Abstract
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is a malignant tumor originating from the epithelium of nasopharyngeal mucosa. 1 It is characterized by hidden onset, deep lesions, and atypical early symptoms. Therefore, once diagnosed, the condition has been in the middle or advanced stage, and among some patients, tumor metastasis has appeared. Radiotherapy (RT), chemotherapy, and surgery are commonly used for the treatment of NPC. Since this is mostly squamous cell carcinoma, RT is considered as the first choice. 2 However, the RT may damage important organs and tissues around the nasopharynx and neck. This treatment often leads to adverse reactions, such as salivary gland injury, dry mouth, radioactive oropharyngeal mucositis, radioactive dermatitis, and hemotoxic reactions, which reduce the life quality of the patients. 3 and even interrupts the treatment. In some patients whose lesions are under control, local recurrence or distant metastasis may appear later. 4
At present, traditional Chinese medicine (TCM) has been extensively applied against cancers at all stages. It may improve the curative effect of chemoradiotherapy and reduce the occurrence of toxic side effects which has been proved clinically. 5 –7 Aidi injection (Z52020236, China Food and Drug Administration) is an important TCM injection, which is widely used in the adjuvant therapy of malignant tumors in China. 8 (Table 1). As an anticancer TCM drug, Aidi injection is often used with chemoradiotherapy as an adjuvant therapy for lung cancer, liver cancer, stomach cancer, and so on.9–11 Aidi injection can reduce adverse reactions caused by tumor chemoradiotherapy, for example, gastrointestinal reactions and hepatorenal toxicity.12,13 It is safe, and may significantly improve patients’ life quality after chemoradiotherapy.12,14 Aidi injection is also being broadly used in nasopharyngeal cancer.
Pharmacological Mechanisms of Chinese Herbs in Aidi Injection.
A randomized controlled trial (RCT) 23 reported that Aidi injection combined with RT could improve the life quality score and reduce toxic and side effects. There are more and more similar reports but it is difficult to draw a reliable conclusion from them for lack of systematic evaluation on the efficacy. This paper conducted a meta-analysis on RCTs of Aidi injection combined with RT against NPC so that relevant clinical work and scientific research may have reliable treatment protocols and an evidence-based basis.
Methods
Study Registration
The paper was written under the guidance of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). 24 See statement (Supplementary Table S1). The study protocol was registered at https://osf.io/j4uyh/ (DOI 10.17605/OSF.IO/J4UYH).
Eligibility Criteria
Type of Studies
We included all the RCTs only and excluded the reviews and other studies such as cohort, case–control, case series, and qualitative studies. The published abstracts were also excluded when unable to get the details.
Types of Participants
The diagnostic criteria refer to the diagnosis of NPC in Radiotherapy of Oncology. The patient was clearly diagnosed with NPC, whose diagnostic criteria were referred to Radiotherapy of Oncology. 25 The international tumor-node-metastasis (TNM) system was used for tumor grading. 26
Interventions
Treatment group: Aidi injection combined with RT; control group: only the same RT as the treatment group.
Types of Outcome Measures
Primary Outcomes
Primary outcomes are as follows: (1) Karnofsky Performance Status Scale (KPS) and (2) total effective rate (TER). For evaluation criteria, refer to the WHO efficacy evaluation criteria 27 : (i) Complete response (CR): Disappearance of all targeted lesions in two observations no less than 4 weeks interval; (ii) Partial response (PR): a >50% decrease in sum of products of the greatest diameters (SPD) of all lesions; without new lesions nor progression of any lesions; (iii) Stable disease (NC): a 50% decrease or 25% increase in SPD of one or more lesions; and (iv) Progressive disease (PD): a >25% increase over the smallest measurement in the single lesion, or the appearance of new lesions. TER = (CR + PR)/total number *100%.
Secondary Outcome
Secondary outcome indicators are as follows: Toxic and side effects (refer to Radiation Therapy Oncology Group [RTOG]): Criteria for the classification of acute radiation injury) including leukopenia, hemoglobin reduction, thrombocytopenia, oropharyngeal mucosal injury (OMI), nausea, vomiting, and skin injury.
Exclusion Criteria
Repetitively published researches;
Studies of abstracts only or conference papers without available data;
Studies with noticeable errors or incomplete and unavailable data;
Studies with high bias risk 28 ;
Studies with other TCM therapies in treatment group such as acupuncture or other formula;
Studies without outcomes.
Data Sources and Search Strategy
The PubMed, Cochrane Library, CNKI, EMBASE, Web of Science, CBM, Wanfang, and VIP data were thoroughly searched from the database establishment to February 2021 with MeSH terms and free terms. Also, we searched for the potentially unpublished literature both in Google Scholar and Baidu. The words for search in English were Aidi, NPC, carcinoma, and nasopharyngeal, which in Chinese were Ai Di Zhu She Ye (Aidi injection), Bi Yan Ai (NPC), Bi Yan Lin Ai (nasopharyngeal squamous cell carcinoma). Supplementary Table S2 shows the search strategy in PubMed.
Study Selection and Data Extraction
The literature was independently screened by Renhong Wan and Anlan Zhao with the following procedures: (1) Remove the duplicated papers in EndNote X9. (2) Review titles and abstracts for preliminary selection. (3) Screen the full text in accordance with the criteria of inclusion and exclusion. Another investigator, Min Zhong, would resolve the discrepancies. The missing data or information was obtained by contacting the corresponding authors when necessary. All the following information was extracted into Microsoft Excel 2013: (1) General information of the first author, publication year, samples in each group, baseline information, tumor stage, and functional status score; (2) Interventions and course; (3) Outcome measures; and (4) Risk or bias assessment factors in RCTs.
Bias Risk Assessment
Bias risk assessment and results cross-check were performed by Renhong Wan and Anlan Zhao independently. Another investigator, Min Zhong, would resolve the discrepancies. The recommended tool for bias risk assessment in Cochrane Mnual 5.1.0 was adopted. 29
Statistical Analysis and Data Synthesis
The RevMan5.3 of Cochrane Collaboration was applied for meta-analysis. The weighted mean difference (WMD) was used for continuous variables if the measuring instruments and the units were consistent, otherwise standard mean difference (SMD) was used. Relative risk (RR) was used to analyze the dichotomous variables, and the 95% CI was provided for each effect size. Heterogeneity between the results was analyzed by the chi-square test, and quantitatively determined in combination with I2. P≥0.10 and I2<50% indicated no significant heterogeneity between studies, and a fixed effect model (FEM) was used for the meta-analysis. P < .10 and I2≥50% indicated significance between studies. In this case, the sources of heterogeneity need be discussed by both subgroup analysis and sensitivity analysis. Only if significant clinical and methodological heterogeneity are excluded, a random effect model may be used for the meta-analysis. The sensitivity analysis was used for the influence of a single study on the combined effect size, as well as the stability of the meta-analysis results. Funnel plot would be used for publication bias for major outcome measures if >10 researches were included, and potential publication bias would be evaluated by Egger's and Begg's tests.
Results
Description of the Included Studies
Preliminary screening obtained 83 relevant articles, and 38 remained after duplicate ones were removed. Through reading the titles and abstracts, 24 articles remained, and then, 15 of the 24 articles were excluded through full-text reading. At last, 9 articles were included.23,30–37 The screening procedure is shown in Figure 1. Table 2 shows the basic features of the included studies.

Flowchart.
Basic Features of the Included Studies.
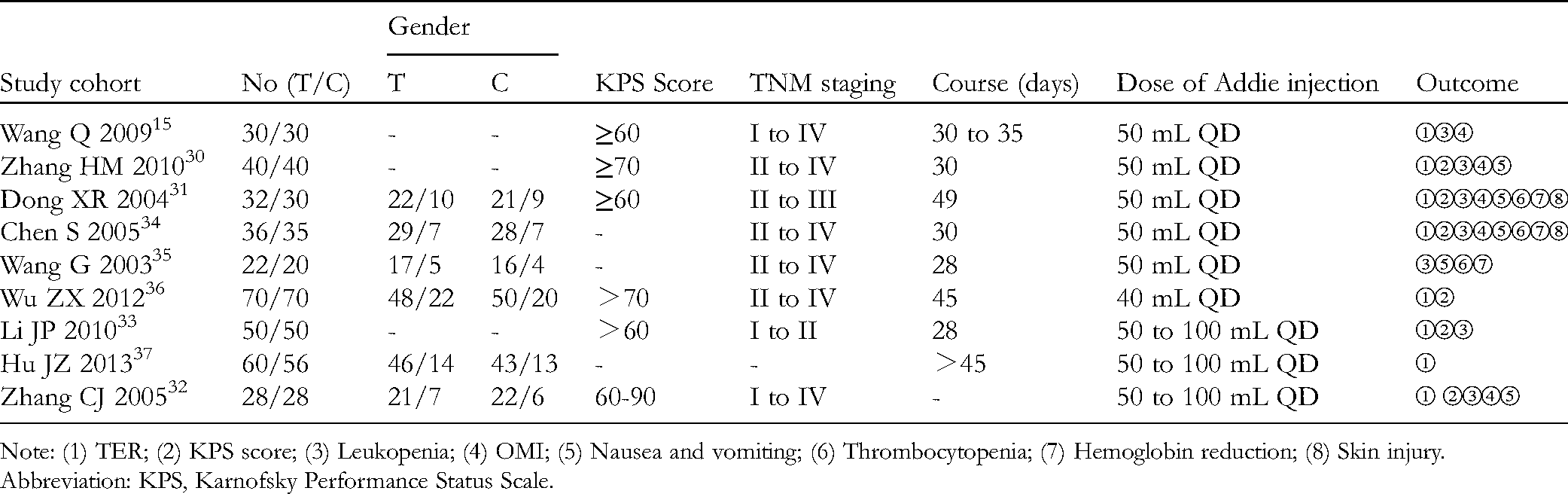
Note: (1) TER; (2) KPS score; (3) Leukopenia; (4) OMI; (5) Nausea and vomiting; (6) Thrombocytopenia; (7) Hemoglobin reduction; (8) Skin injury.
Abbreviation: KPS, Karnofsky Performance Status Scale.
Bias Risk Assessment
The risk assessment tool by Cochrane Manual 5.1.0 was used to evaluate the quality of 9 included articles.23,30–37 One article 36 used the method of random sequence generation correctly. None of the studies mentioned blinding. Combined with the intervention method, all the studies were evaluated as high risk; all studies were evaluated as low risk in the distribution of hidden, incomplete outcome indicators, selective report, etc. The results of bias risk assessment are shown in Figure 2.

Summary of bias risk.
Outcome Measures
Primary Outcome Indicators
TER
Eight RCTs were included,23,30–34,36,37 including 680 patients. Heterogeneity test: P = .13, I2 = 38%, without statistical heterogeneity. FEM results showed that for the TER, there existed no statistical difference between the 2 groups [RR = 1.03, 95% CI (0.98, 1.08), P = .20 > .05], (Figure 3). A subgroup analysis was performed according to the dosage of Aidi injection. The results showed that there was no heterogeneity for the three subgroups, and no statistically significant between-group difference (P > .05) (Figure 4). In the Meta-analysis for separate re-evaluation on the complete remission rate (CRR, the No. of complete remission cases/total No. of cases *100%), heterogeneity test: P = .68, I2 = 0%, without statistical heterogeneity. FEM meta-analysis results showed that the CRR of the 2 groups was [RR = 1.17, 95% CI (1.04, 1.31), P = .009], with significant difference (Figure 5).

Meta-analysis for comparison of total effective rate (TER) between the treatment group and the control group.
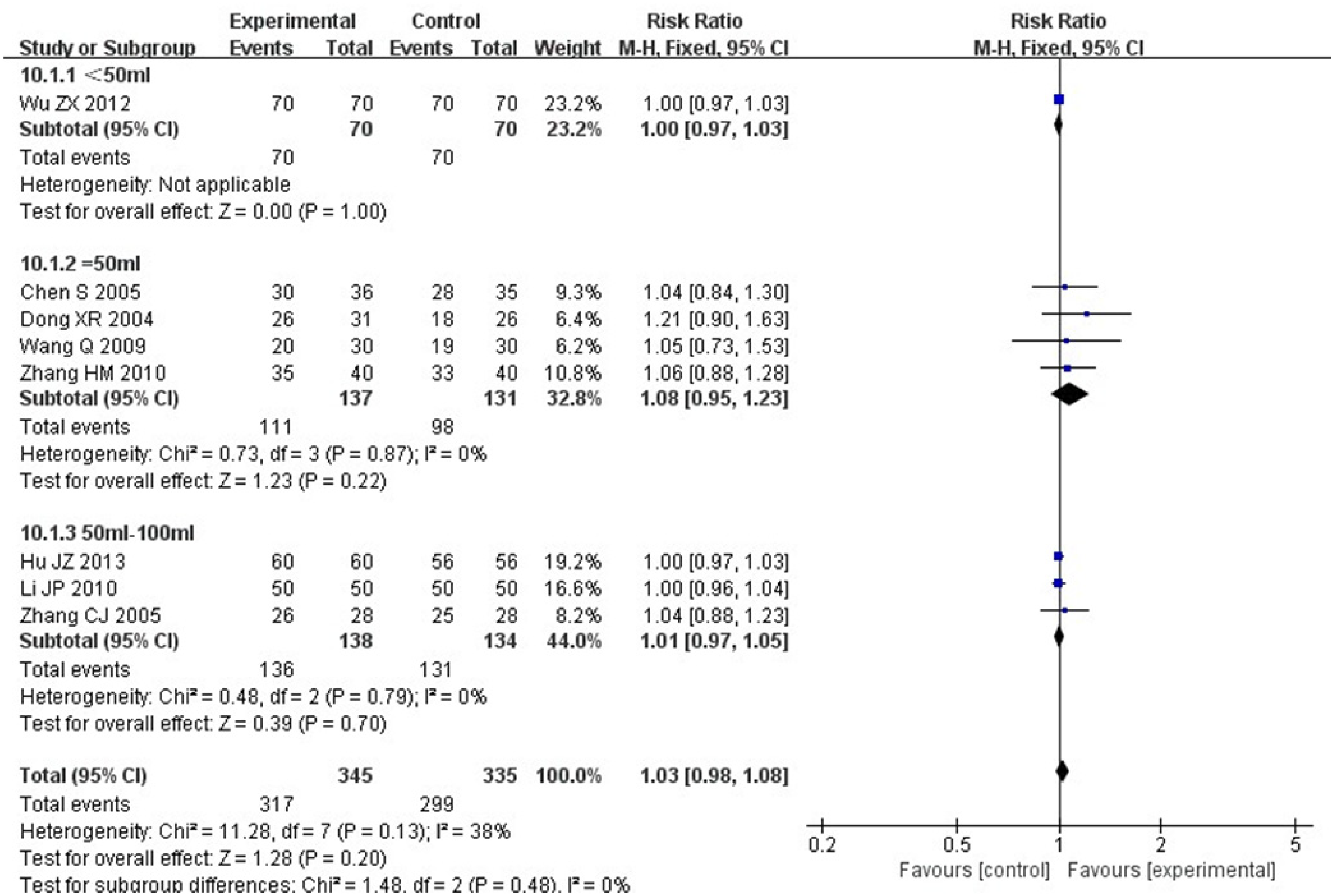
Meta-analysis for total effective rate (TER) subgroups of treatment group and control group.

Meta-analysis for comparison of complete remission rate (CRR) between the treatment group and the control group.
KPS Score
Six RCTs, including 504 patients,30–34,36 reported a KPS score. Heterogeneity test: P = .26, I2 = 23%, without statistical heterogeneity. The FEM results showed that for the KPS score, there was no statistically significant between-group difference [RR = 1.30, 95% CI (1.19, 1.42), P < .00001] (Figure 6). A subgroup analysis was performed according to the dose of Aidi injection. The results showed that all the 3 subgroups were homogeneous. The FEM meta-analysis results were as follows: [RR = 1.29, 95% CI (1.08, 1.55), P = .005]; [RR = 1.17, 95% CI (1.05, 1.30), P = .004]; and [RR = 1.56, 95% CI (1.25, 1.94), P < .00001]. The difference was statistically significant (Figure 7).

Mate analysis for a comparison of Karnofsky Performance Status Scale (KPS) score between groups.
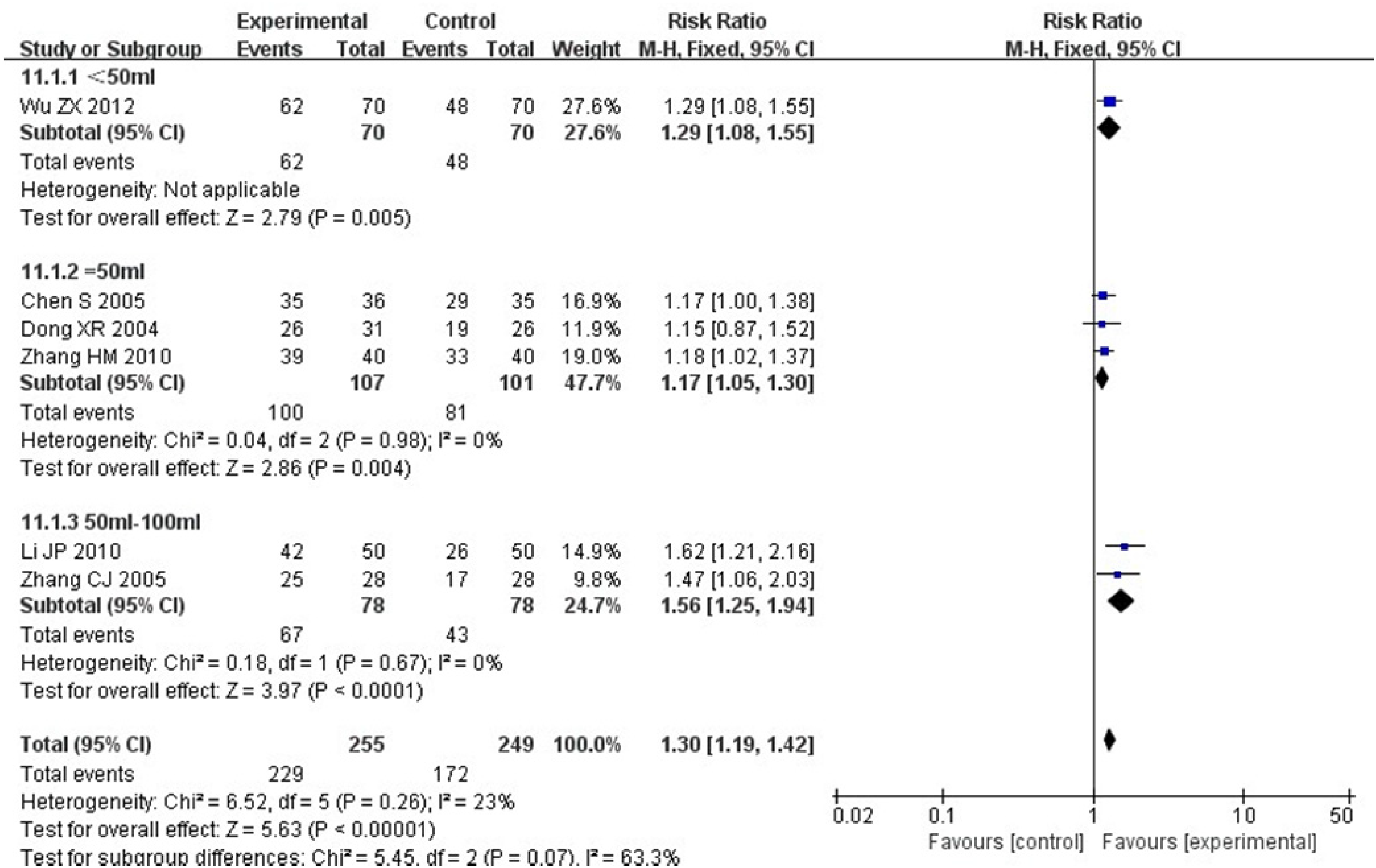
Mate analysis for Karnofsky Performance Status Scale (KPS) score subgroups of the 2 groups.
Secondary Outcome Indicators
Leukopenia
Seven RCTs including 466 patients23,30–35 reported leukopenia. Heterogeneity test: P = .77, I2 = 0%, without statistical heterogeneity. The results of FEM meta-analysis showed that for the leukopenia, there was statistically significant between-group difference [RR = 0.65, 95% CI (0.54, 0.78), P < .00001] (Figure 8).
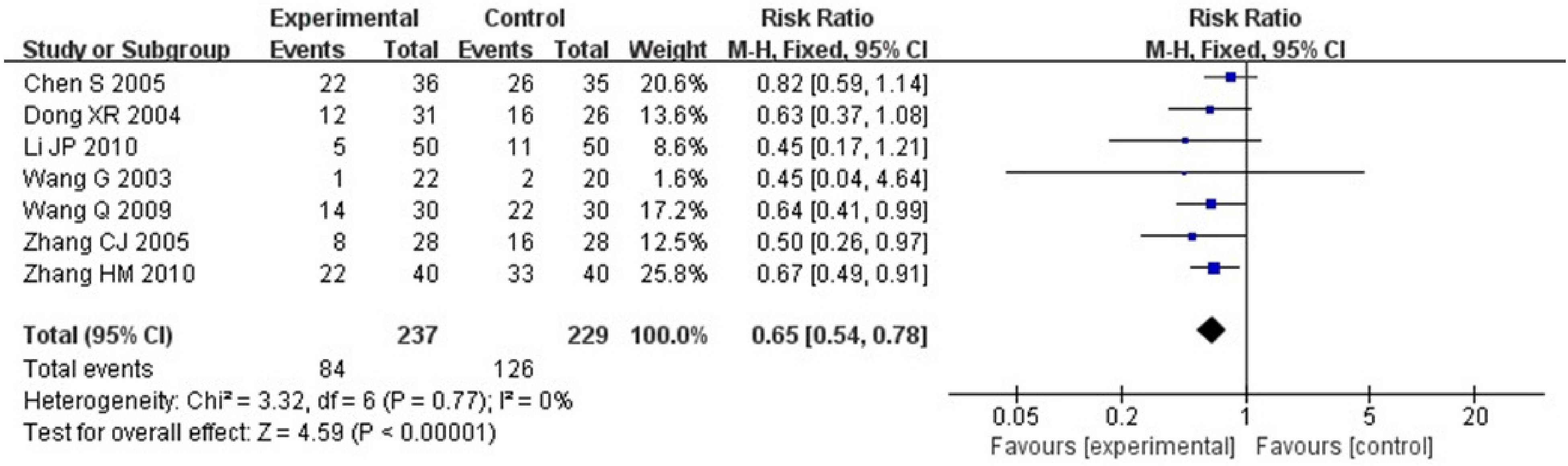
Mate analysis for a comparison of leukopenia between groups.
Thrombocytopenia
Three RCTs including 170 patients30,34,35 reported thrombocytopenia. Heterogeneity test: P = .46, I2 = 0%, without statistical heterogeneity. The results of the FEM meta-analysis showed that for the thrombocytopenia, there was no statistically significant between-group difference [RR = 0.78, 95% CI (0.52, 1.16), P = .22 > .05] (Figure 9).

Mate analysis for a comparison of thrombocytopenia between groups.
Hemoglobin Reduction
Three RCTs including 170 patients30,34,35 reported hemoglobin reduction. Heterogeneity test: P = 0.98, I2 = 0%, without statistical heterogeneity. The FEM results showed that for the hemoglobin reduction, there was statistically significant between-group difference [RR = 0.60, 95% CI (0.43, 0.84), P = .003] (Figure 10).

Mate analysis for a comparison of hemoglobin reduction between groups.
Nausea and Vomiting
Five RCTs including 306 patients30–32,34,35 reported nausea and vomiting. Heterogeneity test: P = .61, I2 = 0%, without statistical heterogeneity. The FEM results showed that for nausea and vomiting, there was statistically significant difference between the 2 groups [RR = 0.60, 95% CI (0.45, 0.80), P = .0004] (Figure 11).

Mate analysis for a comparison of nausea and vomiting between groups.
Oropharyngeal Mucosal Injury
Five RCTs including 324 patients23,30–32,34 reported OMI. Heterogeneity test: P = .76, I2 = 0%, without statistical heterogeneity. The FEM meta-analysis results showed that for OMI, there was statistically significant difference between the 2 groups [RR = 0.25, 95% CI (0.15, 0.44), P < .00001] (Figure 12).

Mate analysis for a comparison of oropharyngeal mucosa injury (OMI) for 2 groups.
Skin Injury
Two RCTs including 123 patients31,34 reported skin injury. Heterogeneity test: P = .97, I2 = 0%, without statistical heterogeneity. The FEM meta-analysis results showed that for the skin injury, there was no statistically significant between-group difference [RR = 0.81, 95% CI (0.64, 1.04), P = .1 > .05] (Figure 13).
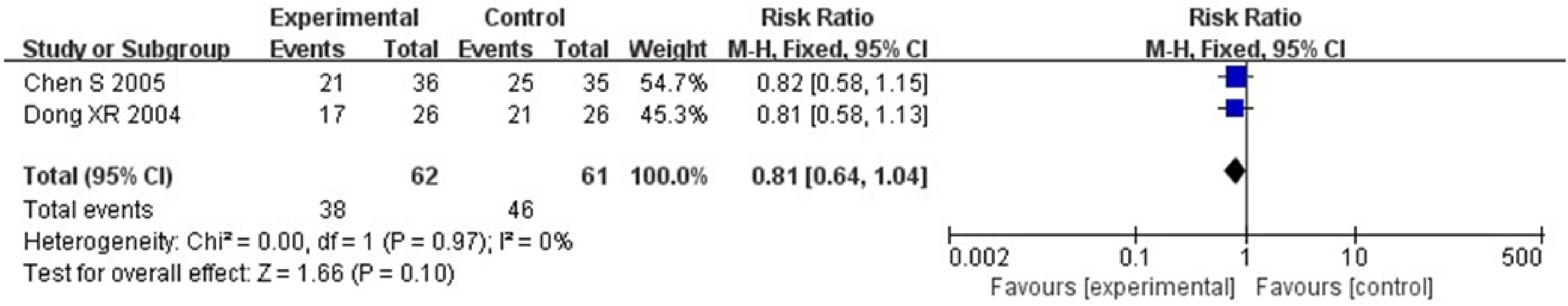
Mate analysis for a comparison of skin lesions between the treatment group and the control group.
Sensitivity Analysis
The sensitivity analysis was carried out by a one-by-one elimination method, and effect size and P value changes were observed after one-by-one elimination of included studies. The effect size of outcome indicators did not change significantly after excluding any article, indicating the results were reliable and stable.
Publication Bias
Considering that total 9 studies were included, we investigated the publication bias through Egger's test and Begg's test, and the results showed that there was no publication bias (P = .1920 > .05, P = .1078 > .05, respectively).
Evaluation of Quality of Evidence
Evidence quality was assessed by the Grading of Recommendations Assessment, Development, and Evaluation (GRADE). After being assessed by the GRADE evidence grading system, the KPS score, the degree of leukocyte, and hemoglobin reduction, the incidence of OMI, and the incidence of nausea and vomiting were all low, and the above outcome indicators all met the 2 downgrading criteria: (1) Risk of bias: Only one study accurately used the random method and none of the studies specifically described the allocation concealment and blinding. As it was an objective indicator, it was downgraded one level. (2) Imprecision: Downgrading was due to the small No. of included studies and wide confidence intervals (Table 3).
Grading of Recommendations Assessment, Development, and Evaluation (GRADE) Evidence Profiles of the Included Studies.

The basis for the assumed risk (eg, the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).
Only one study accurately used the random method, and none of the studies specifically described the allocation concealment and blind method. As it is an objective indicator, it is downgraded one level.
Downgrading a notch was conducted because the number of included studies is small, and the confidence interval is wide.
Abbreviations: CI, confidence interval; KPS, Karnofsky Performance Status Scale; RR, risk ratio.
Discussion
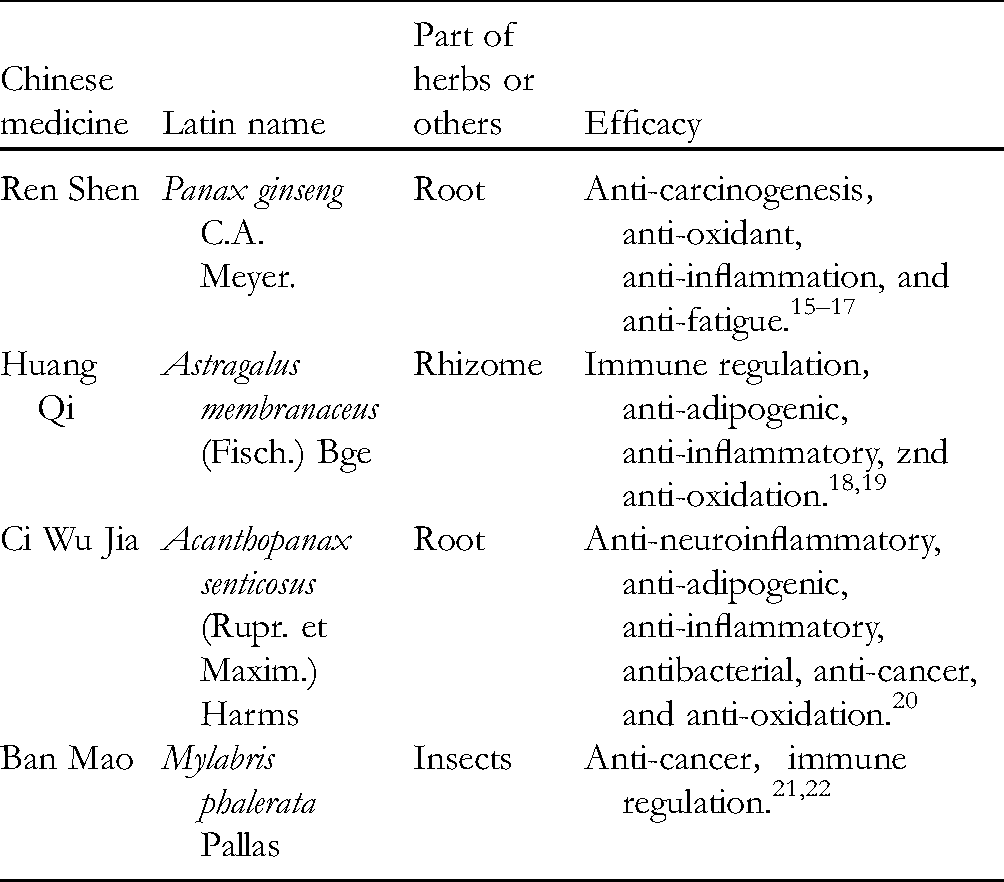
The etiology of NPC is still unclear, but it is generally believed to be highly correlated with Epstein Barr (EB) virus, 38 environmental factors, dietary habits, and genetic susceptibility.1,39 Traditional medicine assigned it to the categories of “sinusitis (Bi Yuan)", “ upper stony flat abscess (Shang Shi Ju)", and “glanders (Bi Ju)". 40 According to the classification of TCM, the syndrome types of NPC can be divided into lung heat, blood stagnation, phlegm cagulation, and blood stagnation-phlegm coagulation. 41 The TCM pathogenesis is deficiency in origin and excess in superficiality. Aidi injection is composed of cantharis (Mylabris phalerata Pallas), Radix Astragali (Astragalus membranaceus (Fisch.) Bge), ginseng (Panax ginseng C. A. Meyer), and acanthopanax root (Acanthopanax senticosus [Rupr. et Maxim.] Harms). 42 Ginseng (Ren Shen) can greatly invigorate primordial qi, promote production of fluid, and benefit lung; Radix Astragali (Huang Qi) invigorates qi for consolidating superficies and promotes pus discharge and tissue regeneration; Acanthopanax Root (Ci Wu Jia) can replenish qi and invigorate the spleen; Cantharis (Ban Mao) can remove blood stasis. All medicinal materials above are combined to treat both manifestation and root cause of the disease. 43
Modern pharmacological studies have shown that Aidi injection has antitumor and immunomodulatory effects. 44 The roles of active ingredients in it are as follows: ginsenoside can enhance the function of T and B cells, induce interferon and interleukin, and increase the activity of LAK and NK cells 45 ; astragalus polysaccharide can enhance reticuloendothelial phagocytosis, as well as the anticancer activity of T cells, NK cells, LAK cells, and IL-II. It can also enhance the ability to kill tumor cells 46 ; acanthopanax root (Ci Wu Jia) extract contains a variety of glycosides and acanthopanax polysaccharide, which can improve immunity, increase white blood cells, improve the adaptability of the body to stress, intoxication, and other nonspecific damages 47 ; norcantharidin can increase active oxygen level, mediate SKOV3 cell apoptosis, as well as G2/M phase periodic arrest. 48 An animal test has confirmed that Aidi injection can inhibit tumor metastasis by reducing the expression of Vimentin and VEGF-A in tumor tissues and increasing the expression of cadherin-1. 44
For this paper, a meta-analysis was conducted on 9 RCTs of Aidi injection combined with RT for NPC (422 patients). The results showed that compared with RT alone, Aidi injection combined with RT was not different in efficacy for local lesions of NPC (based on the TER). Subgroup analysis on the dose of the TCM injection also showed no difference, indicating that Aidi injection may not reduce the tumor volume. In addition, we evaluated the CRR of patients separately, and it was demonstrated that the experimental group was superior to the control group in the CRR, which indicated that Aidi injection had certain advantages in patients with obvious effects. Considering that the No. of studies is limited, the results are to be further verified by more relevant studies.
KPS score is an indicator reflecting patients’ physical functioning status. In this meta-analysis, the significant difference in KPS score between the 2 groups indicates that Aidi injection did improve the life quality and daily behavior ability of patients.
In terms of adverse reactions, the Aidi injection combined with RT group has more advantages than the RT group. Aidi injection can reduce the toxic and side effects caused by RT. It can improve leukopenia and hemoglobin reduction, and reduce the incidence of OMI, nausea and vomiting. However, it cannot improve the adverse reactions of thrombocytopenia and skin damage.
All the results showed good homogeneity, and the sensitivity analysis showed good stability. The study showed that compared with the RT alone group, the Aidi injection combined with RT group had more advantages in improving the symptoms of patients with NPC and alleviating adverse reactions.
This study has some limitations. First, limited by the quality and quantity of the included trials, more high-quality studies are needed to confirm the above conclusions. Second, the operation of allocation concealment and blinding was not specifically described, and there might be selection bias, measurement bias, or implementation bias. Third, the included studies were all from China, with regional limitations and poor universality.
Conclusion
Current evidence suggests that Aidi injection combined with RT for NPC treatment can improve patients’ KPS score and reduce some toxic and side effects. However, the difference of TER between 2 groups is still yet to be evaluated, and Egger's test and Begg's Test did not indicate publication bias.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221096174 - Supplemental material for Efficacy of Aidi Injection Combined With Radiotherapy for the Treatment of Nasopharyngeal Carcinoma: A Systematic Review and Meta-Analysis
Supplemental material, sj-docx-1-npx-10.1177_1934578X221096174 for Efficacy of Aidi Injection Combined With Radiotherapy for the Treatment of Nasopharyngeal Carcinoma: A Systematic Review and Meta-Analysis by Anlan Zhao, Renhong Wan, Min Zhong, Xiaojing Guo, Shuyuan Zou, Jiajing Zhou, Zuojing Zeng and Pei Chen in Natural Product Communications
Supplemental Material
sj-doc-2-npx-10.1177_1934578X221096174 - Supplemental material for Efficacy of Aidi Injection Combined With Radiotherapy for the Treatment of Nasopharyngeal Carcinoma: A Systematic Review and Meta-Analysis
Supplemental material, sj-doc-2-npx-10.1177_1934578X221096174 for Efficacy of Aidi Injection Combined With Radiotherapy for the Treatment of Nasopharyngeal Carcinoma: A Systematic Review and Meta-Analysis by Anlan Zhao, Renhong Wan, Min Zhong, Xiaojing Guo, Shuyuan Zou, Jiajing Zhou, Zuojing Zeng and Pei Chen in Natural Product Communications
Footnotes
Author Contributions
Conception and design: Renhong Wan, Anlan Zhao.
Design of data synthesis analysis scheme: Anlan Zhao,Min Zhong, Shuyuan Zou, and Xiaojing Guo.
Manuscript writing: Renhong Wan, Anlan Zhao, and Shuyuan Zou.
Final approval of manuscript: Pei Chen, Zuojing Zeng, and Renhong Wan.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
