Abstract
Objective
Breast cancer stands as one of the most prevalent cancers globally, representing around 31% of new cancer diagnoses in women. The emergence of drug resistance undermines the efficacy of chemotherapy, highlighting the urgent need for alternative treatments. Ginger (Zingiber officinale) is a dietary plant known for its different biological effects. Our goal was to isolate the cytotoxic compounds from the petroleum ether extract of dried ginger.
Methods
Through bioactivity-guided isolation and comprehensive phytochemical analyses, the cytotoxic effects of different concentrations (ranging from 3.91 to 500 µg/mL) of ginger petroleum ether extract and its fractions were examined against MCF-7, SW 480, and A549 cancer cell lines, as well as 3T3 normal cells using the MTT assay. Fractions were obtained using column chromatography, and those showing significant toxicity were further fractionated using a bioactivity-guided separation method. The most cytotoxic component was purified using plate chromatography, and its toxicity was evaluated. Additionally, molecular docking was performed on gamma-secretase, PPARγ, MMP-9, tubulin, proteins Notch1, HES1, and cyclin D1.
Results
Fractions showed toxicity against all the tested cell lines, with a greater effect on MCF-7. A subfraction, FsA7, exhibited an impressive IC50 value of 1.68 ± 0.36 µg/mL against MCF-7. It also showed selective toxicity towards cancer cells compared to 3T3 cells. Upon analysis using GC/MS, it was identified as 6-shogaol based on the retention index (RI) and library search programs. Molecular docking studies indicated that 6-shogaol interacts with critical cancer-related targets, especially MMP-9, shedding light on its mechanism of action.
Conclusion
These findings highlight the therapeutic potential of 6-shogaol as a complementary treatment in breast cancer. This research provides foundational insights into the use of ginger-derived compounds in enhancing current cancer treatment strategies. Pre-clinical and clinical evaluation of cytotoxic effects of 6-shogaol in combination with anti-cancer drugs can be considered for future studies.
Introduction
Cancer is the second leading cause of death after cardiovascular diseases in Western countries. In 2020, approximately 19.3 million people worldwide were diagnosed with cancer, and about 10 million people died of cancer. 1 Breast cancer is one of the most common cancers worldwide; accounting for approximately 31% of all newly diagnosed cancers in women, and is the leading cause of cancer-related deaths. Despite the many advantages of chemotherapy, resistance to chemotherapeutic agents occurs through multiple mechanisms, including genetic mutations that alter drug targets, increased drug efflux by overexpressed transport proteins, and increased DNA repair capacity of cancer cells. As a result, many patients experience disease relapse and reduced response to conventional therapies. 2 Breast cancer is a complex disease with various pathologies, histological features, treatment responses, metastatic spread patterns, and patient outcomes, and remains a leading cause of cancer-related mortality, reflecting the critical need for effective therapeutic strategies. Sex, aging, family history, and reproductive factors such as early menarche, late menopause, late age at first pregnancy, and low parity, as well as endogenous and exogenous estrogens, modern lifestyles including excessive alcohol consumption and high dietary fat intake, are the most significant known risk factors for breast cancer progression. 3 Despite the proven impact of family history on increased risk of breast cancer incidence, cases without a family history are also common. 4 It has been investigated that matrix metalloproteinases (MMPs) play important roles in metastasis in many cancers. For example, overexpression of MMP2 and MMP9 is consistent with distant and lymph node metastasis in breast cancer. Their key roles in cell proliferation, inflammation, angiogenesis, and metastasis highlight their importance in breast cancer diagnosis and management. 5 Tobacco smoking, family history, diet, alcohol consumption, chronic lung inflammation, ionizing radiation, occupational exposure to asbestos, silica, radon, heavy metals, polycyclic aromatic hydrocarbons, and air pollution are all considered risk factors for the incidence of lung cancer. 6 Colorectal cancer is the third most common cancer among both men and women. 7 The etiology of this cancer may be hereditary or related to underlying inflammatory bowel disease. Factors such as a high-fat diet, consumption of red meat and alcohol, and underlying diseases like obesity and diabetes increase the risk of colorectal cancer. 8 Several strategies such as surgery, chemotherapy, radiotherapy, hormone-therapy, immunotherapy, and even target-therapy are available for cancer treatment. Despite being a common and favorable choice of cancer therapy, chemotherapy has some drawbacks, particularly the toxicity of chemotherapeutic drugs to normal cells and the emergence of drug resistance in tumor cells that ultimately leads to treatment failure. 9 Therefore, it is imperative to develop alternative therapies that can combat these challenges. 10
Numerous studies have demonstrated the potential synergy between antineoplastic drugs and medicinal plants in the treatment of certain cancers, offering hope for the discovery of novel medicinal agents in natural products. There is a wealth of literature on the cytotoxicity of medicinal plants, which provides a valuable resource for promising plants in this field.11,12 Today, plants possessing medicinal and cosmetic effects or health benefits besides their nutritional values are more popular and appealing than ever. Zingiber officinale Roscoe, commonly known as ginger, is one of the earliest plants in the Zingiberaceae family. In addition to its medicinal effects, ginger is widely used in the food industry as a key ingredient and spice in various food items, soft drinks, beverages, and medicinal products. The plant's rhizomes contain a diverse array of secondary metabolites, including phenolic compounds, terpenoids, flavonoids, essential oils, lipids, and polysaccharides. 13 Gingerols and shogaols are the plant's major pharmacologically active phenolic compounds.14,15 In dried ginger, 6-shogaol is a significant component with potent biological activity and high stability. 16 This compound is known for its anti-inflammatory, anti-cancer, analgesic, antipyretic, and antioxidant properties.17,18
The pharmacological activities of ginger and its constituents have been extensively studied, including anti-microbial, antioxidant, neuroinflammation modulation, anti-inflammatory, and anti-cancer activity.19,20 Ginger is considered a promising chemopreventive dietary agent with anti-tumorigenic and antiproliferative effects on gastric, liver, prostate, and leukemia cancer cells (HL-60). It also inhibits the activities of cyclooxygenase and lipoxygenase enzymes. 21 Previous studies have reported the cytotoxicity of the plant rhizomes using the Brine Shrimp cytotoxicity assay. 11 Despite the widespread use of ginger and its phytochemicals, safety concerns remain for researchers. This study aims to investigate the toxicity of the petroleum ether extract of ginger and its components, as well as to evaluate their safety on normal cells using the MTT assay. The next step involves a docking study of the isolated component(s) on potential targets such as gamma-secretase, MMP-9, PPARγ, tubulin, protein Notch1, HES1, and cyclin D1. The selection of these proteins, which have critical roles in developing cancer, as targets was based on the previous studies performed on the mechanism of action of 6-shogaol.22–26 γ-secretase is a protease that cleaves different types of proteins. Relating to cancer, it can break down Notch, cadherins, and other proteins, leading to functions such as cell fate regulation and cell adhesion. The inhibitors of this enzyme can act as anti-cancer. 27 The role of MMP-9 (Matrix Metalloproteinase-9) in the formation and growth of cancer is established. Its increased levels have been observed in various types of cancers, and especially are related to tumor stage and weak treatment response. 28 PPARγ (peroxisome proliferator-activated receptor-γ) is a factor involved in the onset and progression of some cancers; however, its role is controversial due to its diverse mechanisms of action.29,30 Tubulin protein is another key point for cancer treatment. Based on its essential role in cell division, tubulin inhibition can result in the induction of apoptosis. 31 Notch1 (Neurogenic locus notch homolog protein 1) is a protein involved in proliferation, differentiation, and apoptosis, which describes its role in cancer. 32 HES1 (Hairy and enhancer of split homolog-1) is recognized as one of the key players in malignancies. It is an upregulator of genes involved in cancer cell proliferation, migration, metastasis, and drug resistance. Therefore, it can be considered an important target for cancer therapeutics. 33 Cyclin D1 is a key regulator of the cell cycle, and its disregulation leads to uncontrolled cell proliferation and cancer progression. Its upregulation is reported in breast, colorectal, non-small-cell lung, and several other cancers. 34 Activity-guided fractionation alongside molecular studies successfully isolates and traces the active compounds of a plant step-by-step and delineates the interactions between 6-shogaol and critical cancer-related targets such as MMP-9. While previous studies have explored the anti-cancer properties of ginger and its constituents, particularly 6-shogaol, this study distinguishes itself through a comprehensive approach combining bioactivity-guided fractionation, GC-MS identification, and in silico molecular docking to elucidate the interaction of 6-shogaol with matrix MMP-9 in MCF-7 breast cancer cells, a relationship not extensively investigated in prior research. Through the integration of these methodologies, this study aims to provide a nuanced understanding of the molecular mechanisms governing 6-shogaol's anti-cancer effects. This deeper insight has the potential to inform the development of innovative therapeutic strategies designed to circumvent the limitations inherent in current chemotherapeutic regimens. Consequently, this approach not only illuminates the therapeutic promise of ginger-derived compounds but also contributes to the broader effort of improving cancer treatment outcomes.
Materials and Methods
Chemicals and Cell Lines
Dulbecco's Modified Eagles Medium (DMEM) and Fetal Bovine Serum (FBS) were purchased from Biosera, France, and Gibco (USA), respectively; additionally, 3-[4, 5-dimethylthiazol-2-yl]-2, 5 diphenyltetrazolium bromide (MTT), Phosphate Buffered Saline (PBS) and trypsin were provided by Sigma, USA. All other chemicals used in the research were of analytical grade.
MCF-7 (IBRC C10082), A549 (IBRC C10080), and NIH3T3 (IBRC C10100) cell lines were obtained from the Iranian Biological Resource Center (IBRC, Tehran, Iran), and SW 480 (CCL-228) cell line was purchased from the Pasteur Institute of Iran (Tehran, Iran).
Plant Materials
Ginger rhizomes were purchased from the market and authenticated by Dr Mirtadzadini from Bahonar University, and a voucher for the rhizomes was deposited in the Herbarium Center of the School of Pharmacy, at the University of Medical Sciences, Kerman, Iran (KF2006).
Quality Control of the Plant
For quality control, the water content, total ash, acid-soluble ash, and the extraction value (%) of the dried plant were determined, as previously explained. 35
Preliminary Phytochemical Screening
The plant underwent a preliminary study to detect the presence of various secondary metabolites such as alkaloids, flavonoids, terpenoids, and saponins using methods described in previous studies. 36 The petroleum ether extract of the plant was used to measure cytotoxicity, which had shown significant cytotoxicity in previous studies.11,12
Extraction and Fractionation
Dried ginger rhizomes (100 g) were subjected to extraction using a petroleum ether maceration method for 24 h, repeated three times with 200 mL of solvent each time. The extract was concentrated and further processed through column chromatography, yielding 21 fractions (F1 to F21). Fractions exhibiting similar Thin-Layer Chromatography (TLC) profiles and enhanced cytotoxicity were purified using a gradient solvent system of petroleum ether, chloroform, and ethyl acetate. The final purified subfraction, designated FsA7, underwent analysis via Gas Chromatography-Mass Spectrometry (GC-MS) to confirm its composition and identity as 6-shogaol based on retention indices and fragmentation patterns. The process is illustrated in Figure 1.

The Flowchart of the Fractionation and Biological Assay of the Ginger Extract.
Cell Culture
The cytotoxic effect was evaluated using MCF-7 (breast cancer), SW 480 (colorectal cancer), A549 (lung cancer), and 3T3 (normal mouse fibroblast) cell lines. The cells were cultured in DMEM supplemented with 10% FBS and an antibiotic solution (100 U/mL penicillin and 100 μg/mL streptomycin). The cells were then incubated at 37 °C, 5% CO2, and 95% relative humidity. Once the cell monolayers reached 80% confluency, they were trypsinized and seeded in flat-bottom 96-well microplates (100 µL of a 105 cell/mL suspension). The microplates were then incubated for 24 h to allow the cells to attach and form a monolayer. The following day, the attached cells were treated with different concentrations of fractions (3.91, 7.81, 15.62, 31.25, 62.5, 125, 250, and 500 µg/mL), subfractions, and the bioactive component. DMEM with DMSO (0.5%) served as the solvent control, and doxorubicin (2, 4, 8, 16, and 32 µg/mL) was used as the standard, as it is one of the most potent anti-cancer drugs. Each compound and concentration was tested in at least three wells. After 24 h of incubation, the cells were examined under an inverted microscope (Micros, Austria) and then exposed to 10 µL MTT (5 mg/mL) for 3 h at 37 °C in the dark. The formed formazan crystals were dissolved in 100 μL DMSO, and the absorbance was measured at 570 nm and 620 nm (as the reference). The viability percentage was calculated by dividing the average absorbance of the treated wells by the average absorbance of the solvent control wells. The experiment was repeated two or three times for each compound. The most potent fraction was subfractionated, and the subfractions and isolated bioactive component were tested on MCF-& and 3T3 cell lines. It should be mentioned that in each repetition of the experiment, at least one concentration of doxorubicin (a concentration about the calculated IC50) was used as a control to detect possible errors.
Gas Chromatography-Mass Spectrometry (GC/MS) Analysis
The GC-MS method was conducted on the pure compound prepared at a concentration of 1 mg/mL. The compound was injected into an Agilent 7890B series gas chromatograph combined with an Agilent 5977A series mass spectrometer equipped with a capillary HP-5MS column (30 m × 250 μm I.D., film thickness 0.25 μm). The carrier gas (Helium) flow rate was set at 1 mL/min, and the temperature program was as follows: 10 min at 50 °C, raised to 140 °C (3 °C/min), 10 min at 140 °C, and held isothermally for 10 min. The split ratio was 1:20, and electron ionization mode at 70 eV using full-scan mode from m/z 40–500 was employed for MS detection. The ion source and quadrupole temperatures were set at 230 °C and 150 °C, respectively.
Molecular Docking
The ligand was visualized and depicted as a 2D structure using ChemDraw 19.1 software, then imported into the Molecular Operating Environment (MOE 2019) software for subsequent optimizations. 37 The target receptors acquired in PDB format from the RCSB Protein Data Bank (https://www.rcsb.org) were imported into the MOE software. 38 The initial phase included the automatic preparation of the ligand and receptor, which was accomplished using the Quickprep function. Any minor discrepancies were resolved using the “structure preparation” tool. The “energy minimize” tool was used with the Amber10: EHT forcefield, an R-field value of 1.80, and a cut-off range of 8–10 to refine the 3D structures of the ligands and optimize the receptor in terms of atomic energy levels. 39 The binding site of a receptor was located using the “site finder” tool. The primary site, typically identified as the most prominent, was labeled as “Dummies.” The docking procedure followed the preparation of ligands and receptors and the identification of the primary binding site with “Receptor atoms” selected in the receptor section and “Dummies” designated in the site section. The settings for placement and refinement were set as “Triangle Matcher” and “Induced Fit,” respectively. For each ligand, the scoring methods were specified as “London dG,” with a pose count of 120:1. 40 The BindingDB server was used to find a dataset of compounds with a backbone similar to 6-shogaol. The structure was drawn, and the similarity was set at 70%. After the search, 81 compounds (including duplicates) were found and downloaded for further molecular modeling processes.41–45
Statistical Analysis
Results are shown as mean ± SEM The inhibitory concentration 50 (IC50) values were calculated using GraphPad Prism®. The selectivity index (SI) was calculated by dividing the average IC50 value on 3T3 (the normal cell line) by the average IC50 value on the cancer cell lines. 46 ANOVA (one-way analysis of variance) followed by Tukey's post hoc test was used to compare the differences with a significance level set at 0.05 or less.
Results
Extraction, Quality Control, and Phytochemical Study
The extraction yielded 4.15% (w/w) dried plant material, with a water content of 9.67% and total ash of 0.4% (w/w). Alcoholic and water extracts of the plant were determined to be 13.4% and 9.6% w/w, respectively. Phytochemical screening revealed the presence of saponins (+++), terpenoids (+++), and flavonoids (+++) in order of abundance; alkaloids and tannins were absent.
GC-MS Analysis
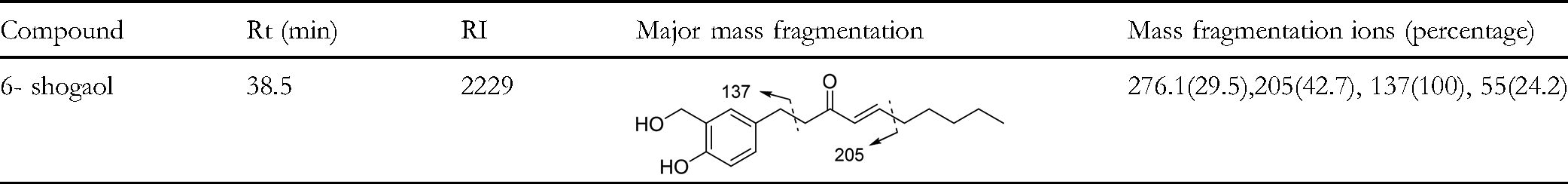
Gas chromatography combined with mass spectrometry (GC–MS) is the primary technique utilized for identifying sulfide compounds because of its outstanding resolution and capability for mass identification in GC–MS. 47 To identify any toxic compounds present in the fractions and to search for novel compounds, we conducted GC/MS analysis. The result showed that even when using non-polar solvents, the extract contains a high enough amount of 6-shogaol to cause cytotoxicity. Therefore, following purification, Subfraction FsA7, which exhibited significant toxicity, underwent final purification through plate chromatography using a solvent system of toluene: ethyl acetate: formic acid (9: 1: 2) the compound with the lowest IC50 value on the MCF-7 cell line was analyzed using the GC-MS method (Figure 2). The compound was identified by comparing the retention index (RI) and using library search programs such as NIST and Wiley (Table 1). As shown in Figure 2, the identified compound was 6-shogaol, with the main molecular ion (m/z) peaks related to 6-shogaol being 136 (base peak), 55, 119, 151, 205. The fragmentation patterns are described in Table 1.

The GC/MS Chromatogram of 6- Shogaol.
Identification of 6- Shogaol and its Mass Fragmentation Ions.
Cytotoxicity Assay
The results of the MTT assay indicated cytotoxicity in the majority of the isolated fractions on the examined cancer cell lines. Interestingly, most of these fractions exhibited little toxicity on the normal (3T3) cell line. Consequently, some of these fractions achieved large selectivity index (SI) values. As shown in Table 2, MCF-7 was the most sensitive cell line to the fractions. According to the National Cancer Institute (NCI) classification of cytotoxicity, compounds with IC50 ≤ 20 µg/mL are highly toxic, IC50 21 to 200 µg/mL are moderately toxic, IC50 201 to 500 µg/mL are weakly toxic, and IC50 > 500 µg/mL are non-toxic.48,49 Based on this classification, most fractions are moderately toxic for the studied cell lines. More precisely, fractions F5, F8, F10, F11, F16, and F20 were highly toxic for MCF-7 cells. Fraction F10 was highly toxic for A549, and none of the fractions were highly toxic for SW 480. Whereas, the number of fractions with moderate toxicity on MCF-7, A549, and SW 480 cell lines were respectively 14, 15, and 17 fractions.
IC50 (Mean ± S.E.M, µg/mL) and SI Values of Primary Fractions Separated by Column Chromatography from Petroleum Ether Extract of Ginger.

The survival percentage of cytotoxic fractions on two cell lines, MCF-7 and 3T3, is shown in Figure 3. These fractions have demonstrated the most toxicity on the MCF-7 cell line. The IC50 values of the subfractions Fs1 to Fs5 were > 500, 87.03 ± 2.99, 4.99 ± 0.85, 4.83 ± 0.19, and 89.80 ± 6.41 µg/mL, respectively. Fs3 and Fs4 exhibited the highest cytotoxicity on MCF-7 cells and were further purified (Figure 4).

Evaluation of the Cell Viability (%) of MCF-7 and 3T3 Cells Treated with the Six Major Highly Toxic (for MCF-7) Fractions of Z. officinale Extract on a Logarithmic Scale (Mean ± SD, n = 3).

The Survival of MCF-7 Cells Treated with Subfractions Fs1 to Fs5 (Mean ± SD, n = 3). Results for only Three Concentrations are Shown. Statistically Non-Significant Differences are Indicated by “ns”, While the Other Comparisons are Statistically Significant (p < 0.05).
Figure 5 displays the survival percentages of subfractions FsA1 to FsA12. Based on these results, FsA7 was selected for final purification and characterization.

Viability Percentage of Subfractions FsA1 to FsA12 at a Concentration of 50 µg/ mL on MCF-7 Cells (Mean ± SD, n = 3). The Same Letter at the top of Each Column Shows the Statistically Significant Differences (p < 0.05).
The IC50 values of the pure component were 1.68 ± 0.36 µg/mL (6.08 µM) and 6.08 ± 0.96 µg/mL (22.00 µM) for MCF-7 and 3T3 cell lines, respectively, resulting in a selectivity index of 3.62. The viability percentages for MCF-7 and 3T3 cells treated with 6-shogaol are also displayed in Figure 6. As shown in the figure, 6-shogaol at low concentrations induces greater suppression of proliferation in MCF-7 cells compared to 3T3 cells. However, at higher concentrations, this selectivity is no longer evident.

Viability Percentages of MCF-7 and 3T3 cells after 24 h Treatment with Different Concentrations of A) 6-shogaol (Final Sub-Fraction) and B) Doxorubicin (Mean ± SD, n = 3). ***: p < 0.001.
Molecular Docking
Since we observed high toxicity of 6-shogaol on the cultured cells, we conducted a molecular docking analysis to identify a potential target for it. This information can be valuable for future studies on drug design. With the exploration of previous studies,22–24,50 it was concluded that targets such as gamma-secretase (PDB ID: 7C9I), PPARγ (PDB ID: 6MS7), matrix metalloproteinase-9 (MMP-9) (PDB ID: 6ESM), tubulin (PDB ID: 4TV9), proteins Notch1 (PDB ID: 5FM9), HES1 (PDB ID: 7C4O), and cyclin D1 (PDB ID:6P8F) could be considered as suitable candidates in the target prediction step. The results of the molecular docking of 6-shogaol against these targets are documented in Table 3.
The Scores of 6-Shogaol Versus Multiple Predicted Receptors in Molecular Docking. Smaller Numbers Indicate Higher Binding Energy.

Among the receptors evaluated within an acceptable scoring range, three—namely MMP-9, the primary functional site of cyclin D1, and tubulin—demonstrated the highest docking scores. Notably, MMP-9 exhibited superior ligand positioning and formed a greater number of molecular interactions, accompanied by a significantly higher docking score compared to the others. Scores mentioned in Table 3 indicate that MMP-9 (PDB ID: 6ESM) could be potentially a suitable receptor for the justification of its mechanism of action (Figure 7). Based on this hypothesis, MMP-9 can be targeted by ligands similar to 6-shogaol, so that the best available small molecule can be selected from the dataset of similar compounds. The exact results of molecular docking scores of 81 similar compounds against MMP-9 are documented in a separate supplementary data file, but to put it in a nutshell, after omitting the duplicate ligands, two ligands (including 6-shogaol itself, showing the best score among the data set) showed significant scores (lower than −16) and can be suitable candidates for drug design procedure (Table 4). The existence of several donor and acceptor hydrogenic bonds in the side chains and main backbone of the molecules, alongside strong π-H stackings in the placement of phenol rings, has led to the establishment of such a nicely fitted ligand-receptor complex.

The Placement of 6-Shogaol (Green Ligand) and the Other Pioneer Compond (Yellow Ligand) in the Main Site of MMP-9.
The Molecular Structure of two Pioneer Ligands Alongside Their Docking Scores and Interactions.

While MMP-9 exhibited the most favorable molecular docking results, the binding affinities observed for tubulin and the first functional site of cyclin D1 were also notable. These findings suggest that 6-shogaol may exert its anti-cancer activity through multiple molecular pathways. Interaction with tubulin implies a potential to disrupt microtubule dynamics, thereby impairing mitotic spindle formation and inhibiting cancer cell proliferation. Similarly, binding to cyclin D1, a key regulator of the G1 to S phase transition in the cell cycle, may interfere with cell cycle progression and promote cell cycle arrest. 51 Collectively, these interactions support a multifaceted mechanism of action, contributing to the compound's cytotoxic potential against cancer cells.
Discussion
The World Health Organization has identified breast cancer as the leading cause of death for women worldwide in 2020. An array of medicinal plants and their phytochemicals have shown inhibitory activity on MCF-7 cancer cell lines in laboratory conditions.52,53 6-Shogaol, the main pungent constituent of dried and chopped ginger, is the dehydration product of 6-gingerol and has a wide range of biological activities, including anti-cancer effects. 54 It has been demonstrated that 6-shogaol exhibits an inhibitory effect on different cancer cell lines and animal disease models. 55 The primary antitumor properties of 6-shogaol include inhibiting cancer cell growth, preventing cancer metastasis, inducing apoptosis in cancer cells, and promoting cancer cell differentiation.56–58 To investigate the effectiveness of 6-shogaol on breast cancer stem cells, Ali et al created spheroids resembling stem cells from two types of breast cancer cell lines, namely ER/PR positive MCF-7 and triple-negative MDA-MB-231. The findings suggested that the inhibitory effect of 6-shogaol on spheroid growth and sustainability is due to the down-regulation of Notch signaling mediated by γ-secretase. 24 Ling et al observed that among the shogaol derivatives, 6-shogaol (5 to 20 μM), which has the shortest alkyl side chain, exhibits an inhibitory effect on PMA-induced invasion of MDA-MB-231 breast carcinoma cells. This inhibitory effect was associated with a reduction in the extracellular secretion of MMP-9. 22 Moreover, it has been shown that 6-shogaol can act as a ligand for PPARγ, which activates PPARγ and subsequently suppresses NFκB activity in MCF-7 and HT29 cells. These findings suggest that the antitumor effects of 6-shogaol are partially attributed to the activation of PPARγ-dependent signaling pathways. 23 Several in vitro and in vivo studies have confirmed that 6-shogaol shows little or no toxic effect on normal cells/tissues while effectively inhibiting breast cancer stem cells, including monolayers and spheroids, at non-toxic doses. In a breast cancer model, Ray Anas et al showed that the induction of autophagy by 6-shogaol leads to the death of breast cancer cells. The production of a large number of cytoplasmic vesicles and a lipid shift from LC3-I to LC3-II after 48 h of exposure of MCF-7 cells to 6-shogaol indicated that autophagy had occurred. 24 Additionally, the expression of target genes D1 and Hes1 by 6-Shogaol blocked Notch signaling in MCF-7 cells and decreased the survival of breast cancer cells. 25 In this breast cancer model, 6-shogaol in combination with γ-secretase DAPT showed a more significant inhibitory effect on Notch and its target proteins compared to each of these compounds alone, which indicates that one of the possible mechanisms of 6-shogaol is the inhibition of Notch signaling mediated through the inhibition of γ-secretase. 26 In the study by Bawadood et al, the cytotoxic effects of 6-shogaol were partially attributed to the inhibition of cells in S-phase and G2/M-phase. By inhibiting autophagy, 6-shogaol forces breast cancer cells to undergo apoptosis/necrosis. 22 Reduction of MMP-9 expression through NF-kB activation and PPAR-γ-dependent apoptosis in MCF-7 cells by 6-shogaol has also been reported. This compound has indicated microtubules as a possible target of 6-shogaol by interacting with sulfhydryl groups of cysteines in tubulin. 23
In this study, the reason for choosing petroleum ether extract was based on our previous study, which compared the cytotoxicity of different ginger extracts. The results revealed that the most cytotoxic extract was the petroleum ether extract (data not yet published). The results were consistent with previous studies on ginger. 59 In previous studies, polar solvents like ethanol and methanol were utilized for the extraction and isolation of gingerol and shogaol derivatives from ginger. 10 However, in our study, we opted to use petroleum ether as the solvent for extraction. Petroleum ether is a non-polar solvent that is capable of extracting non-polar compounds.
Molecular docking studies identified three receptors—MMP-9, the primary functional site of cyclin D1, and tubulin—as potential targets for elucidating the functional pathways of the investigated compounds. Comparative analysis of ligand positioning within each receptor's binding pocket, along with docking scores and the number of key molecular interactions critical for ligand fit, suggested a stronger likelihood of compound interaction with MMP-9 (Figure 8). Nevertheless, the possibility that these compounds exert multifaceted effects by simultaneously targeting multiple receptors remains a plausible and worthy consideration for further investigation. The favorable docking scores observed within the dataset of structurally similar compounds, comprising 81 analogs of the original molecule 6-shogaol, provide partial validation of the binding interactions. Notably, 6-shogaol itself achieved the highest docking score, stabilized by two hydrogen bond acceptors interacting with tyrosine 248 and arginine 249, along with an arene-hydrogen bond positioned deep within the target binding pocket. Additionally, a structurally analogous compound, presented in Table 4, exhibited a comparable binding orientation and a docking score closely matching that of 6-shogaol. These findings hold significant implications for rational drug design and the strategic development of molecular structures based on this scaffold.

The Proposed Mechanism of Action of 6-Shogaol Against Tumor via Inhibiting MMP-9.
Our study has some limitations that can be considered for future studies. The limitations of the in vitro study include the reliance on established cancer cell lines, which do not fully capture the biological complexity and variability of living organisms. The focus on 6-shogaol as the primary cytotoxic compound may overlook the synergistic interactions among other phytochemicals present in ginger. Additionally, the use of cell lines may not accurately represent the responses of primary tumor cells or the tumor heterogeneity found in clinical settings. Although in many studies, 3T3 cells are considered normal cells to compare the effect of drugs on normal and cancer cells, using a human normal cell line, which we did not include in our study, can be more valuable. Other limitations involve focusing on specific dosing regimens, sourcing ginger samples from a single geographic location, and the need for more sophisticated analyses to strengthen conclusions. Therefore, further research is necessary to validate and expand upon these findings, addressing the noted limitations and paving the way for future inquiries into phytochemical analysis and herbal therapeutics, particularly regarding the medicinal properties of ginger.
Conclusions
6-Shogaol was successfully isolated from ginger petroleum ether extract using chromatography methods based on a bioactivity-guided approach. Structural identification was performed using GC-MS, and its potential as an anti-cancer agent was evaluated. This indicates that ginger petroleum ether extract contains bioactive compounds, particularly 6-shogaol, capable of inhibiting the proliferation of cancer cells. While these findings underscore 6-shogaol's efficacy, variations in extraction techniques, sample selection, and biological models across studies highlight the need for rigorous standardization in assessing the anti-cancer potential of natural products. Molecular docking studies suggest that 6-shogaol's anti-cancer activity may involve interaction with and inhibition of MMP-9. However, it is crucial to consider that excessive daily intake of ginger products may pose health risks.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251375168 - Supplemental material for Integrative Computational and Bioactivity-Guided Strategies for Isolating Cytotoxic Phytochemicals in Ginger (Zingiber officinale Roscoe)
Supplemental material, sj-docx-1-npx-10.1177_1934578X251375168 for Integrative Computational and Bioactivity-Guided Strategies for Isolating Cytotoxic Phytochemicals in Ginger (Zingiber officinale Roscoe) by Faegheh Farhadi, Mohammad Amin Langarizadeh, Marjan Noormohammadi, Saman Dehghan, Ali Movahedinezhad, Ali Mohammadi, Yasaman Abolhassani, Salehe Sabouri and Fariba Sharififar in Natural Product Communications
Footnotes
Acknowledgment
We would like to express our appreciation to Kerman University of Medical Sciences for the financial support of this project.
CRediT Authorship Contribution Statement
Data Availability Statement
The authors confirm that the data supporting the findings of this study are available within the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study is approved by the ethical committee of Kerman University of Medical Sciences (code: IR.KMU.REC.1398.331).
Funding
This work was supported by the Research Council of Kerman University of Medical Sciences under Grant No. 98000407.
Statement of Human and Animal Rights
This study does not involve human and animal subjects, and the statement of human and animal rights is not applicable.
Statement of Informed Consent
There are no human subjects in the article, and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
