Abstract
Keywords
Introduction
Psoriasis is a chronic, relapsing, immune-mediated papulosquamous skin disease.It is characterized by the presence of scaly red plaques on the skin surface. 1 In the United States, psoriasis affects approximately 3.2% of adults and 0.13% of children, with an incidence rate of approximately 80 new cases per 100,000 individuals annually.2,3 Globally, approximately 125 million individuals are affected by psoriasis, and the prevalence of the condition varies significantly across regions, ranging from 0.5% in parts of Asia to 8% in Norway. 4 Reports on the prevalence of the disease vary by country, with the overall prevalence estimated to range between 0.14% and 1.99%. 5 The quality of life for most psoriasis patients is significantly impaired due to the disease, with many experiencing major negative impacts on mental health and a reduced life expectancy resulting from disease complications. Compared to the general population, psoriasis patients are at a higher risk of developing depression (up to 20%) and exhibiting suicidal thoughts or behaviors.6,7
Psoriasis is clinically classified into four types: pustular, erythrodermic, plaque, and arthritic, with plaque psoriasis being the most common. Plaque psoriasis accounts for approximately 80% to 90% of all psoriasis cases. Plaque psoriasis can occur on any part of the body, with commonly affected areas including the scalp, trunk, buttock folds, and extensor surfaces, such as the elbows and knees. Lesions of plaque psoriasis range from small, red, scaly papules to large, thick plaques. The affected areas typically have well-defined borders and are often symmetrically distributed. Additionally, it may appear at sites of trauma, such as those caused by scratching, cuts, or pressure. When the scales on the plaque surface are removed, pinpoint bleeding may occur, resulting in the formation of the Auspitz sign. 8 Treatment options for psoriasis include topical treatments and phototherapy for mild cases, as well as systemic drug therapies for moderate to severe cases. 9 Local treatments include targeted phototherapy, 10 corticosteroids, 11 and non-corticosteroid therapies. 12 For systemic treatment, immunosuppressive drugs, such as methotrexate, 13 may be considered, while biologics, such as tumor necrosis factor-alpha (TNF-α), interleukin-17 (IL-17), and IL-23 inhibitors, are used for refractory cases.14,15 Although many conventional medicines (CM) are available, several issues still need to be addressed in the systemic treatment of psoriasis. For instance, acitretin capsules may cause mild side effects, including dose-dependent hair loss and dryness. 16 Long-term use of corticosteroids is also associated with adverse reactions, such as nephrotoxicity and bone marrow suppression. 17 Although biologics are reported to be more effective than traditional oral medications, a significant proportion of patients may still experience allergic reactions or gastrointestinal issues. 14 However, the cost of these drugs is a significant factor that reduces treatment adherence and accessibility. Therefore, further research on new drugs is of considerable value, particularly to ensure that their efficacy and safety are comparable to existing CM.
Recent studies have demonstrated that traditional Chinese medicine (TCM) significantly improves clinical symptoms and enhances the quality of life in psoriasis patients. Traditional Chinese patent medicines are widely used in clinical practice for treating psoriasis due to their advantages, including convenience, multiple ingredients, multi-target action, diverse effects, and low side effects, and have been recommended in relevant clinical guidelines. 18 Combining TCM with CM can effectively control erythema, thickening, and scaling symptoms, maintain long-term efficacy, reduce recurrence rates, prevent complications, such as psoriatic arthritis, and achieve a holistic treatment effect. Multiple meta-analyses indicate that the combination of various traditional Chinese patent medicines and conventional Western medications produces unmatched effects in improving PASI scores and inflammatory markers in psoriasis patients.19–21 However, due to the wide variety of traditional Chinese patent medicines and the lack of clinical efficacy comparisons between different drugs, determining the optimal treatment approach remains challenging. Therefore, this study makes full use of existing clinical research data, with psoriasis patients as the study subjects. The control group will receive conventional Western medicine treatment, while the experimental group will receive a combination of traditional Chinese patent medicines with conventional Western medicine in a randomized controlled trials (RCTs). A network meta-analysis will be conducted to assess the effectiveness and safety of different traditional Chinese patent medicines in treating psoriasis, providing evidence-based support for psoriasis treatment.
Materials and Methods
This study strictly adhered to the latest guidelines and items from the systematic review and meta-analysis (PRISMA) 22 and has been registered on the PROSPERO (No. CRD420250655020).
Inclusion Criteria
Study Type: Published clinical RCTs evaluating the efficacy and safety of traditional Chinese patent medicines in treating plaque psoriasis, with no restrictions on language or publication date. Study Participants: Patients with a clear diagnosis of plaque psoriasis based on the psoriasis diagnosis and treatment guidelines, with no restrictions on age, race, gender, or region.23–25 Since the focus of this review is plaque psoriasis, clinical trials involving other subtypes of psoriasis, such as psoriatic arthritis, guttate psoriasis, palmoplantar pustulosis, and erythrodermic psoriasis, are excluded from the study. Interventions:The experimental and control groups must receive consistent treatments for other underlying conditions, such as antihypertensive, antidiabetic, and lipid-lowering therapies. The control group will receive CM treatments recommended by guidelines, while the experimental group will receive either a combination or no combination of approved traditional Chinese patent medicines alongside the control treatment, with no restrictions on the method, dosage, or duration. Outcome Measures: The primary outcome will be the Psoriasis Area and Severity Index (PASI) score, with secondary outcomes including the total effective rate, TNF-α, IL-17, and CD4+/CD8+ levels. Additionally, to assess the safety of the interventions, adverse event rates will be included as secondary outcomes. At least one outcome measure must be reported in the studies for inclusion.
Exclusion Criteria
(a)Non-RCT studies; (b)studies involving non-Western conventional treatments in the interventions; (c) non-clinical studies; studies unrelated to plaquesoriasis or associated diseases; (d)studies with incomplete or erroneous data; (e)duplicate publications (only the most complete data retained); (f)studies with inaccessible full texts; (g) reviews, meta-analyses, articles, and commentaries.
Search Methods and Strategy
A computer search was performed on PubMed, Web of Science, Cochrane Library, EMbase, CNKI, VIP, Wanfang, and SinoMed for RCTs involving the treatment of psoriasis with traditional Chinese patent medicines, covering the period from the inception of the databases until January 3, 2025. The search was conducted using keywords such as “plaque psoriasis”, “Chinese patent medicine”, “randomized controlled trial”, “tablets”, “powders”, “pills”, “capsules”, “granules”, and other related terms in both Chinese and English databases, combining subject terms and free words. Tables S1-5 summarize the search strategies employed in the aforementioned databases.
Literature Screening and Data Extraction
Two researchers independently screened the literature and extracted data, importing the retrieved citations into NoteExpress software for deduplication, with some articles requiring manual removal of duplicate entries. The articles were screened according to the inclusion and exclusion criteria, and only RCTs were included. In case of disagreement between the two researchers, a third party was involved to resolve the issue. Data extraction and entry were carried out using Excel sheets, with the extracted information including the first author's surname, publication year, study design, blinding, number of participants, disease characteristics, average age, interventions, treatment duration, and outcome measures.
Quality Assessment of Included Studies
Two researchers used RevMan 5.4.1 statistical software to perform risk of bias assessments on the included studies using the Cochrane Risk of Bias tool from the 5.3 version of the Cochrane Handbook to evaluate the methodological quality of the included studies. 26 The results were cross-checked, and in case of disagreements, a third party assisted in the decision-making. The assessment included the randomization process, deviation from the intended interventions, completeness of outcome data, outcome assessment process, selective reporting bias, and others, with each aspect containing 1 to 7 specific questions. The results were classified as “high risk,” “low risk,” or “some risk.”
Statistical Analysis
Stata 16.0 was used to perform network meta-analysis. For dichotomous variables, relative risk (odds ratio, OR) was used as the effect measure, while for continuous variables, mean difference (MD) or standardized mean difference (SMD) was used. The 95% confidence interval (CI) were calculated and presented. If the 95% CI for dichotomous variables did not include 1, or for continuous variables did not include 0, the intergroup differences were considered statistically significant. The surface under the cumulative ranking curve (SUCRA) was used to rank the efficacy of various interventions, with a higher SUCRA indicating better efficacy. A league table was constructed to display the OR/MD values and 95% CIs from direct pairwise comparisons of interventions. If the 95% CI includes 0 or 1, the result is considered non-significant. If OR > 1 or MD < 0, the row intervention is considered superior to the column intervention, and vice versa. A “comparison-correction” funnel plot was used to assess the presence of small sample effects or publication bias, with visual confirmation of asymmetry. Additional tests, including the Egger and Begg tests, were also conducted.27,28
Results
Literature Search and Basic Characteristics
The PRISMA flow diagram illustrates the process of literature screening, study selection, and reasons for exclusion, as shown in Figure 1. The preliminary search identified 2434 articles. After removing duplicates through automatic and manual deduplication using EndNote X9, 1088 articles were retained. Irrelevant and clearly non-eligible articles, such as animal cell studies, surgical-related research, case reports, and reviews, were excluded based on title and abstract, and 324 articles were downloaded for further screening. After reviewing the full texts, studies that did not meet the inclusion criteria were excluded, and 50 RCTs were finally included. A total of 1719 patients were included, all clinically diagnosed with plaque psoriasis. The treatment group consisted of 860 patients, and the control group consisted of 859 patients, with an average age ranging from 25 to 45 years. All included studies focused on plaque psoriasis as the subtype, and in each study, CM was used as the control group. The following is a list of CM used as control treatments: Acitretin, Compound Amino Peptide Preparation, Methotrexate, Roxithromycin, Penicillin, Vitamin A, corticosteroids, and other topical medications. The treatment duration in all eligible studies ranged from 8 to 12 weeks. A total of eight traditional Chinese patent medicines were involved: Total Glycosides of Paeony Capsule (TGP), Compound Glycyrrhizin Capsules (CGZ), Compound Qingdai Capsules (CQC), Koyin Pills (KYP), Tripterygium Wilfordii Glycosides Tablets (TWG), Xiaoyin Capsules (XY), Psoriasis Capsules (PC), and Zidan Psoriasis Capsules (ZDP). In terms of outcome indicators, a total of 44 studies assessed the total efficacy rate, 37 studies evaluated the PASI score, 17 studies assessed TNF-α, 23 studies evaluated IL-17, 14 studies evaluated CD4+/CD8+ levels, and 41 studies assessed adverse reactions. The basic characteristics of the included RCTs are presented in Table 1.

Literature Screening Process.
Basic Characteristics of Included Articles.
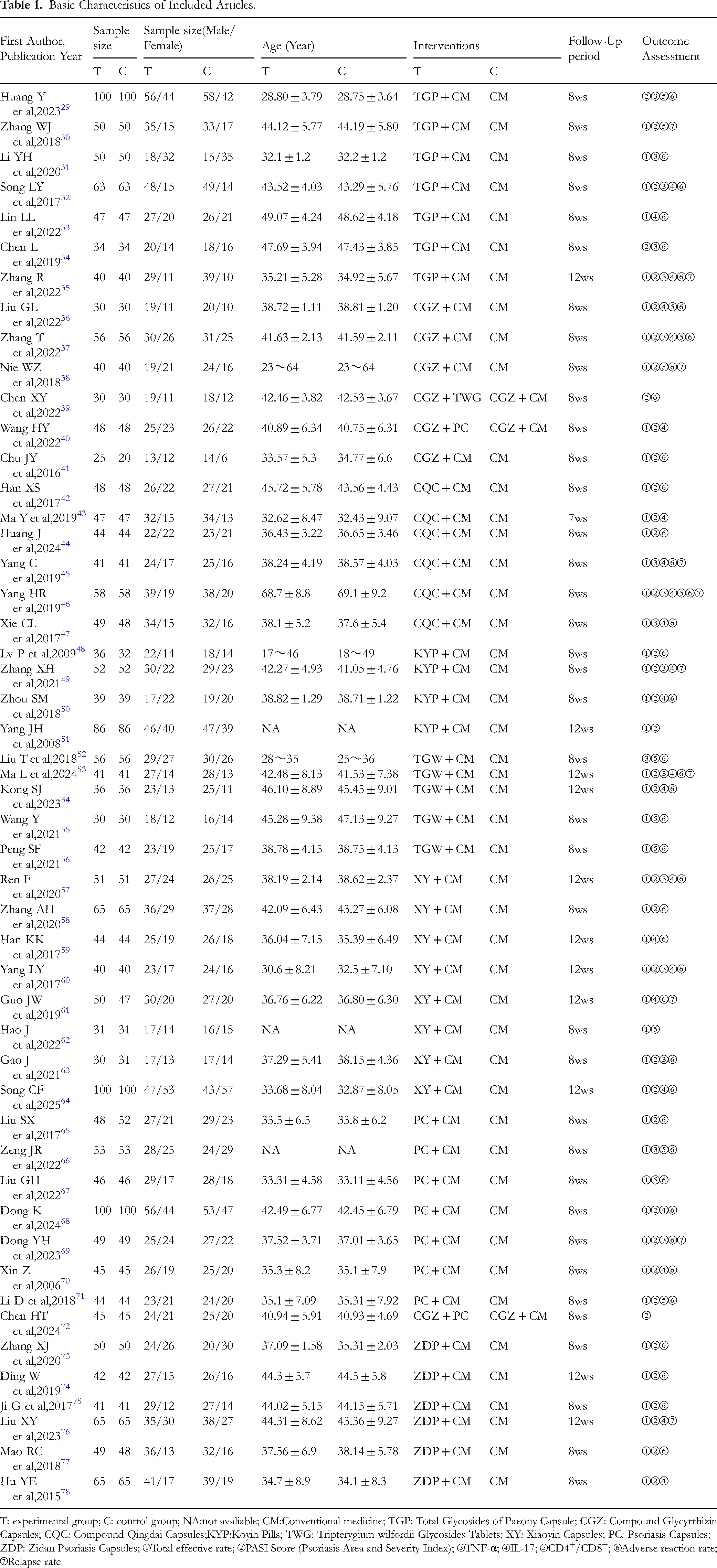
T: experimental group; C: control group; NA:not avaliable; CM:Conventional medicine; TGP: Total Glycosides of Paeony Capsule; CGZ: Compound Glycyrrhizin Capsules; CQC: Compound Qingdai Capsules;KYP:Koyin Pills; TWG: Tripterygium wilfordii Glycosides Tablets; XY: Xiaoyin Capsules; PC: Psoriasis Capsules; ZDP: Zidan Psoriasis Capsules; ①Total effective rate; ②PASI Score (Psoriasis Area and Severity Index); ③TNF-α; ④IL-17; ⑤CD4+/CD8+; ⑥Adverse reaction rate; ⑦Relapse rate
Quality Assessment of Included Studies
The 50 included studies were all RCTs. Regarding random sequence generation, 24 studies29–32,34,37–50,58,61,63–68 used the random number table method, 1 study 33 used random drawing, and 1 study 72 used stratified block randomization. These studies were all assessed as “low risk.” One study 56 grouped based on treatment plan differences, and two studies69,76 based on the order of patient visits, both of which were assessed as “high risk.” Other studies did not report the randomization method and were assessed as “unclear risk.” Regarding allocation concealment, four studies32,61,68,73 used the envelope method, rated as “low risk.” The remaining studies did not report allocation concealment methods and were rated as “unclear risk.” In terms of blinding, five studies34,58,64,68,76 used double-blind methods, rated as “low risk,” while the remaining studies did not mention blinding and were rated as “unclear risk.” The data in the included studies were complete, and the outcome evaluations were reliable, rated as “low risk.” Regarding selective reporting, no study protocols were found in clinical trial registries, and all were rated as “unclear risk.” Regarding other potential sources of bias, no study reported funding sources, and all were rated as “unclear risk.” The result figures were generated using RevMan 5.4.1 software, as shown in Figure 2.
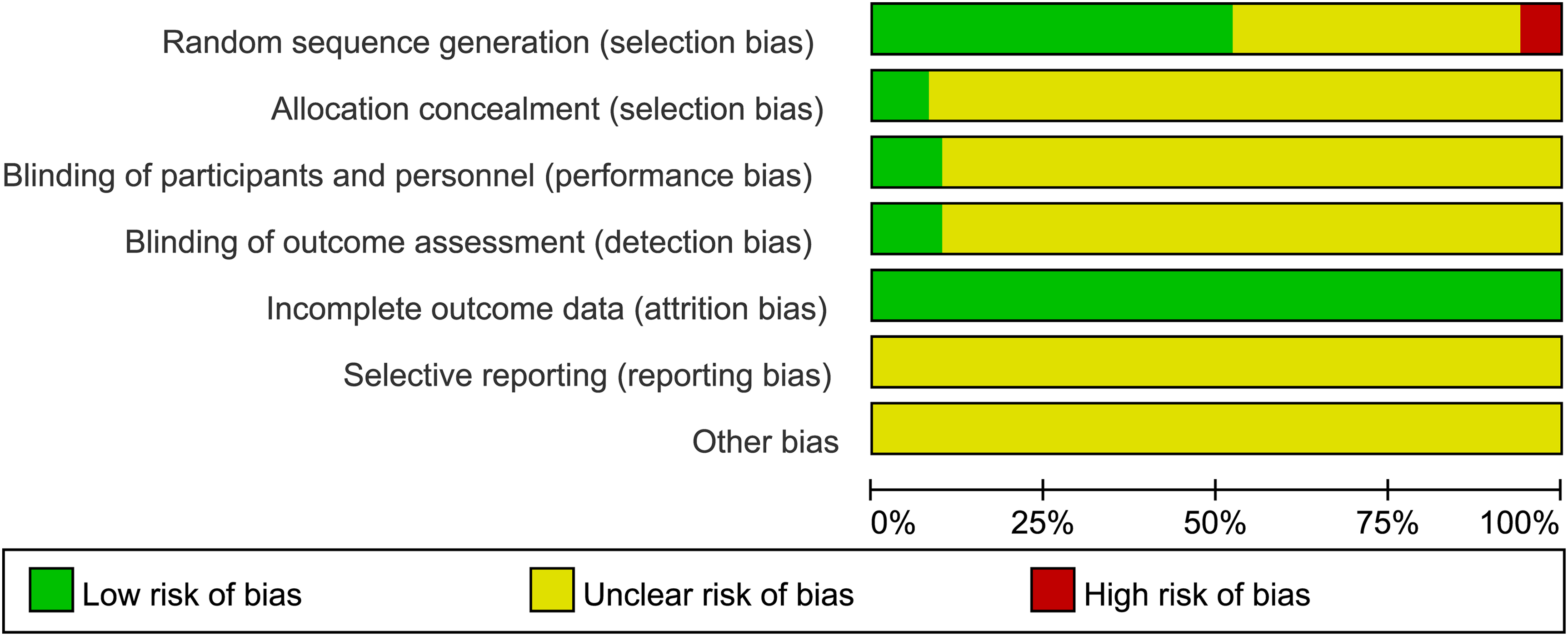
Percentages of Included Literatures that Produced Risks of Bia.
Total Effective Rate
Evidence Network
Forty-four RCTs30–33,35–38,40–51,53–71,73–78 reported the total effective rate, involving nine interventions: TGP, CGZ, CQC, KYP, TWG, XY, and PC combined with CM and CM alone. The total sample size was 4307 participants. The evidence network was centered on CM, and no closed loops between interventions were identified, making inconsistency tests unnecessary, as shown in Figure 3. In the evidence network diagram, each circle represents a different intervention, with the size of the circle corresponding to the sample size for each intervention. The thickness of the lines between the circles represents the number of studies comparing the interventions, and the straight lines represent direct or indirect comparisons between pairs of interventions. There is evidence of both direct and indirect comparisons between interventions, fulfilling the basic conditions for network meta-analysis (as detailed below).

Evidence Network Diagram of Different Outcome Indicators.
Network Meta-Analysis and SUCRA Ranking
Pairwise comparisons were conducted among the nine interventions, resulting in 36 comparisons. Network meta-analysis showed that TGP + CM (OR = 1.13, 95% CI [1.05, 1.22]), CGZ + CM (OR = 1.27, 95% CI [1.13, 1.43]), CQC + CM (OR = 1.15, 95% CI [1.06, 1.24]), KYP + CM (OR = 1.24, 95% CI [1.11, 1.39]), TWG + CM (OR = 1.17, 95% CI [1.06, 1.29]), XY + CM (OR = 1.26, 95% CI [1.17, 1.36]), PC + CM (OR = 1.19, 95% CI [1.11, 1.27]), and ZDP + CM (OR = 1.19, 95% CI [1.10, 1.28]) all outperformed CM in improving total efficacy. Furthermore, XY + CM was superior to TGP + CM (OR = 1.12, 95% CI [1.00, 1.24]), and the difference was statistically significant (P < 0.05), while the other interventions showed no statistically significant differences, as shown in Figure 4.

Network Meta-analysis Comprising the Effects of Total Effective Rate.
In studies reporting total efficacy, the cumulative ranking chart for the nine interventions is shown in the figure. The effectiveness of these interventions is ranked according to SUCRA values, with higher SUCRA values indicating better efficacy in improving total effectiveness. The probability ranking is as follows: CGZ + CM (83.1%) > XY + CM (82.3%) > KYP + CM (74.4%) > PC + CM (51.9%) > ZDP + CM (51.9%) > TWG + CM (45.0%) > CQC + CM (33.8%) > TGP + CM (27.6%) > CM (1.6%), as shown in Table 2.
Ranking of SUCRA Probabilities for Each Outcome Indicator.

PASI Score
Evidence Network
Thirty-seven RCTs29,30,32,34–44,46,48–51,53,54,57,58,60,63–65,68–78 reported PASI scores, involving 11 interventions with a total sample size of 3763 participants. The evidence network was centered on CM, and no closed loops were formed between interventions, making inconsistency testing unnecessary, as shown in Figure 3.
Network Meta-Analysis and SUCRA Ranking
Pairwise comparisons of the 11 interventions resulted in 55 comparisons. Network meta-analysis showed that TGP + CM (MD = -1.88, 95% CI [-2.66, −1.10]), CGZ + CM (MD = -1.16, 95% CI [-2.03, −0.29]), KYP + CM (MD = -1.35, 95% CI [-2.21, −0.49]), XY + CM (MD = -1.21, 95% CI [-1.97, −0.44]), PC + CM (MD = -1.59, 95% CI [-2.36, −0.82]), CGZ + TWG (MD = -2.22, 95% CI [-4.17, −0.27]), and ZDP + CM (MD = -1.48, 95% CI [-2.18, −0.78]) outperformed CM in improving PASI scores. The differences were statistically significant (P < 0.05), while the other interventions showed no significant differences, as shown in Figure 5.

Network Meta-analysis Comprising the Effects of PASI Score.
In studies reporting PASI scores, the cumulative ranking chart for the 11 interventions is shown. The effectiveness of these interventions is ranked according to SUCRA values, with higher SUCRA values indicating better improvement in PASI scores. The probability ranking is as follows: CGZ + TWG (81.2%) > TGP + CM (79.1%) > PC + CM (68.1%) > ZDP + CM (62.1%) > KYP + CM (55.2%) > CGZ + PC (51.4%) > XY + CM (47.7%) > CGZ + CM (45.3%) > CQC + CM (45.3%) > TWG + CM (45.3%) > CM (2.9%), as shown in Table 2.
TNF-α
Evidence Network
Seventeen RCTs29,31,32,34,35,37,45–47,49,52,53,57,60,63,66,69 reported TNF-α, involving eight interventions with a total sample size of 1726 participants. The evidence network was centered on CM, and no closed loops were formed between interventions, making inconsistency testing unnecessary, as shown in Figure 3.
Network Meta-Analysis and SUCRA Ranking
Pairwise comparisons of the 10 interventions resulted in 45 comparisons. Network meta-analysis indicated that, compared with CM, TGP + CM (MD = -1.32, 95% CI [-2.55, −0.08]) and PC + CM (MD = -3.65, 95% CI [-5.63, −1.66]) were more effective in reducing TNF-α levels. Furthermore, PC + CM was superior to XY + CM (MD = -2.56, 95% CI [-5.11, −0.01]) and CQC + CM (MD = -2.94, 95% CI [-5.48, −0.39]), with statistically significant differences (P < 0.05), while the other comparisons did not show significant differences, as shown in Figure 6.

Network Meta-analysis Comprising the Effects of TNF-α.
In studies reporting TNF-α, the cumulative ranking chart for the eight interventions is shown. The effectiveness of these interventions is ranked according to SUCRA values, with higher SUCRA values associated with a reduction in TNF-α levels. The probability ranking is as follows: PC + CM (94.8%) > KYP + CM (70.6%) > TGP + CM (54.6%) > TWG + CM (53.4%) > XY + CM (46.5%) > CQC + CM (35.4%) > CGZ + CM (33.4%) > CM (11.3%), as shown in Table 2.
IL-17
Evidence Network
Twenty-three RCTs32,33,35–37,40,43,45–47,49,50,53,54,57,59–61,64,68,70,76,78 reported IL-17, involving ten interventions with a total sample size of 2410 participants. The evidence network was centered on CM, and no closed loops were formed between interventions, making inconsistency testing unnecessary, as shown in Figure 3.
Network Meta-Analysis and SUCRA Ranking
Pairwise comparisons of the 10 interventions resulted in 45 comparisons. Network meta-analysis showed that, compared with CM, TGP + CM (MD = -1.03, 95% CI [-1.77, −0.30]), CQC + CM (MD = -0.70, 95% CI [-1.33, −0.07]), KYP + CM (MD = -3.35, 95% CI [-4.31, −2.39]), XY + CM (MD = -0.69, 95% CI [-1.25, −0.12]), PC + CM (MD = -1.14, 95% CI [-2.02, −0.25]), and ZDP (MD = -1.13, 95% CI [-2.01, −0.25]) were more effective in reducing IL-17 levels. Additionally, KYP + CM was superior to CQC + CM (MD = -2.65, 95% CI [-3.80, −1.50]), CGZ + CM (MD = -2.63, 95% CI [-3.94, −1.31]), TGP + CM (MD = -2.32, 95% CI [-3.52, −1.11]), TWG + CM (MD = -2.46, 95% CI [-3.78, −1.14]), XY + CM (MD = -2.66, 95% CI [-3.78, −1.55]), PC + CM (MD = -2.22, 95% CI [-3.52, −0.91]), CGZ + PC (MD = -2.52, 95% CI [-4.34, −0.70]), and ZDP (MD = -2.22, 95% CI [-3.52, −0.92]), with statistically significant differences (P < 0.05). No significant differences were found for other interventions, as shown in Figure 7.

Network Meta-analysis Comprising the Effects of IL-17.
In studies reporting IL-17, the cumulative ranking chart for the ten interventions is shown. The effectiveness of these interventions is ranked based on SUCRA values, with higher SUCRA values associated with a reduction in IL-17 levels. The probability ranking is as follows: KYP + CM (95.0%) > PC + CM (63.3%) > ZDP + CM (62.8%) > TGP + CM (58.3%) > TWG + CM (49.7%) > CGZ + PC (46.3%) > CGZ + CM (40.5%) > CQC + CM (38.8%) > XY + CM (37.5%) > CM (3.0%), as shown in Table 2.
CD4+/CD8+
Evidence Network
Fourteen RCTs29,30,36–38,46,52,55,56,62,66,67,71 reported CD4+/CD8+ ratios, involving seven interventions with a total sample size of 1272 participants. The evidence network was centered on CM, and no closed loops were formed between interventions, making inconsistency testing unnecessary, as shown in Figure 3.
Network Meta-Analysis and SUCRA Ranking
Pairwise comparisons of the seven interventions resulted in 21 comparisons. Network meta-analysis showed that TGP + CM (MD = 2.10, 95% CI [0.84, 3.37]), CGZ + CM (MD = 2.10, 95% CI [1.05, 3.15]), CQC + CM (MD = 2.27, 95% CI [0.47, 4.07]), TWG + CM (MD = 1.27, 95% CI [0.23, 2.31]), and PC + CM (MD = 1.13, 95% CI [0.09, 2.16]) were more effective than CM in improving the CD4+/CD8+ ratio, with statistically significant differences (P < 0.05), while the other comparisons showed no statistically significant differences, as shown in Figure 8.

Network Meta-analysis Comprising the Effects of CD4+/CD8+.
In studies reporting CD4+/CD8+ ratios, the cumulative ranking chart for the seven interventions is shown. The effectiveness of these interventions is ranked according to SUCRA values, with higher SUCRA values associated with improvement in CD4+/CD8+ ratios. The probability ranking is as follows: CQC + CM (75.2%) > CGZ + CM (74.7%) > TGP + CM (72.9%) > XY + CM (49.1%) > TWG + CM (41.2%) > PC + CM (35.3%) > CM (1.6%), as shown in Table 2.
Adverse Reaction Rate
Evidence Network
Forty-one RCTs29,31–50,52–61,63–71,73–78 reported the incidence of adverse reactions, involving ten interventions with a total sample size of 3955 participants. A total of 424 patients experienced adverse reactions, primarily including skin dryness, pruritus, dizziness, headache, desquamation, and abdominal discomfort, as shown in Table 3. The evidence network was centered on conventional Western medicine, and no closed loops were formed between interventions, making inconsistency testing unnecessary, as shown in Figure 3.
Occurrence of Adverse Reactions.

Network Meta-Analysis and SUCRA Ranking
Pairwise comparisons of the ten interventions resulted in 45 comparisons. Network meta-analysis showed that, compared with CM, CGZ + CM (OR = 0.45, 95% CI [0.22, 0.92]), XY + CM (OR = 0.64, 95% CI [0.44, 0.94]), and PC + CM (OR = 0.56, 95% CI [0.35, 0.91]) were more effective in reducing adverse reaction rates, with statistically significant differences (P < 0.05). The other traditional Chinese medicines combined with CM showed comparable effects to CM in reducing adverse reactions, as shown in Figure 9.

Network Meta-analysis Comprising the Effects of Adverse Reaction Rate.
In studies reporting adverse reaction rates, the cumulative ranking chart for the ten interventions is shown. The effectiveness of these interventions is ranked according to SUCRA values, with higher SUCRA values associated with a reduction in adverse reaction rates. The probability ranking is as follows: CGZ + CM (83.8%) > PC + CM (72.4%) > KYP + CM (64.2%) > XY + CM (62.1%) > ZDP + CM (58.9%) > CGZ + TWG (48.1%) > TGW + CM (42.8%) > CQC + CM (24.4%) > TGP + CM (22.8%) > CM (20.6%), as shown in Table 2.
Publication Bias
In this study, publication bias was analyzed for each outcome indicator, and a “comparison-correction” funnel plot was generated to assess bias. The dots in the funnel plot represent pairwise comparisons between two interventions. The color of the dots represents different interventions, and the number of dots of each color represents the number of RCTs reporting the comparison. If there is no influence from small sample sizes or publication bias, the dots in the funnel plot will be symmetrical and evenly distributed on both sides of the vertical line (X = 0). The results showed that the studies included for the total effective rate, PASI score, IL-17, CD4+/CD8+, and adverse reaction rate outcome indicators were generally symmetrically distributed in the upper-middle part of the inverted funnel plot, around both sides of the central line, suggesting a low likelihood of publication bias. The funnel plot for the TNF-α outcome indicator showed poor symmetry, with some dots scattered outside the 95% CI region, suggesting that the included literature might have publication bias or small sample effects, as shown in Figure 10.

Funnel Plots of Each Outcome Indicator.
Discussion
Psoriasis is a chronic, recurrent, inflammatory, and systemic immune-mediated disease triggered by the interaction between genetic, environmental, and immune factors. Psoriasis is often associated with various comorbidities, including cardiovascular diseases, diabetes, hypertension, and metabolic syndrome, which significantly impair the quality of life of patients. 79 Genetic, immune, and environmental factors play critical roles in the development of psoriasis. Plaque psoriasis accounts for 80% to 90% of all psoriasis cases and is the most prevalent form. It is characterized by well-defined red plaques ranging from 1 to 5 cm in diameter, with variation in number. The plaques may be few and scattered, or multiple plaques may merge into larger ones, potentially covering the entire body. 80 Currently, immunosuppressants and biologics targeting the immune system can effectively control the onset and persistence of psoriasis, although the long-term efficacy of these treatments remains unsatisfactory. 81 TCM, based on syndrome differentiation and treatment, has demonstrated good clinical efficacy and long remission periods. The combination of TCM and Western medicine offers significant advantages and considerable potential for the treatment of this disease. Existing systematic reviews suggest that understanding the optimal timing and unique strengths of both TCM and Western medicine can significantly improve overall efficacy.However, with the increasing use of traditional Chinese patent medicines, most studies focus solely on the effects of specific patent medicines, lacking comprehensive comparisons. Selecting the most effective and safest medicines for treating plaque psoriasis from the wide variety of products has been an ongoing concern for clinicians. Therefore, this study conducted a network meta-analysis to evaluate the efficacy and safety of traditional Chinese patent medicines used in the clinical treatment of plaque psoriasis, based on the total effective rate, PASI score, TNF-α, IL-17, CD4+/CD8+ ratios, and adverse reaction rates, providing evidence-based recommendations for treatment regimens for plaque psoriasis.
In this study, a network meta-analysis was conducted, incorporating 50 RCTs with diverse baseline characteristics. A total of eight traditional Chinese patent medicines combined with conventional Western medicine for the treatment of plaque psoriasis were included, each demonstrating unique clinical benefits. The results indicate that, in terms of improving the total effective rate, CGZ combined with CM is most likely to be the optimal intervention, followed by XY and KYP. In improving PASI scores, CGZ combined with TWG is most likely to be the best intervention, followed by TGP and PC combined with CM. In reducing TNF-α levels, PC combined with CM is most likely to be the best intervention, followed by KYP and TGP. In reducing IL-17 levels, KYP combined with CM is most likely to be the best intervention, followed by PC and ZDP. In improving CD4+/CD8+ ratios, CQC combined with CM is most likely to be the best intervention, followed by CGZ and TGP. In reducing adverse reaction rates, CGZ combined with CM is most likely to be the best intervention, followed by PC and KYP.Regarding adverse reactions, overall, the occurrence of adverse reactions was low, and the safety of the treatments was high. None of the studies reported severe adverse reactions. The majority of reported adverse reactions were mild, such as skin dryness, pruritus, dizziness, headache, and desquamation, which can be alleviated by discontinuing the medication, resting, or providing basic symptomatic treatment. This study reports the adverse reactions of traditional Chinese patent medicines for treating plaque psoriasis, which is significant in guiding clinical practice. It helps to comprehensively assess their safety and provides more reliable decision-making support for clinical practice.
To link the efficacy of traditional Chinese patent medicines revealed in this study for treating plaque psoriasis with the development of new drugs, further exploration of their mechanisms and core medicinal ingredients is needed.Traditional Chinese patent medicines are usually administered in the form of compound medications.The chemical components of these medicines not only have multi-target, multi-pathway action characteristics that produce better synergistic effects, but they also reduce the side effects of single drugs, decrease recurrence and adverse reactions, and affect the complex underlying mechanisms of various diseases.82,83 The main formulation principle of traditional Chinese patent medicines that makes this possible can be described as “Jun, Chen, Zuo, and Shi”. 84 In this method, the medicinal herbs responsible for the primary effects are placed in the positions of “Jun” and “Chen” with higher dosages, while the herbs used to reduce side effects or enhance synergistic effects are placed in the positions of “Zuo” and “Shi,” with relatively smaller doses.This approach predicts that properly combined herbal formulas will be more effective than single herbs. 85 In outcome measures like PASI Score and inflammation, CGZ, TWG, and KYP ranked as the top three most effective treatments. Further comparative and analytical data mining of herbal prescriptions revealed five specific herbal ingredients frequently used in the treatment of psoriasis.For example, baicalin, which was frequently used in this study, is one of the main flavonoid compounds purified from the root of Scutellaria baicalensis. It has anti-inflammatory and immune-regulatory effects, which can reduce inflammation levels in psoriasis and improve skin lesions. 86 Wu 87 demonstrated that baicalin could reduce the upregulation of TNF-α, IFN-γ, IL-22, and IL-17 expressions induced by TNF-α in HaCaT cells, exerting an anti-inflammatory effect. It also decreased the expression of apoptosis-related proteins Bcl-2 and Caspase-3, inducing apoptosis in HaCaT cells, lowering cell viability, and inhibiting cell proliferation. This effect might be mediated by the suppression of STAT3/NF-κB-mediated inflammation and keratinocyte proliferation. 87 Ziziphirol, a cycloartane glycoside isolated from the root of Rehmannia glutinosa, has significant anti-inflammatory and antioxidant effects. 88 Research indicates that the beneficial effects of ziziphirol in psoriasis may depend on its inhibitory activity on the SIRT1-mediated MAPK/NF-κB signaling pathway. Ziziphirol treatment significantly suppresses oxidative stress and inflammation both in vivo and in vitro, as evidenced by reduced oxidative stress markers (MDA, GSH, SOD, CAT) and pro-inflammatory cytokines (IL-6, TNF-α, IL-1β, IL-22, IL-17), thereby improving the pathological phenotype of psoriasis. 89 To link the efficacy of traditional Chinese patent medicines revealed in this study for treating plaque psoriasis with the development of new drugs, further exploration of their mechanisms and key medicinal ingredients is needed. Traditional Chinese patent medicines are typically administered as compound medications. The chemical components of these medicines exhibit multi-target, multi-pathway actions that produce enhanced synergistic effects, while also reducing the side effects of single drugs, decreasing recurrence and adverse reactions, and impacting the complex underlying mechanisms of various diseases.82,83 The main formulation principle of traditional Chinese patent medicines that facilitates this can be described as “Jun, Chen, Zuo, and Shi”. 84 In this method, the medicinal herbs responsible for the primary effects are placed in the “Jun” and “Chen” positions with higher dosages, while the herbs used to reduce side effects or enhance synergistic effects are placed in the “Zuo” and “Shi” positions, with relatively smaller doses. This approach suggests that properly combined herbal formulas are more effective than single herbs. 85 In outcome measures such as PASI score and inflammation, CGZ, TWG, and KYP ranked as the top three most effective treatments. Further comparative and analytical data mining of herbal prescriptions identified five specific herbal ingredients frequently used in the treatment of psoriasis. For example, baicalin, which was frequently used in this study, is a major flavonoid compound purified from the root of Scutellaria baicalensis. It has anti-inflammatory and immune-regulatory effects, which can reduce inflammation levels in psoriasis and improve skin lesions. 86 Wu 87 demonstrated that baicalin could reduce the upregulation of TNF-α, IFN-γ, IL-22, and IL-17 expressions induced by TNF-α in HaCaT cells, exerting an anti-inflammatory effect. It also decreased the expression of apoptosis-related proteins Bcl-2 and Caspase-3, inducing apoptosis in HaCaT cells, lowering cell viability, and inhibiting cell proliferation. This effect may be mediated by the suppression of STAT3/NF-κB-mediated inflammation and keratinocyte proliferation. 87 Ziziphirol, a cycloartane glycoside isolated from the root of Rehmannia glutinosa, has significant anti-inflammatory and antioxidant effects. 88 Research indicates that the beneficial effects of ziziphirol in psoriasis may depend on its inhibitory activity on the SIRT1-mediated MAPK/NF-κB signaling pathway. Ziziphirol treatment significantly suppresses oxidative stress and inflammation both in vivo and in vitro, as evidenced by reduced oxidative stress markers (MDA, GSH, SOD, CAT) and pro-inflammatory cytokines (IL-6, TNF-α, IL-1β, IL-22, IL-17), thereby improving the pathological phenotype of psoriasis. 89 Ligustrazine, an amide alkaloid monomer extracted from Ligusticum chuanxiong, can reduce inflammatory damage in psoriasis. 90 Jiang confirmed that inhibiting the activation of the TRAF6/c-JUN/NF-κB signaling pathway may be a key mechanism by which ligustrazine reduces psoriasis-related inflammation, leading to the downregulation of downstream genes associated with psoriasis, such as S100A7/A8 and CCL2/20. 91 Another in vitro experiment also demonstrated that ligustrazine regulates the secretion of IL-6, IL-17, IL-4, eosinophil chemotactic factors, and factors involved in activating normal T cell expression by inhibiting the NF-κB signaling pathway in HaCaT cells. Both studies indicate that ligustrazine provides effective protective activity against psoriasis-related skin inflammation. 92 Additionally, licorice, the dried root and rhizome of the leguminous plant Glycyrrhiza, and its extracts such as glycyrrhizin and glycyrrhizic acid have significant anti-inflammatory and immune-regulatory pharmacological effects.93,94 Research has shown that glycyrrhizin can increase serum IL-4 and IL-17 levels in psoriasis rats, inhibiting mitosis in estrogen-phase vaginal epithelial cells, thus exerting anti-psoriasis effects. 95 Glycyrrhizic acid can reduce the expression of IL-17, IL-6, and IFN-γ in psoriasis lesions of mice, improving the area of lesions, erythema, and the size and number of scales, thereby achieving the goal of treating psoriasis. 96 Meanwhile, another key herbal ingredient, Tripterygium wilfordii, is derived from the whole root or peeled root bark of the deciduous shrub of the Celastraceae family. Studies have shown that Tripterygium wilfordii has anti-inflammatory and immune-suppressive pharmacological effects, 97 and its main component, triptolide, can reduce serum IL-17 levels, improve lesion inflammation, and effectively exert anti-psoriasis effects. 98 To explore the optimal effective dose of triptolide for treating psoriasis, a study found that administering 10, 20, and 40 mg of triptolide to psoriasis mice showed that 20 mg had the best antioxidant effect. Both 20 and 40 mg significantly reduced serum IL-6, IL-17, IL-23, and TNF-α levels, among other inflammatory factors. 99 These preliminary studies suggest that single herbal extracts can inhibit the occurrence and development of psoriasis through multiple pathways and targets, such as anti-inflammatory and immune-regulatory effects, thereby affecting biological processes such as inflammation, angiogenesis, oxidative stress, cell proliferation, and apoptosis.In contrast to the pharmacological predictions of single drugs, considering the interactions between multiple herbs may lead to enhanced therapeutic effects. This finding supports the hypothesis that in the traditional Chinese patent medicine formulas used in this study, the core medicinal ingredients not only act on the mechanisms of individual herbs but also follow the “Jun, Chen, Zuo, and Shi” formulation principle. Therefore, tracking the synergistic effects of core herbal combinations and identifying the best herbal combinations that can maximize these synergistic interactions may serve as a target for future drug candidate research.This study used network meta-analysis to compare the efficacy and safety of eight traditional Chinese patent medicines combined with CM in the treatment of plaque psoriasis, providing a rational, targeted, and personalized drug selection plan for clinical treatment of psoriasis patients. However, it should be noted that this study has several limitations. Firstly, due to the strict inclusion and exclusion criteria for selecting literature, the number of studies corresponding to some outcome indicators for traditional Chinese patent medicines is relatively small, and the number of studies included for different traditional Chinese patent medicines within the same outcome indicator varies greatly, which may influence the results.Secondly, some studies had poor quality, with some failing to describe blinding and allocation concealment, which could lead to small sample effects and publication bias, thus impacting the efficacy of the findings. Thirdly, the treatment duration for traditional Chinese patent medicines is not standardized, and most are used in a generalized manner without considering the evaluation of relevant TCM syndrome elements, which may interfere with the overall assessment of the patient's efficacy. It is recommended to standardize the treatment duration in future studies.Fourthly, due to the unclear categorization of all included RCTs, this review did not conduct subgroup analysis on major risk factors such as age, gender, and etiology, which significantly affect psoriasis. Finally, no closed loops were formed between different interventions, and there was a lack of direct comparisons between traditional Chinese patent medicines, which may affect the accuracy of the results. Caution should be exercised when interpreting the results.
Conclusion
This systematic review supports the use of traditional Chinese patent medicines combined with conventional medicine as an effective and safe treatment for psoriasis. The meta-analysis shows that traditional Chinese patent medicines have significant advantages over the control group in various efficacy indicators, including total effective rate, PASI score, TNF-α, IL-17, and CD4+/CD8+ ratios in psoriasis patients. Additionally, traditional Chinese patent medicines significantly reduced the adverse reaction rate compared to the CM control group. In other words, traditional Chinese patent medicines are considered to actively improve skin symptoms, inflammation status, quality of life, and medication adherence in psoriasis patients.Further analysis of the outcome indicators revealed the top three traditional Chinese patent medicine prescriptions, which identified five high-frequency core herbs: Scutellaria baicalensis, Rehmannia glutinosa, Ligusticum chuanxiong, Glycyrrhiza, and Tripterygium wilfordii. These herbs are generally considered to inhibit the development of psoriasis through multiple pathways and targets, such as anti-inflammatory and immune-regulatory effects. Therefore, in future drug development related to this subject, combining these five herbal categories into traditional Chinese patent medicines to maximize synergistic immune-modulating effects may serve as a promising research hypothesis.
Although this study has some limitations, it still provides a reasonable overview and suggestions for clinical application in plaque psoriasis. Therefore, it is recommended that future studies conduct more large-sample, multi-center, double-blind, high-quality RCTs with long-term follow-up to verify the results and provide more accurate evidence-based support.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251352975 - Supplemental material for Comparison of the Efficacy and Safety of Eight Traditional Chinese Patent Medicines in the Treatment of Plaque Psoriasis: A Systematic Review and Network Meta-Analysis
Supplemental material, sj-docx-1-npx-10.1177_1934578X251352975 for Comparison of the Efficacy and Safety of Eight Traditional Chinese Patent Medicines in the Treatment of Plaque Psoriasis: A Systematic Review and Network Meta-Analysis by Qiunan Teng, Hong Yang, Chenghao Zheng, Dongyu Kuang and Ying Sun in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X251352975 - Supplemental material for Comparison of the Efficacy and Safety of Eight Traditional Chinese Patent Medicines in the Treatment of Plaque Psoriasis: A Systematic Review and Network Meta-Analysis
Supplemental material, sj-docx-2-npx-10.1177_1934578X251352975 for Comparison of the Efficacy and Safety of Eight Traditional Chinese Patent Medicines in the Treatment of Plaque Psoriasis: A Systematic Review and Network Meta-Analysis by Qiunan Teng, Hong Yang, Chenghao Zheng, Dongyu Kuang and Ying Sun in Natural Product Communications
Supplemental Material
sj-docx-3-npx-10.1177_1934578X251352975 - Supplemental material for Comparison of the Efficacy and Safety of Eight Traditional Chinese Patent Medicines in the Treatment of Plaque Psoriasis: A Systematic Review and Network Meta-Analysis
Supplemental material, sj-docx-3-npx-10.1177_1934578X251352975 for Comparison of the Efficacy and Safety of Eight Traditional Chinese Patent Medicines in the Treatment of Plaque Psoriasis: A Systematic Review and Network Meta-Analysis by Qiunan Teng, Hong Yang, Chenghao Zheng, Dongyu Kuang and Ying Sun in Natural Product Communications
Footnotes
Acknowledgements
Everyone who contributed significantly to this study has been listed.
Author Contributions
We confirm that all listed authors meet the authorship criteria, and all authors are in agreement with the content of the manuscript. QiunanTeng and Ying Sun had the original idea for the study and proposed the study design. Hong Yang and Chenghao Zheng conducted the literature search, screened and selected the studies initially identified. Chenghao Zheng and Dongyu Kuang read and evaluated the quality of the studies included. QiunanTeng and Ying Sun conducted the meta-analysis. QiunanTeng wrote the initial manuscript and serves as guarantor. Ying Sun revised the manuscript. All authors contributed to interpreting the study findings and to the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
All data are fully available without restriction.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
