Abstract
Objective
This study aimed to evaluate the cytotoxic potential of schisantherin A, a bioactive lignan isolated from Schisandra chinensis (Turcz.) Baill., against human liver cancer cell lines through both in vitro and in silico approaches.
Methods
The cytotoxic activity of schisantherin A was assessed in vitro using three human liver cancer cell lines as HepG2, Hep3B, and Huh7. In silico studies involved molecular docking to evaluate the binding affinity of schisantherin A to two key endoplasmic reticulum stress-related proteins IRE1α and PERK. Molecular dynamics simulations were subsequently performed to assess the stability and interaction profile of the ligand-protein complex.
Results
Schisantherin A exhibited notable cytotoxicity against the tested liver cancer cells, with IC50 values of 6.65 µM (HepG2), 10.50 µM (Hep3B), and 10.72 µM (Huh7). Molecular docking predicted favorable binding to IRE1α and PERK, with docking scores of −7.6 kcal/mol and −5.5 kcal/mol, respectively. MD simulations revealed a stable binding conformation of schisantherin A within the IRE1α binding pocket, mediated by multiple non-covalent interactions involving key amino acid residues.
Conclusion
The in vitro and in silico findings suggest that schisantherin A may exert its cytotoxic effects via targeting IRE1α-mediated pathways, supporting its potential as a lead compound for liver cancer therapy.
Introduction
Schisandra chinensis (Turcz.) Baill. is a medicinal herb and edible plant belonging to the Schisandraceae family. 1 It is also known as “wu-wei-zi”, meaning “five-flavor fruit”, because its berries contain the five basic tastes bitter, sweet, salty, sour, and spicy. 2 It is a dioecious climbing plant with morphological characteristics including heights of 0.5 to 25 m.1,2 S. chinensis is found in the natural habitats of Korea, North-Eastern China, Japan, and Russia, where the plant thrives in conditions of moderate humidity and light. In traditional medicine, the deep red S. chinensis berries have been utilized in the treatment of gastrointestinal ailments, sleep deprivation, kidney infections, asthma, and coughing. Furthermore, they exhibit several biological effects, including antimicrobial, antivirus, anticancer, anti-obesity, antidiabetic, and anti-inflammatory actions. 3 The fruits of S. chinensis are mainly known for their hepatoprotective activity; in traditional Chinese medicine, liver- and kidney-related health conditions have been treated using S. chinensis as a tonic. Recent research has increasingly suggested that extracts from S. chinensis can protect liver cells from damage and inflammation and increase their abilities to detoxify substances, promote tissue regeneration, and reduce the progression of fibrosis.1,2 Due to these beneficial properties, S. chinensis represents a viable herbal remedy meriting further study in terms of its long-term therapeutic effects on the liver. 1
Previously, a wide range of bioactive compounds have been identified in S. chinensis, mainly including lignans, flavonoids, triterpenes, polysaccharides, phenolic acids, and essential oils. 2 Dibenzocyclooctadiene lignans are the most abundant group in the fruits of S. chinensis1,2; among these, schisantherin A (or gomisin C) is a key lignan exhibiting hepatoprotective bioactivity via an antioxidant mechanism and by stimulating regeneration. 4 Schisantherin A, with the molecular formula of C30H32O9 and a molecular weight of 536.6 g/mol, is a complex compound comprising a hydroxyl group and multiple methoxy groups attached to a lignan skeleton. 5 It exhibits significant lipophilicity, indicated by a partition coefficient (LogP) of approximately 5, as computed by XLogP3 3.0 (PubChem release 2021.10.14). This property facilitates bioavailability and cellular membrane permeability. 6 Dibenzocyclooctadiene lignans derived from the Schisandra genus are, in general, capable of triggering programmed cell death (apoptosis) and possess hepatoprotective properties. 2
Hepatocellular carcinoma (HCC) is a primary liver tumor that often develops against a backdrop of chronic liver disease and cirrhosis. A critical factor in the progression from cirrhosis to HCC is the role of the hepatic stellate cell; this is activated during liver injury and differentiates into a contractile myofibroblast-like cell, contributing to the deposition of extracellular matrix proteins. 7 Given the pivotal role of activated stellate cells in the onset and progression of HCC, targeting their activation has emerged as a potential therapeutic strategy for patients diagnosed with this cancer. One approach to inhibit stellate cell activation involves blocking the inositol-requiring transmembrane kinase/endoribonuclease 1α (IRE1α) pathway of the unfolded protein response (UPR). 7 In addition to inositol-requiring enzyme 1 (IRE1), protein kinase R-like endoplasmic reticulum kinase (PERK) is a key transmembrane sensor of the UPR activated by endoplasmic reticulum (ER) stress. PERK plays a significant role in human UPR-related diseases, including tumorigenesis and neurodegenerative disorders. Studies have shown that it promotes tumor growth, metastasis, autophagy, and resistance to radiation therapy, making it a promising target for future therapeutic interventions in addressing treatment failures. 8 Previous studies have shown that schisantherin A is a potential candidate for the therapeutic treatment against liver cancer.9,10 Thus, the present study hypothesizes the anticancer effects of schisantherin A and aims to evaluate the cytotoxic activity of schisantherin A against liver cancer and use models to predict its interaction with specific proteins involved in the pathway of programmed cell death, including IRE1 and PERK.
Materials and Methods
Plant Materials and Schisantherin A Isolation
Schisandra chinensis fruit samples were purchased from Hai Thuong Lan Ong, Hochiminh city in September 2022, and identified by MH Tran, PhD. from School of Medicine and Pharmacy. The identified sample has the code TMH-2022-NVTN-001 and is stored in the Laboratory of Pharmacognosy and Drug Quality Control, School of Medicine and Pharmacy, The University of Danang. Isolation of schisantherin A: see supporting information.
Cancer Cells Cultures and In Vitro Proliferation Activity Assay
The human liver cancer HepG2, Hep3B, Huh7 and human embryonic kidney HEK293 cell lines were generously provided by Prof. Jeong-Hyung Lee from Kangwon National University, Korea. The cell lines were cultured at 37 °C in Dulbecco's Modified Eagle Medium (DMEM), supplemented with 10% Fetal Bovine Serum (FBS), 100 U/mL penicillin, and 100 mcg/mL streptomycin. The cells were maintained in a 5% CO2 incubator for 48 h. The antiproliferative activity of the compounds was assessed on cancer cell lines using a slightly modified Dojindo kit (Dojindo Molecular Technologies, Inc., Rockville, MD, USA). 11
Selected Ligand and Target Proteins
The molecular docking was implemented between schisantherin A and the two protein targets, which were taken from RCSB Protein Data Bank (https://www.rcsb.org/) with the identification code as follow IRE1α (ID: 6XDB) and PERK (ID: 4X7H).
ADMET Predictions
The druggability of the candidates was also evaluated, based on ‘Lipinski's rule of five’10. Particularly, SwissADME tool was used to calculate the physicochemical properties of schisantherin A. Besides, DL-AOT prediction server was applied to estimate acute oral toxicity of the target chemical compound.12,13
Molecular Dynamic Simulation
To support the docking interaction, molecular dynamics simulation (MD) of the compound and the targeted proteins was performed, based on Newton's law of motion, which estimates more precisely the protein-ligand recognition and binding due to changes in conformation and geometrical interactions in time-dependent manner. The estimation simulates an in vivo-like environment via force-field based model. In this study, MD simulation (by Schrodinger Inc.) was estimated between schisantherin A and the two protein targets involved in the apoptosis pathway IRE1α and PERK.14,15
Results
In Vitro Cytotoxic Activity
First, the cytotoxic activity of schisantherin A against human liver cancer cell lines as HepG2, Hep3B, and Huh7, and human embryonic kidney HEK293 cells was tested. The cells were cultured for 48 h and exposed to various concentrations of schisantherin A (1-30 µM). A dose-dependent trend was observed in the viability of liver cancer cells in the treated group when compared with vehicle-treated cells. Schisantherin A displays the most potent cytotoxicity against HepG2, with an IC50 of 6.65 ± 0.32 µM, and also exhibits cytotoxic effects against both Hep3B and Huh7 cells, with IC50 values of 10.50 ± 0.81 and 10.72 ± 0.46 µM, respectively. Camptothecin, a positive control, shows good cytotoxic activity against HepG2, Hep3B, and Huh7 cells, with IC50 values of 1.5 ± 0.08, 2.5 ± 0.17, and 1.2 ± 0.10 µM, respectively. In addition, schisantherin A exhibits no cytotoxic effect on human embryonic kidney cell lines (Table 1).
Cytotoxic Activity of Schisantherin A and Camptothecin.

The experiments were conducted in triplicate; the cytotoxicity effect is expressed as the mean ± standard deviation (SD) of three replicates.
Positive anticancer reference.
(–) no test; >30 µM implies weak activity.
In Silico Results
Molecular Docking Simulations
Among the target proteins, IRE1α demonstrates a relatively stable binding conformation with schisantherin A (Figure 1A), achieving a docking score of −7.57 kcal/mol (Table 2). Conventional hydrogen bonds (green dashed lines) indicate interactions between schisantherin A and specific residues, and pi–cation and pi–anion bonds (orange lines) represent interactions with charged residues. Hydrophobic interactions (pi–alkyl; pink/violet lines) illustrate nonpolar contacts between the ligand and amino acid side chains of, for example, LEU A577 and ALA581. Key residues including HIS692, LYS599, and ASP711 play significant roles in stabilizing the ligand via hydrogen bonding and electrostatic interactions, while hydrophobic interactions with residues LEU577 and VAL586 contribute to the overall binding affinity of schisantherin A.
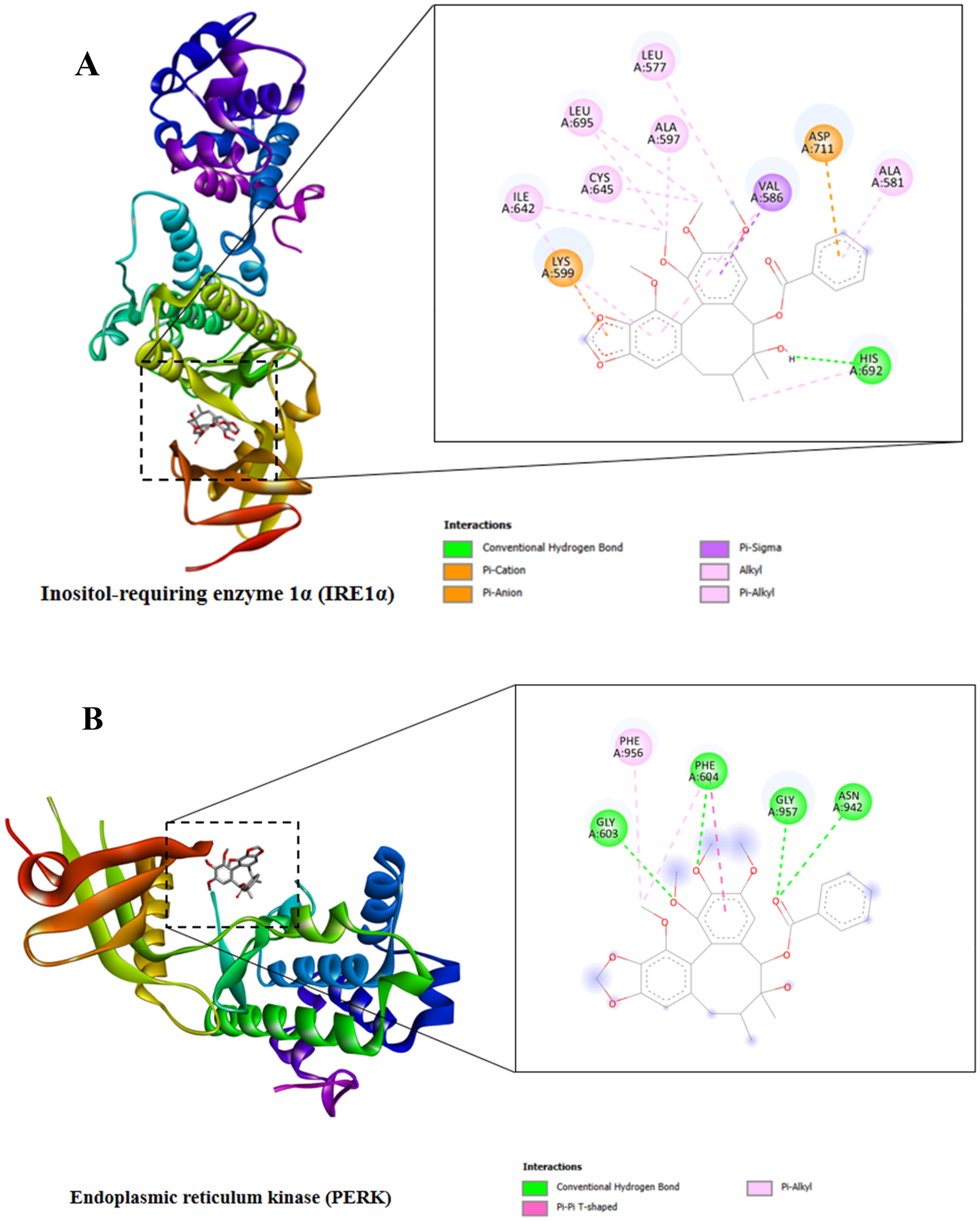
Interactions Between Schisantherin A and IRE1α (A) and PERK (B).
Interactions and Binding Affinity of Schisantherin A.

In this study, schisantherin A exhibits a weak docking stability with PERK (−5.50 kcal/mol; Table 2). Schisantherin A demonstrates direct conventional hydrogen bonds (green dashed lines) with amino acids GLY603, GLY957, PHE604, and ASN942, contributing to the stability and specificity of ligand binding (Figure 1B). Additionally, π-π T-shaped interactions (pink dashed lines) between the aromatic rings of schisantherin A and phenyl groups of PHE956 were noted, indicating their significance in stabilizing the ligand within the binding pocket.
ADMET Predictions
To examine the potential for schisantherin A and protein interactions, the physicochemical properties, bioactivity, and pharmacokinetics were evaluated using ADMET predictions (Table 3). Schisantherin A violates only one rule as the molecular weight exceeds 500 Da; other than this, with regard to polarity, its topological polar surface area (TPSA) is 101.91 Ų, falling within the desirable range of 20 to 130 Ų. This specific factor is indicative of the ability to cross biological barriers such as brain access and absorption. Regarding solubility, the LogS value (calculated via the ESOL model) does not exceed 6, and in terms of flexibility, the number of rotatable bonds does not exceed 9. The molar refractivity (MR) measures the total polarizability of one mole of a substance and should fall in the range of 40 to 130—the result for schisantherin A (142.96) is higher than the upper limit of the desired range.
Physicochemical Properties, Lipophilicity, and Water Solubility of Schisantherin A, Analyzed Using SwissADME.

MW: molecular weight; LogP: log of octanol/water partition coefficient; nHBD: number of hydrogen bond donors; nHBA: number of hydrogen bond acceptors; TPSA: topological polar surface area; MR: molar refractivity; LogS: log of solubility; nRotB: number of rotatable bonds.
In terms of pharmacokinetics, schisantherin A possesses a low skin permeability coefficient (LogKp), indicating the likelihood that it is not skin permeable. The BOILED-Egg model was utilized to evaluate gastrointestinal absorption and blood–brain barrier (BBB) permeability (Table 4). While the absorption of schisantherin A is high, it is unable to pass the BBB. The data of the permeability glycoprotein (P-gp) substrate shows that schisantherin A is a substrate capable of passing through biological membranes. Among the five major isoforms of cytochrome P450 (CYP), schisantherin A exhibits inhibition against CYP2C9, CYP2D6, and CYP3A4. This inhibition can be considered the cause of toxicity or any undesirable adverse effects due to the accumulation of drug molecules or their metabolites. 13 Regarding the oral toxicity evaluation via the DL-AOT platform, schisantherin A was classified as falling within the yellow “caution” level of toxicity. The prediction of the regression consensus model yielded an LD50 value of 2.92 mg/kg, indicating the concentration of schisantherin A lethal to 50% of the tested animals of a particular species (Table 5).
Pharmacokinetic Predictions of Schisantherin A Computed via SwissADME.

Toxicity Predictions Using DL-AOT.

Molecular Dynamics Simulations of Schisantherin A with IRE1α
Ligand–Protein Analysis
As mentioned above, molecular docking analysis was conducted on two proteins able to induce apoptosis. However, only IRE1α was chosen to further evaluate the molecular dynamics (MD) properties because PERK yielded a lower docking score, indicative of possible weak protein–ligand interactions. The MD simulation results provide insights into the stability of the interaction between schisantherin A and the IRE1α protein over 100 ns (Figure 2). The root mean square deviation (RMSD) analysis illustrates that the Cα backbone of IRE1α fluctuates consistently within the range of around 2.0 to 3.2 Å throughout the simulation, indicative of a stable protein structure under binding conditions (Figure 2A). This stability suggests that the protein's conformation does not undergo significant changes upon the binding of schisantherin A. Meanwhile, the RMSD of the complex (red) remains relatively stable in the early phase of the simulation, suggesting that the ligand maintains a steady position within the binding pocket. However, after 70 ns, the RMSD values show a notable increase, reaching a peak toward the end of the simulation. This late-stage fluctuation may suggest conformational adjustments as schisantherin A repositions itself within the binding site for optimal interactions, hinting at a potentially complex binding process requiring a longer simulation time to reach a stable equilibrium. The behavior observed in the RMSD plots points, overall, to a stable interaction, with minor adjustments by the ligand that might indicate flexibility within the binding site.

RMSD (A) and RMSF (B) Analysis.
Root mean square fluctuation (RMSF) analysis was performed to understand the flexibility of specific residues in IRE1α during the binding of schisantherin A. Notably, the peaks around certain residue indices indicate regions of the protein that exhibit higher flexibility. These peaks could correspond to loop regions or peripheral parts of the protein that are more prone to movement when accommodating the ligand (Figure 2B). The data suggest that, despite these localized flexibilities, the core structure of IRE1α remains stable, as reflected by consistently low RMSF values across most of the residue indices. This stability supports the RMSD analysis and implies that the protein maintains its structural integrity while forming interactions with schisantherin A.
Protein–Ligand Contact, Histogram, and Timeline Analysis
Figure 3 presents the 2D interaction analysis between schisantherin A and IRE1α during the 100 ns simulation. Among the key interactions, Tyr753 interacts with schisantherin A, contributing approximately 22% to the overall contact profile (Figure 3A). This involvement indicates a strong hydrophobic or pi-stacking interaction that enhances the binding affinity. His723 and Arg820 contribute around 13% and 10%, respectively, with Arg820 showing the potential for water-mediated interactions, indicating flexibility in accommodating schisantherin A within the binding pocket. Gln818 and other residues are also involved in maintaining hydrogen bonds and bridging interactions that support ligand stability. These findings suggest that schisantherin A engages in a diverse set of hydrophobic, hydrogen bonding, and water-mediated interactions that collectively stabilize its position within the IRE1α binding site. The prominent role of Tyr753 in forming hydrophobic and pi-stacking interactions may be critical for the compound's potential efficacy in modulating the activity of IRE1α.

Protein–Ligand Contact (A), Histogram (B), and Timeline (C) Analysis.
Figure 3B presents a comprehensive view of the types of interactions between schisantherin A and IRE1α. Tyr753 prominently contributes via hydrophobic interactions and plays a crucial role in anchoring schisantherin A within the binding pocket. Arg820 and His723 contribute to hydrogen bonding and water-mediated interactions, providing additional stability through polar contacts. Gln818 supports binding via water bridges, indicating the importance of solvent interactions in maintaining the ligand's positioning. Phe962 demonstrates a combination of hydrophobic and water-mediated interactions, adding to the complexity and stability of the binding network. The diversity of interactions—comprising hydrophobic, hydrogen bonding, and water-mediated forms—demonstrates the compound's versatile binding mechanism, supporting the hypothesis of its potential efficacy in cancer pathway modulation.
The contact timeline between schisantherin A and specific IRE1α residues is depicted in Figure 3C. The top graph shows that the overall number of contacts between schisantherin A and IRE1α fluctuates within a stable range over most of the simulation, indicating consistent ligand–protein interactions. Peaks in the total contact count suggest moments of enhanced binding stability, while dips might reflect minor conformational adjustments of the ligand. The discussion that follows highlights the contribution of individual residues to the binding over time. Residues such as Tyr753, Arg820, and His723 appear to maintain frequent contact with schisantherin A, as indicated by their sustained presence across the timeline. The red color shows where multiple contacts are consistently maintained, reinforcing their importance in the binding stability. Certain residues, including Gln818 and Phe962, exhibit intermittent contacts, reflecting adaptable interactions that may support ligand positioning and flexibility within the binding site. The timeline analysis complements the previously discussed RMSD and RMSF results, confirming persistent and versatile interactions between schisantherin A and IRE1α. The presence of both stable and dynamic contacts suggests that the ligand can maintain strong binding while adapting to slight structural shifts in the protein.
Ligand Flexibility Analysis
The RMSF plot for schisantherin A provides insights into the flexibility of individual atoms within the ligand while interacting with IRE1α (see Supporting information Figure S1A). The RMSF values indicate the extent to which different parts of the ligand fluctuate over the 100 ns simulation and range between approximately 10 and 12 Å, suggesting a relatively consistent level of flexibility throughout the ligand structure. This degree of fluctuation indicates that certain segments of schisantherin A adjust dynamically within the binding pocket, possibly to maintain optimal contact with the protein. Further, the fluctuations may reflect conformational flexibility that aids in accommodating structural shifts in IRE1α and stabilizing interactions. Regarding the ligand property, RMSD analysis (Figure S1B) showed stable values below 0.8 Å, confirming that schisantherin A maintains a steady conformation throughout the simulation. Similarly, the radius of gyration (rGyr) fluctuated minimally (3.92-4.08 Å), reflecting the ligand's compact and structurally coherent state. No intramolecular hydrogen bonds (intraHB) were detected, suggesting the ligand's conformation is primarily stabilized by interactions with IRE1α and the surrounding solvent. The molecular surface area (MolSA) remained stable (405-450 Ų), while the solvent-accessible surface area (SASA) exhibited moderate increases between 60-100 ns (200-600 Ų), indicating potential conformational changes that enhanced solvent exposure. The polar surface area (PSA) was consistently maintained between 75-105 Ų, supporting sustained polar interactions with the protein and solvent. These findings suggest that schisantherin A exhibits high conformational stability, adaptable flexibility, and favorable binding characteristics when complexed with IRE1α.
IRE1α Secondary Structure Elements and Timeline Analysis
The secondary structure profile of IRE1α during the 100 ns simulation is illustrated in Figure S2. The histogram (see Supporting Information Figure S2A) shows stable distributions of α-helices (blue) and β-strands (red) across key residue segments, indicating well-preserved secondary structural elements. These results suggest that binding of schisantherin A does not disrupt the native fold of IRE1α. In addition, the SSE timeline (Figure S2B) further confirms this structural stability. The top plot indicates a consistent ∼45% secondary structure content over time, while the bottom panel displays stable α-helical and β-strand regions throughout the simulation. The persistence of these elements highlights that IRE1α maintains its structural integrity upon ligand binding, with no significant conformational rearrangements observed.
Ligand Torsion Analysis
Torsional analysis of schisantherin A (see Supporting Information Figure S3) revealed key rotational bonds that enable conformational adjustments during binding with IRE1α. These flexible torsion sites, highlighted in various colors, facilitate optimal ligand accommodation within the dynamic protein binding pocket. Such flexibility likely contributes to the formation and stabilization of hydrogen bonds, hydrophobic contacts, and other noncovalent interactions essential for binding affinity. Dihedral angle distributions show predominant peaks in the histograms and circular plots, indicating stable conformational preferences at specific bonds. Notably, torsion angles highlighted in light blue and red exhibit narrow, defined ranges, suggesting favored conformations supporting tight binding. In contrast, broader angle distributions in plots such as orange and green indicate greater conformational freedom, which may be critical for adapting to local fluctuations in the binding site. These findings underscore the structural plasticity of schisantherin A and its ability to maintain stable interactions with IRE1α throughout the simulation.
Discussion
According to the International Agency for Research on Cancer (IARC), liver cancer ranked sixth in global incidence in 2022 but remained among the top three causes of cancer-related mortality, following lung and breast cancers. 16 Hepatocellular carcinoma (HCC), the predominant subtype, involves a complex pathogenesis characterized by genetic and epigenetic alterations, chromosomal instability, oncogene activation, and tumor suppressor gene inactivation. 17 Contributing factors such as oxidative stress, hypoxia, and chronic inflammation—especially from persistent hepatitis B or C infection—further accelerate malignant transformation. One of the key hallmarks of HCC is endoplasmic reticulum (ER) stress, which activates the unfolded protein response (UPR) through three major transducers: inositol-requiring enzyme 1 alpha (IRE1α), protein kinase R (PKR)-like ER kinase (PERK), and activating transcription factor 6 (ATF6). 14 Depending on the degree of ER stress, these pathways either restore cellular homeostasis or initiate apoptosis. Among them, IRE1α and PERK have emerged as promising therapeutic targets for HCC due to their dual roles in promoting survival or triggering apoptosis under stress conditions.18,19
Lignans from Schisandra chinensis, such as schisandrin A, schisandrin B, and gomisin A, have previously been reported to exert anticancer effects by inducing apoptosis, modulating cell cycle progression, and enhancing antioxidant capacity. 20 Schisantherin A, a structurally related dibenzocyclooctadiene lignan, has been relatively less studied. Prior work suggests that it suppresses HCC cell proliferation and migration by regulating glycolysis and arresting the G0/G1 phase. Moreover, it has been shown to increase the oral bioavailability of lenvatinib, a first-line HCC treatment, by inhibiting intestinal P-glycoprotein, thereby supporting its pharmacological relevance. 21 In the present study, schisantherin A exhibited dose-dependent cytotoxicity against three liver cancer cell lines: HepG2 (IC50 = 6.65 ± 0.32 µM), Hep3B, and Huh7 (both ∼10.5 µM). These IC50 values fall within the range considered moderate to promising in natural product-based screening studies. Notably, the compound showed no cytotoxicity toward the noncancerous HEK293 cell line up to 30 µM. Based on these data, the calculated selectivity index (SI)—defined as the ratio of IC50 in normal cells to cancer cells—was >4 for HepG2 and >2 for Hep3B and Huh7. These values reflect a moderate to favorable degree of selectivity, suggesting that schisantherin A may preferentially target tumor cells while sparing normal tissues. Although HEK293 cells are not liver-derived, they are widely accepted as a general noncancerous model in early cytotoxicity screening due to their stable growth and human origin. Future investigations should include non-tumorigenic hepatocyte-derived cell lines (eg, THLE-2 or LO2) to better assess hepatic tissue-specific selectivity. To gain mechanistic insights, molecular docking revealed that schisantherin A binds favorably to the ER stress-related proteins IRE1α and PERK, with binding affinities of −7.66 kcal/mol and −5.50 kcal/mol, respectively. A 100 ns molecular dynamics (MD) simulation of the schisantherin A–IRE1α complex confirmed the stability of this interaction, with key residues such as Tyr753, Arg820, and His723 contributing to hydrogen bonding and hydrophobic stabilization. These findings suggest that schisantherin A may modulate IRE1α-mediated apoptotic signaling; however, further validation at the protein level is necessary to confirm this hypothesis.
In addition, camptothecin was used as a positive control to benchmark anticancer efficacy. However, its cytotoxicity against HEK293 cells was not evaluated, and therefore a Selectivity Index for camptothecin could not be computed. This limits direct comparison of therapeutic selectivity between schisantherin A and the reference drug. We acknowledge this as a study limitation and recommend that future investigations include both tumor and non-tumor cell lines for all reference and test compounds to accurately determine SI values. Additional limitations of this study include the absence of experimental data validating in silico findings. Although docking and MD simulations suggest strong binding to IRE1α, functional confirmation via Western blot, qPCR, or proteomics is needed to verify downstream pathway modulation. Furthermore, a 100 ns simulation may not fully capture long-term conformational changes or rare binding events extended simulations could provide more comprehensive insights. Lastly, this study focused only on IRE1α and PERK, broader target profiling including other apoptotic markers will be required to clarify the compound's multifaceted effects in HCC.
Conclusion
In conclusion, our study demonstrated that schisantherin A, derived from Schisandra chinensis, exhibits cytotoxic effects against hepatocellular carcinoma cell lines HepG2, Hep3B, and Huh7, with IC50 values of 6.65 ± 0.32, 10.50 ± 0.81, and 10.72 ± 0.46 µM, respectively. Molecular docking analysis revealed that schisantherin A binds to key apoptotic regulators, IRE1α and PERK, with binding affinities of −7.66 and −5.50 kcal/mol, respectively. Molecular docking and dynamics simulations revealed strong and stable binding of schisantherin A to the ER stress regulator IRE1α, suggesting a potential mechanism involving UPR pathway modulation. These findings may support the potential of schisantherin A as a promising lead compound for liver cancer therapy. Further studies are warranted to validate its molecular mechanism and therapeutic efficacy through in vitro protein expression analyses and in vivo evaluations.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251383035 - Supplemental material for Cytotoxic Activity of Schisantherin A Against Liver Cancer Cells: In Vitro and In Silico Insights
Supplemental material, sj-docx-1-npx-10.1177_1934578X251383035 for Cytotoxic Activity of Schisantherin A Against Liver Cancer Cells: In Vitro and In Silico Insights by Viet An Thi Nguyen, Thuy Mi Pham Lam, Phu Tran Vinh Pham, Hieu Phu Chi Truong, Tan Khanh Nguyen, Hai Dang Nguyen and Manh Hung Tran in Natural Product Communications
Footnotes
Acknowledgements
MHT and the research group are grateful for the technical supported by School of Medicine and Pharmacy, The University of Danang.
Ethical Approval Statement
This study did not require ethical approval as it did not involve human participants and animal studies.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Authors Contributions
VATN and TMPL and MHT designed the study; VATN and TMPL carried out the experiments; PTVP, HPCT, TKN conducted experiments; TKN analyzed the data; HDN and MHT revised the manuscript. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
