Abstract
Aquilaria crassna Pierre ex Lecomte, Thymelaeaceae, is cultivated for producing resinous heartwood, also called agarwood. Its leaves are a source of herbal tea in Vietnam due to its rich content of polyphenols. However, the α-glucosidase inhibition activity and the contents of phenolic compounds in leaves of different ages have not yet been determined. In the current study, 7 polyphenols [iriflophenone 3,5-C-β-D-diglucoside (
Introduction
Aquilaria is one of the most widespread genera of the Thymelaeaceae family, which is native to Southeast Asian countries such as Cambodia, China, Malaysia, Indonesia, Laos, Thailand, and Vietnam. Three species—Aquilaria crassna, Aquilaria malaccensis, and Aquilaria sinensis—are primarily being cultivated on thousands of hectares to produce agarwood, which comes from the resinous heartwood. 1 In 1993, the Rainforest Project Foundation began to support Vietnamese farmers in cultivating Aquilaria plants in several provinces in southern Vietnam. That project supported plantations of more than one hectare; later the scheme was expanding into central Vietnam. The primary purpose of the planting was to produce the precious commercial product (used in perfumes and incense) known as agarwood, which is almost extinct, from 5- to 10-year-old plants. Aquilaria crassna has become the most cultivated plant in several farms in southern Vietnam, including in the provinces of An Giang, Kien Giang, and Binh Phuoc, alongside 2 other species—A. banaensae, and A baillonii. 1 However, inoculation methods are not always successful from field to field. Some farmers have started to cut down the trees following their failure to produce agarwood. However, in other places, the farmers have changed their cultivation techniques in order to grow this plant to yield leaves by trimming the tree to keep it small, similar to a tea tree.4,5
Apart from agarwood, the leaves of Aquilaria have traditionally been used as a herbal tea for laxative, anti-inflammatory, sedative, and pain-relieving purposes. 6 It has been widely reported that Aquilaria leaves are a very rich source of polyphenols, such as xanthones, flavonoids, and benzophenones. Mangiferin, iriflophenone 3-C-β-D-glucoside, iriflophenone 2-O-α-rhamnoside, genkwanin 5-O-β-primeveroside, genkwanin 4′-methyl ether 5-O-β-primeveroside, and genkwanin are the major components of A agallocha, A sinensis, and A crassna.2,3 Recently, A crassna was reported to have activity on lipopolysaccharide-induced tumor necrosis factor-alpha production by human polymorphonuclear cells and the cell-signaling response. 6 The water extract of A crassna has been shown to exhibit antipyretic, analgesic, and anti-oxidative activities.6,8 Iriflophenone 2-O-α-L-rhamnoside and iriflophenone 3-C-β-D-glucoside have had a positive effect on reducing blood glucose levels. Genkwanin 5-O-β-D-primeverosid and mangiferin have been proven to have laxative effects. In addition, genkwanin has been shown to have antibacterial and anti-inflammatory properties, in addition to anticancer activity against HT-29 and SW-480 human colorectal cancer lines in vitro, and on APC Min /+ mice in vivo.6,8,9
Several HPLC methods have been studied to determine the content of phenolic compounds in the leaves of A sinensis and A crassna. 10 In our preliminary screening of the Vietnamese natural and medicinal plants against diabetes, A crassna showed an inhibitory effect on α-glucosidase activity. Our preliminary study on the polyphenol content of young and old leaves also found that the content was much higher in the young leaves compared to the old leaves. Consequently, it became necessary to understand the dynamic changes in the significant phenolic compounds in the different ages of Aquilaria leaves. Hence, in this study, we isolated the major phenolic compounds as markers for establishing a quantification method to determine the major phenolic compound content in different-aged leaves of A crassna. This work provides valuable information that is essential for generating standardized materials for in vivo and in vitro studies, and for the authentication of the species as a food product.
Materials and Methods
Plant Materials and Chemicals
Branches of A crassna were collected from the Evergreen Forest Co. Ltd in Dong Phu District, Binh Phuoc Province, Vietnam (11°37′42.2″N 106°59′34.3″E) in March 2015 and identified by Dr Vo Van Chi, Faculty of Pharmacy, University of Medicine and Pharmacy at Ho Chi Minh city. The leaves were detached and sorted into sets of 10, from the youngest (first leaf) to oldest (tenth leaf) leaves that were then dried at 50 °C. The first 3 leaves were used for extraction and isolation. Voucher specimens (UMP-2015-AQ.001-010) were deposited at the Faculty of Pharmacy, University of Medicine and Pharmacy at Ho Chi Minh City, Vietnam. Each leaf (from the first to the tenth) was weighed, and then pulverized into powder of <355 µm. For extraction and isolation purposes, leaves were directly extracted without pulverization. The HPLC solvents were purchased from Merck and Labscan.
Apparatus
Preparative HPLC was performed on an LC-8A Shimadzu instrument, using a Discovery HS C18 column (25 cm × 21.2 mm, 10 µm). The injection volume was 10 mL, and the analysis was performed at room temperature. The flow rate was maintained at 10 mL/min, and the absorbance was measured at a wavelength of 330 nm. HPLC was performed on an ACQUITY UPLC H-Class system (Waters), equipped with a PDA detector (330 nm) and a Phenomenex Gemini C18 (150 mm × 4.6 mm, i.d., 3 µm) (Phenomenex), connected to Empower software. The separation was achieved using a mobile phase of 0.2% acetic acid/acetonitrile (A) and 0.2% acetic acid (B) in the following sequence: 0 to 1 min: 10% A; 1 to 5 min: 13% A; 5 to 10 min: 13% A; 10 to 13 min: 25% A; 13 to 14 min: 25% A; 14 to 15 min: 30% A; 15 to 18 min: 35% A; 18 to 19 min: 64% A; 19 to 22 min: 65% A; 22 to 23 min: 65% A; 23 to 28 min: 85% A; and 28 to 31 min: 10% A. The column temperature was set at 40 °C. The HPLC flow rate was 1 mL/min. A 4-μL sample solution was injected into the ultra-performance liquid chromatography (UPLC) system. TLC was performed using silica gel F254 (40-63 µm, Merck). Compounds were detected by UV light at 254 nm and 365 nm, and then were sprayed with 10% H2SO4 in EtOH, followed by heating. Column chromatography (CC) was performed using silica gel (200-300 mesh), along with liquid chromatography using LiChroprep RP-18 silica gel (40-63 µm) (Merck). Silica gel 60 (40-63 µm, 230-400 mesh ASTM) for the CC was purchased from Scharlau. The other chemicals were of the highest grade available. 1H and 13C NMR spectra were recorded in DMSO-d6 using a Bruker AM-500 (500 MHz) spectrometer. ESI-MS were recorded on a MSQ_PLUS mass spectrometer (THERMO).
Extraction and Isolation of Standard Compounds
The isolation of polyphenolic compounds was targeted, with major polyphenol peaks from the total extract from UPLC chromatography occurring at 330 nm. The dried young leaves of A crassna (0.9 kg) were extracted under reflux using 100% MeOH (2 m 10 L) at 65 °C. The total MeOH extract was then cooled. A yellow precipitate resulted, which was filtered to obtain 70 g of residue. This was then crystallized several times using hot MeOH to obtain mangiferin (50 g, 3). The filtered extract was evaporated in vacuo to produce a MeOH extract (200 g), which was dissolved in water, and partitioned using CH2Cl2. A precipitate appeared in the CH2Cl2 extract, and was filtered and crystallized in CHCl3-MeOH (8:2) to obtain genkwanin (15 mg, 7). The CH2Cl2 fraction was evaporated in vacuo to obtain 25 g of extract. The water layer was extracted using EtOAc. The precipitate appeared as an EtOAc extract after removing 50% of the solvent. It was then filtered to yield 1.5 g of residue (Fraction E1). The EtOAc fraction was evaporated in vacuo to obtain 45 g of extract. The water layer was extracted using watersaturated n-butanol to give 15 g of n-butanol extract. The aqueous layer was then purified using Diaion HP-20, and eluted using 100% water and 100% methanol. The methanol eluate was evaporated in vacuo to obtain 12 g of dried extract. E1 (1.5 g) was passed through a silica gel column and eluted using CHCl3:MeOH (85:15) to obtain 7 fractions (E1-E7). Fraction E6 was continuously chromatographed on a silica-gel column and eluted with EtOAc-MeOH (9:1) to obtain genkwanin 4'methoxy 5-O-β-primeveroside (35 mg, 6). Fraction E7 was recrystallized in MeOH-H2O (5:5) to yield genkwanin 5-O-β-primeveroside (15 mg, 5). The EtOAc fraction (45 g) was subjected to a silica gel column, and eluted using EtOAc-MeOH (100:0, 100:5, 100:15) to obtain 9 fractions (EA1-EA9). Fraction EA3 (12.5 g) was crystallized in EtOAc to obtain iriflophenone 2-O-α-L-rhamnoside (5.13 g, 4). The n-BuOH fraction was then passed through a silica gel column and eluted using CHCl3-MeOH (10:1; 8:1; 5:1) to obtain 5 subfractions (Bu1-Bu5). Subfraction Bu5 (350 mg) was further separated by RP-18 and eluted using MeOH-H2O (4:6) to obtain 6 further fractions (Bu5.1-Bu5.6). Fraction Bu5.6 (55 mg) was further purified by prep-HPLC, using MeOH-H2O (24:74) as a mobile phase, to yield iriflophenone 3-C-β-D-glucoside (15 mg, 2). A 100% methanol extract (5 g), obtained from the Diaion HP-20 column, was subjected to the RP-18 column, and eluted with a gradient of water/methanol. The 100% MeOH fraction (200 mg) was then separated by prep-HPLC, eluted using MeOH-H2O (15:85), to yield iriflophenone 3,5-C-β-diglucoside (32 mg, 1). After confirming the purity (>95%) by HPLC-UV analysis and recorded 1H and 13C NMR spectra, each isolated compound was authorized as an analytical standard.

Isolation procedure of primary polyphenols from Aquilaria crassna leaves.
Preparation of Standard Solution
A stock solution containing iriflophenone 3,5-C-β-diglucoside (
Optimization Extraction Solvent for HPLC
To obtain better extraction rates for the 7 polyphenols under investigation from A crassna leaves, different solvents and extraction methods were applied, including water and MeOH (30%, 60%, 80%, and 100%), with reflux and sonication also being employed, as described in Table 1. All extracts were filtered through a 0.2-µm membrane before injection into the UPLC.
Extraction Methods.

Each extraction was conducted in triplicate.
Preparation of HPLC Samples
A 100 mg aliquot of A crassna leaf powder for each different age was placed in a volumetric flask, to which 25 mL MeOH 100% was added. This was then shaken on a Vortex Genie (Scientific Industries, Inc) for 10 s before being subjected to ultrasonication (Branson 5510) for 30 min at 55 °C. The solution was filtered through a 0.22-µm membrane before UPLC analysis.
Method Validation
The precision of the analytical method was determined by intra- and inter-day variations. A 100 mg aliquot of A crassna leaves was extracted and analyzed, as described above. The intraday precision was performed by triplicate extraction and analysis on a single day. The inter-day precision was carried out on 3 different days. Variations were expressed as a percent relative standard deviation (% RSD). A recovery test was used to evaluate the accuracy of this quantification method. A precise amount of each polyphenol was added to a 100-mg A crassna leaf sample, and then extracted and analyzed, as described above. The following formula determined the average recovery: recovery (%) = (observed amount – original amount)/spiked amount × 100%, with % RSD = (SD/mean) × 100%. Using the validated method, samples from 10 groups of leaves were successfully analyzed. Each type of leaf was prepared as an extract, following the procedure described above, and was filtered before injection into the UPLC system. The contents of the 7 polyphenols obtained were calculated, using the formula:
α-Glucosidase Inhibitory Assay
α-glucosidase (EC 3.2.1.20) isolated from Saccharomyces cerevisiae (750 UN) and p-nitrophenyl-α-
Aliquots of 1.5 mM p-nitrophenyl-α-
Protein and Ligand Preparation
The 3D structure of α-glucosidase (Protein Data Bank [PDB 2ZE0]) was obtained from the PDB (http://www.rcsb.org/pdb). The selected 2ZE0 was fixed using a CHARMM (Chemistry at Harvard Macromolecular Mechanics) force field in DS 2.5 (DS, Accelrys Software), which added up the hydrogen atoms, partial charges, and missing residues that are appropriately used for the processes of molecular docking. The 3D structures of 3 natural compounds—mangiferin (
Molecular Docking
Virtual screening of the bioactive compounds on α-glucosidase [PDB 2ZE0] was carried out using AutoDock Vina software.14,15 A grid box covering the active protein site was generated using the following parameters: center_x = 8.24; center_y = 26.90; center_z = 23.69; size_x = 25; size_y = 25; and size_z = 25. The docking scores are reported in kcal/mol, and the compounds were ranked by their docking scores. Finally, the molecular interactions between the proteins and selected ligands were visualized using Discovery Studio Visualizer software.
Statistical Analysis
The statistical significance was evaluated using an analysis of variance and SPSS software version 20. A P value of <.05 was considered to be statistically significant.
Results and Discussion
Isolation of Phenolic Compounds
Seven polyphenolic compounds were isolated from A crassna leaves using liquid–liquid partitioning, followed by normal-phase CC, RP-18 silica-gel CC, and/or semi-prep HPLC. The structures of the isolated compounds [iriflophenone 3,5-C-β-diglucoside (

Structures of major polyphenols isolated from Aquilaria crassna leaves.
α-Glucosidase Inhibitory Activity of the Isolated Compounds and Extracts
The α-glucosidase inhibitory activity of compounds
In the next study, the extracts from leaves 1 to 10 were tested for their α-glucosidase inhibitory activity using 2 concentrations of 1500 and 750 µg/mL. The results are presented in Table 2.
Screening for α-Glucosidase Inhibitory Effects of the Extracts of First—10th Leaves 1 to 10.

Experiments carried out in 3 replicates.
L1: first leaves.
Positive compound.
The α-glucosidase inhibition activity of the leaves indicates a gradual decrease in activity from leaf 1 to leaf 10 in both of the 2 concentrations (1500 and 750 µg/mL). These results are understandable because the highest polyphenol contents were found in leaves 1 and 2, and gradually decreased to leaf 10. The younger leaf samples (1-3) showed α-glucosidase inhibition of 89.5%, 92.4%, and 73.7%, respectively. This might indicate a correlation between the polyphenol content of the leaf samples and α-glucosidase inhibitory activity. Since leaves 1 to 3 showed the most potent α-glucosidase inhibitory activity, they were subsequently tested for their IC50 values.
Leaf sample 1 exhibited the most potent α-glucosidase inhibiting effect, with an IC50 value of 169.8 µg/mL, followed by leaf 2, with an IC50 value of 248.0 µg/mL, and leaf 3, with an IC50 value of 507.2 µg/ µg/ µg/mL (acarbose, positive control 50.0 ± 2.3 µg/ µg/mL) . These results are consistent with the screening results that indicated that the α-glucosidase inhibitory effect increased with the polyphenol content in the leaf samples. In terms of tea quality, this suggests that for a high polyphenol content and vigorous α-glucosidase inhibitory activity, the leaves should be collected from the ends of the branches.
Molecular Docking Results
α-Glucosidase inhibitors are common oral antidiabetic drugs used to control carbohydrates, typically by converting them into simple sugars, allowing them to be absorbed by the intestines. The 3D structures of the most potent compounds, such as mangiferin (

Molecular docking of α-glucosidase using 2ZEO active site. (A) 2D and (B) B 3D structures of acarbose. (C) 2D and (D) 3D structure of genkwanin. (E) 2D and (F) 3D structure of genkwanin 5-O-β-D-primeveroside. (G) 2D and (H) 3D structure of mangiferin.
Molecular Docking Simulation Results for Inhibitory Complexes Between the Ligands and Targeted Protein (PTP1P-2ZE0).
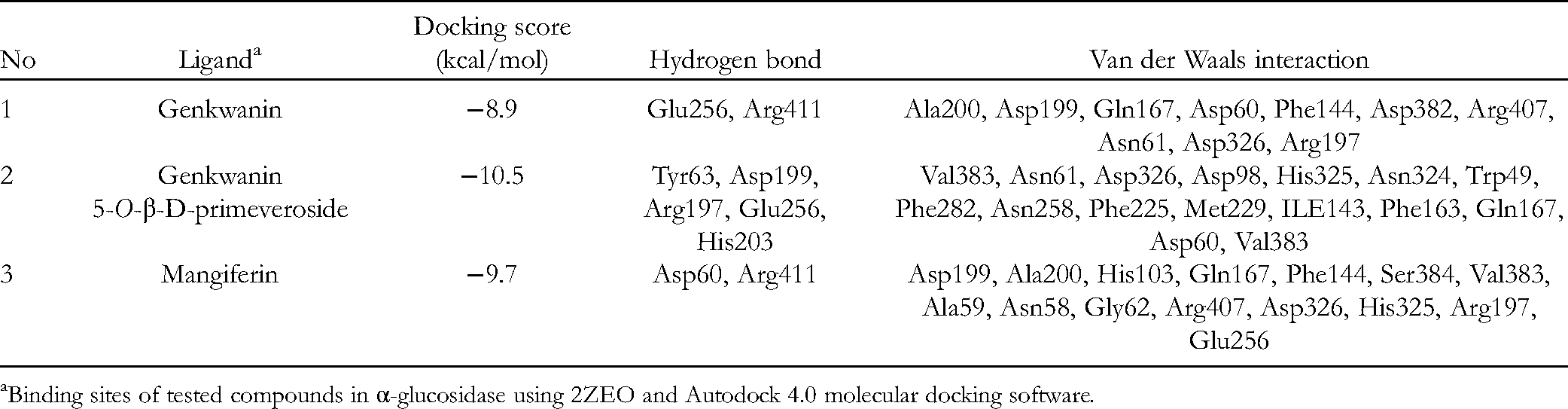
Binding sites of tested compounds in α-glucosidase using 2ZEO and Autodock 4.0 molecular docking software.
Polyphenol Content of Different-Aged Leaves
Extraction Method Development
In order to obtain an effective extraction method, ultrasonication and reflux were used in tandem with the variables involved in the procedure, including methanol at different concentrations (30%, 60%, 80%, and 100%), temperature (55, 60, 80, and 100 °C), extraction duration (30-60 min), and extract repetition (3 times). The results showed that reflux using 60% MeOH gave a better extraction yield than ultrasonication with MeOH 100% and was suitable for extracting almost all the major polyphenolic compounds (data not shown). However, in the reflux method using 60% MeOH, mangiferin precipitated after the extract cooled. Therefore, 100% MeOH with ultrasonication is the most appropriate extraction method for the 7 primary polyphenols in A crassna leaves.
Optimization of HPLC Conditions
The optimal mobile phase composition for the analysis of iriflophenon 3,5-C-β-D-diglucoside (
Validation of a Developed Method
The detection method calibration curves exhibited good linear regressions under the optimized chromatographic conditions, as shown in Table 4.
Validated Analytical Parameters for the UPLC/UV Quantification of 7 Major Polyphenols in Aquilaria crassna Leaves.

Abbreviation: UPLC, ultra-performance liquid chromatography.
Polyphenol Content of A crassna Leaves
The major polyphenolic components of the leaves included compounds
Polyphenol Contents (mg/g) in Leaves of Different Ages.

Data are presented as the mean ± SD of results from 3 independent experiments.
Conclusion
In this study, 7 phenolic compounds [iriflophenon 3,5-C-β-D-diglucoside (
Footnotes
Acknowledgments
This study was supported by the University of Medicine and Pharmacy at Ho Chi Minh city 2019.
Authors’ Note
HVTL, PLN, and MHT designed the research and revised the manuscript; TNTN, TDL, DHN, HVTN, and TNN performed the experiments; and TKN, HVTL, and MHT wrote the article together. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
