Abstract
Introduction
The immune system functions as the principal detector of pathogenic microorganisms invading the host organism. It subsequently initiates phagocytosis to eradic these pathogens from the body. 1 Furthermore, the immune system coordinates a variety of immune responses, including anti-inflammatory, anti-tumor, and anti-infection mechanisms, to maintain the physiological homeostasis and stability of the organism.2–4 However, the immune system can also inflict harm on the organism when abnormal immune function is triggered by certain conditions. Examples of such conditions include autoimmune diseases, hypersensitivity reactions, and tumors. 5 Globally, the prevalence of immune disorders is steadily increasing, due to a range of congenital immune deficiencies and acquired nutritional deficiencies. 6 These conditions undermine the development of robust immune defenses, rendering an increasing number of individuals susceptible to various diseases. 7 Therefore, a healthy and stable immune system is crucial for the prevention of many diseases.
Panax notoginseng, initially documented in Li Shizhen's Ming Dynasty ‘Compendium of Materia Medica’, is knowned for its therapeutic properties, including the resolution of blood stasis, hemostasis, activation of blood circulation, and analgesia. 8 Contemporary pharmacological research has revealed that Panax notoginseng exhibits anti-tumor 9 and anti-inflammatory properties, 10 enhances immune function, 11 aids in the management of diabetes mellitus, 12 and possesses anti-aging effects. 13 Several studies have indicated that Panax notoginseng total saponins (PNS) modulate the body's immune system and maintain homeostasis at multiple levels, including cellular and humoral immunity, as well as the regulation of natural killer cell functions.14,15 The primary active constituents of Panax notoginseng are saponins, with over 70 distinct saponin monomers identified to date.16,17 These rare ginsenosides not only retain the fundamental efficacy of conventional ginsenosides but also exhibit unique properties, demonstrating significant antitumor, neuroprotective, and hepatoprotective effects.18–21
During the initial phase of Cordyceps sinensis fermentation, the research team acquired the fermented Panax notoginseng saponin extract. Subsequent in vivo experiments demonstrated that this fermented extract exhibited enhanced biological activity compared to the non-fermented Panax notoginseng saponin extract. Furthermore, the composition and concentration of Panax notoginseng saponins were analyzed pre- and post-fermentation using High-Performance Liquid Chromatography (HPLC). The analysis indicated that, post-fermentation, the concentrations of three saponin components-ginsenoside Rb3, Rk3, and Rh4-were significantly elevated, surpassing those in the pre-fermentation Panax pseudoginseng saponin extract and accounting for approximately 74.2% of the total post-fermentation saponins. To assess the potential role of ginsenosides Rb3, Rk3, and Rh4 as active components post-fermentation, these ginsenosides, which were present in the highest concentrations, were combined with three types of rare saponins to create formulations of Panax pseudoginseng rare saponin compositions. In this study, the ginsenosides Rb3, Rk3, and Rh4 were combined to formulate a rare saponin mixture derived from Panax pseudoginseng, administered at dosages of 148 mg/kg, 74 mg/kg, and 37 mg/kg, respectively. These dosages were determined based on their proportional content ratios of 26.3%, 19.4%, and 28.5%. An immunocompromised mouse model was established to investigate the effects of this ginsenoside combination on murine immune function. The findings offer a scientific basis for the development and application of rare saponins from Panax notoginseng, promoting the sustainable and efficient use of this medicinal plant resource. This research thereby contributes to the sustainable advancement of the pharmaceutical industry and the conservation of ecological environments.
Methods
Animal Grouping, Modelling and Treatment
A total of 48 mice were randomly divided into six groups of eight mice, including the normal group, the model group, the astragalus polysaccharide group (250 mg/kg), The dosage of astragalus polysaccharide is referenced in the literature37, 22 and the high, medium, and low dose groups of Panax notoginseng rare saponin combination. (Rk3, Rb3, and Rh4) (doses of 148 mg/kg, 74 mg/kg, and 37 mg/kg, respectively). To induce immunosuppression, cyclophosphamide was administered intraperitoneally at a dose of 100 mg/kg for three consecutive days in all groups except the control group, which received saline injections. Both the control and model groups were gavaged with saline, while the experimental groups received the respective drug dosages via gavage for 14 consecutive days. The mice were kept on a standard diet with normal water intake, and their body weights were recorded on days 4, 7, 10, 13 and 17. The protocol was approved by the Animal Ethics Committee of Dali University. 23
Determination of Organ Index
A dissection was taken of the thymus, spleen, and liver of mice and fat and fascial tissues were removed. At the end of the experiment, the dissections were weighed and an organ index was calculated.
24
Determination of Peripheral Blood Cell Counts with a Whole Blood Cell Analyser
One hour following the final administration, blood samples were collected from mice via enucleation. Subsequently, 20 µL of whole blood was added to a pre-dilution solution for analysis. The white blood cell count (WBC), as well as the absolute counts of lymphocytes, neutrophils, and monocytes in peripheral blood, were determined using a cell analyzer in pre-dilution mode. 25
Measurement of Phagocytosis Rate in Peritoneal Macrophages
Mice were euthanized by cervical dislocation 1 h after the final administration and surface-sterilized by immersion in 75% ethanol for 3 min. Subsequently, 4 mL of sterile phosphate-buffered saline (PBS) containing 10% fetal bovine serum (FBS) and 2 mL of air were injected into the peritoneal cavity. The abdomen was gently massaged for 3 min to dislodge macrophages, followed by aseptic extraction of peritoneal lavage fluid. The fluid was transferred to centrifuge tubes and centrifuged at 1200 ×g for 5 min. The supernatant was discarded, and the pelleted macrophages were washed twice with PBS. Cell density was adjusted to 5 × 109 cells/L using RPMI-1640 medium supplemented with 10% FBS.The cell suspension (100 μL/well) was seeded into a 96-well plate and incubated at 37 °C under 5% CO2 for 3 h to allow adherence. Non-adherent cells were removed by washing with PBS. Adherent macrophages were incubated with 200 μL of 0.1% Neutral Red solution per well for 3 h. After discarding the supernatant, cells were washed three times with PBS to remove unphagocytosed dye. To lyse cells and release phagocytosed Neutral Red, 200 μL of acidic ethanol lysis buffer (1:1 v/v ethanol and acetic acid) was added to each well, followed by overnight incubation at 4 °C. Absorbance was measured at 520 nm using a microplate reader.
26
The phagocytosis rate (%) was calculated as:
Histopathological Analysis of the Liver and Spleen of Immunocompromised Mice
Liver and spleen tissues were fixed in 10% neutral formaldehyde solution for 48 h. After gradient ethanol dehydration, xylene removal, and paraffin embedding, liver and spleen tissues were processed. The paraffin sections were deparaffinized to water, stained with hematoxylin, and dehydrated with gradient alcohol, Stained with eosin and sealed for observation to assess liver and spleen tissue damage. 27
Immunohistochemical Analysis
Tissue sections of liver and spleen were deparaffinised to water prior to immunohistochemistry (IHC). The sections were immersed in sodium citrate buffer solution at pH 6.0, treated with antigen repair in a microwave oven for 20 min, and tested for inhibition of endogenous peroxidase activity. For 30 min at room temperature, sections were immersed in 3% hydrogen peroxide solution and covered with 5% goat serum treated with a closure solution. The sealing solution was gently removed and a certain concentration of Nrf2 (1:200), HO-1 (1:200), CD4 (1:1000) and CD8 (1:2000) mixed with PBS was added dropwise to the sections.28,29 We then incubated the sections overnight at 4 °C in a humidified environment, washed three times in PBS at pH 7.4 and gently shaken to remove excess liquid. A secondary antibody (HRP-labelled) corresponding to the primary antibody was applied evenly to the tissue sections, and they were shaken dry. After 50 min at room temperature, the sections were exposed to diaminobenzidine (DAB) for color development. Hematoxylin was applied for 3 min, and the sections were finally dehydrated and sealed.
Immunofluorescence Analysis
The spleen samples were first dewaxed using an environmentally friendly dewaxing solution, followed by gradient ethanol dehydration and rinsing with distilled water. Microwave antigen retrieval was then performed using citrate buffer, followed by washing with PBS. To prevent non-specific binding, a 3% BSA solution was employed before overnight incubation of a mixture of CD3 and Nrf2 primary antibodies in a wet box at 4 °C. After washing with PBS, the corresponding secondary antibody was added dropwise, and the nuclei were counterstained with DAPI. The samples were processed to quench tissue autofluorescence and subsequently sealed. Finally, the co-localization of CD3+ T cells and Nrf2 proteins was observed using fluorescence microscopy at specific wavelengths. The positive signal intensity was quantified with ImageJ software, and the results were expressed as mean fluorescence intensity (MFI).30,31
Statement
For ARRIVE: ‘The reporting of this study conforms to ARRIVE 2.0 guidelines. 32
Statistical Methods
Data were analyzed using SPSS 25.0. The final results of the data were expressed as mean ± standard deviation (
Results
Effects of Rare Saponin Combinations of Panax notoginseng on Body Mass of Immunocompromised Mice
The study revealed that the body weights of the model group on days 7, 10, 13, and 17 were significantly lower than those of the normal group (P < 0.01). In contrast, the body weights of mice in both the Astragalus polysaccharide group and the Panax notoginseng rare saponin combination group showed significant increases on days 10, 13, and 17 (P < 0.01 or P < 0.05) compared to the model group. These findings are illustrated in Figure 1. Within the model group, there was a significant decrease in both the thymus index and spleen index (P < 0.01), while the liver index increased, although not significantly (P > 0.05).

Effect of Rare Saponin Combinations of Panax notoginseng on Body Weight, thymus, Spleen and Liver Indices in Immunocompromised Mice. (a) Changes in Body Weight of Mice. (b) Thymic Index of Mice (c) Spleen Index of Mice (d) Liver Index of Mice Quantitative Data are Shown as the Mean ± Standard Deviation, n = 8. PNRSC: Panax notoginseng Rare Saponin Combinations; APS: Astragalus Polysaccharide; *P < 0.05; **P < 0.01 Versus Normal Group; #P < 0.05; ##P < 0.01 Versus Model Group.
Effects of Panax notoginseng Rare Saponin Combinations of on Immune Organ Indices in Immunocompromised Mice
Compared to the model group, the spleen index in the Astragalus polysaccharide group and the Panax notoginseng rare saponin combination group, across all dose levels, exhibited a significant increase (P < 0.01). Conversely, the liver index demonstrated a statistically significant decrease (P < 0.01). Additionally, the thymus index in the Astragalus polysaccharide group and the Panax notoginseng rare saponin combination group, particularly at medium and high doses, showed a statistically significant increase (P < 0.01 or P < 0.05), as depicted in Figure 1.
Effects of a Rare Saponin Combination of Panax notoginseng on Peripheral Blood Routine in Immunocompromised Mice
Peripheral blood routine indices are standard assessments employed to evaluate immune system health and overall physiological status. In the model group, the absolute counts of monocytes, lymphocytes, neutrophils, and leukocytes were significantly reduced compared to the normal group (P < 0.01). In the high-dose group receiving the Panax notoginseng rare saponin combination, mice in the model group exhibited significantly increased levels of leuko-lymphocytes (P < 0.05). Furthermore, there was a notable increase in monocytes, neutrophils, and leukocytes in mice treated with Astragali polysaccharide, as well as those administered medium- and high-dose Panax notoginseng rare saponin combination. The study revealed that both the absolute value of granulocytes and the WBC were significantly elevated (P < 0.05), as depicted in Figure 2a-d.
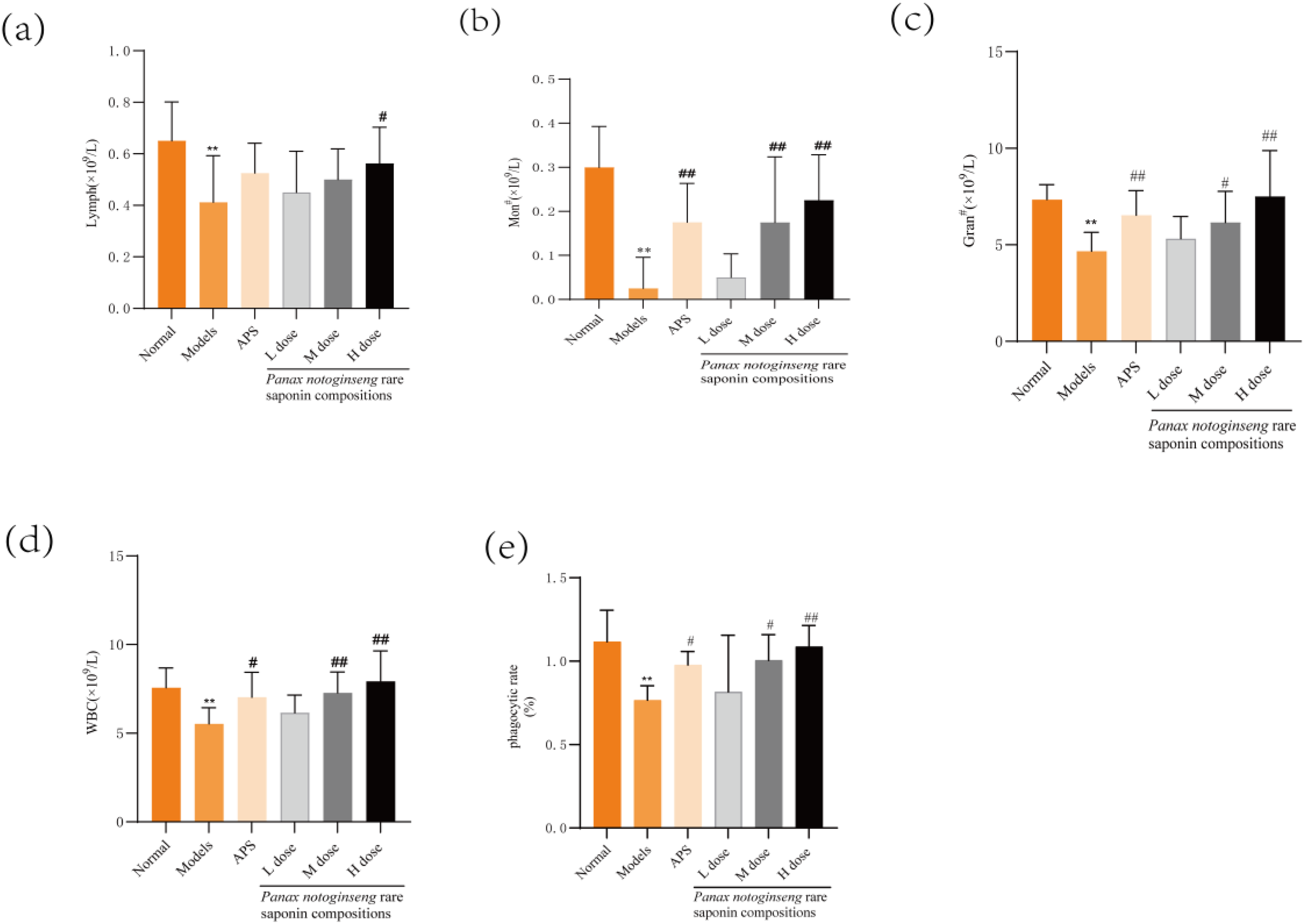
Effect of Panax notoginseng Rare Saponin Combination on Peripheral Blood Routine and Phagocytosis of Peritoneal Macrophages in Immunocompromised Mice. (a-e): Measurement of White Blood Cell Count (WBC), Absolute Value of Lymphocytes, Absolute Value of Neutrophils, Absolute Value of Monocytes and Phagocytosis Rate of Abdominal Macrophages in Mice by Using a Blood Cell Analyser. Quantitative Data are Shown as the mean ± standard deviation, n = 8. APS: Astragalus Polysaccharide; *P < 0.05; **P < 0.01 versus Normal Group; #P < 0.05; ##P < 0.01 versus Model Group.
Effects of a Rare Saponin Combination of Panax notoginseng on Phagocytosis of Peritoneal Macrophages in Immunocompromised Mice
The phagocytosis rate of abdominal macrophages is typically used to express their phagocytic capacity. In model mice, this phagocytosis rate was significantly lower than that observed in normal mice (P < 0.01). The administration of astragali polysaccharides in conjunction with rare saponins of Panax notoginseng at medium and high dosages significantly enhanced the phagocytic activity of abdominal macrophages compared to the model group (P < 0.01 or P < 0.05), as depicted in Figure 2 e.
Effects of a Rare Saponin Combination of Panax notoginseng on the Pathological Morphology of the Liver in Immunocompromised Mice
Figure 3a illustrates the hematoxylin and eosin (HE) staining results of mouse liver tissue. In the control group, the hepatocyte architecture is well-defined, exhibiting intact cellular morphology with no indications of hepatocyte degeneration. Conversely, the model group demonstrates a disorganized and irregular liver cell structure, characterized by oedematous degeneration, significant hepatocyte enlargement, and an indistinct orientation of the hepatic cords. In the medium- and high-dose groups, administration of the Panax notoginseng rare saponin combination leads to a gradual and orderly arrangement of liver structures, with the orientation of the cellular cords becoming increasingly discernible.

Panax notoginseng Rare Saponin Combination Attenuates Hepatic and Splenic Pathological Changes in a Cyclophosphamide-Induced Immunodeficiency Mouse Model Representative Images(50 µm). (a) Representative Images of Hematoxylin and Eosin Stained Liver Tissue; (b) Representative Images of Hematoxylin and Eosin Stained Spleen Tissue. APS: Astragalus Polysaccharide.
Effects of a Combination of Panax notoginseng Dilute Saponins on the Pathological Morphology of the Spleen in Immunocompromised Mice
The histological examination of HE stained microscopic sections of the spleen, as depicted in Figure 3b, reveals that the spleen in the control group exhibited a clear and complete histological architecture. The splenic nodules were well-developed, with distinct red and white pulp regions, and the boundaries of the marginal zones were clearly delineated.In the experimental group, the demarcation between the red and white pulp of the spleen exhibited blurring, and the structural integrity of the splenic nodules was compromised. Additionally, splenomegaly was observed, with a significant reduction or complete disappearance of the splenic nodules. These findings indicate the successful establishment of the immunocompromised model. Compared to the model group, the splenic nodules in the middle- and high-dose Panax notoginseng rare saponin combination treatment groups exhibited restoration. The structural integrity of the spleen, including the delineation of the red and white medullas, was progressively clarified. These findings suggest that the Panax notoginseng rare saponin combination exerts a protective effect on the spleen in immunocompromised mice.
Effects of Rare Saponin Combinations of Panax notoginseng on CD4+ and CD8+ Protein Expression in Spleen of Immunocompromised Mice
This study investigates the effect of the Panax notoginseng rare saponin combination on the expression of CD4+ and CD8+ proteins in the spleen of an immunocompromised mouse model using immunohistochemical techniques. Compared to the normal group, the model group shows a significant reduction in the expression levels of both CD4+ and CD8+ proteins in the spleen (P < 0.01). Furthermore, the astragalus polysaccharide group, along with the low, medium, and high dose groups of Panax notoginseng rare saponin combinations, effectively attenuated the reduction in CD8 + and CD4 + protein expression in the spleen (P < 0.01),The results are shown in Figure 4.
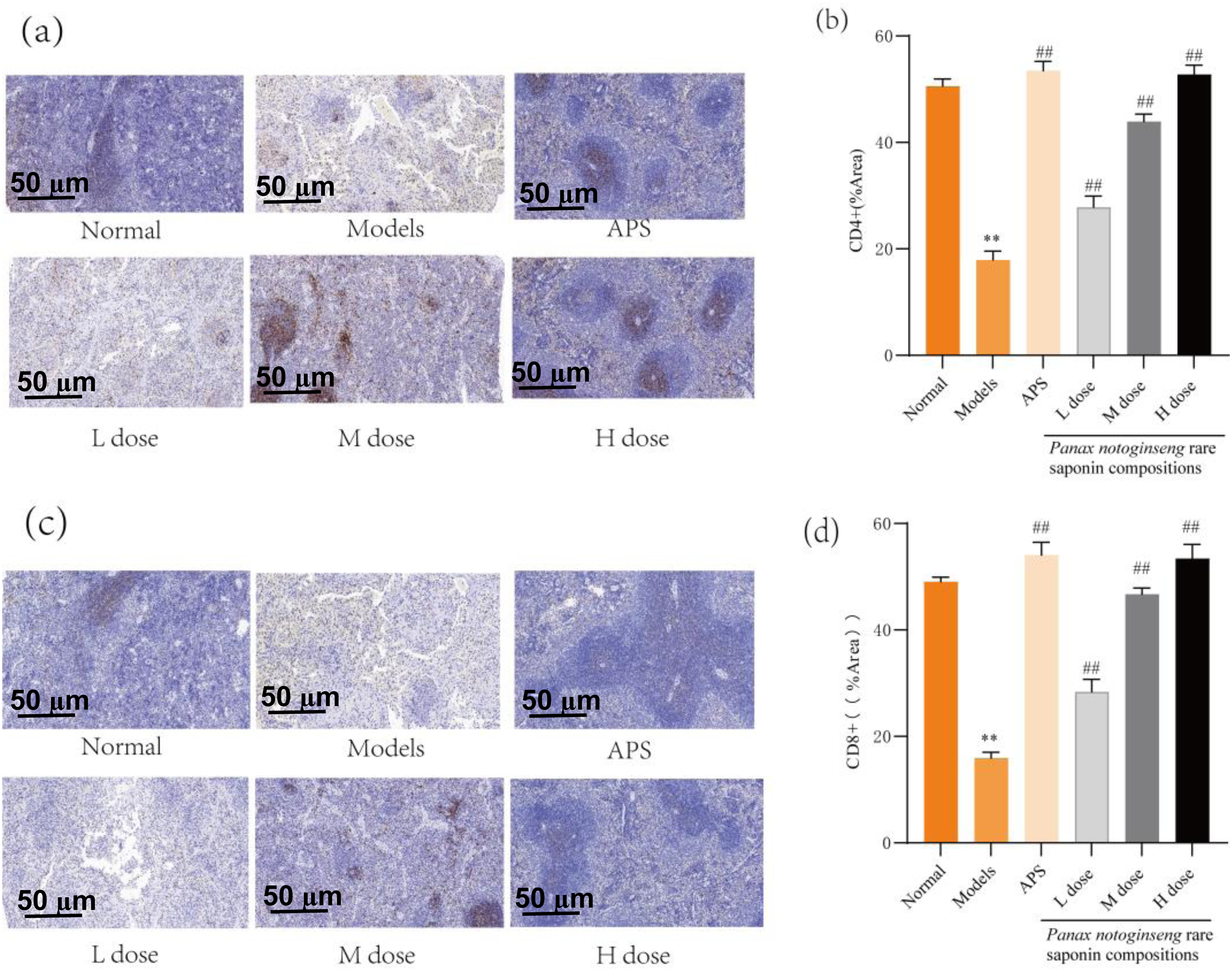
Effect of Panax notoginseng Rare Saponin Combinations on CD4+ and CD8+ Protein Expression in Spleen of Immunocompromised Mice (50 µm). (a-b): Expression Level of CD4+ Protein in the Spleen Of Mice. (c-d): Expression Levels of CD8+ Protein in the Spleens of Mice. Quantitative Data are Expressed as mean ± standard deviation n = 6.APS: Astragalus Polysaccharide; *P < 0.05; **P < 0.01 versus Normal Group; #P < 0.05; ##P < 0.01 versus Model Group.
Effects of Rare Saponin Combinations of Panax notoginseng on the Expression of Nrf2 and HO-1 Proteins in the Liver of Immunocompromised Mice
Figure 5 illustrates the results of the immunohistochemical analysis of hepatic Nrf2 and HO-1 protein expression in mice. The model group exhibited significantly decreased expression levels of Nrf2 and HO-1 proteins in the liver compared to the normal group (P < 0.01). In contrast, the astragali polysaccharides group and the high-dose Panax notoginseng rare saponin combinations group demonstrated a substantial increase in Nrf2 and HO-1 levels relative to the model group (P < 0.01).

Effect of Rare Saponin Combination of Panax notoginseng on the Expression of Nrf2 and HO-1 Proteins in the Liver of Immunocompromised Mice(50 µm). (a-b): Expression Level of Nrf2 Protein in Mouse Liver. (c-d): Expression Levels of HO-1 Protein in Mouse Liver.Quantitative Data are Shown as the Mean ± Standard Deviation, n = 8.APS: Astragalus Polysaccharide; *P < 0.05; **P < 0.01 Versus Normal Group; #P < 0.05; ##P < 0.01 Versus Model Group.
Effects of Rare Saponin Combinations of Panax notoginseng on CD3 + and Nrf2 Immunofluorescence in Spleen of Immunodeficient Mice
To further validate the effects of the rare saponin combination from Panax notoginseng on CD3+ T cells and Nrf2 expression in spleen tissues of immunocompromised mice, immunofluorescence staining was performed on splenic sections. Reduced CD3+ expression indicated immunosuppression, as shown in Figure 6a-b. Compared with the normal group, the model group exhibited a significant decrease in CD3+ levels (P < 0.01). Treatment with astragalus polysaccharides and medium/high doses of the saponin combination markedly restored CD3+ expression (##P < 0.01 vs model group). Similarly, Nrf2 immunostaining revealed that its expression in the model group was significantly suppressed (**P < 0.01 vs normal group). In contrast, both astragalus polysaccharides and medium/high doses of the saponin combination substantially upregulated Nrf2 levels in splenic tissues (##P < 0.01 vs model group), The results are shown in Figure 6.

Effects of the Rare Saponin Combination from Panax notoginseng on Nrf2 and CD3+ Co-localization in Spleen Tissues of Immunocompromised Mice. (a) Fluorescence Imaging of CD3+ (T-cell marker, red) and Nrf2 (green) Co-localization in Spleen Sections. Nuclei Were Counterstained with DAPI (blue) (50 µm). (b) Quantification of CD3 + Expression Levels in Splenic Tissues. (c) Quantification of Nrf2 Expression Levels in Splenic Tissues. Quantitative Data are Shown as the mean ± standard deviation, n = 6.APS: Astragalus polysaccharide; *P < 0.05; **P < 0.01 versus Normal group; #P < 0.05; ##P < 0.01 versus model group.
Discussion
The current study identifies several immunosuppressive drugs, including cyclophosphamide, dexamethasone, hydrocortisone, and cyclosporin A.33–35 Cyclophosphamide (CTX), an alkylating agent, is utilized in chemotherapy for various tumors and cancers, as well as in bone marrow transplants, to reduce bacterial translocations, infections, and complications arising from the concurrent administration of chemotherapy and antibiotics. 36 CTX has been shown to inhibit both cellular and humoral immunity, affect cell proliferation, and interfere with the maturation and development of the immune system37–39 Consequently, an immunosuppressive mouse model is frequently established using CTX to assess its immunomodulatory properties. Within an organism's immune system, parameters such as body weight, the weight of immune organs, and the indices and expression levels of splenic lymphocytes are critical. 22 These indices generally reflect the intensity of the body's intrinsic immune function. Suppression of immune function can lead to atrophy of immune organs and a reduction in their indices.
The continuous administration of Panax notoginseng rare saponin compositions via gavage has been observed to significantly increase the thymus and spleen indices in mice, thereby preventing cyclophosphamide (CTX)-induced atrophy of these immune organs. This observation suggests that Panax notoginseng rare saponin compositions have the potential to enhance the immune function of immunocompromised mice, thereby supporting their beneficial effects in immunomodulation. The spleen, a large secondary lymphoid organ, performs a wide range of immune functions, with B and T cells playing a pivotal role in the adaptive immune system. 40 Additionally, the thymus is essential for immune regulation as it is the site of T-cell differentiation and maturation. 41 Therefore, the weight and index of the spleen and thymus serve as indicators of an organism's non-specific immunity, and changes in splenic lymphocytes can reflect the organism's immune function. Within the immune system, T lymphocytes in the spleen are crucial, playing avital role in the induction of cellular immunity and acting as key regulators of humoral immunity.
T lymphocytes represent a highly specialized group of cells that can be further categorized into CD4+ and CD8+ subsets, distinguished by the specif receptors expressed on their surfaces. 42 Mature T lymphocytes typically express CD3+ molecules; thus, CD3+ expression is frequently regarded as a significant marker of mature T cells 43 representing the total T cell population. 44 CD8+ T cells play a pivotal role in mediating cellular immunity, particularly through their functions as T suppressor and T cytotoxic cells. These cells are capable of secreting a diverse array of cytokines to target and eliminate pathogenic cells, thereby constituting a crucial component of the body's defense mechanisms. However, these immune responses are susceptible to disruption by external factors. 45 The immune system is predominantly composed of CD4+ T lymphocytes. 46 As the central cells in the immune response, 46 CD4+ T lymphocytes serve as an accurate measure of immune function, reflecting the extent of damage to the body's immune system has been damaged. 47 The CD4+/CD8+ ratio is a critical component of the body's immunomodulatory network. 48
Under physiological conditions, CD4+ and CD8+ T cells engage in a coordinated and balanced interaction, collectively facilitating an immune response that effectively eliminates foreign antigens while minimizing damage to host tissues. 48 An imbalance between CD4+ and CD8+ T cells can lead to immunosuppression and increased susceptibility to infections. 49 These T-cell subsets collaborate and regulate each other, playing a pivotal role in organism-specific immunity. 50 CD4+ T cells modulate humoral immunity by secreting various cytokines, which can directly influence cellular immune responses or enhance B cell-mediated antibody production. In contrast, CD8+ T cells exhibit cytotoxic capabilities, allowing them to directly eliminate target cells, thereby playing a critical role in defening against viral infections and tumorigenesis. 51 In this study, we examined the expression levels of CD4+ and CD8+ proteins in the liver and found that administering a Panax notoginseng rare saponin combination resulted in the upregulation of CD4+ and CD8+ protein expression. Our findings indicate that the Panax notoginseng rare saponin combination effectively promotes the increase in CD4+ and CD8+ T cell expression.
In this study, we examined the expression levels of CD4+ and CD8+ proteins in the liver and found that the administration of a Panax notoginseng rare saponin combination led to the upregulation of CD4+ and CD8+ protein expression. Our results suggest that the Panax notoginseng rare saponin combination effectively enhances the expression of CD4+ and CD8+ T cells. Furthermore, abdominal macrophages, known for their excellent phagocytic capabilities, exhibit a significant ability to eliminate foreign substances from the body. While macrophages possess limited phagocytic capacity when unactivated, upon stimulation, they differentiate into activated macrophages with enhanced phagocytic abilities. 52 The experimental findings demonstrated that the synergistic effect of abdominal macrophages and the Panax notoginseng rare saponin combination significantly augmented macrophage phagocytic function. Specifically, the Panax notoginseng rare saponin combination increased the phagocytosis rate of abdominal macrophages in immunocompromised mice.
These findings indicate that Panax notoginseng rare saponin can enhance the vitality of abdominal macrophages and significantly improve their phagocytic capacity against invading organisms. Immunocompromised individuals are more prone to infections and diseases, with autoimmune conditions being associated with leukopenia and an increased risk of infection. 53 The fluctuation in WBC is indicative of the body's health status, as leukocytes play a crucial role in the immune system. 54 Among leukocytes, neutrophils constitute the largest component of the innate immune system. 55 Neutrophils are central to phagocytosis in the bloodstream, exhibiting remarkable deformability and potent phagocytic capabilities. 56 Upon bacterial invasion, neutrophils are recruited to the site of inflammation in response to chemokine signaling. These neutrophils, rich in lysosomal enzymes, participate in the phagocytosis and eradication of bacteria, as well as the breakdown of tissue debris.
This activity is essential for effectively mitigating the spread of infection within the body. 56 A reduction in absolute neutrophil count is frequently associated with various infectious diseases, particularly those caused by certain viral infections. Research suggests that combinations of rare saponins derived from Panax notoginseng can increase both WBC and absolute neutrophil levels in immunocompromised mice. These findings imply that rare saponin combinations from Panax notoginseng may enhance immune function, strengthen disease defense mechanisms, and reduce the risk of infection.
Free radicals can impair the body's immune system, as natural killer cells and cytotoxic lymphocytes are highly sensitive to oxidative stress.57,58 The activation of oxidative stress results in cellular structure damage, exacerbates inflammation, and accelerates the aging process. The Nrf2/HO-1 signaling pathway is pivotal in regulating antioxidant and anti-inflammatory responses by mitigating mitochondrial damage, modulating calcium ion fluxes, and controlling cell death. 59 Oxidative stress, precipitated by external factors, results in an elevated production of reactive oxygen species (ROS), which subsequently inflict damage on lymphocytes, culminating in hypo-immunity. A study investigating hepatic Nrf2/HO-1 expression reported an upregulation of both proteins, suggesting that rare saponin combinations from Panax notoginseng may contribute to the modulation of oxidative stress and immune function.
This study investigated the immunomodulatory effects of Panax notoginseng rare saponin combinations in cyclophosphamide-induced immunocompromised mice, highlighting the pivotal role of the Nrf2/HO-1 oxidative stress pathway in regulating immune cell function. Results demonstrated that Panax notoginseng rare saponin combinations significantly upregulated hepatic Nrf2 and HO-1 protein expression, alongside elevated expression of CD4+, CD8+, and CD3+ T-cell markers in splenic tissues. Immunofluorescence analysis further revealed enhanced co-localization of Nrf2 with CD3+ T cells in the spleen, suggesting that the Nrrf2/HO-1 pathway may improve T-cell functionality through antioxidant mechanisms, thereby amplifying immune responses.
As a central transcription factor in antioxidant defense, Nrf2 activates downstream targets such as HO-1 to scavenge ROS and mitigate oxidative damage. Oxidative stress is known to impair immune cell activity, including the proliferation and function of natural killer (NK) cells and T lymphocytes. In this study, cyclophosphamide-induced oxidative stress in the model group markedly suppressed hepatic Nrf2/HO-1 expression and reduced splenic CD4+, CD8+, and CD3+ T-cell populations. Conversely, Panax notoginseng rare saponin combinations treatment counteracted ROS-mediated lymphocyte damage by restoring Nrf2/HO-1 levels, which correlated with normalized CD4+, CD8+ ratios and enhanced phagocytic activity. These findings align with prior evidence that Nrf2 activation preserves immune cell viability by stabilizing mitochondrial function and calcium homeostasis, thereby preventing oxidative stress-induced apoptosis. 59
The dosing regimen spanned 14 days, which enabled observation of acute immunomodulatory responses but precluded evaluation of long-term efficacy or potential toxicity associated with Panax notoginseng rare saponin combinations. While cyclophosphamide-induced immunosuppression is a well-established model, its mechanism-primarily through DNA synthesis inhibition-may not fully recapitulate immune dysfunction arising from alternative etiologies, such as genetic defects or chronic infections. 24 These mechanistic discrepancies across immunosuppression models could constrain the generalizability of findings to broader clinical contexts. The findings of the current study demonstrated that the rare saponin combination of Panax notoginseng (ginsenosides Rb3, Rk3, and Rh4) enhances the immune function in immunocompromised mice. While the potential mechanism of action was preliminarily explored, the precise and comprehensive immune mechanism warrants further investigation. Although the immunomodulatory effects of Panax notoginseng's rare saponin combinations (ginsenosides Rb3, Rk3, and Rh4) have been initially demonstrated, the exact mechanism of action remains elusive. Immunohistochemistry may prove fortuitous in verifying specific mechanisms, and it is imperative to further investigate the relevant molecular signaling pathways using flow cytometry and Western blotting to elucidate the regulatory mechanisms comprehensively. Future studies should integrate in vitro cellular experiments (such as T cell differentiation into Th1/Th2/Th17 subsets) with molecular interaction investigations to further elucidate the underlying mechanisms.
Conclusion
The rare saponin combination of Panax notoginseng to immunocompromised mice resulted in the recovery of body weight and an increase in the indices of the thymus, spleen, and liver. In addition, phagocytosis of peritoneal macrophages increased, as did the number of lymphocytes, monocytes, neutrophils, and leukocytes. The rare saponin combination of Panax notoginseng markedly improved the immune function of immunosuppressed mice. Furthermore, this saponin combination elevated the levels of Nrf2 and HO-1 proteins in liver tissue, and CD4+ and CD8+ proteins in spleen tissue, The rare saponin combination of Panax notoginseng significantly upregulated the fluorescence intensity of CD3+ T-cell markers and Nrf2 antioxidant proteins in splenic tissues of immunocompromised mice.
Footnotes
Acknowledgments
Special thanks to the support of Yunnan Province's major science and technology projects and Wenshan College's Sanqi School of Medicine.
Ethical Considerations
This study was approved by the Dali University Laboratory Animal Ethics Committee (2021-PZ-202); Dali University Laboratory Animal Ethics Committee Address: 2 Hongsheng Road, Dali City, Dali Bai Autonomous Prefecture, Yunnan Province, China; Approval date: 6 December 2021;
Author Contributions
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Major science and technology special plan of Yunnan Province, (grant number 202002AA1000053, 202102AA310045).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All dataset were generated or analyzed in the current study.
Human and Animal Rights Statement
All procedures in this study were conducted in accordance with the Laboratory Animal Ethics Committee of Dali University(2021-PZ-202)* approved protocols.


