Abstract
Memory disorders are the main symptoms of aging and Alzheimer’s disease and seriously affect the quality of life. Schisandra, as a famous traditional Chinese medicine, has been used for modulating “the internal organs” for a thousand years. The total lignans from Schisandra have been scientifically proved to improve learning and memory ability. Since it is unclear which monomer in Schisandra total lignans exerts such a function, we evaluated the potential effects of Schisantherin A (SCA), the main monomer from Schisandra, on improving learning ability and memory in amyloid β-protein (Aβ1-42)-induced Alzheimer’s disease (AD) model mice. We found that SCA (5 mg/kg) significantly prolonged the latency and reduced the number of errors in a step-through test. SCA significantly shortened the time of finding the platform and increased the number of crossing the platform and the residence time in a Morris water maze test. SCA increased superoxide dismutase activities and reduced the Malondialdehyde level of the hippocampal tissue, suggesting its role in reducing oxidative stress in the AD mice. Furthermore, we found that SCA significantly decreased the hyperphosphorylation of Tau by altering glycogen synthase kinase-3β (GSK-3β) phosphorylation on Tyr216 and Ser9. Our results revealed the mechanism underlying SCA-mediated learning and memory improvement by regulating GSK-3β activity and lowering the hyperphosphorylation of Tau protein in the hippocampus of AD mice.
Alzheimer’s disease (AD) is a progressive and ultimately fatal neurodegenerative disease. Its main clinical manifestation is the deficiency of memory cognition. 1 -4 Studies have shown that the hyperphosphorylation of Tau is one of the mechanisms for AD. 5 -7 Schisandra, a traditional Chinese medicine, is the dried ripe fruit of Schisandra chinensis Turcz. Baill. of Magnoliaceae. Schisandra has been described to possess various functions according to the theory of traditional Chinese medicine, such as astringing lung, nourishing kidney, promoting the secretion of body fluid, arresting sweating, astringing essence, keeping calm, and relieving mental stress. “Bao Pu Zi” written by the Jin Dynasty scholar Ge Hong (283, 343 AD) recorded that the long-term use of Schisandra could help to rejuvenate and prolong life. Recent studies have found that Schisandra lignans can significantly improve the learning and memory ability of the aging mice. 8 -11 Schisantherin A (SCA, named by Jiasen Liu) is one of the main active lignans derived from Schisandra and has been reported to significantly improve the learning and memory impairment induced by amyloid β-protein (Aβ) in mice. 12 -14 However, its underlying mechanism remains elusive. In this study, an AD model of the mouse was established by administering Aβ1-42, and the effect of SCA on improving the learning and memory was investigated in the AD model mice. This study revealed that SCA executed these functions by modulating the Tau-glycogen synthase kinase-3β (GSK-3β) signaling pathway.
The step-through test is a standard method to assess short-term or long-term memory of mice. 15 By using the step-through test, we examined the effects of SCA on the learning and memory in an AD model mice (Figure 1). AD model mice were produced by Aβ1-42. On the second day, the darkness avoidance latencies of Aβ1-42-treated mice were significantly shortened in comparison with untreated control mice (P < 0.01) and the number of errors within 5 minutes was significantly increased (P < 0.01) in AD model (MOD) group. Whereas, SCA prolonged the darkness avoidance latency of Aβ1-42-treated mice (P < 0.05), reduced the number of errors significantly (P < 0.05) in the SCA (M) group (SCA-treated AD mice) in comparison with MOD mice (Figure 2).

Experimental procedure. Aβ, amyloid β-protein; AD, Alzheimer’s disease; CON, control; SCA, schisantherin A; MOD, model; SCA(C), SCA-treated normal mice; SCA(M), SCA-treated AD mice; SOD, superoxide dismutase; MDA, malondialdehyde.

Effects of SCA on the latency and number of errors of AD mice in the step-through test. (a) Effects of SCA on the latency of Aβ1-42-induced AD mice. (b) Effects of SCA on the number of errors in AD mice. Data are expressed as means ± standard deviation (n = 10). ## P < 0.01 in relation to CON group. *P < 0.05 in relation to MOD group. Aβ, amyloid β-protein; AD, Alzheimer’s disease; CON, control; SCA, schisantherin A; MOD, model; SCA(C), SCA-treated normal mice; SCA(M), SCA-treated AD mice.
The Morris water maze is a classic test for analysis of spatial memory and consists of a navigation trial and probe trial. 16 -18 We employed this test to examine the effect of SCA on the learning and memory of Aβ1-42-treated mice. The results of the time of finding the platform gradually shortened from the second to fifth day in all groups, but increased in the MOD group in comparison with that in the control group (P < 0.01) (Figure 3). However, in the group treated with SCA, the time of finding the platform in the training on the fifth day was significantly shortened in comparison with that of the MOD group (P < 0.01) (Figure 3).

Effects of SCA on the time of finding the platform in Aβ1-42-induced AD mice in the place navigation test. (a) Effects of SCA on the time of finding the platform in Aβ1-42-induced AD mice. (b) Effects of SCA on the swimming path of Aβ1-42-induced AD mice. Data are expressed as means ± standard deviation (n = 10). # P < 0.05 and ## P < 0.01 in relation to control (CON) group. *P < 0.05 and **P < 0.01 in relation to MOD group. Aβ, amyloid β-protein; AD, Alzheimer’s disease; CON, control; SCA, schisantherin A; MOD, model; SCA(C), SCA-treated normal mice; SCA(M), SCA-treated AD mice.
On the sixth day after Aβ1-42-administration, the platform was removed and the probe trial was conducted. The results of the probe trial are shown in Figure 4. The residence time in the target quadrants was significantly shortened (P < 0.01) and the number of crossing the original platform was significantly reduced (P < 0.05) in the MOD group in comparison with the control group. However, in SCA (C) group (SCA-treated normal mice), the residence time in the target quadrants was not significantly different (P > 0.05), but the number of crossing platforms was significantly increased (P < 0.05) in comparison with that of control mouse group. In the SCA (M) group, the residence time in the target quadrants was significantly prolonged in comparison with that of MOD group (P < 0.05) and the number of crossing the platform was increased significantly in comparison with that of MOD group (P < 0.05).

Effects of SCA on the time of finding the platform in Aβ1-42-induced AD mice. (a) The residence time in target quadrants. (b) The number of passing the platform. Data are expressed as means ± standard deviation (n = 10). ## P < 0.01 in relation to CON group. *P < 0.05 and **P < 0.01 in relation to MOD group. Aβ, amyloid β-protein; AD, Alzheimer’s disease; CON, control; SCA, schisantherin A; MOD, model; SCA(C), SCA-treated normal mice; SCA(M), SCA-treated AD mice.
Oxidative damage is a key factor in the pathogenesis of AD. The activity of superoxide dismutase (SOD) and the content of Malondialdehyde (MDA) are often used to examine the balance between oxidative damages and antioxidant effects. In order to determine the antioxidative effect of SCA, SOD activity and MDA content of the hippocampus in Aβ1-42-treated mice were measured. As shown in Figure 5, in the hippocampus of MOD group mice, SOD activities decreased and MDA contents increased significantly (P < 0.01) in comparison with those of the control group. However, in the SCA (M) group (SCA-treated AD mice), SOD activities increased and MDA contents decreased significantly (P < 0.01, P < 0.05) in comparison with those of the MOD group.

Effects of SCA on SOD activities and MDA contents of the hippocampus in Aβ1-42-induced AD mice. Data are expressed as means ± standard deviation (n = 10). ## P < 0.01 in relation to CON group. *P < 0.05 and **P < 0.01 in relation to MOD group. Aβ, amyloid β-protein; AD, Alzheimer’s disease; CON, control; SCA, schisantherin A; MOD, model; SCA(C), SCA-treated normal mice; SCA(M), SCA-treated AD mice; SOD, superoxide dismutase; MDA, malondialdehyde.
To elucidate the mechanism underlying SCA-mediated improvement of the learning and memory in AD mice induced by Aβ1-42, the expression of phosphorylation of Tau protein in the hippocampus of mice was analyzed. Western analysis showed that the expression of Tau-5 was not significantly changed in the MOD group (P>0.05), while the phosphorylations of Tau at Ser-199, Tau Ser-396, and Ser-404 were significantly increased in MOD group (P < 0.01). However, after treated with SCA, the phosphorylations of Tau at Ser-199, Tau Ser-396, and Ser-404 were significantly decreased in the hippocampus in both SCA (C) group and SCA (M) group (P < 0.01) (Figure 6(a, b)).
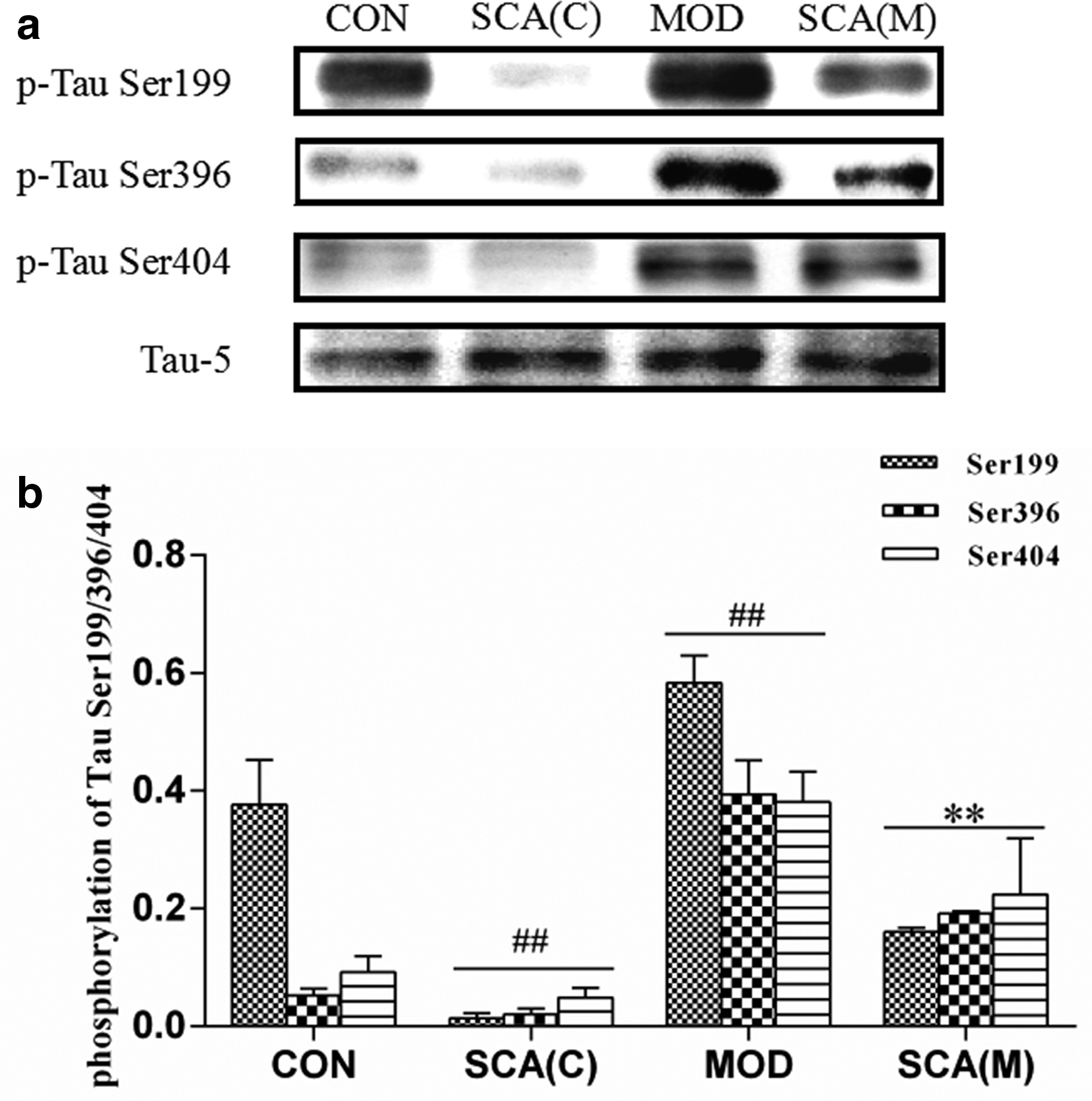
Expressions and its phosphorylations after SCA treatment in the hippocampus of AD mice (mean ± standard deviation). (a) The images of the expression level of Tau-5, p-Tau Ser199, Ser396, and Ser404 of the hippocampus in control (CON vs SCA [C]) and Aβ1-42-induced AD mice (MOD vs SCA [M]). (b) The phosphorylation ratio of Tau Ser199, Ser396, and Ser404 to Tau-5. Aβ, amyloid β-protein; AD, Alzheimer’s disease; CON, control; SCA, schisantherin A; MOD, model; SCA(C), SCA-treated normal mice; SCA(M), SCA-treated AD mice.
In addition, we also examined the expression of phosphorylation of GSK-3β, which is a kinase responsible for Tau hyperphosphorylation. The result showed that the expression of GSK-3β in the hippocampus of mice was not significantly different (P>0.05) in all groups, but the expressions of phosphorylation of GSK-3β at Tyr-216 and Ser-9 were significantly increased (P < 0.01) and decreased (P < 0.01), respectively, in MOD group. SCA treatment lowered the expression of phosphorylated protein of GSK-3β at Tyr-216 (P < 0.01) and elevated the expression of phosphorylated protein of GSK-3β at Ser-9 in the hippocampus of both normal (SCA [C]) and AD (SCA [M]) mice (P < 0.01) (Figure 7(a, b)). Since phosphorylation on Tyr216 of GSK-3β is required for maximum activity, 19 while phosphorylation on Ser-9 diminishes primed substrate phosphorylation by GSK-3β. 20 Our results revealed that SCA was able to lower the GSK-3β kinase activity and then reduced the hyperphosphorylation of Tau protein.

Effect of SCA on the expression levels of GSK-3β and its phosphorylation at Tyr216 and Ser9 in the hippocampus of AD mice. (a) The images of the expression levels of GSK-3β and its phosphorylation at Tyr216 and Ser9 in the hippocampus of AD mice. (b) The phosphorylation ratio of GSK-3β Tyr216 and GSK-3β Ser9 to GSK-3β. ## P < 0.01 in relation to CON group. **P < 0.01 in comparison with MOD group. AD, Alzheimer’s disease; GSK-3β, glycogen synthase kinase-3; CON, control; SCA, schisantherin A; MOD, model; SCA(C), SCA-treated normal mice; SCA(M), SCA-treated AD mice.
The pathological progression of AD has been linked to many factors, including inflammation, oxidative stress, and environmental factors. Two classic histopathological hallmarks of AD are the deposition of the amyloid β protein- (Aβ-) loaded senile plaques and accumulation of the neurofibrillary tangles (NFT), which are composed of the aggregated form of the filament protein tau. 21 -23 Aβ can activate Tau protein kinase 1, which phosphorylates Tau protein to the formation of paired helical filament and NFT, eventually resulting in neurodegeneration, synapse loss, and learning and memory impairment. 24,25 Many researches reported that a synthetic Aβ peptide, Aβ1-42, has been used in rodents to induce AD. 26 -28 Our results also showed that the intracerebroventricular injection of Aβ1-42 caused the hyperphosphorylation of Tau protein and abnormal phosphorylation of GSK-3β protein at two phosphorylation sites in the hippocampus of mice, and, as a consequence, significantly impaired learning and memory ability of the mice. Therefore, the mice treated by Aβ1-42 have been proven to be a useful animal model for AD study and testing anti-AD drug candidates.
The most direct factor to cause the excessive accumulation of Aβ roots is oxidative stress. 29,30 SOD is one of the main antioxidant enzymes and plays an important role in the balance between the oxidation and antioxidation in the body. 31,32 MDA, an important product of lipid peroxidation, is continuously increasing in the brain tissue and peripheral blood along with aging, serving as an indicator of the free radical-caused body injuries. 30 SCA could significantly increase the activity of SOD and decrease the content of MDA in the hippocampus of AD mice. Due to the antioxidant nature of SCA, our result suggested that the antioxidant ability of the body might be enhanced and then the oxidative injury be prevented, which were consistent with the results reported by Li et al. 12
Tau protein is widely expressed in neurons as a microtubule-associated protein involved in the construction of the cell skeleton. In AD, Tau protein is hyperphosphorylated at sites Ser199, Ser396, and Ser404. The hyperphosphorylation or other abnormal modification of Tau protein let it lose the binding ability to microtubules, damage the normal cytoskeletal structure, and disintegrate the nerve fibers. Hyperphosphorylation of Tau protein makes the protein accumulated to form straight filaments or double helix, leading to the death of neurons and ultimately AD. 33 -37 GSK-3β is one of the key kinases involved in the hyperphosphorylation of AD-like Tau, and its elevated activity may cause the abnormal hyperphosphorylation of Tau protein. The phosphorylation of GSK-3β at Tyr216 is the basis of the regulatory protein activities and renders GSK-3β in a state of activation in quiescent cells, thereby aggravating the phosphorylation of Tau protein. The phosphorylated GSK-3β at Ser9 site negatively regulates GSK-3β protein activity by causing the N-terminal tail of GSK-3β self-association in the primed-substrate binding pocket, thus diminishing primed-substrate phosphorylation by GSK3β. 38 -41 Our result showed that SCA significantly increases the phosphorylation of GSK-3β at Ser9 site and decrease that of GSK-3β at Tyr216, therefore reducing the hyperphosphorylation of Tau in the hippocampus of AD mice. As a result, SCA improved the learning and memory ability and exhibited an anti-AD activity in AD mice (Figure 8).

Schisantherin A improves the learning and memory by regulating GSK-3β activity and then reducing the phosphorylation of Tau protein of the hippocampus in AD mice. Aβ, amyloid β-protein; AD, Alzheimer’s disease; GSK-3β, glycogen synthase kinase 3β.
In the current study, we tested SCA in the Aβ induced model and focused on the hyperphosphorylation of Tau protein and its kinase GSK-3β. Therefore, it remains unclear if SCA is effective in AD induced by other factors under other mechanisms. Furthermore, there are several other regions in the brain that are involved in spatial learning and memory (such as entorhinal and perirhinal cortices, prefrontal cortex, the cingulate cortex, and the neostriatum). We would like to know if SCA also improves the spatial learning and memory ability associated with these regions of the brain other than the hippocampus. These issues deserve further studies on SCA.
In the current study, we confirmed that SCA was able to improve the learning and memory in AD mice induced by Aβ. These effects might attribute to its antioxidation activity by reducing the phosphorylation level of Tau and regulating the kinase activity of GSK-3β in the hippocampus.
Experimental
Animals
Male healthy C57BL/6 mice, weighing (20 ± 2) g, were provided by The Experimental Animal Research Center of Jilin University, and the experimental animal production license number was SCXK (Ji): 2016-0003. The mice were raised in separate cages, under the diurnal cycle of 12 hours:12 hours and with free access to food and water. All procedures were approved by the Ethics Committee for Use of Experimental Animals at Beihua University (Jilin, China).
Animal Administration and Experimental Protocol
C57BL/6 mice were randomly divided into 4 groups, namely CON group (normal saline by intracerebroventricular injection and distilled water by intragastrical gavage); MOD group (3 μL of 1 mg/mL Aβ1-42 by intracerebroventricular injection and distilled water by intragastrical gavage), SCA (M) group (3 μL of 1 mg/mL Aβ1-42 by intracerebroventricular injection and 5 mg/kg SCA by intragastrical gavage), and SCA (C) group (normal saline by intracerebroventricular injection and 5 mg/kg SCA intragastrical gavage), 15 in each group. The mice were given the corresponding agents intragastrically once daily for 14 days, in which Aβ1-42 was given by intracerebroventricular injection on the first day, 14 and the Morris water maze test was performed on the ninth day. On the 14th and 15th day, the step-through test was performed, the activity of SOD and the level of MDA were detected, and the protein levels were detected by Western blot. The experimental procedure was shown in Figure 1.
Step-Through Test
The mice were placed in the bright chamber of a step-through instrument with their backs to the entrance of the dark chamber, and at the same time, the timer was started. The time required for the mice to get into the dark chamber from the entrance was recorded as the darkness avoidance latency. The mice were trained for 5 minutes in this test and the number of errors was recorded within 5 minutes. The second test was performed at an interval of 24 hours, and the latency and the number of errors within 5 minutes for each mouse were recorded. 15
Morris Water Maze Test
The mice were placed in a WMT-100 Morris water maze video tracking test system, the training time for the mice was set as 120 seconds, and the time more than 120 seconds when the mice could not reach the terminal was recorded as 120 seconds. The mice were trained once every 24 hours and the experimental data on the 15th day were collected. The mice were placed near the platform just before the first training and allowed to climb on the platform 3 times independently, then the mice were placed near the platform before each training and allowed to climb the platform once independently, and on the sixth day, the platform was removed for the observation of the spatial searchability of mice, including the number of crossing the platform and the number of crossing the effective area. 16 -18
Detection of SOD Activities and MDA Levels in the Hippocampus in AD Mice
Ten mice from each group were randomly selected, sacrificed at 30 minutes after the last administration. Their hippocampal tissue and serum samples were collected and the activity of SOD and the content of MDA were detected by WST-1 and TBA methods according to the instructions of the kits, respectively.
Western Blot Detection of Tau Phosphorylation-Related Protein Expressions in the Hippocampus
The hippocampal tissues of 3 mice from each group were taken at 30 minutes after the last administration, added with the lysis buffer to be lysed on ice for 1 hour, and then the cytolysate was centrifuged by a high-speed centrifuge. The supernatant protein concentration was determined by bicinchoninic acid assay and the target proteins were separated by a 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis. Two hours later, the proteins were transferred to polyvinylidene difluoride membranes. The membranes were rinsed with Tris buffer (TBST) for 5 minutes and then blocked with a blocking buffer (TBST buffer containing 5% skim milk powder) for 1 hour. After the incubation at room temperature, the blocking buffer was discarded and the corresponding first antibodies were added onto the membranes, respectively. After that, the membranes were incubated at 4°C overnight and washed 5 times with TBST, 5 minutes each time, then added with the corresponding second antibodies (1:2000), incubated for 2 hours and washed 5 times with TBST, 5 minutes each time, and finally added with the color-substrate solution for the ECL color development.
Statistical Analysis
The statistical analysis was performed using SPSS 19 software. The latency in the step-through test, the number of errors, the number of crossing the platform and the effective area, and the time of staying at the target quadrant in the Morris water maze test, the activity of SOD and the content of MDA in the hippocampus of mice, and the experimental data of Western Blot were expressed in mean ± standard deviation. Comparisons between multiple groups were assessed by one-way analysis of variance with post hoc test, and the multiple groups of Morris water maze tests were assessed by two-way analysis of variance. P < 0.05 was considered to be statistically significant.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Natural ScienceFoundation of Jilin Province(20150101230JC), the Education Department of Jilin Province (JJKH20180376KJ), and the Health Department of Jilin Province (2018J089).
