Abstract
Mesangial proliferative glomerulonephritis (MsPGN) is characterized by mesangial cell proliferation, inflammation, and extracellular matrix deposition in the mesangial area, which develops into glomerulosclerosis and contributes to end-stage renal disease. Justicidin B is a bioactive compound isolated from
Mesangial proliferative glomerulonephritis (MsPGN) is the most common renal disease in China, which is characterized by a pathological injury pattern that involves the proliferation of glomerular mesangial cells (MCs), an inflammatory response, and the accumulation of extracellular matrix (ECM). 1 In other research, MC proliferation was shown to play a crucial role in the inflammatory response and matrix accumulation in the glomerulus. 2 The abnormal proliferation of MCs results in the release of inflammatory cytokines, which leads to subsequent ECM deposition and interstitial fibrosis and glomerulosclerosis, and ultimately, end-stage renal disease. 3 Therefore, inhibiting the proliferation of MCs, inflammation, and ECM accumulation could decrease or delay the development of MsPGN. 4
Platelet-derived growth factor (PDGF) family members are the best-known growth factors involved in mediating the proliferation of MCs. Mesangial cells express PDGF subunit B (PDGF-B) chain mRNA and secrete gene products in the form of homodimers in vitro. 5 Platelet-derived growth factor-BB is an important mitogen in MCs activated by the glomerulonephritis-related inflammatory response. 6 Studies have confirmed that PDGF-BB promoted the production of ECM. In addition, the vascular smooth muscle cell proliferation induced by PDGF-BB increased the activity of reactive oxygen species (ROS), 7 and thus, antioxidants can inhibit the PDGF-BB-induced proliferation of vascular smooth muscle cells and the ROS signaling pathway. Antioxidants have also been shown to inhibit glomerulosclerosis and MC proliferation in glomerular disease models. 8 Therefore, antioxidants may be potential therapeutics for kidney diseases.
Recent studies have shown that oxidative stress plays an important role in the occurrence of MsPGN by affecting MC proliferation, phenotypic transformation, ECM deposition, and the inflammatory response.
9
Nuclear factor erythroid 2-related factor 2 (Nrf2) is known as an important receptor of oxidative stress and a key regulator of antioxidant reaction components. The expression of Nrf2 in tissue cells can promote the production of antioxidant enzymes to exert antioxidant stress and maintain the stability of the body’s internal environment.
10
Nuclear factor erythroid 2-related factor 2 is not only a transcription factor for cell self-protection under physiological conditions but is also a receptor in the body playing an important role in antioxidative stress and preventing the invasion of exogenous toxic substances under pathological conditions.
11
Under normal physiological conditions, Nrf2 is regulated by a Kelch-like ECH associated protein 1 (Keap1), which circulates between the cytoplasm and nucleus, and maintains the low concentration and transcriptional activity of Nrf2. Under oxidative stress, activated Nrf2 dissociates from Keap1 and translocates into the nucleus, where it binds to Maf protein and then to basal antioxidant-response elements to initiate the expression of antioxidant enzymes, including heme oxygenase 1 (HO-1),
The mammalian target of rapamycin (mTOR) is a giant serine/threonine protein kinase that plays a key role in protein synthesis, cell survival, proliferation, apoptosis, and activation. 15 Mammalian target of rapamycin activation has been confirmed in rat models of various chronic kidney diseases, including IgA nephropathy, 16 compensatory renal hypertrophy, 17 diabetic nephropathy, 18 membranous nephropathy, 19 and anti-glomerular basement membrane (anti-GBM) glomerulonephritis. 20 The PI3K/Akt/mTOR signaling pathway is one of the classical signaling pathways involved by mTOR. Reportedly, the Akt/mTOR signaling pathway is mainly activated in MCs. 21 Activation of the Akt/mTOR pathway can induce mesangial hypertrophy, whereas inhibition of this pathway can suppress MC hypertrophy. 22 In addition, accumulating evidence suggests that Akt signaling pathway can promote MC proliferation. The phosphorylation of Akt was involved in MC proliferation in an anti-Thy1 MsPGN rat model. 23,24

Information of Justicidin B: (a) Plant of
Previous studies have shown that PDGF-BB is essential for the proliferation of MCs before the development of glomerulosclerosis in an experimental glomerulonephritis model. 26 In this study, we used 40 ng/mL PDGF-BB to stimulate MCs and studied the effect of Justicidin B on their proliferation. The cells were treated with different concentrations of Justicidin B (0, 5, 10, 20, and 40 µg/mL) for 12, 24, and 48 hours. The Cell Counting Kit-8 (CCK-8) assay showed that PDGF-BB increased the proliferation of glomerular MCs, compared to the control group. Justicidin B inhibited the PDGF-BB-induced MC proliferation. With increasing concentrations of Justicidin B, glomerular MC proliferation was inhibited by Justicidin B in a dose-dependent manner, with an IC50 of approximately 29.51 µg/mL (Figure 1(c)). Therefore, treatment with 20 µg/mL Justicidin B for 24 hours was used in the subsequent experiments.
In Figure 2 , proliferating cell nuclear antigen (PCNA) is a nuclear protein associated with the cell cycle and serves as a marker of cell proliferation. Western blots (Figure 2(a)) and immunofluorescence assays (Figure 2(b)) were used to detect the expression of PCNA. The results showed that PDGF-BB increased the level of PCNA in HBZY-1 cells and that Justicidin B inhibited the expression of PCNA. To study whether the antiproliferative effect of Justicidin B was attributed to the induction of cell cycle arrest, the HBZY-1 cells were treated with Justicidin B for 24 hours and the cell cycle distribution was analyzed by flow cytometry (Figure 2(c)). Platelet-derived growth factor-BB reduced the percentage of cells in the G1 and S phases but increased those in the G2 phase, showing that PDGF-BB could promote cell cycle progression. These results showed that Justicidin B could block PDGF-BB-induced cell cycle progression by inhibiting S-G2 phase transition and arresting cells in the G1 phase.

Effect of Justicidin B on the proliferation of mesangial cells. (a) The expression with estern blot analysis of PCNA; β-actin is used as a loading control. The relative quantitation of PCNA protein, expressed as the ratio of PCNA/β-actin (*
The occurrence and development of MsPGN are also accompanied by an inflammatory response. Activated and proliferated MCs can produce a variety of inflammatory factors and chemokines, thereby promoting the proliferation of MCs. 27 Therefore, Western blots were performed to detect the expression of TNF-α and MCP-1. As shown in Figure 3, the inflammatory factors, TNF-α and MCP-1, were increased in HBZY-1 cells treated with PDGF-BB. Justicidin B reversed the inflammatory response by decreasing levels of the inflammatory factors. Therefore, our results demonstrated that Justicidin B inhibited the inflammatory response in the PDGF-BB-induced proliferation of MCs.

Expressions of TNF-α and MCP1 in mesangial cells by Western blot analysis; β-actin is used as a loading control. The relative quantitation of TNF-α and MCP1 protein (*
Mesangial cells are the most active intrinsic cells of the glomerulus. When stimulated, cell proliferation is the most common response. One of the main functions of MCs is to synthesize ECM (composed of collagen, glycoprotein, and proteoglycan). The oversynthesis and deposition of ECM are the key mechanisms of MsPGN. Collagen IV (Col-IV) and fibronectin (FN) are components of the ECM. Immunofluorescence staining (Figure 4(a)) and Western blot analysis (Figure 4(b)) showed that the expression levels of Col-IV and FN were upregulated significantly in PDGF-BB-treated HBZY-1 cells, compared to the control group. Justicidin B downregulated the expression of Col-IV and FN significantly. These results suggested that Justicidin B could reduce the accumulation of ECM by inhibiting the proliferation of HBZY-1 cells induced by PDGF-BB.

Justicidin B inhibited extracellular matrix accumulation. (a) Immunofluorescence staining of fibronectin and collagen IV (×200) in HBZY-1 cells. (b) Western blot analysis of fibronectin and collagen IV in HBZY-1 cells; densitometric results of collagen IV and fibronectin. Error bars represent SEM.
In other research, inflammatory reactions and oxidative stress were activated in glomerular MCs stimulated by various factors, such as high glucose, 28 angiotensin II (Ang-II), and aldosterone. 29 Excessive ROS can lead to hypertrophy and apoptosis, ECM aggregation, and cytokine secretion in MCs, resulting in the dysfunction and structural damage of kidney. 30 Superoxide dismutase, as a downstream target gene of Nrf2, is the key enzyme involved in scavenging oxygen free radicals. Malondialdehyde (MDA) is the end-product of lipid peroxidation induced by free radicals in vivo, which can cause cross-linking polymerization of proteins, nucleic acids, and other life macromolecules, resulting in cytotoxicity. Malondialdehyde content is an important parameter reflecting the body’s potential antioxidant capacity. Thus, we analyzed the concentrations of SOD, MDA, and T-AOC in the MCs. The results demonstrated (Figure 5(a)) that after intervention with Justicidin B, the SOD activity and T-AOC content of the MCs increased, whereas the MDA concentration decreased. To further understand the mechanism of Justicidin B in antioxidants, Western blot analysis was used to detect the expression of key proteins in oxidative stress-related signaling pathways in the cells. Our study demonstrated that the protein levels of Nrf2 and HO-1 were decreased compared to the control group. Justicidin B dramatically increased the protein levels of Nrf2 and HO-1 (Figure 5(b)). These results suggested that Justicidin B could inhibit oxidative stress, ECM deposition, and inflammation by enhancing the Nrf2/HO-1 signaling pathway.
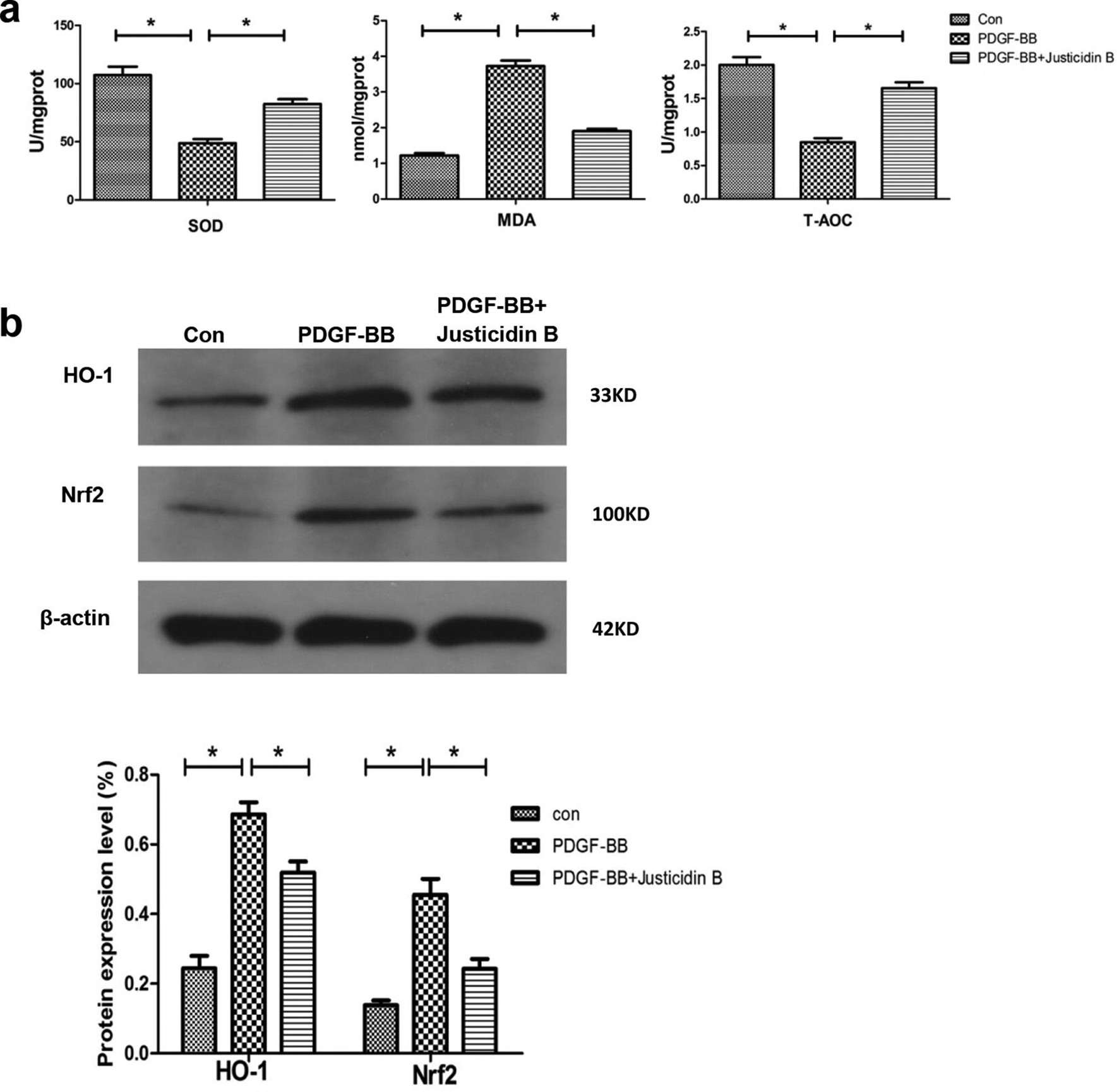
Justicidin B reduced the effects on platelet-derived growth factor-BB induced oxidative stress injury in mesangial cells: (a) superoxide dismutase activity, malondialdehyde, and T-AOC (*
Previous studies reported that the Akt/mTOR and MAPK/ERK signaling pathways could promote MC proliferation and play a crucial role in inflammation.
31,32
Thus, in order to further confirm the molecular mechanisms underlying the antiproliferative and anti-inflammatory effects of Justicidin B in PDGF-BB-induced HBZY-1 cells, the protein levels of protein kinase B (Akt), phosphorylated- protein kinase B (p- Akt); mammalian target of rapamycin (mTOR), phosphorylated-mammalian target of rapamycin (p-mTOR) and extracellular regulated protein kinases (ERK), phosphorylated- extracellular regulated protein kinases (p-ERK) were analyzed by Western blots to investigate the effects of Justicidin B on the Akt/mTOR and MAPK/ERK signaling pathways. As shown in Figure 6, the protein expression of p-Akt, p-mTOR, and p-ERK was significantly increased in the PDGF-BB group (

Effects of Justicidin B on Akt/mTOR and MAPK/ERK signaling pathways in platelet-derived growth factor-BB-induced HBZY-1 cells. Activation of Akt, p-Akt; mTOR, p-mTOR; and ERK, p-ERK were detected by Western blot assay; β-actin was used as a loading control. Relative quantitative analysis of p-Akt/Akt and p-mTOR/mTOR, MAPK/ERK and p-ERK/ERK (*
In this study, we preliminarily elucidated the mechanism of the anti-MsPGN effect of Justicidin B. The inhibition of MC proliferation was confirmed in vitro. This was the first study to report that Justicidin B inhibited the proliferation of MCs.
Mesangial proliferative glomerulonephritis is a very common chronic nephritis disease, and MC proliferation contributes to mesangial matrix homeostasis. 33 Mesangial cell proliferation and matrix expansion are biological responses to glomerular diseases or injuries. 34,35 Justicidin B was found to inhibit the proliferation of MCs induced by PDGF-BB in a dose-dependent manner. Compared to the model group, the expression of PCNA protein in the intervention group decreased. A decrease in the number of immunofluorescently staining cells was also seen. Further flow cytometry analysis showed that the cells were mainly concentrated in the G1 phase. The inflammatory response and ECM accumulation are the basic manifestations of MsPGN. After the Justicidin B treatment of PDGF-BB-stimulated MCs, the expression of TNF-a and MCP1 decreased significantly, indicating that the inflammatory response decreased. The immunofluorescent staining and protein expression of FN and Col-IV showed that Justicidin B could significantly reduce ECM accumulation induced by PDGF-BB stimulation. We further investigated the oxidative stress response of PDGF-BB-stimulated cells and found that Justicidin B could increase the activity of SOD and the content of T-AOC and decrease the content of MDA. Its antioxidant effect was mediated by the Nrf2/HO-1 signaling pathway. The results further confirmed that the effect of Justicidin B on MsPGN was mediated by the Akt/mTOR signaling pathway.
In conclusion, Justicidin B showed a positive effect on inhibiting proliferation, ECM accumulation, oxidative stress, and the inflammatory response induced by PDGF-BB in HBZY-1 cells. Most importantly, we found that the underlying mechanism of the positive effects may be associated with the ability of Justicidin B to enhance the Nrf2/HO-1 pathway and suppress the Akt/mTOR pathway. Therefore, the findings of this studies identify a potential drug candidate for the treatment of MsPGN.
Experimental Methods
Cell Culture
The rat glomerular MC line (HBZY-1) was purchased from the Wuhan Academy of Life Sciences, Wuhan, China. The cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM, Gibco, United States) containing 10% fetal bovine serum (FBS, BI, United States) at 37°C in a 5% CO2 atmosphere. First, HBZY-1 cells were treated with different concentrations of PDGF-BB containing 2% FBS. Second, after pre-incubation in serum-free DMEM for 12 hours, the cells were treated with 40 ng/mL PDGF-BB for 24 hours in DMEM containing 2% FBS with different concentrations of Justicidin B (0, 5, 10, 20, and 40 µg/mL) for 12, 24, and 48 hours. For the following experiments, the cells were incubated in serum-free DMEM for 12 hours and then divided into 3 groups as follows: (a) control, where the cells were exposed to DMEM with 2% FBS; (b) PDGF-BB, where the cells were cultured in DMEM with 2% FBS containing 40 ng/mL PDGF-BB; and (c) the PDGF-BB + Justicidin B (20 µg/mL) group. The cells were exposed to the above conditions for 24 hours.
Cell Proliferation Assay
HBYZ-1 cell proliferation was detected by the CCK-8 (BIOSHARP) assay. Briefly, cells in the logarithmic phase were selected and the cell concentration was adjusted to 5 × 103/mL. HBZY-1 cells were seeded into 96-well plates and cultured until they were completely adherent. After pre-incubation in serum-free DMEM for 12 hours, the cells were treated with 20 ng/mL PDGF-BB (Peprotech) for 24 hours. After treatment with PDGF-BB or Justicidin B, 10 µL of CCK-8 solution was added to each well for an additional 4 hours. The number of viable cells was measured using a microplate reader (Molecular Devices, United States) at an absorbance of 450 nm and growth curves were generated.
Cell Cycle Analysis
A total of 5 × 105 cells/well were seeded in 6-well plates and starved in serum-free DMEM at 37°C. After 12 hours of starvation, the cells were treated with PDGF-BB containing 2% FBS for 24 hours and then Justicidin B was added for 24 hours. The cells were trypsinized, washed with phosphate-buffered saline (PBS), and stained using the Cell Cycle Test Kit (Nanjing Kaiji Biotech, China) according to the manufacturer’s instructions. The cell cycle was analyzed by flow cytometry (Beckman, United States).
Enzyme-Linked Immunosorbent Assay
Changes in the SOD, MDA, and T-AOC activity in the MC supernatant were detected according to the instructions of the corresponding Enzyme-Linked Immunosorbent Assay kits (Nanjing Jiancheng, PR China).
Immunofluorescence
HBZY-1 cells cultured in the plate were immersed in PBS (Gibco, United States) for 3 times for 3 minutes each. The slides were fixed in 4% polyformaldehyde for 15 minutes, immersed in PBS 3 times. The cells were permeabilized in 0.5% Triton X-100 (Biyuntian, China) at room temperature for 20 minutes (the antigen expressed on the cell membrane omitted this step). The slides were immersed in PBS 3 times, dried by absorbent paper, and normal goat serum (Wuhan Boshide, China) was dripped on the slides. Absorbent paper sucks off the sealing liquid and it does not require further wash; then, enough diluted primary reactance was added to each slide drop and put into the wet box, and incubated at 4°C overnight. The fluorescent secondary reactant was added and immersed in phosphate buffered saline tween-20 (PBST) climbing tablets 3 times, after using an absorbent paper to absorb the excess liquid on the climbing tablets. Then, Col-IV antibody (1:100; Abcam, United Kingdom), FN antibody (1:100; Abcam, United Kingdom), or PCNA antibody (1:100; Abcam, United Kingdom) was added to the slides and incubated at 20°C to 37°C in a wet box for 1 hour, the slides were immersed in PBST 3 times, each time for 3 minutes, and 4’,6-diamidino-2-phenylindole (DAPI) (Biyuntian, China) was dropped on and the slides were incubated in the dark for 5 minutes to dye the specimen. The slide was washed with PBST, the remaining liquid was absorbed with a piece of absorbent paper, the slides were sealed with sealing liquid containing antifluorescence quenching agent. The results were observed under fluorescence microscopy and the images were collected.
Western Blot Analysis
The HBZY-1 cells were treated with PBS and placed on ice, then lysate was added. After lysing, the cells were scraped off with a clean spatula and centrifuged at 12 000 rpm for 5 minutes at 4°C. The centrifuged content was stored at −20°C. Three parallel holes were made, each of which had a volume of 20 µL. Phosphate-buffered saline was added to 2 parallel holes as blank controls. bicinchoninic acid (BCA) from the kit was added and the optical density (OD) 568 nm was measured. The prepared glue was fixed on the electrophoresis tank, the electrophoresis solution was poured into the storage tank, and the membrane transfer tank was filled with electric fluid to start the transfer.
The polyvinylidene fluoride (PVDF) membrane was soaked in tris-buffered saline tween-20 (TBST) containing 5% skim milk powder and sealed in a shaker at room temperature for 2 hours. The phosphorylated protein was blocked with 1% bovine serum albumin. The antibodies against MCP1 (1:1000), TNF-α (1:1000), PCNA (1:2000), HO-1 (1:1000), Nrf2 (1:1000), Akt (1:1000), p-Akt (1:10 000), mTOR (1:1000), p-mTOR (1:1000), ERK (1:1000), p-ERK (1:1000), and β-actin (1:1000; Abcam, United States) were diluted with blocking solution and incubated overnight at 4°C. The PVDF membrane was fully washed in TBST 5 times. The secondary antibody (1:50 000) labeled with horseradish peroxidase was added and incubated in a shaker at 37°C for 2 hours. The PVDF membrane was fully washed in TBST. The reinforcement solution and a stable peroxidase solution of the electrochemiluminescence (ECL) reagent were mixed in a proportion of 1:1 and the working solution was dropped onto the PVDF membrane. Filter paper was used to absorb the excess substrate solution, the membrane was covered with preservative film, then X-ray film, and then successively put into developer and fixer to develop the film. The film was dried, scanned, and the grayscale values of the film were analyzed with impact point prediction (IPP).
Statistical Analysis
The data are expressed as the mean ± SEM of at least 3 repeated experiments. Statistical significance was analyzed by one-way factorial ANOVA, followed by IBM SPSS 22.0 software. Statistical significance was defined for
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (Grant no. 31570343).
