Abstract
Introduction
Sustainable and efficient food systems must provide healthy food for a growing population, estimated at 9.15 billion by 2050, 1 while also helping to create more livable environment. Ruminants are the population that provide meat, milk, wool, and other fibers. 2 Therefore, to maximize animal performance and production potential, it is necessary to apply appropriate feeding practices that assess the animals’ needs in terms of feed value as well as quantity and quality. On the other hand, it is also important to strike a balance between increasing production and reducing the negative impact on the environment.
The use of toxic chemical agents such as ionophore antibiotics, halogenated compounds, and nitrates3,4 in the animal feed sector increased the presence of chemical residues in human food products. It is therefore important to replace synthetic antioxidants with plant-based feed additives 5 in animal nutrition to improve livestock productivity and product quality. 6
Natural extracts, essential oils, and plant by-products contained bioactive compounds that positively influence the health status and production parameters of ruminants. These biomolecules are powerful natural stimulants due to their diverse antibacterial, antiparasitic, antioxidant, anti-inflammatory, and immunostimulant properties. 7 The quality of bioactive substances contained in phytobiotics, such as alkaloids, terpenoids, polyphenols, etc, can vary depending on the cultivation method, different soil abundances, or even atmospheric conditions. 8 They prevent inflammation and diarrhea, increase digestive enzyme activity, and promote digestion in ruminants. 9 The nutritional potential of ruminant feeds can be determined based on their nutritional and energy values and feed efficiency, regardless of the nature of the substrate or the animal species studied.
Dill (Anethum graveolens L.) is a versatile plant used as a source of food, medicinal, and pharmaceutical products. This plant is used not only as an aromatic herb and spice in home cooking but also in the food and canning industries. The water-soluble polysaccharides from dill seeds were used as meat preservatives. 10 A. graveolens contained essential oils, fatty oils, proteins (15.68%), carbohydrates (36%), fiber (14.80%), polyphenols, furanocoumarin, and minerals. 11 According to the study of Zeyneb et al 12 A. graveolens has shown in vitro potential as an ecological and environmental-friendly antifungal agent. In addition, due to its wealth of active ingredients and natural substances, 13 this plant can be considered a support for the body, particularly in traditional medicine to treat some gastrointestinal disorders such as bloating, indigestion, and colic. Indeed, the results of an ethnopharmacological survey carried out by Brinsi et al 14 proved that the use A. graveolens was efficient to treat digestive disorders (40.7%).
Therefore, the nutritional benefits of A. graveolens in ruminant production deserve to be studied and evaluated, as dill can be an excellent feed supplement to encourage the consumption of environmental-friendly and anti-methanogenic plants. Therefore, this study aims to evaluate the phytochemical and bioactive potential of A. graveolens, in particular, its antioxidant activity and its effect on ruminal fermentation in vitro using the gas production technique in sheep and goats.
Results
Nutritional Value, Phytochemical Analysis of A. graveolens
The results of the chemical composition of A. graveolens are presented in Table 1. Fruits have the highest dry matter content, followed by flowers and leaves, respectively. Leaves, on the other hand, contained the lowest levels of organic matter and carbon.
Chemical Composition (% DM) of various Parts of Anethum graveolens.

Note: Data are represented as mean ± SD (n = 3). Means in the same column with no common superscript differ significantly (P < 0.05). Abbreviations: Dry matter (DM), Mineral matter (MM); Organic matter (OM); Total lipids (TL); Total phosphorus (P); Total nitrogen (N).
Total nitrogen and total lipid contents did not significantly differ between the different parts of A. graveolens. Leaves and flowers had the highest phosphorus concentrations, around 74 ppm. However, the results of total lipid content did not significantly (P < 0.05) vary in A. graveolens leaves, flowers, and fruits (Table 1).
Parietal Constituents
Analyzing parietal constituents, we found that fruits had the highest level of neutral detergent fiber (NDF), acid detergent fiber (ADF), acid detergent lignin (ADL), lignin and hemicellulose, as compared to leaves and flowers. These results also showed that the content of true raw cellulose (TRC) did not significantly differ (P < 0.05) between the three extracts (Table 2).
Parietal Composition (% DM) of various Parts of Anethum graveolens.

Note: Data are represented as mean ± SD (n = 3). Means in the same column with no common superscript differ significantly (P < 0.05). Abbreviations: Neutral Detergent Fiber (NDF); Acid Detergent Fiber (ADF); Acid Detergent Lignin (ADL); Hemicellulose (HC); True raw cellulose (TRC).
Quantification of Phenolic Compounds
The phenolic compounds content of A. graveolens extracts is represented in Table 3. The results showed that there were significant differences (P < 0.05) between the different extracts, where the highest contents in extraction yield, total phenols, flavonoids and total tannins were recorded in the aqueous extract of flowers. In contrast, the highest concentration of condensed tannins was observed in leaves.
Contents of the Samples in Secondary Metabolites of various Extracts of Anethum graveolens.

Note: Data are represented as mean ± SD (n = 3). Means in the same column with no common superscript differ significantly (P < 0.05). Abbreviations: Extraction yield (EY); Total polyphenols (TP); Total flavonoids (TF); Total tannins (TT); Condensed tannins (CT); AGLEAE: A. graveolens leaves aqueous extract; AGFLAE: A. graveolens flowers aqueous extract AGFRAE: A. graveolens fruits aqueous extract.
Evaluation of Antioxidant Activities
Radical scavenging activity on 1, 1-diphenyl-2-picrylhydrazyl (DPPH•)
Concerning the DPPH• radical scavenging activity of the various A. graveolens extracts, the present results showed significant differences (P < 0.05) between the samples examined (Table 4). Indeed, fruits’ extract (AGFRAE) showed the best antioxidant activity (IC50 = 41.71 ± 2.23 µg/ml) compared to those obtained from flowers (AGFLAE) and leaves (AGLEAE). Moreover, the IC50 of AGFRAE was close to that of BHT (IC50 = 41.04 ± 1.26 μg/mL), used as a reference antioxidant molecule.
Concentration of 50% Inhibition of DPPH● Free Radicals (IC50) of Anethum graveolens Aqueous Extracts Compared to Reference Molecules.

Note: Data are represented as mean ± SD (n = 3). Means in the same column with no common superscript differ significantly (P < 0.05). Abbreviation: Butylatedhydroxytoluene (BHT).
Ferric (Fe3+) Reducing Power
The ability of A. graveolens extracts to reduce Fe3+ to Fe2+ was measured (Figure 1). The obtained results showed a significant increase (P < 0.05) in the absorbance value for the different extracts. AGFLAE showed the highest ferric (Fe3+) reducing power (2.79) compared with the other extracts. The significant reducing power of A. graveolens extracts can be explained by their wealth of secondary metabolites, which can act in the same way as reductones, donating electrons to free radicals to convert them into more stable products.

Antioxidant activity of various Anethum graveolens extracts using the Ferric ions (Fe3+) reducing power assay. Data are represented as mean ± SD (n = 3). a, b, c Different letters indicate significant differences between the same concentration in different Anethum graveolens extracts (P < 0.05).
In Vitro Fermentation and gas Production Kinetics
Kinetic parameters for in vitro fermentation of various substrates are shown in Table 5 and Figure 2. In vitro fermentation of A. graveolens was based on three phases (Figure 2A and Figure 2B). The first one (I) is the Lag phase which was indicated by the negative value of the soluble fraction “a”. This latent phase explains the low degradation due to the time required for the microorganisms to attach to and colonize the dietary fibers. The second phase (II) is an exponential phase, during which the production is potential “b”, influencing the speed at which food passed through the rumen. Finally, the third phase (III) is a stationary state during which the gas production is minimal, called “c”, and represented the level of digestibility of the “b” fraction.

Kinetics gas production in vitro (ml/300 mg DM) of the various parts from Anethum graveolens during 48 h of incubation in sheep (A) and goats (B) and the volume of methane released at the end of ruminal fermentation (C).
The Effect of Anethum Graveolens In Vitro on Equation Y = a + b (1-exp-ct) and the Characteristic Parameters of gas Production Kinetics.

Note: Data are represented as mean ± SD (n = 3). Means in the same column for the same species (sheep or goat) and different plant parts with no common superscript differ significantly (P < 0.001; P < 0.01 and P < 0.05). *: significant at an error threshold α =5%; **: significant at an error threshold α =1%; ***: significant at an error threshold α =0.1%. Abbreviations: Organic matter digestibility (OMD); Metabolizable energy (ME); Volatile fatty acids (VFA).
Fermentation kinetics showed that there was no significant difference (P < 0.001) between the two species in terms of gas production. Depending on the nature of the substrate and the speed of enzymatic degradation, the results showed that fraction “a” was significantly higher in fruits, “b” in flowers, and “c” in leaves for sheep and goats. A highly significant difference (P < 0.001) was observed between the different A. graveolens substrates for gas production after 24 h incubation.
The volume of gas produced after 48 h of incubation was significantly higher for leaves (33.5 ml in sheep and 41.5 ml in goats) and flowers (39 ml in sheep and 48 ml in goats). These data lead to deduce that fruits produce the least gas and were therefore the easiest substrate to be digested compared with other substrates in ruminants.
Digestibility parameters such as organic matter digestibility, metabolizable energy, and volatile fatty acids are shown in Table 5. The results demonstrated that fruits were characterized by the lowest levels of organic matter digestibility. The same data was also recorded for metabolizable energy and volatile fatty acids, which showed that fruits gave the lowest levels compared to leaves and flowers, in both studied animal models. At leaf level, methane concentrations were low in sheep and goats. However, from an ecological point of view, we can conclude that they were the most environmental friendly and the safest for nature (Figure 1C).
Nutritional Value
The nutritional and energy values of the A. graveolens substrates studied are shown in Table 6. The results showed that fruits have the highest value of Feed Unit for Lactation (UFL) compared with leaves and flowers. The levels of digested proteins in the small intestine when rumen-fermentable nitrogen is limiting (PDIN) and when rumen-fermentable energy is limiting (PDIE) did not differ significantly between the different plant substrates.
Prediction of the Food Values of various Parts of Anethum Graveolens.
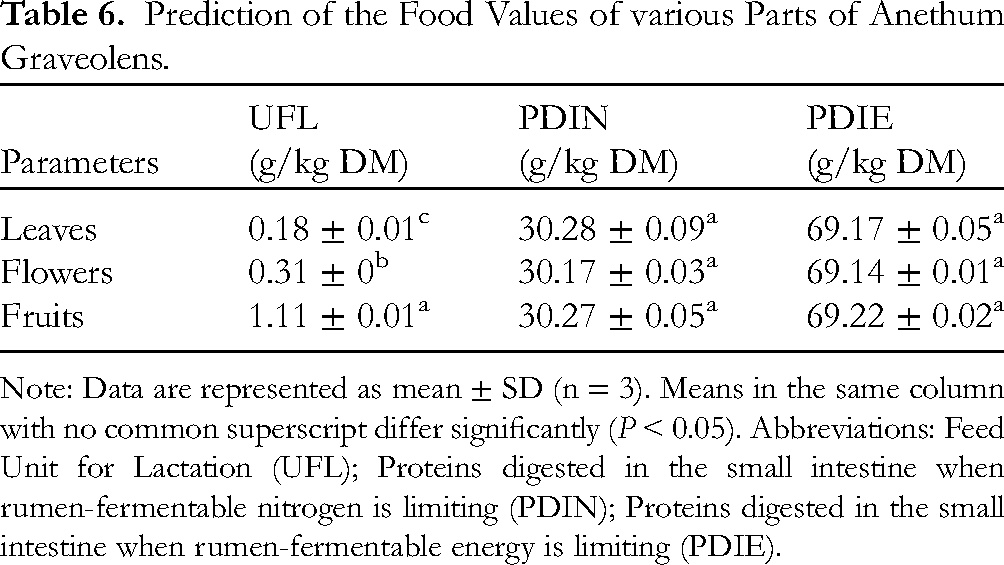
Note: Data are represented as mean ± SD (n = 3). Means in the same column with no common superscript differ significantly (P < 0.05). Abbreviations: Feed Unit for Lactation (UFL); Proteins digested in the small intestine when rumen-fermentable nitrogen is limiting (PDIN); Proteins digested in the small intestine when rumen-fermentable energy is limiting (PDIE).
Discussion
The phytochemical characterization and the bioactive activities of different substrates of A. graveolens were evaluated and their effects on ruminal fermentation using the in vitro gas production technique in sheep and goats were determined. Using organic alternatives such as A. graveolens, the farmer can benefit not only from the plant nutritional and energetic properties but also from its nutraceutical side due to its antioxidant and anti-inflammatory properties on animal health.
Phytochemical analysis of the various plant parts (leaves, flowers and fruits) showed that the fruits had the highest levels of organic matter and total carbon, while the leaves had the highest phosphorus concentrations. Dietary lipid intake is considered a promising avenue, particularly when ruminant diets are rich in polyunsaturated fatty acids. 15 Although these results showed significant total lipid contents, these were not significantly different between the different substrates. Similarly, the presence of high total nitrogen content in the samples supports the in vitro digestibility that was positively correlated with nitrogen content. According to the study of Selmi et al 16 the abundance of total lipids and total nitrogen can be beneficial and positive for digestibility. To meet animal needs, the efficiency of nitrogen and energy metabolism depends on the degree of microbial digestion. 17
Parietal constituents such as neutral detergent fiber, acid detergent fiber and acid detergent lignin are used by animal nutritionists as good indicators of feed intake (volume) and energy (digestibility). Allover results showed higher NDF values in fruits compared with leaves and flowers. Both parietal and non-parietal carbohydrates are digested by the rumen. 18 As a result, fibers can be a limiting factor in gas production.
Phytobiotic plants are attracting growing interest as potential sources of natural growth stimulants and bioactive molecules in ruminant feed. 19 These secondary metabolites, namely alkaloids, saponins, terpenoids, and phenolic compounds, 20 can also be altered by differences in atmospheric conditions, cultivation methods or even different soil properties. 8 In vitro culture of A. graveolens is advantageous thanks to the valuable therapeutic properties of its phenolic compounds including antioxidant, anti-inflammatory, immunostimulant and anticancer properties.21,22 Interestingly, the present results revealed valuable phenolic profile in the secondary metabolites of different samples, where the highest total polyphenol levels were found in flowers and fruits.
Edible tannins (water-soluble and condensed) are phenolic secondary compounds naturally present in plants. Their chemical composition and concentration depend on the plant species, plant parts, and environmental factors such as climate, soil, topography and phenological stage.23,24 Considered antinutritional and/or toxic compounds, the addition of hydrolyzable or condensed tannins to ruminant feed can prevent flatulence, reduce methanogenesis 25 and inhibit the growth of parasites such as nematodes. 26 They also protect lipids from biohydrogenation in plants.27,28 The results of present study indicated that the presence of tannins did not limit the in vitro fermentation of the various parts of A. graveolens. This is confirmed by several recent systematic studies demonstrating the positive role of tannins in animal nutrition and health. 25 For a better understanding of their beneficial and harmful effects, the use of tannin-rich plants should be recommended according to their composition, concentration, and origin. 29 Their role in nitrogen utilization is rarely negative, and sometimes positive. Tannins also bind to proteins (forming protein-tannin complexes), often protecting them from degradation in the rumen without limiting intestinal digestibility.
There is a clear link between the onset of some pathologies and the decrease in antioxidant status in animals. 30 Therefore, it is important to improve meat quality parameters by maintaining a good antioxidant status, improving vitamin E content and reducing lipid peroxidation.31,32 In addition, Jinesh et al 33 studied the antioxidant activity of various A. graveolens extracts and found a positive correlation with their phenolic content. Another study showed that A. graveolens extract had antioxidant activity comparable to that of DL-α-tocopherol and quercetin. 34 The findings of this study showed that fruit extracts had stronger antioxidant activity than leaf and flower extracts, and values were close to those given by the reference molecules, ascorbic acid and, BHT. According to the ferric-reducing power test, the flowers had the best ability to reduce Fe3+ to Fe2+. Yung-Shin et al 35 confirmed this finding, showing that flowers had higher antioxidant activity than leaves and fruits. Other studies confirmed that plant extracts such as rosemary 36 and officinal sage 37 improved the in vitro fermentation in ruminants thanks to their antioxidant potential.
According to the “Substrate + Inoculum” Association, “rumen biotransformation” is a concept that summarizes the phenomena of food decomposition and fermentation (degradation, synthesis, and transformation) by microorganisms within the rumen. It is explained by the degradation of proteins into peptides and NH3, parietal and non-parietal carbohydrates into soluble carbohydrates and volatile fatty acids (VFA), CO2, and CH4. 17 These phenomena are governed by three laws: chemical kinetics, thermodynamics and microbial population dynamics. The influence of different parts from A. graveolens on rumen fermentation is related to substrate composition, phenolic compound content, rumen degradation rate, and rumen residence time. 38 The present results showed that the amount of gas produced at 24 and 48 h after incubation was greater in goats than that in sheep. The rumen of goats is equipped with a microbial ecosystem capable of digesting fibers, notably entodiniomorph type b ciliate protozoa (Epidinium and Eudiplodinium) 39 that contain most of the enzymes involved in cellulolysis and play an active role in rumen food digestion.
The in vitro digestibility study determined the influence of different A. graveolens substrates on ruminal fermentation. The volume of gas produced after 24 and 48 h of incubation was significantly higher for flowers and leaves in sheep and goats. The fiber content of the fruit was the limiting factor in gas production kinetics. Flowers and leaves also showed the highest concentrations of organic matter digestibility, metabolic energy and volatile fatty acids in sheep and goats. Acetate, propionate, and butyrate are the main sources of energy for animals and are produced during the breakdown of carbohydrates. 40 Goats have the best results compared to sheep, as they are the main producers of metabolic energy and VFAs. In general, the various A. graveolens substrates had interesting nutritional and energy values that could improve in vitro digestibility in ruminants, but fruits were the best.
Methane is a powerful greenhouse gas produced during digestive fermentation and released into the atmosphere through belching. Thus, to limit global warming, its emission must be reduced. 41 On an environmental level, the craze for the use of biomolecules 42 present in plants is due to their potential as nutraceuticals and their advantages in reducing methane (CH4) emissions. 43 The illustrated results showed that sheep and goats had the lowest methane levels during leaf digestibility. According to an ethnopharmacological survey, 14 conducted in different regions of the Tunisian North-East, leaves were the most used part, according to the order's respondents (49.2%), which may be advantageous. The high level of condensed tannins in leaves can also contribute to methane reduction compared with other substrates. This data has been confirmed in vitro and in vivo where dietary tannins showed an effect on the methanogenesis through mitigation of methane (CH4) production. 44
A. graveolens also showed promising results as a natural organic feed additive in ruminant production, due to its positive effects on the nutrition and health of small ruminants. The analyzed plant substrates could play an active role in improving their antioxidant status. Therefore, improving the functioning of microbial digestion and mitigating the release of pollutants such as methane by animals will reduce their negative impact on the environment.
Limitations and Future Perspectives
The study of A. graveolens substrates in vitro on small ruminants is only a preliminary study, that need to be accomplished with the verification of its beneficial effects in vivo and the identification of rumen modifications. These studies can be done nutritionally and therapeutically in animals (ruminal fluid, blood, and urine). Thanks to artificial intelligence, scientific research can contribute to progress in livestock farming using connected tools capable of detecting health disorders in animals, such as the generation of alerts based on changes in animal health or changes in rumination activity. There is therefore an opportunity to collect and analyze not only ruminal fluid but also blood and urine to study the effects of phytobiotics in vivo and to determine their therapeutic effects on animals.
Conclusion
The present study highlighted the phytochemical and biochemical importance of the different substrates of A. graveolens. The quantity of phenolic compounds was different, with the highest levels were recorded in the flowers. AGFRAE showed the most significant antioxidant properties. Moreover, the various parts of this phytobiotic-rich plant represent a source of functional ingredients that could be beneficial to enhance digestibility and therefore used as a food additive for ruminants. However, A. graveolens substrates have to undergo more in vivo tests for their efficacy and safety on animals.
Materials and Methods
Chemicals and Reagents
Standards of phenolics and flavonoids, Folin–Ciocalteu reagent, vanillin, quercetin and DPPH (2,2-diphenyl-1-picrylhydrazyl) were purchased from Thermo Fisher (Germany). Hydrochloric acid, sulfuric acid, nitric acid, NaOH, and chloroform were purchased from Carlo Erba Reagents (Valde Reuil, France). KH2PO4, sodium lauryl sulfate, ammonium meta-vanadate, ethanol, and methanol were purchased from Prolabo (France).
Plant Material
Leaves, flowers, and fruit substrates of A. graveolens were collected at the Beja study site, supplied by organic farmers, in the wild near their wheat fields. The choice of this species was based on its abundance in the study region and its palatability to ruminants as it was appreciated for its strong odor and appetizing aroma. Sampling was carried out according to specific periods. Leaves appeared from April to May, and flowers and fruits from early June to September. After drying in ambient air and being protected from light, samples from each plant part were crushed and stored in vials until further analysis.
Animal Material
Rumen juice was obtained from donor animals (sheep and goats) from the Tabarka municipal slaughterhouse (Tunisia). It was directly stored in a thermos at 39 °C until it reached the laboratory to prevent alteration of the inoculum.
Physicochemical Analysis of A. graveolens
Dry, Mineral and Organic Matters
The determination of the dry, mineral, and organic matter (DM, MM and OM, respectively) contents of various A. graveolens samples was carried out by the Association of Official Agricultural Chemists (AOAC).
45
Dry matter was determined after drying fresh samples (leaves, flowers, and fruit) in an oven at 105 °C until a constant weight was reached. Mineral content was determined after calcining 1 g of each sample tested in a muffle furnace at 550 °C for 4 h. Organic matter and carbon contents were determined according to the following formulas, respectively: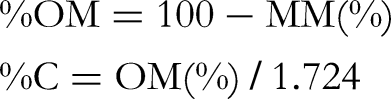
Total Quantity of Nitrogenous Matter
Total nitrogen was measured using the Kjeldahl method (AOAC). 45 This method comprises three stages: mineralization, distillation, and titration. In the presence of a catalyst (CuSO4), the sample is hot mineralized using sulfuric acid (density: 1.84 g/cm3, purity: 95-98%) and hydrogen peroxide. Organic nitrogen is converted to ammoniacal nitrogen (NH4+). This is transferred with 30 ml NaOH 50%, vaporized as ammonia (NH3), and then recovered in a boric acid solution (4%) containing two color indicators (methyl red and bromothymol blue). Finally, the homogenate was titrated with a solution of hydrochloric acid (0.1 N). The total amount of nitrogenous material was calculated by multiplying the amount of nitrogen by the factor 6.25. 46 All samples were analyzed in three technical replicates.
Total Phosphorus Level
The phosphorus content was determined according to the method by Olsen and Sommer. 47 After incineration, the ash obtained was treated with hydrochloric acid. The ultraviolet (UV)-visible spectrophotometer (DU 640 B; Beckman Coulter, Indianapolis, Indiana, USA) measured transmittance at 430 nm against a blank containing different concentrations of KH2PO4. Each sample was examined three times. All samples were analyzed in three technical replicates.
Total Lipid Content
Total lipid extraction was carried out using the method described by Bligh and Dyer
49
and modified by Cequier Sánchez et al
50
Total sample lipids (1 g of dry matter from ground plant samples) were fixed in boiling water for 10 min to inhibit phospholipases. The plant sample was crushed in a porcelain mortar and pestle. Chloroform-methanol was used to extract lipids in a 2/1 (v/v) ratio. The total quantity of solvent used was 15 ml of solvents/g of dry matter. The mixture was then decanted and the chloroform layer containing the total lipids was recovered and placed in a binder-ventilated oven at a temperature of 60 °C for 48 h. Following evaporation, the total lipid content (TL) was calculated using the following formula:
Study of Parietal Components
The determination of parietal constituents: neutral detergent fiber (NDF), acid detergent fiber (ADF) and acid detergent lignin (ADL), was carried out according to the method of Van Soest et al 48 using the semi-automatic FIBRESTEST RAYPA device.
The cell content was solubilized by the action of sodium lauryl sulfate detergent and pectic substances by the action of a chelating agent in a buffered medium, resulting in the formation of the cell wall (NDF). Theoretically, the fraction consists of cellulose, hemicelluloses and lignin. Sulfuric acid was used to determine the ADL content by treating the ADF fraction, where the true raw cellulose content (TRC) represented the difference between ADF and ADL contents. The amount of hemicellulose was calculated based on the difference between NDF and ADF. All samples were analyzed in three technical replicates.
Preparation of A. graveolens Extracts
A portion of plant powder (1 g) was macerated in 20 ml of distilled water for 24 h using a magnetizing system. The macerated extracts were filtered through Whatman filter paper and the extraction yield (EY %) was measured and then the extract was stored at 4 °C until use.
Secondary Metabolites Determination
Total polyphenol levels were determined using the Folin-Ciocalteu method of Singleton et al 51 500 µl of Folin-Ciocalteu reagent (10%) and 1 ml of an aqueous solution of sodium carbonate Na2CO3 (7.5%) were added to 500 µl of diluted extract, and the mixture was stirred rigorously. After incubation in the dark for 1 h, the absorbance was measured at 760 nm. Total phenolic content was expressed as mg gallic acid equivalents per g of dry matter (mg GAE/g DM).
The estimation of total flavonoids was determined according to the method of Yi et al 52 1 ml of the diluted aqueous extract was added to 1 ml of a 2% methanolic solution of aluminum chloride AlCl3 and the mixture was stirred rigorously. After incubation in the dark for 15 min, absorbance was measured at 430 nm. Total flavonoid content was expressed as mg quercetin equivalent per g dry matter (mg QE/g DM).
Condensed tannins levels were determined using the vanillin method in an acid medium, as described by Price et al 53 50 µl of diluted aqueous extract, 3 ml of vanillin solution (4%), and 1.5 ml of concentrated H2SO4 were mixed and stirred rigorously. After incubation in the dark for 15 min, the absorbance was measured at 550 nm. Condensed tannins (CT) content was expressed as mg catechin equivalent per g dry matter (mg CE/g DM).
Total tannins levels were determined using the method of Kujala et al 54 500 µl of Folin-Ciocalteu reagent (50%) were added to 0.5 ml of extract and 1 ml of Na2CO3 (20%). The absorbance was measured at 730 nm, and total tannin (TT) content was expressed as mg tannic acid equivalent per g of dry matter (mg TAE/g DM).
All samples were analyzed in three technical replicates.
Free Radical-Scavenging Activity on DPPH•
The antioxidant activity of A. graveolens extracts was assessed by the DPPH• radical scavenging test described by Ammar et al
55
1 ml of aqueous extract at various concentrations (10, 25, 50, 100, 150, 200, 250 µg/ml) was added to 1 ml of 0.06 mM DPPH methanolic solution (2.4 mg/100 ml). At the same time, a negative control was prepared by mixing 1 ml of distilled water with 1 ml of the DPPH solution. Ascorbic acid and BHT were used as reference molecules. After incubation, the violet coloration of the DPPH turns yellow, and this decoloration will be measured spectrophotometrically. The mixture was placed in the dark at room temperature for 30 min, and the optic density was determined at 517 nm. Inhibition (I) percentages were estimated according to the following equation:
The IC50, which correspond to the extract concentration required for the inhibition of 50% of the DPPH radical, was calculated from the graph plotted based on the percentage of DPPH inhibition as a function of the extract concentration.
Ferric Reducing Antioxidant Power (FRAP)
The antioxidant activity of the various A. graveolens extracts by the ferric reducing power test was carried out according to the method of Yildirim et al 56 A volume of 0.5 ml of each extract (ranging from 1 to 5 mg/ml) was mixed with 1.25 ml 0.2 M potassium phosphate buffer (pH 6.6) and 1.25 ml potassium ferricyanide solution (1%). This mixture was kept at 50 °C in a water bath for 30 min. Once cooled, 0.5 ml of 10% trichloroacetic acid was added and centrifuged at 1000 × g for 10 min whenever necessary. 1.25 ml of the supernatant was then mixed with 1 ml of distilled water and 0.25 ml of freshly prepared ferric chloride (0.1%). After 10 min of incubation, the absorbance of the resulting solutions was measured at 700 nm using a spectrophotometer (Jenway, Stone, UK). The presence of antioxidants in A. graveolens extracts would, in the reducing power test, cause the reduction of Fe3+ to Fe2+, which can be regulated by measuring the formation of Perl Prussian blue (Fe4[Fe(CN)6]3) at 700 nm. All samples were analyzed in three technical replicates.
In Vitro Fermentation of Substrates
Ruminal fermentation of A. graveolens extracts was studied using the in vitro gas production technique based on the method of Menke et al 57 This method consists of incubating a substrate in the presence of ruminal inoculum and a buffer for a defined period under anaerobic condition at (39 °C). The inoculum (rumen juice) of goats and sheep was collected from the municipal slaughterhouse in Tabarka (Tunisia). They were used following the local ethics committee of Tunis University on the use and care of animals and the NIH recommendation. The protocol was approved by the ‘Comited’Ethique Bio-medicale (CEBM)’ (JORT472001) of the ‘Institut Pasteur de Tunis’. In the laboratory, the inoculum was homogenized and filtered through 4 layers of surgical gauze. The fermentation medium was obtained by mixing in each syringe: 10 ml filtered rumen juice, 20 ml artificial saliva, and 300 mg powdered A. graveolens leaves, flowers, or fruits (3 replicates per sample). Syringes were placed in a water bath at 39 °C. Following incubation, the gas volume was read every two hours, and at the end 10 ml of NaOH were injected into each syringe, absorbing the CO2 and causing the piston to retract. The difference between the total volume of gas produced and CO2 volume represented the amount of methane produced. 58
Parameters characteristic of gas production were deduced from the exponential model proposed by Orskov and Mcdonald.
59
The digestibility of organic matter (OMD) was measured according to the formula proposed by Menke et al,
57
while metabolizable energy (ME) and total volatile fatty acids (VFA) were determined using the method of Makkar.
60
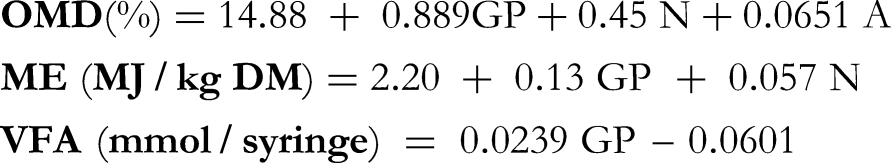
GP: volume of gas produced (ml/300 mg DM) at 24 h of incubation
N: total nitrogenous matter (g/100 g DM)
A: mineral matter (g/100 g DM)
The metabolizable energy was expressed in (kcal/kg DM) where 1 MJ = 239 kcal.
All samples were analyzed in three technical replicates.
Feed Value Prediction
The determination of the energy and nitrogen values of the various A. graveolens extracts, expressed respectively in Feed Unit for Lactation UFL (g/kg DM) and PDI (g/kg DM), was predicted by the equations adopted by Sauvant
61
:
PDIN is protein digested in the small intestine when rumen-fermentable nitrogen is limiting and PDIE when rumen-fermentable energy is limiting
TRC: true raw cellulose
TL: total lipids
All samples were analyzed in three technical replicates.
Statistical Analysis
Data were analyzed by a Post Hoc Tukey analysis of variance (Anova One Way) using IBM SPSS Statistics for Windows, version 25 (IBM Corp., Armonk, N.Y., USA) and expressed as means ± standard deviation of the mean (SD). All statistical tests were two-tailed, and a P value of 0.05 or less was considered significant.
Measured parameters were subjected to an analysis of variance using the GLM procedure in SAS software (1989), and compared using Duncan's multiple rank tests (1955).
The model equation to compare the metabolizable energy, the concentration of the volatile fatty acids, and the digestibility of the organic matter according to the species is given as follows:
Yi: measured parameter, μ: overall mean, Ai: effect of the ith species, Eij: residual error.
Parameters characteristic of gas production kinetics are predicted according to the model of Orskov and Mc Donald (1979).
The model equation:
Y: volume of gas produced after each incubation time (ml)
a: gas production from the readily fermentable soluble fraction (ml)
b: gas production from the potentially fermentable insoluble fraction (ml)
c: gas production rate (h−1)
t: incubation time (h)
Footnotes
Acknowledgements
The financial support of the Tunisian Ministry of Higher Education is appreciatively acknowledged.
Author Contribution Statement
C.B., A.D., H.S., and S.J. performed the phytochemical composition and in vitro digestibility experiments as well as statistical analysis. C.B., S.J., and H.S. provided reagents and materials (eg, fermentation syringes) and performed the in vitro digestibility analysis and statistical analysis experiments. C.B. and H.S. participated in data processing and experimental planning. The final draft was read and approved by all authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
All maintenance and sacrifice procedures were used following the local ethics committee of Tunis University regarding the use and care of animals and the NIH recommendation. The protocol was approved by the ‘Comité d'Ethique Bio-medicale (CEBM)’ (JORT472001) of the ‘Institut Pasteur de Tunis.’
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
