Abstract
Objective
This work aims to study the volatile constituents and quantify their relative abundances in the non-polar extracts of saffron (
Methods
Non-polar extracts of saffron petals and stigmas were acquired through ultrasonic extraction utilizing chloroform, petroleum ether, and hexane as solvents. The extracts’ volatile compounds were analyzed via gas chromatography-mass spectrometry (GC/MS). The numerous compounds were examined for their potential cytotoxic activity
Results
The results indicate a significant variation and abundance of bioactive compounds in the petals and stigmas of
Conclusion
The GC/MS analysis demonstrated a significant variety of volatile compounds in the petals and stigmas of saffron. Due to their favorable binding interactions with the target protein, 14b-pregnan and vitamin E may be regarded as effective phytochemicals in up-regulating the essential protein (p53) that maintains cellular homeostasis and repairs cellular damage. The volatile compound valeric acid, identified for the first time in the saffron petals of the Algerian cultivar, may serve as a biomarker for distinguishing the geographic origins of saffron.
Introduction
Saffron is derived from autumn crocus.
However, it is now commonly used in the healthcare field. Saffron has a variety of therapeutic properties, 2 including antidepressant, anxiolytic, hypolipidemic, anti-atherogenic, and anti-hypertensive effects. Saffron has remarkable health benefits, thanks to its main active ingredients: picrocrocin (which gives it its bitter taste), safranal (which gives it its scent), and crocin (which gives it its color). Saffron's diverse range of medicinal properties is most likely due to the synergistic action of crocin, crocetin, and safranal, as well as other minor compounds. Their potent antioxidant and anti-inflammatory properties appear to be central to many of these effects, which include neuroprotection, cardiovascular health, mood regulation, and potential anticancer properties. 3 Several studies have investigated the chemical components of saffron.
A study 4 reported that the phytochemical composition of saffron petals and stigma included tannins, terpenes, steroids, alkaloids, flavonoids, anthocyanins, and saponins. The chemical composition of saffron varies greatly between regions, with key markers such as crocin, picrocrocin, and safranal playing an important role in determining quality. Ongoing research aims to establish reliable chemical markers for geographical origin, contributing to the quality and authenticity of saffron. 5 The chemical composition of saffron is analyzed using advanced analytical techniques like high-performance liquid chromatography and gas chromatography-mass spectrometry (GC-MS).6,7 GC-MS has not been fully investigated in the case of saffron. It provides various experimental measurements with high sensitivity, low detection limits, and the ability to rapidly analyze a large number of volatile compounds.
In this study, GC/MS analysis was used to identify the volatile constituents of saffron samples grown in the El Outaya region of Biskra, Algeria, using an ultrasonic-assisted extraction method. The objective is to investigate the impact of non-polar solvent extraction on the volatile components and their relative abundances, assessing its potential as a biomarker for distinguishing saffron based on geographic origins. Additionally, the toxicity profile of the selected compounds from the non-polar extracts was analyzed by forecasting their bioavailability and pharmacokinetic properties using an
Materials and Methods
Plant Material
Extract Preparation
Both petals and stigmas were dried away from light, and six grams (6 g) of each were crushed separately in a pestle mortar to form a powder. Petal and stigma powders were divided into three equal parts (2 g) and ultrasonically extracted separately with 20 mL of solvents (chloroform, petroleum ether, and hexane). The extraction was repeated three times, with the solvent renewed each time. The contents were then filtered through filter paper Whatman N°1. The filtrate was evaporated at 40 °C using a rotary evaporator and kept in a refrigerator at 4 °C until analysis.
GC-MS Analysis
The GC analysis was done on an Agilent Technologies GC 17A gas chromatograph with a gross-linked HP 5MS column (30 m*0.25 mm, film thickness 0.25 μm). The oven temperature was set to isothermal at 60 °C for 8 min, with helium as the carrier gas at a rate of 0.5 mL per minute. An HP Agilent Technologies 6800 plus mass selective detector was used for the GC/MS analysis. The operating conditions were identical to those for the analytical GC. The MS operating parameters were as follows: ionization potential, 70 ev, ionization current, 2 A, ion source temperature, 280 °C, resolution 1000, scan time, 5 s, scan mass range, 34-450 u, spit ratio, 50:1, and injected volume, 1.0 µL. Compounds were identified by comparing their retention times to matching peaks in the National Institute of Standards and Technology (NIST) and Wiley mass spectral libraries, as well as comparing the fragmentation pattern of the mass spectra and retention indices to those reported in the literature by the NIST Standard Reference Database (version 2.4).
Bioavailability and Pharmacokinetics
In Silico ADME-Toxicity Prediction Studies
The drug-like and pharmacokinetic properties of selected compounds from non-polar extracts of
Molecular Docking
The ligand interactions with the receptor (PDB ID: 1TUP) structure, which represents the p53 protein's central domain, were investigated using Chimera and AutoDock Vina.8-11 This domain, which includes residues 102 to 292, is critical for specific DNA binding, which is frequently compromised by cancer-related mutations. In AutoDock Vina, the docking grid was configured to target key residues, most notably Arg248 and Arg273, which interact directly with DNA, while Arg175 and His179 help stabilize p53 central domain. Tumor-associated mutations frequently affect these residues.11,12 Prior to performing molecular docking, both the protein and ligand files must be thoroughly prepared. This process includes retrieving 1TUP structure from the Protein Data Bank (https://www.rcsb.org/), removing water molecules and extraneous cofactors, adding polarizable hydrogens to increase atomic charges, and converting the structure to PDBQT format with AutoDockTools. 10 Similarly, the ligand structure was refined with Chimera, assisted model building with energy refinement was used for energy minimization, Gasteiger charges were assigned, and the data was converted to PDBQT format. The molecular docking results were analyzed using Discovery Studio 2020. 13
Results
GC-MS of non-Polar Extracts
The components of non-polar extracts of
Gas Chromatography-Mass Spectrometry (GC-MS) Analysis of Volatile Compounds Extracted from Petals and Stigmas Saffron.

Bioavailability and Pharmacokinetics
SwissADME and ProTox-II were used to evaluate the selected compounds ADMET (absorption, distribution, metabolism, excretion, and toxicity) properties, including bioavailability and toxicokinetic characteristics. The analysis was conducted using the laws that establish the qualities of a drug, such as Lipinski, 14 Egan, 15 and Veber. 16 The compound must not violate more than one Lipinski rule, with a molecular weight (MW) < 500, number of hydrogen bond donors ≤ 5, number of hydrogen bond acceptors ≤ 5, topological surface area (TPSA) < 140, and water partition coefficient (WLOGP) ≤ 5.88. All fifteen compounds chosen for this study followed Lipinski's, Egan's, and Verber's rules, indicating favorable pharmacological properties (Table 2).

Abbreviations: ADMET, absorption, distribution, metabolism, excretion and toxicity, Lipophilicity a,Pharmacokinetics b, Drug Likeliness c, TPSA, topological polar surface area; nRB, no. of rotable bonds; nOHA, no. of H-bond acceptor; nOHD, no. of H-bond donor; WLOGP, water partition coefficient; GI absorption, gastrointestinal absorption; BBB, blood–brain barrier; P-gp, permeability glycoprotein; CYP, cytochrome P450; Entry 1: Valeric acid, 2: Linoleic acid, 3: Nonadecane,4: 1,3-Bis(trimethylsilyl) benzene, 5: Nonacosane, 6: Eicosane, 7: Heptacosane, 8: Cyclotrisiloxane, hexamethyl-, 9: E and Z isomers of 1-(26,6-Trimethyl-1-cyclohexen-1-yl)-3-methyl-2- heptane, 10: Safranal,11: 1,9 Tetradecadienne, 12: Z-14-Nonacosane, 13: Vitamin E, 14: 14b-Pregnane, 15: Hexadecyloxirane.
The results (Table 3) show that twelve of the fifteen compounds tested were not hepatotoxic, carcinogenic, cytotoxic, immunotoxic, or mutagenic. The bioavailability hexagons (Figure 1) and boiled egg prediction (Figure 2) were also used to demonstrate the drug-like properties and GI absorption of the selected compounds derived from

Bioavailability Hexagons of the Major Petals and Stigmas of

Boiled-Egg Graph of the Selected Phytoconstituents. 1: Valeric Acid, 2: Linoleic Acid, 3: Nonadecane,4: 1,3-Bis(Trimethylsilyl) Benzene, 5: Nonacosane, 6: Eicosane, 7: Heptacosane, 8: Cyclotrisiloxane, Hexamethyl-, 9: E and Z isomers of 1-(26,6-Trimethyl-1-cyclohexen-1-yl)-3-Methyl-2- Heptane, 10: Safranal, 11: 1,9 Tetradecadienne, 12: Z-14-Nonacosane, 13: Vitamin E, 14: 14b-Pregnane, 15: Hexadecyloxirane.
Toxicological Properties of Selected Compounds from Petals and Stigmas of Saffron.

Pr: Prediction, Pb: Probability, Ac: Active, In: Inactive Toxicity class: (Class I: fatal if swallowed (LD50 ≤ 5), Class II: fatal if swallowed (5 < LD50 ≤ 50), Class III: toxic if swallowed (50 < LD50 ≤ 300), Class IV: harmful if swallowed (300 < LD50 ≤ 2000), Class V: may be harmful if swallowed (2000 < LD50 ≤ 5000), Class VI: non-toxic (LD50 > 5000)).
Molecular Docking
Because it is the most frequently altered tumor suppressor gene in human malignancies, the protein p53 is often referred to as the “guardian of the genome”. It performs a number of regulatory functions by receiving, processing, and transmitting signals through a variety of pathways, including those governing cellular senescence, metabolism, inflammation, autophagy, and other processes that control the survival and eradication of aberrant cells.
17
This study focuses on the interaction of selected bioactive compounds derived from non-polar extracts of

Illustration of the Interactions and Binding Modes of Major Compounds with 1TUP.
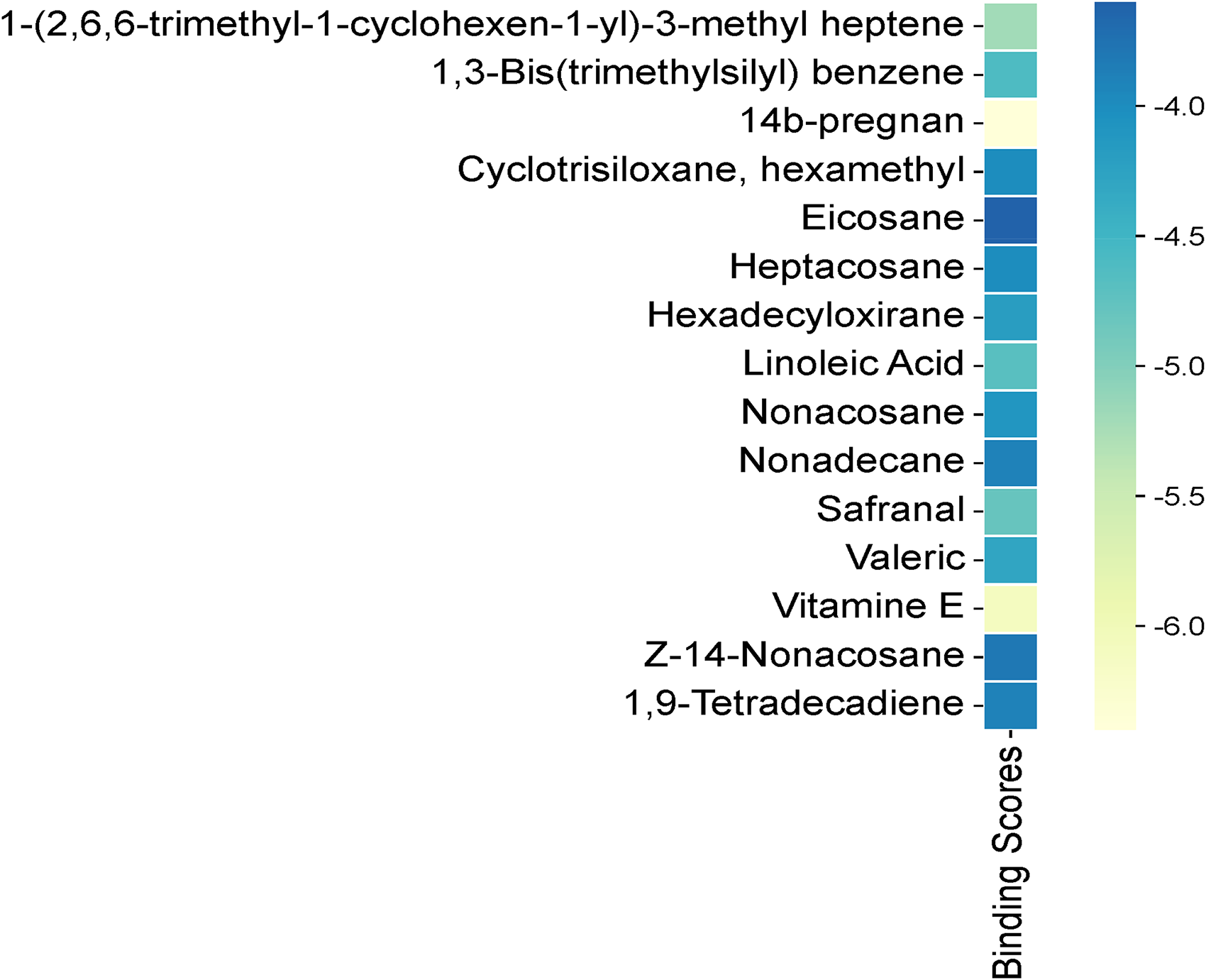
Heat Map of Binding Scores of Major Compounds Interacting with p53 Protein.
Discussion
Conventional extraction techniques have been used, including ultrasonic-assisted extraction with various solvents such as chloroform, petroleum ether, and hexane. Because no research had been done on saffron extract production using this method, this study was conducted to produce saffron extracts with different chemical compositions using three non-polar solvents, With the goal of developing an extraction method that can be used in the detection of new biomarkers to distinguish the origins of saffron from its geographical map.18,19 The GC/MS analysis revealed differences in chemical composition between the samples, which could be attributed to the different solvent systems. 20 However, in the GC/MS analysis of the petals and stigmas, we discovered a common component between the petal solvents, Eicosane, and another common component between the stigma solvents, 1,9-Tetradecadiene. Thus, all extracts (petals and stigmas) share a common component, Cyclotrisiloxane, Hexamethyl, albeit in varying amounts.
The results show that, for petals extracts, the presence of Heptacosane, Linoleic acid, and Nonacosane as main components is in partial agreement with the previous study; 21 in fact, their amounts are respectively 14.33, 11,37, and 9,93% against 2,96, 3,21, and 26,21% in our study. Linoleic acid, heneicosane, 1-docosene, and pentacosane were among the components found in trace amounts. When we compare our results to previous studies, 22 we find a significant disagreement, with only four common components found in varying amounts: Safranal, Eicosane, Isophorone, and Nonadecane (7.12, 5, 59, 0.60, and 1.55 against 38.17, 0.12, 0.18, and 0.17%, respectively). Another study 23 on polar stigma extract found that Heptacosane was present in a minor amount (0.48%), whereas this component was major in our study. Furthermore, the amount of Safranal is high in comparison to our study (7.12 vs 60%), as are Isophorone, Octadecane, Nonadecane, and Eiosane (0.6, 2.58, 1.55, and 5.59% vs 17.3, 0.1, 0.1, 0.1%, respectively).
According to the literature, the chemical composition of chloroformic, petroleum ether, and hexane extracts from
A comparison of the presented results reveals that a more non-polar stigmas extract contained the most volatile compounds; all of the above-mentioned compounds were discovered for the first time in the petals and stigmas grown in the El Outaya region.
Furthermore, the compounds (8 of 15) with LD50 > 2000 mg kg−1 suggest that they can be used for drug delivery or administering a pharmaceutical composition to achieve a therapeutic effect at the target site of the human body. Except for the compounds that inhibit the isoform CYP1A2, the selected compounds have been shown to be effective for drug delivery, transportation, and distribution. The compounds did not inhibit the majority of cytochrome P450 (CYP) isoforms, including CYP1A2, CYP2C19, CYP2C9, CYP2D6, and CYP3A4. The boiled egg model (Figure 2) was used to predict pharmacokinetic characteristics by evaluating passive gastrointestinal absorption (HIA) and brain penetration (BBB) as a function of the chosen compounds’ position in the WLOGP versus TPSA plot. 32 The findings also show that the compounds (3, 6, 9, 10, and 11) are in the gray area, indicating that they are unlikely to be well absorbed or cross the BBB. The compounds (1, 2, 4, 8, and 10) appear in the yellow ellipse, indicating their ability to cross the BBB. Five molecules (5, 7, 12, 13, 15) are not predicted to be absorbed or BBB permeant because they fall outside the range of the WLOGP versus-TPSA plot (TPSA = 0- 38.83 Å and WLOGP > 5).
Except for the out-of-range compounds, all of the selected compounds have a red point, indicating that they are not substrates of the P-gp efflux transporter, which plays an important role in drug absorption, distribution, and excretion. The ability of a compound to cross the BBB is an important consideration in determining its potential neurological impact. While it has the potential to treat central nervous system disorders, it also carries the risk of neurotoxicity and other adverse effects. As a result, careful assessment of BBB permeability is critical in drug development and understanding the effects of various substances on the nervous system. 33 The lack of adequate datasets restricts the computational methods for toxicity prediction. Toxicology prediction techniques must be improved as new compounds are introduced, and long-term exposure concerns grow. The toxicological profiles of chemical compounds can be predicted more quickly and affordably using molecular docking, which can be a useful substitute for dealing with large amounts of data. 34
The results show that 14b-pregnan (−6.4 kcal/mol) and vitamin E (−6.1 kcal/mol) have the highest binding affinities to targeted p53 (1TUP), indicating stable and energetically favorable interactions. Other compounds, such as 1-(26,6-trimethyl-1-cyclohexen-1-yl)-3-methyl heptene (−5.2 kcal/mol) and safranal (−4.8 kcal/mol), exhibit strong affinities. In contrast, some hydrocarbons, such as eicosane (−3.6 kcal/mol) and nonadecane (−3.9 kcal/mol), have lower docking scores, indicating weaker protein interactions
Similarly, 1,3-Bis(trimethylsilyl) benzene primarily forms π interactions with Arg174 and Pro177, indicating that this region is suitable for aromatic interactions. 14b-pregnan has π interactions with hydrophobic and aromatic residues, whereas cyclotrisiloxane has both conventional hydrogen bonds with Ile195 and Leu194 and π interactions with His214, indicating a combination of hydrophobic and hydrophilic interactions. Certain compounds, such as eicosane and heptacosane, interact with multiple hydrophobic residues using π interactions, indicating binding within extended hydrophobic pockets. Hexadecyloxirane exhibits π interactions with hydrophobic residues and forms a carbon-hydrogen hydrogen bond with Arg174, demonstrating a hybrid interaction pattern.
Linoleic acid forms a conventional hydrogen bond with Arg175 and interacts with hydrophobic residues via π interactions, indicating a region where hydrophilic and hydrophobic forces coexist. Nonacosane, nonadecane, and Z-14-nonacosane interact primarily through π interactions with hydrophobic residues, which strengthens their affinity for hydrophobic regions. Safranal binds to Arg174 and Tyr163 through π interactions and forms carbon-hydrogen hydrogen bonds with Val172 and Glu171, indicating an interaction between aromatic and hydrophilic residues. Meanwhile, valeric acid forms hydrogen bonds with Arg273 and Val274, as well as π interactions with Pro250, indicating a preference for a hydrophilic environment. Finally, 1,9-tetradecadiene interacts with multiple hydrophobic residues via π interactions, indicating preferential binding in a hydrophobic or semi-hydrophobic environment. Overall, π interactions and hydrogen bonds, whether conventional or carbon-hydrogen, are important determinants in ligand recognition and binding to the central domain of targeted p53, influencing the stability and function of the resulting complexes.
Conclusion
This study employed ultrasonic extraction and GC/MS analysis to assess the qualitative and quantitative chemical variation in the non-polar extracts of saffron (
Footnotes
Acknowledgements
We would like to thank MESRS (Ministère de l’Enseignement Supérieur et de la Recherche Scientifique, Algeria) and DGRSDT (Direction Générale de la Recherche Scientifique et du développement Technologique, Algeria) for their financial support.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data are available from the corresponding author.
