Abstract
Breast cancer is the leading cause of cancer mortality in women. In this study, liriodenine and lysicamine from Goniothalamus elegans Ast. were investigated for their anti-breast cancer activity based on their molecular interactions with three proteins related to breast cancer. Liriodenine had predicted binding affinities for BRCA1, BRCA2, and estrogen receptor alpha of −6.2, −7.9, and −8.3 kcal/mol, respectively. Lysicamine had predicted binding affinities of −5.8, −7.2, and 7.6 kcal/mol. To evaluate the biological activity of liriodenine and lysicamine, we studied their in vitro cytotoxic effects on MCF-7 cells. These alkaloids showed significant inhibitory effects with IC50 values of 33.31 and 70.03 µM. These results suggest that Goniothalamus elegans could be a promising medical plant for breast cancer treatment. Further studies are needed to understand the molecular mechanisms and improve the toxicity of liriodenine and lysicamine for clinical use.
Introduction
According to the WHO, breast cancer is a prevalent cancer in women, with 2.3 million cases diagnosed and 685,000 deaths globally in 2020. Incidence rates are 88% higher in developed countries such as Australia, Western Europe, and North America (55.9 per 100,000 on average). However, developing countries such as Melanesia, Western Africa, and Polynesia have 17% higher mortality rates, at 15.0 per 100,000. 1 Breast cancer is closely associated with BRCA genes and estrogen receptor α (ERα). The risk of developing breast cancer by the age of 70 for BRCA1 and BRCA2 mutation carriers is 65% and 45%, respectively. 2 In addition, approximately two-thirds of breast cancers express ERα.3,4
The BRCA genes BRCA1 and BRCA2 were discovered in mammalian genomes. They were first determined to be breast cancer susceptibility genes in the 1990s.5,6 Since then, further studies have clarified their role and associated morphological patterns in breast cancer. The BRCA1 gene, which is localized to the long arm of chromosome 17, is distributed over approximately 100 kb of genomic DNA, and is composed of 24 exons, and encodes a protein containing 1863 amino acid residues. A sequence near the N-terminus is structurally similar to zinc (RING) finger domains and is able to interact with BARD1 to mediate BRCA1-associated tumor suppression and BAP1 to mediate BRCA1-associated inhibition of breast cancer cell growth. The middle part of BRCA1, encoded by exons 11–13, interacts with various proteins involved in transcription, DNA repair, and cell cycle progression. The C-terminal domain interacts with transcription regulators such as p53 and BACH1, and DNA damage repair proteins such as CtIP and CCDC98. The BRCA2 gene, which is localized to a 6-centimorgan interval on chromosome 13q12–13, is roughly 70 kb in length, is composed of 27 exons, and encodes a protein containing 3418 amino acid residues. The N-terminus of the BRCA2 protein is a transcriptional activation domain. The BRC motif, which is found in the middle part of exon 11, and the C-terminal domain, interact with RAD51 in the context of BRCA2-mediated DNA repair.7-9
BRCA1 and BRCA2 bind to many DNA repair proteins such as RAD51, BASC, and H2AX. For example, BRCA1 co-localizes with RAD51 to facilitate homologous recombination signaling and guarantee that double-strand breaks are not repaired until RAD51 is formed. Simultaneously, BRCA2 binds directly to RAD51 to guide it to sites of DNA damage. When BRCA genes are mutated, DNA is not appropriately repaired, leading to DNA mutation. If too many new mutations arise in cancer driver genes, tumorigenesis can occur. By interacting with transcription factors such as c-myc and TP53, BRCA1 and BRCA2 operate as transcriptional regulators of specific target genes. Patients with either BRCA1 or BRCA2 mutations are more likely to have TP53 mutations, which prevent the expression of p21, allowing BRCA-deficient cells to escape apoptosis and proliferate, resulting in tumorigenesis. 9
Estrogen receptor α (ERα) plays an essential role in the regulation of normal breast epithelium and the development of breast carcinoma, and mediates the effects of estrogen on human cells. 10 The ERα protein consists of 595 amino acids, and the corresponding gene is located on chromosome 6. Three out of six domains of ERα are functionally significant. The C domain (DBD contains two C4-type zinc fingers, which bind to estrogen-response elements (ERE). The N-terminal A/B domain (NTD) is involved in the transcriptional activation activity of AF-1. In contrast, the E domain (LBD) forms a homodimer upon estrogen binding that regulates gene suppression and activation and acts as a transcriptional activator.11,12 ER regulates the expression of some target genes by activating transcription factors in concert with co-activators. ERα can upregulate BRCA2 and TP53, while downregulating CCNG2.12,13 In addition, in the presence of E2, ERα regulates the PI3K/AKT signaling pathway, which leads to cancer progression. 12
Goniothalamus elegans Ast (G. elegans), family Annonaceae, is widely distributed in Southeast Asia. In traditional medicine, the species is used to treat heart disease and bloody diarrhea.14,15 A recent study of G. elegans described the isolation of 15 compounds which are mainly classified into three groups: acetogenins, styryllactones, and alkaloids. Several compounds isolated from this species have antimalarial and antibacterial activities and cytotoxic activity against the cancer cell lines KB, MCF-7, and NCI-H187. Goniothalamin and (+)-altholactone exhibited potent cytotoxic activity against KB and NCI-H187 cell lines compared to the positive control ellipticine. Velutinam was cytotoxic against all three cell lines, whereas aristolactam BII was cytotoxic against only the NCI-187 cell line. 15 In this study, the crude extract and fractions from the aerial parts of G. elegans showed potential cytotoxic activities. Since the cytotoxic activity of the ethyl acetate fraction was the strongest, this fraction was separated by column chromatography to obtain the two alkaloids lysicamine and liriodenine. Molecular simulations were used to predict these compounds’ interactions with BRCA1, BRCA2 gene, and ERα, and their biological properties. In vitro experiments on the human breast cancer cell line MCF-7 and the non-cancer cell line HEK-293 were performed to test our predictions.
Results and Discussion
Phytochemical investigation of the aerial part of G. elegans yielded two oxoaporphine alkaloids known as lysicamine and liriodenine. Oxoaporphines are a class of compounds typically found in the Annonaceae family. 16 Liriodenine has been reported in many Goniothalamus species, such as G. amuyon, G. gitigensis, G. tapis, G. scortechinii, and G. andersonii. 16 However, to our knowledge, the current study is the first to report the isolation of lysicamine and liriodenine from G. elegans.
Molecular Docking Results
3D structures of liriodenine and lysicamine were downloaded from the PubChem database. These compounds were docked to the BRCA1, BRCA2 and ERα structures, to predict the mechanism of breast cancer cell suppression. In this study, the commercial anticancer drug, 5-fluorouracil, was selected as a reference compound.
Interaction and Binding Affinity Between Alkaloids and BRCA1
Our docking studies predicted BRCA1 binding energies of liriodenine and lysicamine higher than −4.3 kcal/mol, which is the binding energy of 5-fluorouracil (Table 1).
Docking Results of Alkaloids Towards BRCA1 Protein.

Liriodenine was predicted to interact with BRCA1 by one hydrogen bond and two hydrophobic interactions at Phe1662. Lysicamine formed four hydrogen bonds at Asp1778, Ile1680, Leu1679, and Lys1702, and two hydrophobic interactions at Leu1701 and Pro1776. (Figure 1).

Interaction between isolated compounds and BRCA1. (A) liriodenine; (B) lysicamine.
Interaction and Binding Affinity Between Alkaloids and BRCA2
Liriodenine and lysicamine had docking energies for BRCA2 significantly higher than −5.2 kcal/mol, the docking energy of 5-fluorouracil (Table 2).
Docking Results of Alkaloids Towards BRCA2 Protein.

Liriodenine was predicted to interact with BRCA2 by one hydrogen bond and two hydrophobic interactions at Phe1662. Meanwhile, lysicamine formed four hydrogen bonds at Asp1778, Ile1680, Leu1679, and Lys1702, and two hydrophobic interactions at Leu1701 and Pro1776. (Figure 2).

Interaction between isolated compounds and BRCA2. (A) liriodenine; (B) lysicamine.
Interaction and Binding Affinity Between Alkaloids and Estrogen Receptor α
Liriodenine and lysicamine had predicted docking energies for ERα higher than −5.2 kcal/mol, the docking energy of 5-fluorouracil (Table 3).
Docking Results of Alkaloids Towards Estrogen Receptor α.

Liriodenine was predicted to interact with ERα with five hydrophobic interactions at Ala350, Leu525, Leu346, Met343, and Met421. Lysicamine formed seven hydrophobic interactions at Ala350, Leu536, Leu354, Lys529, Leu525, Met528, and Trp383 (Figure 3).

Interaction between isolated compounds and estrogen receptor α. (A) liriodenine; (B) lysicamine.
Structurally, the two alkaloids liriodenine and lysicamine have an oxoaporphine framework. Rings in the polyphenol structure were found to form π interactions with key amino acid residues. Carbonyl and hydroxyl groups in the compounds formed strong hydrogen bonds with amino acid residues of the protein targets. It is noteworthy that the heterocyclic nitrogen molecule is similar to the structure of both compounds, which also contributes to strong binding interactions with amino acids at the catalyst site of these proteins.
ADMET Properties
Lipinski's rule of five is a tool to assess the drug-likeness of compounds, to predict whether they will be orally active. The logP, TPSA, MW, HBA, and HBD values reflect both the oral bioavailability of compounds with good membrane permeability and the hydrophobicity of compounds. nRotB and MR values are used to predict the intestinal absorption and oral bioavailability of drugs.
Liriodenine and lysicamine both have acceptable drug-like properties and other drug-likeness parameters (Table 4). 17 We note that both liriodenine and lysicamine satisfied the rule MW ≤ 500 Da, logP < 5, nHBD ≤ 5, nHBA ≤ 10, and TPSA < 140 Å, indicating that they may have good oral bioavailability. In addition, the logS values for liriodenine and lysicamine are −4.25 and −4.27, respectively. This indicates moderate solubility in water. The TPSA value of both compounds is 48.42. 18 Therefore, both compounds were predicted to be absorbed well in the intestine. nRotB is an indication of the stereo-specificity of a drug, and in both cases was <10.
Drug Properties of Isolated Compounds Analyzed with SwissADME.

MW: Molecular weight, LogP: Log of octanol/water partition coefficient, nHBA: Number of hydrogen bond acceptor(s), nHBD: Number of hydrogen bond donor(s), MR-Molar refractivity, nRotB: Number of rotatable bonds, Log S: log of solubility, TPSA: Total polar surface area.
The in silico predictions of absorption, distribution, metabolism and excretion of liriodenine and lysicamine are presented in Table 5. Both compounds were predicted to have high gastrointestinal absorption, and were also predicted to be able to permeate the blood–brain barrier. The logKp, an indicator of skin permeability, was similar for both compounds, at −5.57 cm/s and −5.58 cm/s. 19 A variety of cytochromes (CYPs) affect drug metabolism, with CYP1A2, CYP2C19, CYP2C9, CYP2D6, and CYP3A4 being vital for drug biotransformation. 20 The SwissADME prediction tool suggested that liriodenine may inhibit CYP1A2 and CYP3A4 and be a substrate of P-glycoprotein (P-gp). Lysicamine was predicted to inhibit all cytochromes, but was not predicted to be a P-gp substrate.
ADME Predictions of Isolated Compounds Computed by SwissADME.

Abbreviations: log Kp: Log of skin permeability; GI Abs: Gastro-intestinal absorption; BBB Per: Blood brain barrier permeability; P-gp, P-glycoprotein; CYP, cytochrome-P
Liriodenine and lysicamine have the potential to inhibit cancer cells. However, the activity of these compounds on normal cells should be evaluated to assure cancer cell line selectivity and the potential to develop therapeutics without endangering the health of patients. In this study, the toxicity of liriodenine and lysicamine were predicted by the DL-AOT prediction server.
LD50 values of liriodenine and lysicamine were predicted to be 2.28 mg/kg and 2.87 mg/kg, respectively (Table 6). DL-AOT classifies compounds into four groups: “danger/poison”, “warning”, “caution” and “none required”. Both liriodenine and lysicamine were classified in the “warning” group, meaning that they are predicted to be toxic to normal cells. Our in silico data suggest that liriodenine and lysicamine have the potential to inhibit proteins related to breast cancer. However, they are potentially cytotoxic. Therefore, it is necessary to conduct experimental studies of the biological activity of these compounds.
Toxicity of Isolated Compounds Predicted by DL-AOT Prediction Server.

MCF-7 Cancer Cell and HEK-293 Cell Inhibition Assay
In this study, a crude methanol extract and n-hexane, dichloromethane, ethyl acetate, and aqueous fractions from G. elegans were assessed for their cytotoxic activity. The crude methanol extract was cytotoxic to MCF-7 cancer cells with an IC50 values of 25.04 ± 0.94 µg/mL. The ethyl acetate and dichloromethane fractions were cytotoxic to these cells with IC50 values of 42.08 ± 0.98 and 63.01 ± 1.67 µg/mL, respectively. The n-hexane and aqueous fractions were not cytotoxic to MCF-7 cells. Since the cytotoxic effect of the ethyl acetate fraction was the strongest, this fraction was separated by column chromatography to obtain liriodenine and lysicamine.
In order to validate the docking results, in vitro experiments were performed. Estrogen modulates mitosis in MCF-7 cells. 21 In addition, BRCA1 and BRCA2 are two of the key "high-risk" genes associated with breast cancer. 22 Therefore, to assess the potential of liriodenine and lysicamine, we used the MCF-7 cancer and HEK-293 normal cell lines to test our predictions of toxicity.
The cytotoxicity of the isolated compounds against the growth of MCF-7 and HEK-293 cell lines was tested by a SRB assay, with a slight modification. 15 The results are described in Table 7. Liriodenine showed significant cytotoxicity against MCF-7 cells with an IC50 value of 33.31 ± 3.34 µM, and slightly lower cytotoxicity against HEK-293 cells, with an IC50 value of 67.03 ± 3.89 µM. In contrast, lysicamine exhibited a lesser effect on MCF-7 and strong toxicity to HEK-293 cells, with IC50 values of 70.03 ± 5.42 µM and 58.29 ± 5.22 µM, respectively.
The Cytotoxicity of two Alkaloids Isolated from G. elegans.
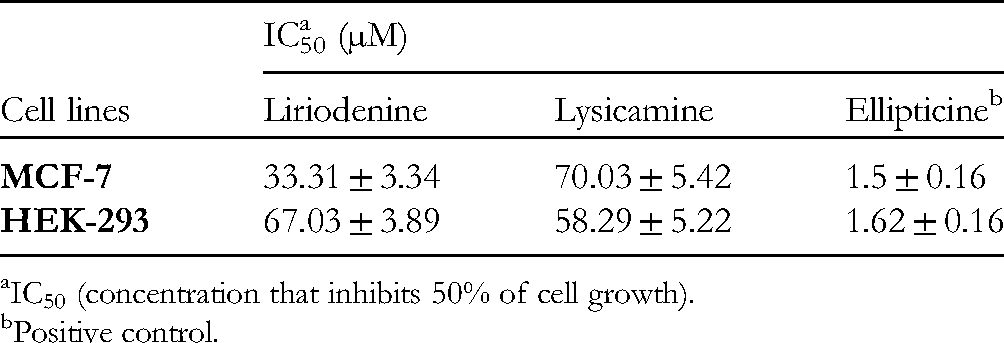
IC50 (concentration that inhibits 50% of cell growth).
Positive control.
An aporphine skeleton is formed by directly bonding the A and D aromatic rings of a benzylisoquinoline nucleus. The nitrogen atom at position 6 is quaternary in the base form. In lysicamine and liriodenine, positions 1 and 2 are substituted by methoxy and methylenedioxy groups, respectively. 23 The methylenedioxy group of liriodenine contributed to its cytotoxic activity against Saccharomyces cerevisiae. Lysicamine, however, lacks this methylenedioxy group, and is thus inactive. 24 The function of this group was demonstrated in other bioactivity studies of aporphine alkaloids.25-27 Lysicamine and liriodenine have cytotoxic activity against various human cancer cell lines.28-30 Topoisomerase II inhibition studies demonstrated that the planar aromatic structure of liriodenine is involved in its anti-tumor activity. 31 Additionally, the oxo-aporphine framework was reported to induce cytotoxicity and inhibit precursor incorporation into DNA during leukemic cell growth. 32
The cytotoxic activity of liriodenine against MCF-7 cells was previously shown to be mediated by several mechanisms. Liriodenine significantly suppressed cell viability, induced apoptosis, and activated caspase-3. Liriodenine was also found to inhibit cyclin D1 protein expression and promoting p53 and VEGF protein expression in MCF-7 cells. 32 Based on the results of our in silico and in vitro studies, liriodenine may be a potential lead for breast cancer treatment, and suggests that further cytotoxicity studies should be performed on other aporphine alkaloids.
Conclusion
Liriodenine and lysicamine are two notable alkaloids with cytotoxic properties. Our study suggests novel biological mechanisms by which these compounds might exert their biological effects, ie inhibition of BRCA1, BRCA2, and ERα. Simulation results were evaluated by in vitro experiments on MCF-7 and HEK-293 cell lines. Liriodenine is a promising candidate for breast cancer therapy. Although lysicamine affects both cancer and normal cells, the results of our research provided information that suggested further studies on aporphine alkaloids.
Materials and Methods
Collection of Plant Material
The aerial parts of Goniothalamus elegans Ast. were collected from Quang Tri province, Viet Nam (N16°44′38.9″ E107°14′51.1″) in May 2020 and were identified by Dr Vu Tien Chinh, Vietnam National Museum of Nature, VAST, Viet Nam and MSc. Le Tuan Anh, Quang Tri Center of Science and Technology, Mientrung Institute for Scientific Research, VAST, Vietnam. A voucher specimen (GE01) was deposited at the Faculty of Pharmacy, Hue University of Medicine and Pharmacy, Hue University, Vietnam.
General Experimental Procedures
The 1D and 2D NMR spectra were obtained using a Bruker Advance 500 spectrometer (Billerica, Massachusetts) with TMS as the internal reference. HRMS-MS/MS data were measured by Sciex X500R - QTOF system, USA. The Agilent 1260 Infinity II system (Agilent Technologies) was used for analytical and preparative HPLC (250 × 9.4 mm). Open column chromatography was performed using silica gel (60 N, spherical, neutral, 40-50 μm, Kanto Chemical Co., Inc., Tokyo, Japan) and YMC RP-18 (Fuji Silysia Chemical Ltd, Kasugai, Aichi, Japan). Analytical TLC was performed on pre-coated silica gel 60F254 and RP-18 F254S plates (0.25 or 0.50 mm thickness, Merck KGaA, Darmstadt, Germany). The cell line MCF-7 (human breast cancer) was used for the cytotoxic activity determinations. Cell culture flasks and 96-well plates were from Corning Inc. (Corning, NY, USA). An ELISA plate reader (Bio-Rad, California) was used to measure the absorbance of the cells in the cytotoxicity assay.
Extraction and Isolation
The dried powder of G. elegans (4.5 kg) was extracted with MeOH (3 times, 10.0 L each) at room temperature to yield 375 g of a dark solid extract. This extract was then suspended in water and successively partitioned with n-hexane, CH2Cl2, and EtOAc (3 times, 5.0 L each) to obtain the n-hexane (117.6 g), CH2Cl2 (91.2 g), EtOAc (83.3 g), and water (74.7 g) fractions after removal of the solvents in vacuo. The EtOAc extract was chromatographed on a silica gel column eluted with a gradient of n-hexane–EtOAc (100:0, 95:5, 90:10, 50:10, 10:10, and 0:100, v/v, 1.0 L each) to obtain 6 fractions (E1–E6). Fraction E3 (11.9 g) was partitioned on a Sephadex LH-20 column eluted with CH2Cl2–MeOH (1:1, v/v) to give 5 sub-fractions (E3A–E3E). Fraction E3C (2.4 g) was chromatographed on a silica gel column eluted with CH2Cl2–EtOAc (8:1, v/v) to obtain 7 sub-fractions (E3C1–E3C7). Fraction E3C5 (280 mg) was partitioned on a YMC RP-18 column eluted with acetone–MeOH–water (3:1:1, v/v) to give 5 additional fractions (E3C5A– E3C5E). Fraction E3C5B (60 mg) was chromatographed on a Sephadex LH-20 column eluted with MeOH to give 4 additional fractions (E3C5B1–E3C5B4). Fraction E2C5B3 (23 mg) was separated by preparative reversed-phase HPLC (Zorbax SB – C18 5 μm, 9.4 × 250 mm) using MeOH–water (70:30, flow rate 2 mL/min) as the eluent to furnish
Computational Studies
Protein and Ligand Preparation
The crystal structures of BRCA1, BRCA2, and estrogen receptor α (PDB ID: 4Y2G, 3EU7, and 3ERT, respectively) were downloaded from Protein Data Bank (RCSB PDB). All the heteroatoms and water molecules of proteins were removed using Discovery Studio 2020. Then, Hydro polar and Kollman charges were added to the proteins using the Autodock tools version 1.5.6. The 3D structures of the two compounds isolated from Goniothalamus elegans were retrieved from the PubChem database. Finally, the macromolecule and ligands were exported into a dockable pdbqt format for molecular docking.
Molecular Docking
Molecular docking processes on BRCA1, BRCA2, and estrogen receptor α were carried out using AutoDock Vina. The grid boxes that cover the active site of proteins were predicted using CASTp server. 35 Docking scores are reported in kcal/mol. Finally, Discovery Studio Visualizer was utilized to visualize the molecular interactions between proteins and ligands.
In-Silico Drug-Likeness and Toxicity
Drug-likeness of compounds was predicted based on an already established concept by Lipinski rule. 36 SwissADME server was used to calculate the ADME properties of ligands which provided information about absorption, distribution, metabolism, and excretion (ADME) properties of the ligands. 37 The acute oral toxicity (T) was predicted using the DL-AOT prediction server. 38
Cytotoxic Assays
The cytotoxic activity of fractions and isolates were tested by SRB assay, 39 with a slight modification, against the growth of human breast cancer (MCF-7) and human embryonic kidney 293(HEK293) cell lines, which were acquired from the Long Island University, USA and the University of Milan, Italy. Briefly, cell lines were grown at a density of 4 × 104 cells/well in 96-well microtiter plates with each well containing 180 μL growth medium. Tested samples were added to each well and the cultivation was continued under the same conditions for another 72 h. Thereafter, the medium was removed and the remaining cell monolayers were fixed with cold 20% (w/v) trichloroacetic acid for 1 h at 4 °C and stained by 1 × SRB staining solution at room temperature for 30 min. Then, the unbound dye was removed by washing repeatedly with 1% (v/v) acetic acid. The protein-bound dye was dissolved in 10 mM Tris base solution for optical density determination at 515 nm on an ELISA Plate Reader. DMSO 1% was used as negative control, with a final concentration of 0.05%. Ellipticine was used as positive control (10, 2, 0.4, and 0.08 μg/mL). The cytotoxicity of samples was measured at doses of 100, 20, 4, and 0.8 μg/mL. Experiments were carried out in triplicate for accuracy of data. TableCurve 2Dv4 software was used for data analysis and calculated as a half maximal inhibitory concentration (IC50) calculation. The inhibition rate (IR) of cells was calculated by the following formula IR% = {100 − [(ODt−OD0)/(ODc−OD0)] × 100 (%)}, in which: IR: Inhibition rate of cell growth, ODt: average optical density value at day 3; OD0: average optical density value at time-zero; ODc: average optical density value of the blank DMSO control sample.
Footnotes
Acknowledgments
This research was funded by the Hue University (Vietnam) under grant number DHH2020-04-112. We are grateful to Mr Anh Tuan Le (Mientrung Inst. for Scientific Research, VAST, Quang Tri, Viet Nam) for collecting the plant material, and Mr Luong Vu Dang (Institute of Chemistry, VAST, Hanoi, Viet Nam) for recording the NMR spectra.
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Hue University-Vietnam grant, (grant number DHH2020-04-112).
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
