Abstract
Objectives
Immortelle has garnered global attention for its cosmetic, medicinal, and culinary applications worldwide. Apart from neryl acetate, α-pinene and γ-curcumene as the most important components for the estimation of the essential oil quality, in recent years a group of β-diketones, known as italidiones, appear as significant quality parameters. However, it is unknown how weather conditions (temperature and precipitation) influence their accumulation in essential oil and hydrosol, nor their anti-inflammatory potential.
Methods
This study investigates the chemical composition of immortelle essential oil and hydrosol over three successive years (2019/20, 2020/21, and 2021/22), by GC-FID and GC-MS analysis. In silico molecular docking model with Vascular Adhesion Protein-1 (VAP-1) was used for the simulation of the anti-inflammatory potential of italidiones, as well as other β-diketones present in immortelle essential oil and hydrosol.
Results
During the three-year trial, it was established that essential oil compounds such as α-pinene, neryl acetate, and italicene were positively correlated with temperature and negatively correlated with precipitation, as well as β-diketones and total italidiones. Moreover, trans-caryophyllene, γ-curcumene, and ar-curcumene were in negative correlation with temperature, and a slight positive correlation with precipitation. On the other side, in the hydrosol the content of both total β-diketones and total italidiones was strongly negatively correlated with temperatures, and positively correlated with precipitation, which was the opposite of essential oil. Additionally, findings indicated that italidiones could bind the human VAP-1 protein in the vicinity of the topiquinone and block its function.
Conclusion
Weather conditions affect variations in the accumulation of volatile compounds in essential oil and hydrosol, which in some cases can affect changes in olfactory characteristics, as well as criteria prescribed by standards, and even changes in biological activities. These results indicate that italidiones, as well as other β-diketones present in immortelle, may be responsible for its anti-inflammatory activity.
Introduction
Immortelle (Helichrysum italicum (Roth) G. Don; fam. Asteraceae), known as everlasting, curry plant or smilje (in ex-Yugoslavian countries), is a perennial xerophytic plant, with a characteristic scent, grows as a small shrub, with elongated hairy leaves and yellow inflorescences appearing from June to August. 1 Although the genus Helichrysum has almost 600 species, only H. italicum has commercial importance and represents the most commonly cultivated species of the genus throughout the world. 2
Flowering aerial parts have been widely used in the Mediterranean region since ancient times for seasoning and flavoring food, in traditional medicine against digestive and liver disorders, and for care and treatment of irritated skin and wounds.3–6 Modern science approved that immortelle possesses antioxidant, antimicrobial, anti-inflammatory, anti-proliferative, anti-collagenase, and anti-elastase properties,7–11 which legitimate the worldwide use of immortelle in the food, cosmetic, and pharmaceutical industries.
In the last decade, the interest of farmers, industry, and consumers in immortelle has been growing. 12 From 2013 to 2021, the areas under this plant cultivated in Serbia have increased from 40 to over 200 ha. 13 Immortelle grown in Serbia and neighborhood regions (ex-Yugoslavia countries), is well adapted to climatic conditions, and have an interesting chemical composition with γ-curcumene, ar-curcumene, α-pinene, and β-selinene as dominant compounds.14,15
The impact of climate change on medicinal and aromatic plants and their biologically active constituents is very significant. 16 Scientists worldwide have already begun to highlight the importance of this problem.17,18 Previous studies reported the influence of weather conditions on the accumulation of volatile compounds in the essential oil of anise, caraway, chamomile, citronella, coriander, hyssop, lavandin, and others.19–26 However, the influence of weather conditions on the accumulation of volatile compounds in hydrosol is much less studied, probably because they are by-products, so there are data only for commercially important hydrolate such as lavandin. 24
Despite the abundant scientific data on immortelle essential oil's chemical composition, no official ISO standard regulates its quality; however, Corsican and Balkan types of essential oil have been unofficially recognized on the market.27,28 Furthermore, only a few studies reported immortelle hydrosol.2,12,29–32 Therefore, this investigation aimed to conduct an analysis of immortelle essential oil and hydrosol obtained by steam distillation on a semi-industrial scale, from the same plantation, during three successive years (2019/20, 2020/21 and 2021/22). In addition, this study investigated the influence of weather conditions (temperature and precipitation) on the accumulation of individual compounds both in the essential oil and the hydrosol, with a focus on italidiones (46,9-trimethyl-8-decene-3,5-dione; 2,4,6,9-tetramethyl-8-decene-3,5-dione; and 3,5,7,10-tetramethyl-9-undecene-4,6-dione). Italidiones, as biologically active molecules, stand out as highly valued. However, scientific data on their activity are limited. Therefore, another goal of this research was in silico molecular simulation docking with Vascular Adhesion Protein-1 (VAP-1) and italidiones, as well as other β-diketones from immortelle essential oil and hydrosol (2,4-dimethylheptane-3,5-dione, and 4,6-dimethyloctane-3,5-dione).
Materials and Methods
Plant Material
Immortelle was grown at the Institute of Field and Vegetable Crops Novi Sad (IFVCNS), Department of vegetable and alternative crops, Bački Petrovac, during three successive seasons (2019/2020, 2020/2021 and 2021/2022) on chernozem-calcareous clay soil. A representative sample of the species was deposited at the herbarium of the University of Novi Sad under voucher number 2-1375.
Under Serbia's agroecological conditions, Immortelle provides two harvests a year: the first in mid-July and the second at the end of August. 14 After harvesting, the plant material was dried in a solar dryer to constant mass and stored in natron bags in a dark, dry place. A mixture of plant material from both harvests within one year (a common practice among immortelle producers in Serbia) was used for steam distillation in a small-scale distillation unit at the IFVCNS.
Weather Conditions
The data for weather conditions used in this study are obtained from meteorological station located at the IFVCNS, Department of vegetable and alternative crops, Bački Petrovac.
Three successive growing seasons (2019/2020, 2020/2021 and 2021/2022) had significantly different sums of precipitations and average monthly temperatures (Figure 1). Overall, the third growing season (2021/22) had the highest average mean daily temperature (13.8 °C), and the lowest amount of precipitation (487.9 mm). The first (2019/20) and second (2020/21) growing seasons had uniform temperatures (13.1 and 13.0 °C, respectively), and the total amount of precipitation (681.3 and 643.5 mm, respectively).

The weather conditions during the three successive growing seasons (Meteorological station at IFVCNS experimental field, Bački Petrovac).
After the harvest in August, the plants prepared for winter dormancy from September to November. This autumn period during the first growing season was the warmest, while the coldest and with the most precipitation was the third growing season. During the winter period (December – February) the average daily temperatures were between 3.5 °C and 5.2 °C, while the sum of precipitations in the first and third growing seasons was uniform (101.7 and 102.8 mm, respectively), and significantly higher in the second growing season (156.7 mm). On average, the coldest weather for the awakening of vegetation (March – May) was in the second growing season (11.1 °C on average), while the warmest conditions with the least precipitation were recorded in the third growing season. The summer period (June – August) was the warmest and had the least precipitation in the third growing season, while the coldest and with the most precipitation was the first growing season (Figure 1).
Essential Oil and Hydrosol Extraction
Dried flowering aerial parts of immortelle were processed by steam distillation. Approximately 100 kg of plant material was placed in a distillation tank made of stainless steel (volume 0.8 m 3 ), and then covered with a lid for every distillation. The lid is closed tightly by nut-bolts and supplied with steam produced by a high-pressure boiler. After passing through the plant material, the steam with volatile compounds goes to the cooler and condenser connected to a receiver flask. The essential oil and hydrosol (aromatic water) collected in the receiver flask were separated based on weight; essential oil floats on the hydrosol surface as it is lighter than water, it is decanted and then cleaned and dried in a separatory funnel with the addition of sodium sulphate. The obtained hydrosol is simply filtered through the filter paper and packed into sterile plastic bottles.
The essential oil yield was calculated as grams of oil per 100 g of dry immortelle flowers. The average immortelle essential oil yield was estimated to be 0.3%, and the mass of 100 ml of essential oil was estimated to be 89.7 g.
The simultaneous distillation-extraction (SDE) technique using the Likens-Nickerson apparatus was applied prior to the analysis of volatile compounds in hydrosol. Briefly, 400 ml of hydrosol was placed in one boiling flask, and 5 ml of dichloromethane was added. After 2 h, volatile compounds were isolated from hydrosol for further qualitative and quantitative analysis.
Analysis of Volatile Compounds
Volatile compounds from immortelle essential oil and hydrosol were analyzed by gas chromatography equipped with a flame ionization detector (GC–FID) and gas chromatography coupled to a mass spectrometry system (GC–MS). The analysis was performed using an Agilent 7890 GC, Agilent 5975C MSD and FID on non-polar HP-5MS fused silica capillary column Agilent 19091S-433 (conditions were mimicked from Adams, 33 ). Helium was used as a carrier gas, with an inlet pressure of 19.6 psi and 1 mL min−1 at 210 °C linear velocity. The injector temperature was set to 250 °C, the injection volume was specified as 1 μL, and the split ratio was 10:1. The source temperature for MS detection was set to 230 °C, and the interface temperature was specified as 315 °C. Electron Impact (EI) mode was set at 70 eV with a mass scan range specified as 40–600 amu. The temperature was programed to change during the analysis, from 60 °C to 300 °C at a rate of 3 °C min−1. The identification of constituents was carried out based on the linear retention index relative to C8–C32 n-alkanes and compared with reference spectra from a database of Faculty of Chemistry, University of Belgrade (Wiley 7, NIST 17 and retention time locked Adams 4 databases, using Automated Mass spectral Deconvolution and Identification System (Amdis 32 ver. 2.73) and NIST search ver. 2.3). The relative abundance of each constituent in the sample was determined by measuring the peak area in the GC-FID chromatogram, and was expressed as percentages by peak area normalization.
Molecular Docking
All simulations were performed using the Schrödinger package (Schrödinger Release 2023-3) and OPLS4 force field. 34 The 3D structure of VAP-1 was retrieved from the Protein Data Bank database (PDB ID: 4BTX). 35 The downloaded protein structure was further prepared for molecular modeling analysis by the addition of hydrogen atoms, determination of protonation types, and termini treatment (Schrödinger Release 2023-3). The tridimensional structures of examined compounds from hydrosol were constructed using the Maestro program in the Schrödinger package. The geometry optimizations of compounds used for molecular docking simulation and VAP-1 protein were performed using the OPLS4 force field and the Powell conjugated gradient algorithm method, with a convergence criterion of 0.01 kcal/(mol Å) and a maximum number of 1000 iterations.
The molecular docking simulations were performed with the program Glide, 36 with extra precision mode and flexible ligands. Epik state penalties were used to calculate the docking scores. Ligand binding affinities were calculated using the MM-GBSA method and the VSGB 2.0 solvation model. 37 The residues within a distance of 4.0 Å from the ligand were categorized as flexible. The results obtained were visualized using the programs Maestro and PyMol.
Statistical Analysis
A linear regression model was formed to predict the immortelle essential oil and hydrosol volatile compound content and hydrosol composition during three years according to temperature and precipitation data, and the appropriate regression coefficients were calculated using the linear formula: x = a + KT T + KP P, where x was essential oil active compound content or hydrosol active compound content, a was the intercept, KT and KP were temperature and precipitation coefficients, while T and P were temperature and precipitation amounts, respectively. The collected data were processed statistically using the software package STATISTICA 10.0.
Results and Discussion
Volatile Constituents from Immortelle Essential Oil
The volatile compounds of immortelle essential oil from three successive growing seasons determined by GC-MS analyses are shown in Table 1. There were 71 identified compounds in 2020 representing 96.2%, 73 compounds in 2021 comprising 96.6%, and 72 in 2022 comprising 96.0%. The main compounds were γ-curcumene (16.3-20.5%), followed by β-selinene (6.8-9.1%), trans-caryophyllene (6.5-7.1%), neryl acetate (5.9-8.1%), ar-curcumene (5.6-8.4%), α-pinene (4.6-6.8%) and italicene (4.6-5.1%). Compounds such as α-pinene, neryl acetate, and italicene were in positive correlation with temperature (2.479, 1.438 and 0.290, respectively) and in negative correlation with precipitation (−0.098, −0.104 and −0.005, respectively), while β-selinene was in positive correlation with both weather condition parameters (Kt = 0.580 and Kp = 0.015). On the other side, trans-caryophyllene, γ-curcumene, and ar-curcumene were in negative correlation with temperature (−0.784, −1.653 and −3.853, respectively), and slight positive correlation with precipitation (0.035, 0.001 and 0.182).
Volatile Compounds of Immortelle Essential Oil.
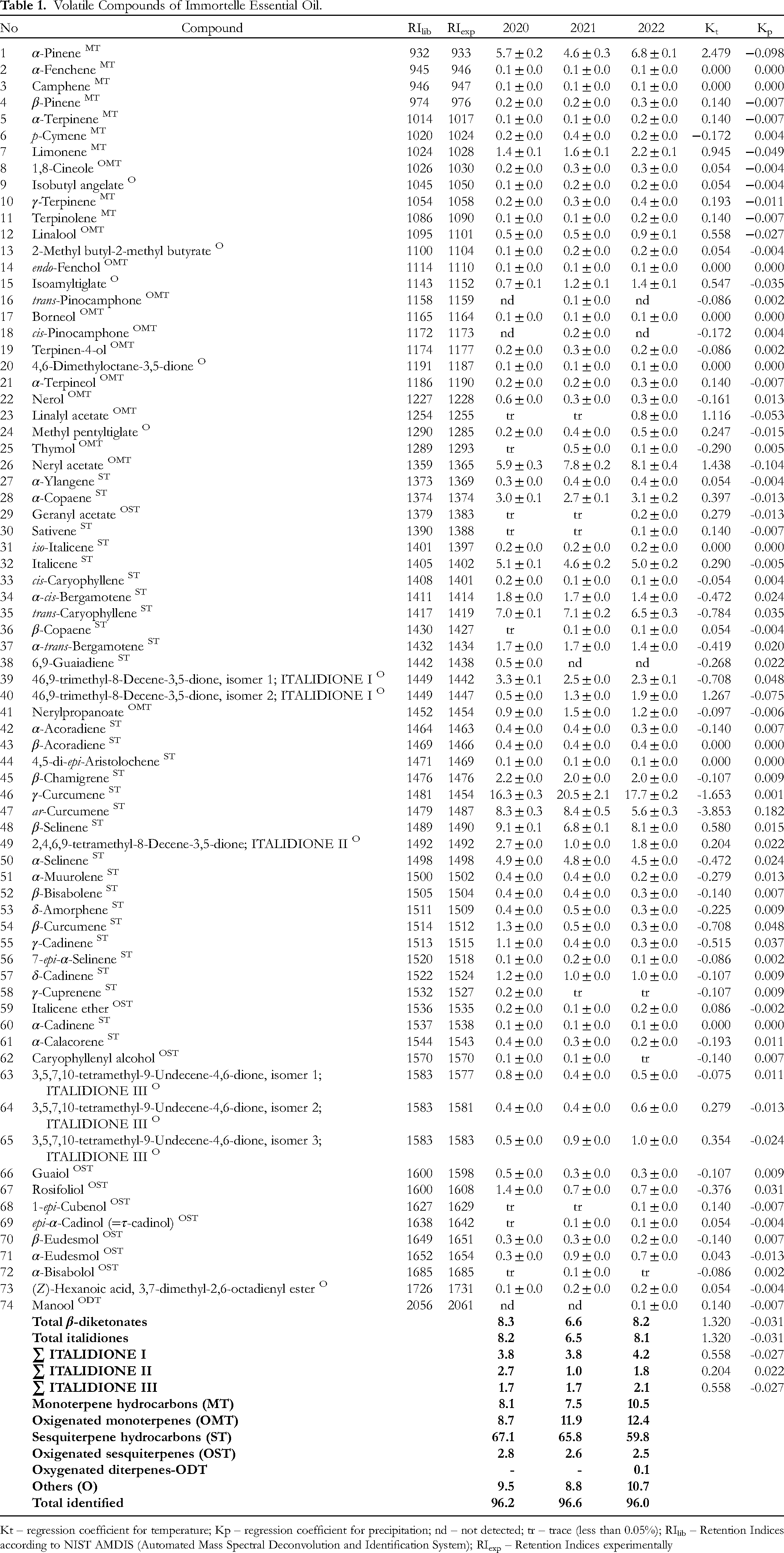
Kt – regression coefficient for temperature; Kp – regression coefficient for precipitation; nd – not detected; tr – trace (less than 0.05%); RIlib – Retention Indices according to NIST AMDIS (Automated Mass Spectral Deconvolution and Identification System); RIexp – Retention Indices experimentally
A total of seven β-diketones and their isomers were determined in essential oil (comprises between 6.6 and 8.3%), of which six belong to italidiones (6.5-8.2%); two isomers italodione I, one isomer italodione II and three isomers italodione III. In general, it can be said that temperatures were strongly positively correlated with β-diketones and total italidiones (Kt = 1.320), while precipitation was slightly negatively influenced (Kp = -0.031). A similar regularity can be observed in the sum italidiones I, II and III. However, no regularity can be observed for individual italidiones isomers. Practically, some isomers have a positive correlation with precipitation and a negative correlation with temperatures, while in others, the opposite trend was noticeable.
According to previous studies, immortelle essential oil composition depends on genotype/ecotype (ie origin) and seasonality/environmental conditions, developmental stage, plant organs, and postharvest processing, as well as an extraction technique. 2 Taking into account dominant compounds in immortelle essential oil from this study, this variety belonged to γ-curcumene chemotype, well adapted to continental climatic conditions.14,15 γ-Curcumene as the dominant compound in immortelle essential oil (with more than 15%) was also noted in other Balkan countries such as Croatia,38,39 Bosnia and Herzegovina,40,41 Montenegro, 42 Serbia,10,43 and Bulgaria,9,44 as well as in Italy,45–47 France, 48 and Portugal,31,49 but also introduced to USA. 50
Several authors have previously described seasonal variations of immortelle essential oil, but climatic conditions and stages of development (floral budding and full flowering) give differences in chemical composition.14,44,51–55 Further, investigations showed that plant organs,56,57 as well as postharvest processing and extraction techniques also influence immortelle essential oil composition.58–60
In general, previous studies indicated that environmental factors such as altitude, temperature and precipitation affect the accumulation of volatile compounds in the essential oil, which further determines its biological potential.14,53–55 The widespread chemotype in the western Balkan countries belongs to curcumene rich type,28,61 and spring and summer precipitation positively influenced its accumulation in plants, probably as an adaptation to climatic conditions. 14 This study further confirms that key compounds γ-curcumene, β-selinene, trans-caryophyllene, neryl acetate, ar-curcumene, α-pinene, and italicene show varying correlations with temperature and precipitation, indicating that environmental factors can influence the composition of the essential oil.
Volatile Constituents from Immortelle Hydrosol
The volatile compounds of immortelle hydrosol from three successive growing seasons determined by GC-MS analyses are shown in Table 2. There were 72 identified compounds in 2020 representing 90.2%, 57 compounds in 2021 compromising 98.4% and 56 in 2022 comprising 96.4%. The main compounds were β-diketones: italodione I (in the sum of all isomers from 18.7 to 27.8%), and 4,6-dimethyloctane-3,5-dione (in the sum of all isomers between 8.6 and 13.9%). A total of 20 β-diketones and isomers were determined in hydrosol, comprising between 29.7 and 46.4%. However, 15 of them are italidiones, comprising between 19.2 and 29.1%. Generally, the content of both total β-diketones and total italidiones was strongly negatively correlated with temperatures (−7.846 and −5.828, respectively), and positively correlated with precipitation (0.682 and 0.449, respectively), which was the opposite of essential oil.
Volatile Compounds of Immortelle Hydrosol.
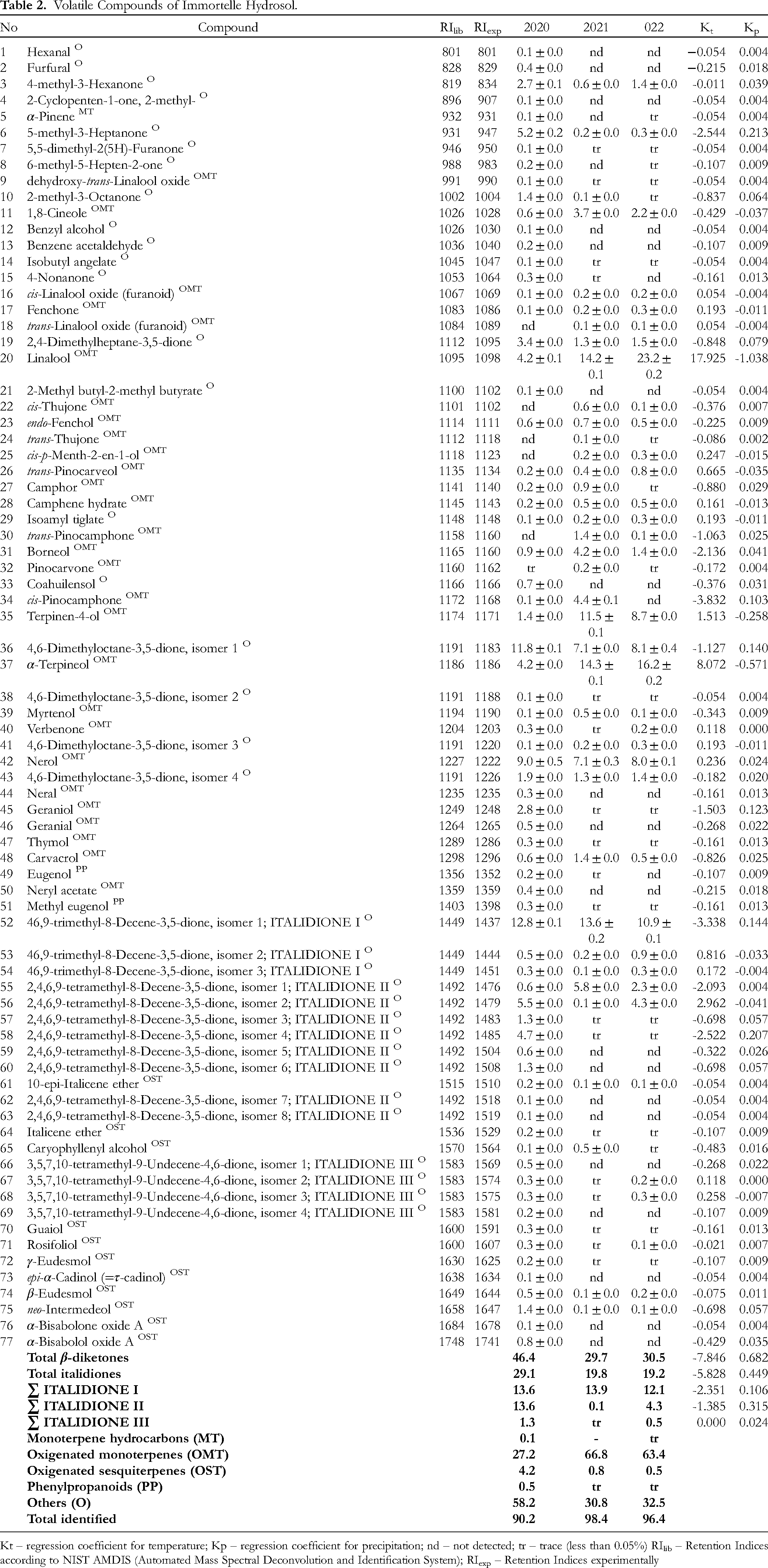
Kt – regression coefficient for temperature; Kp – regression coefficient for precipitation; nd – not detected; tr – trace (less than 0.05%) RIlib – Retention Indices according to NIST AMDIS (Automated Mass Spectral Deconvolution and Identification System); RIexp – Retention Indices experimentally
In a previous study, the compounds detected in five samples of hydrosols from plants cultivated in Slovenia were: 2,2-dimethylnon-5-en-3-one (8.9-18.4%), α-terpineol (7.7-19.6%), 2,6-octadien-1-ol (4.4-19.1%), pentan-3-one (2.6-10.1%), 3-octanone (1.9-12.3%), isopropyl ethyl ketone (1.8-3.0%), 1-(1,1-dimethylethoxy)-2,2-dimethyl-propane (1.6-2.5%), borneol (1.3-5.1%), and 3,6-dimethyl-decane (1.3-3.5%). 12 Further analysis showed that the main compounds in Slovenian immortelle hydrosol were 1,8-cineole (31.5-55.5%), α-terpineol (12.9-16.6%), terpinen-4-ol (13.6-15.1), linalool (8.4-11.2%), eugenol (2.6-4.7%) and 8-decene-3,5-dione,46,9-trimethyl (2.7-4.4%), depending on the method utilized for hydrosol analysis (direct analysis or extraction with organic solvent). 29 In addition, GC-MS analysis of hydrosol obtained from immortelle originating from Bosnia and Herzegovina showed the presence of 19 volatile compounds, and β-himachalene was most abundant with 20.9%, followed by ar-curcumene (13.6%) and neryl acetate (13.0%). 2 On the other hand, immortelle hydrosol accession originating from Corsica, France, contains nerol (23.7%) as a dominant compound, followed by β-fenchyl alcohol (18.4%) and neryl acetate (10.8%). 2 According to these studies, it can be concluded that the composition of essential oils significantly influences hydrosol composition. However, only highly polar (hydrophilic) compounds dissolve and pass into the hydrosol. 62
Italidiones from Immortelle Essential Oil and Hydrosol
β-Diketones (Supplementary Figure 1) are valuable compounds with good antioxidant capability, antimicrobial activity, and prominent anti-cancer properties. Therefore, they are widely used in the treatment of many pathological disorders. 63 All β-diketones occur in many tautomeric forms. 64 They can be found in many plants, widely used in everyday nutrition worldwide and self-medication, such as ginger, 65 curcuma, 66 St. John's wort, 67 and many others.
The italidiones (a group of isomeric β-diketones) are found only in immortelle. 9 They are well soluble in water, 68 therefore, their concentration was significantly higher in the hydrosol. Table 3 shows the presence of β-diketones and italidiones in samples from the available literature. As can be seen, 27 papers refer to these components in immortelle essential oil. On the other hand, Table 4 refers to the presence of β-diketones and italidiones in immortelle hydrosol. Considering that the scientific public's interest in hydrosols is recent, only two papers referred to these components. However, recently Komnenić et al 32 investigated multiple liquid-liquid extraction of dissolved compounds in immortelle hydrosol with four different solvents (chloroform, dichloromethane, hexane, and benzene). They noted presence of italidione I from 1.2 to 118.3 mg/L, italidione II from 0.1 to 33.4 mg/L and italidione III up to 39.6 mg/L.
Literature Review of β-Diketones Detected in Immortelle Essential Oil (%) (from Newest to Oldest).

mixture with α-Farnesene as co-eluting component, nd – not detected
Literature Review of β-Diketones Detected in Immortelle Hydrosol (%) (from Newest to Oldest).

nd – not detected
Apart from italidiones, two more β-diketones were detected in immortelle essential oil and hydrosol: 2,4-dimethylheptane-3,5-dione and 4,6-dimethyloctane-3,5-dione. β-Diketones are a group of secondary metabolites isolated from other plants and animals, while italidiones are characteristic only for plants from the genus Helichrysum. For example, 4,6-dimethyloctane-3,5-dione was present in Tetraclinis articulata, 81 and in this study, its content in immortelle essential oil was low (0.1%), while in hydrosol it was significantly higher (between 8.6-13.9%). On the other side, 2,4-dimethylheptane-3,5-dione was isolated from Anguilla myriaster. 82 However, this compound is responsible for an unpleasant odor of the immortelle stems. 83 On the other hand, both of these compounds refer to specific spice/curry odor. 64 In this study, the presence of 2,4-dimethylheptane-3,5-dione was confirmed only in hydrosol (between 1.3-3.4%), while some other authors referred to its presence in essential oil, too.59,84
Even though hydrosols are by-products of the essential oil distillation process, they possess valuable biological activities – antioxidant, antimicrobial, and antiadhesive potential, but significantly lower than essential oil because of dilution. 30 However, immortelle hydrosol on the European market is mainly sold as cosmetic ingredients or cosmetic products for skin care. 29 Bearing in mind the high amount of italidiones in hydrosol, it can be a valuable raw material for further industrial processing - recovering (which is used for some expensive oils such as rose) or further isolation of pure compounds, or even for direct use in the cosmetic formulation, similar as immortelle essential oil.
Molecular Docking
The essential oil of immortelle is known for its anti-inflammatory effects. 85 Although most research has investigated the anti-inflammatory effects of various extracts, especially essential oils, there is little data on the anti-inflammatory effects of its hydrosol. Immortelle hydrosol is often used as an anti-inflammatory agent, especially for skin treatment. 86 The hydrosol contains bioactive compounds similar to essential oils, but in a different percentage, and additionally some compounds that are not present in the essential oils. The presented results of the composition of the immortelle essential oil and the hydrosol showed differences, especially in the content of italidiones. Therefore, in silico methods to investigate the possibility of the most abundant italidiones in immortelle hydrosol to express anti-inflammatory activity through inhibition of well-known anti-inflammatory target molecule - human vascular adhesion protein 1 (VAP-1) were used.
Analysis of the crystal structure of VAP-1 in complex with the known inhibitor pyridazinone (PDBID: 4BTX) revealed that the hydrophobic phenyl ring binds deeply into the binding pocket and blocks the enzymatic activity of VAP-1 (Figure 2a). Molecular docking simulations revealed that the italidiones identified in samples might bind similarly to the proven VAP-1 inhibitor due to their highly hydrophobic parts as well as the polar groups that interact with the side chain of the protein. In addition, the calculated binding energies indicate strong binding of the analyzed compounds. According to the obtained results, all three italidione compounds can bind the human VAP-1 protein in the vicinity of the topiquinone and block its function (Figure 2). In addition, two β-diketones also have favorable orientations and binding energies.

Human Vap-1 protein in complex with: a) the known inhibitor pyridazinone, crystal structure PDBID: 4BTX; b) italidione II; c italidione I; d) italidione III; e) 2,4-dimethylheptane-3,5-dione; f) 4,6-dimethyloctane-3,5-dione.
Due to the similar overall structure and the similar positions of the OH groups, all examined compounds bind to the VAP-1 protein in a similar orientation. All compounds bind in the hydrophobic pocket, and all can form polar interactions with Thr212. The italidiones can bind deep into the hydrophobic pocket via hydrophobic dimethyl tails (Figure 2). Italidione II has the highest predicted binding energy among the analysed italidiones (−29.4 kcal/mol). In addition to the polar interaction with Thr212, it was able to form hydrophobic interactions with Phe173, Tyr176, Tyr394, Phe389, Leu468, Leu469 and Tyr384 (Figure 2b). Although the second OH group does not interact with the protein, it is orientated towards the solvent, allowing it to form polar interactions with water molecules. Italidione I interacts similarly to italidione II, with the identical residues, but with a lower predicted binding energy (−21.25 kcal/mol) (Figure 2c). Italidione III binds VAP-1 with a predicted binding energy of −18.53 kcal/mol, which is the lowest predicted binding energy among the analysed compounds (Figure 2d). Its binding differs from that of italidiones I and italidiones II as it is able to bind Phe227 but is unable to interact with Phe173. 2,4-Dimethylheptane-3,5-dione has the highest predicted binding energy among the analysed compounds (−38.38 kcal/mol) (Figure 2e). In addition to its polar interaction with Thr212 and hydrophobic interactions with Phe173, Tyr176, Tyr394, Phe389, Leu368, and Leu469, it is the only studied compound that can interact hydrophobically with Met211. Its second OH group does not interact with the protein and can interact with water molecules from the solvent, which is favorable for binding. 4,6-Dimethyloctane-3,5-dione also has a high predicted binding energy (−34.97 kcal/mol) (Figure 2f). In addition to the polar interaction with Thr212 and the hydrophobic interactions with Phe173, Tyr176, Tyr394, Phe389, and Leu469, its second OH group can form polar interactions with Tyr394, making it the only investigated compound where both OH groups can interact with the protein.
These results indicate that italidiones, as well as other β-diketones present in immortelle, may be responsible for its anti-inflammatory activity. Their high concentration in hydrosol, compared to essential oils, recommends it for anti-inflammatory treatments and the production of cosmetics. Moreover, good water suitability makes them suitable for use in water-based cosmetic products such as gels and lotions. 56
Limitations of the Study
The results demonstrated that immortelle, as typical xerophytic plant (plant adapted for life and growth with a limited water supply), is well adapted to different climatic conditions. Compounds such as α-pinene, neryl acetate, and italicene were in positive correlation with temperature and in negative correlation with precipitation, while trans-caryophyllene, γ-curcumene, and ar-curcumene were in negative correlation with temperature and slight positive correlation with precipitation. These results show that climatic factors affect the geographic regionalization of the immortelle; ie the Corsican type (rich in neryl acetate) dominates in Mediterranean climates (warm and rainy winters and hot and dry summers), and the Balkan type (rich in γ- and ar-curcumene) is present in continental climates (characterized by hot summers, cold winters and little rainfall). The main limitation of this study is that only one chemotype was used in the research, and to create a more complete picture, it would be desirable to include more samples and chemotypes.
In addition, the research shows that the hydrolate obtained as a by-product of essential oil distillation can be a very important raw material for further industrial processing due to the high content of β-diketones and italidiones. Given the great interest of the cosmetic industry in these compounds, in silico molecular modeling has shown that they have a great anti-inflammatory potential. This could be considered a good basis for further research into the in vitro and in vivo biological potential of pure compounds. Therefore, further research will be focused on the isolation of italidiones and other β-diketones as potential anti-inflammatory compounds from the immortelle essential oil and hydrolate.
Conclusions
This study comprehensively analyzes immortelle (Helichrysum italicum), focusing on its essential oil and hydrosol. Ther findings demonstrate that the chemical composition of immortelle essential oil remains constant over time, with key constituents such as γ-curcumene, β-selinene, trans-caryophyllene, neryl acetate, ar-curcumene, α-pinene, italicene, and β-diketones. This stability underscores the reliability of immortelle essential oil for various applications. However, the study reveals significant variations in temperature and precipitation across three growing seasons, confirming that γ-curcumin and ar-curcumin have a negative correlation with temperature and a slight positive correlation with precipitation. In contrast, the hydrosol exhibited a unique profile dominated by 20 β-diketones and their isomers, accounting for 29.7% to 46.4% of its composition. This distinct composition points to the hydrosol's potential in different applications, particularly in cosmetic formulations and anti-inflammatory treatments. Molecular docking simulations further support the hydrosol's therapeutic potential, particularly the anti-inflammatory activity of italidiones, which effectively bind to human vascular adhesion protein 1 (VAP-1). These insights into the bioactive components of immortelle hydrosol open new avenues for its use in formulations for anti-inflammatory therapies. This study contributes to the comprehensive understanding of immortelle's chemical dynamics, highlighting its commercial importance and potential applications in various industries. The anti-inflammatory potential of italidiones opens avenues for further exploration in therapeutic formulations, emphasizing the need for continued research in this promising botanical resource.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241284735 - Supplemental material for The Influence of Weather Conditions on the Immortelle Volatile Constituents from Essential oil and Hydrosol with a Focus on Italidiones and Its Molecular Docking Anti-Inflammatory Potential
Supplemental material, sj-docx-1-npx-10.1177_1934578X241284735 for The Influence of Weather Conditions on the Immortelle Volatile Constituents from Essential oil and Hydrosol with a Focus on Italidiones and Its Molecular Docking Anti-Inflammatory Potential by Milica Aćimović, Jovana Stanković Jeremić, Marina Todosijević, Mirjana Cvetković, Biljana Lončar, Vladimir Vukić, Tamara Erceg, Lato Pezo, and Valtcho D. Zheljazkov in Natural Product Communications
Footnotes
Acknowledgements
The authors would like to thank Dr Vladimir Sikora, IFVCNS, Dr Milica Rat, Faculty of Science, University of Novi Sad, and prof. Ljubodrag Vujisić, Faculty of Chemistry, University of Belgrade, for technical support during the experiment.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
This work is financially supported by the Ministry of Science, Technological Development and Innovation of the Republic of Serbia, grant signed with Institute of Field and Vegetable Crops Novi Sad (No: 451-03-66/2024-03/200032), Institute of Chemistry, Technology and Metallurgy, University of Belgrade (No: 451-03-47/2023-01/200026), Faculty of Chemistry, University of Belgrade (No: 451-03-68/2024-14/200168), Faculty of Technology, University of Novi Sad (No: 451-03-47/2024-01/200134), and Institute of General and Physical Chemistry (No: 451-03-66/2024-03/200051).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
