubation time, the cytolytic effect of compounds was analyzed in comparison to negative control. The 50% cytotoxicity concentration (CC50) was defined as the test compound concentration required for destruction in 50% of the cell monolayer compared to the untreated control.
The cells were incubated with dilutions of the natural products in microplates for 72 hours at 37°C in a humidified atmosphere and 5% CO2. This incubation was found to be an optimum time for the evaluation of the cytotoxicity and the inhibition of cell proliferation by finding out the number of viable cells stained with CellTiter-Blue reagent or methylene blue.
For estimating the influence of the natural products on cell proliferation of K-562, the numbers of viable cells present in multiwell plates via CellTiter-Blue assay (PROMEGA) were determined. This uses the indicator dye resazurin to measure the metabolic capacity of cells as indicator of cell viability. Viable cells of untreated control retain the ability to reduce resazurin into resorufin, whereas nonviable cells rapidly lose metabolic capacity and do not reduce the indicator dye. The absorption maximum for resazurin is 605 nm and the absorption maximum for resorufin is 573 nm. Thus, the absorbance measurements at 570 nm using 600 nm as a reference wavelength can be used to monitor the results. Values are compared to blank wells containing CellTiter-Blue reagent without cells.
The adherent HUVEC and HeLa cells were fixed by glutaraldehyde (MERCK 1.04239.0250) and stained with a 0.05% solution of methylene blue (SERVA 29198) for 15 minutes. After gently washing, the stain was eluted with 0.2 mL of 0.33 N HCl in the wells. The optical densities were measured at 660 nm (methylene blue) in SUNRISE microplate reader (TECAN).
Under these experimental conditions, the signals from the methylene blue and CellTiter-Blue reagent are proportional to the number of viable cells.
A repeat determination was conducted in all experiments and 4 replicates were assayed.
The calculations of different values of GI50 and CC50 were performed with the software Magellan (TECAN).
Offline (HPLC-NMR and HPLC-MS) Characterization of Compounds
3-Phenyl-1,2-dihydroacenaphthylen-1,2-diol 7 : 0.7 mg (0.002% dry weight); insufficient amount isolated to obtain a specific rotation; UV (extracted from PDA) (70% CH3CN/D2O) λmax 219, 239, 261, 295 nm; 1H NMR (500 MHz, CDCl3) δ 7.86 (1H, d, J = 8.5 Hz, H-5), 7.78 (1H, d, J = 7.0 Hz, H-6), 7.78 (2H, d, J = 7.0 Hz, H-2′/H-6′), 7.66 (1H, d, J = 8.5 Hz, H-4), 7.60 (1H, m, H-8), 7.58 (1H, dd, J = 7.0, 8.0 Hz, H-7), 7.50 (2H, dd, J = 7.0, 8.0 Hz, H-3′/H-5′), 7.41 (1H, t, J = 7.0 Hz, H-4′), 5.62 (1H, d, J = 5.5 Hz, H-2), 5.52 (1H, d, J = 5.5 Hz, H-1); 13C NMR (125 MHz, CDCl3) δ 142.9 (C, C-8a), 139.7 (C, C-1′), 138.5 (C, C-2a), 136.4 (C, C-3), 135.7 (C, C-8b), 130.4 (C, C-5a), 129.9 (CH, C-4), 128.9 (CH, C-2′/C-6′), 128.7 (CH, C-3′/C-5′), 128.3 (CH, C-7), 127.5 (CH, C-4′), 126.1 (CH, C-5), 124.7 (CH, C-6), 121.4 (CH, C-8), 74.3 (CH, C-1), 72.6 (CH, C-2); HPLC-MS m/z 245.0959 (calcd for C18H13O, 245.0961, [M−OH]+), m/z 285.0886 (calcd for C18H14NaO2, 285.0886, [M+Na]+). HPLC-MS m/z 245[M−OH]+, 285 [M+Na]+.
Supplemental Material
Supplementary Material - Supplemental material for Antimicrobial Evaluation of the Constituents Isolated From Macropidia fuliginosa (Hook.) Druce
Supplemental material, Supplementary Material, for Antimicrobial Evaluation of the Constituents Isolated From Macropidia fuliginosa (Hook.) Druce by Robert Brkljaca, Hans-Martin Dahse, Kerstin Voigt and Sylvia Urban in Natural Product Communications
Footnotes
Acknowledgment
KV wishes to express her gratitude to Mrs Christiane Weigel for her technical assistance in the performance of the antimicrobial activity assays.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
