Abstract
One of the most prevalent cancers in the world, hepatocellular carcinoma (HCC), is extremely resistant to conventional treatment. Chinese herbal medicine has been investigated as a treatment, combining cytotoxic effect against HCC cells with herbs that boost the host immune response. For this purpose, we conducted a meta-analysis of the literature to investigate the impact of Chinese Herbal Medicine (CHM) in combination with chemotherapy on liver cancer therapy. The present systematic review and meta-analysis was performed according to Preferred Reporting Items for Systematic Review and Meta-Analysis guidelines (PRISMA) and was illustrated according to MOOSE (Meta analysis of Observational Studies in Epidemiology) statement. A total of 13 randomized controlled trials (RCTs) were included in this meta-analysis. Data analysis was performed using Cochrane Collaboration Review Manage software (RevMan 5.2, CI = 95%). Quality assessment using the Cochrane risk-of-bias tool revealed that most studies had a low or unclear risk of bias. The results of the study demonstrated a significant increase in tumor response, survival rate, and child pugh score (chi2 = 6.82: 95% CI = 0.02; P < .001) when Chinese herbal medicine (CHM) and chemotherapy were administered together. According to this study, individuals with liver cancer who receive chemotherapy might experience a significant increase in tumor response (chi2 = 1.78: 95% CI = 0.08; P < .001) and survival rate (chi2 = 15.95: 95% CI = −0.03; P < .001) when CHM is added with chemotherapy treatment as opposed to chemotherapy administered alone. Nevertheless, the dearth of large-scale RCTs prevented a definitive conclusion; therefore, further large-scale and high-quality trials are required.
Introduction
Primary hepatocellular carcinoma (HCC), also known as liver cancer, is the second-leading cause of cancer-related deaths worldwide and the sixth most common malignancy in terms of diagnoses. Globally, liver cancer (LC) is the most prevalent cause of cancer-related death. The countries with the highest age-standardized incidence rates of LC worldwide are Thailand, Taiwan, China, Mongolia, Gambia, and 36.1, 35.7, 33.8, and 29.3 per 100 000 people, respectively, in 2008. 1 As the average annual occurrence of cancer rises, it has emerged as one of the most significant health problems affecting people globally. The World Health Organization estimates that there were about 780 000 additional cases of liver cancer reported globally in 2012. The cancer-related death rate was 5.1 in 100,000, ranking second among all cancer-related deaths. The worldwide prevalence was 10.1 per 100 000 individuals. 2 The 5-year survival rate for liver cancer is about 12%, which is a terrible prognostic. 3 The absence of a sensitive test for liver cancer impedes timely diagnosis of the illness in its early stages. Transcatheter arterial chemoembolization (TACE) or chemotherapeutic drugs are the only therapeutic options available for the more than 60% of newly diagnosed instances of advanced liver cancer. 3 Nevertheless, a large number of individuals have poor responses to TACE or have adverse results with traditional systemic cytotoxic treatment. 4 The use of alternatives and complementary therapies in the treatment of liver cancer is rising acceptance globally. Popular in East Asian and Chinese cultures, traditional Chinese medicine (TCM) is an integral part of Taiwan's National Health Insurance (NHI) program's modern healthcare system. Through the inhibition of tumor cell growth and promotion of tumor cell apoptosis, TCM has been shown to target the stimulation of the host immune response for cytotoxic activity against liver cancer, hence mitigating adverse effects associated with gene therapy or chemotherapy. 5 The use of complementary and alternative medicine, or complementary and alternative medicine (CAM), by cancer patients has grown throughout the last few decades. A comprehensive analysis comprising 26 trials of cancer patients from 13 different countries found that the percentage of CAM application ranged from 7% to 64%. (19). CAM was reported by 72% of patients with breast, colon, or prostate cancer who took part in a 2002 survey. Chinese-people have a strong interest for traditional Chinese medicine, or TCM. 6
Chinese herbal medicine is typically most usable TCM. Most widely use of TCM is due to various reasons such as cultural belief's of Chinese communities about effective management of diseases, treatments and prevalence. Since then, TCM has grown in acceptance in popular culture and is widely applied in addition to conventional medical care. TCM has a major role in the modern East Asian and Chinese healthcare systems. Therefore, one important feature of the(National Health Insurance) NHI program is its coverage of both TCM (TCMs)and biomedicine (Western). Following the implementation of the NHI Program in 2003, more than 23 million inhabitants of Taiwan were provided with universal health care coverage. 1
Depending on the medical practices and experience of various specialists, TCM remedies may be added to or removed from medications. This results in a combination that is unstable and challenging to precisely classify. On the other hand, specific formulations of customized Chinese herbal ingredients (CHIs) are only available in China are more suited for meta-analyses and treatment recommendations. It is noted that the combination approach of TCM and Western medicine is much more outcosuccessful in treating liver cancer instead of utilizing conventional therapy. 7 Several systematic evaluations have explored the effectiveness of customized Chinese medicines (CHIs) when used in conjunction with local therapy. 8 In recent years, the application of CHM for liver cancer treatment (PLC) has dew limitations. 9 A vital component of conventional Chinese medicine, PLC and its complications have been managed using physiotherapy. It has been demonstrated to be effective in treating a range of pain issues. 10 One of the most recent research areas of greatest concern, PLC, has progressively shown benefits from the use of Chinese herbal medicine as a supplemental and alternative treatment. 11 However, there isn't any conclusive proof of the efficacy or frequency of PLC adverse effects associated with acupuncture and Chinese herbal medicine. This is another significant reason that keeps it from expanding to the West to treat PLC. Chinese herbal medicine's effectiveness in treating liver cancer will thus be analyzed through this systematic review and meta-analysis. Patients may look for alternative treatments, such as TCM products, either alone or in conjunction with standard of care, despite the poor prognosis associated with certain liver malignancies and the restricted alternatives for treatment outside of surgery. Currently, no researcher has published a thorough, up-to-date analysis of CHM as a liver cancer adjuvant. Therefore, the objective of this study was to conduct a systematic review and meta-analysis of randomized controlled trials (RCTs) to evaluate the efficacy and safety of Chinese Herbal Medicine (CHM) as an adjunctive therapy alongside chemotherapy in the treatment of liver cancer.
Materials and Methods
Protocol and Registration
This systematic review and meta-analysis was conducted in accordance with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines and structured based on the MOOSE (Meta-analysis of Observational Studies in Epidemiology) framework. A protocol outlining the review methodology was developed in line with PRISMA recommendations and was prospectively registered in the PROSPERO database (registration number: CRD42024558228), accessible at https://www.crd.york.ac.uk/prospero. No external funding was received for the completion of this study.
Search Strategy
According to inclusion and exclusion criteria, a comprehensive search was performed of electronic databases including PubMed (MEDLINE), Embase, Cochrane Library, ScienceDirect, and Google Scholar to identify all related literature upto April 2024. The search technique comprised following mesh keywords and synonyms: (“TCM Medicine” OR “Chinese Medicine”, “THM” OR “Herbal drugs” “Medicinal Plants” OR “medicinal herbs” “TCM” AND “liver cancer” “hepatocellular carcinoma” “hepatic carcinoma” AND “chemotherapy” with slight modification for individual searches suitable for instruction of database.
Selection Criteria
The following criteria was used by two unbiased reviewers that filter references which met inclusion criteria: (1) The study contained controlled and treatment outcome group (2) Studies in which CHM was used in combination with chemotherapy as the primary intervention (3)Outcome with effective result (5) Included young individuals, defined as those aged between 21 and 35, (6) The research was published in English. There was no time or country restriction. Excluding criteria (1) Case study, reviews, abstracts, conferences, editorials, incomplete data, studies on animals or cell lines, (2) non RCT studies (control studies and case reports) (3) Non liver cancer patients, patients other tumor, (4) The original data could not be retrievable.
Data Extraction
The following details were gathered about the eligible studies that satisfied the above requirements: primary author, year of publication, study design, sample size, control and treatment group intervention and outcomes. The details were extracted by pairs of reviewers independently using a pre-signed data collection form based on prior publications. The most current publication was included when research or articles from the same population were repeated. The two investigators discussed any discrepancies, and a third party was consulted when necessary to obtain a consensus. The mean and standard deviation were estimated using the quantile estimation method suggested by ref. 12 if the retrieved papers were only provided the median and interquartile range.
Quality Assessment
The methodological quality of the included RCTs was evaluated using the Cochrane risk-of-bias tool, following the guidelines outlined in the Cochrane Handbook for Systematic Reviews of Interventions (version 5.1.0). This assessment focused on six key domains: selection bias (random sequence generation and allocation concealment), performance and detection bias (related to blinding), attrition bias (incomplete outcome data), reporting bias (selective outcome reporting), and other potential sources of bias. Each domain was categorized as having a low, high, or unclear risk. Based on these evaluations, the overall risk of bias for each study was classified into three levels: low (all domains rated as low risk), unclear (at least one domain rated as unclear), or high (at least one domain rated as high risk). The detailed assessments are presented in Figure 1 and Figure 2.

Risk of Bias for Included Studies Graph.
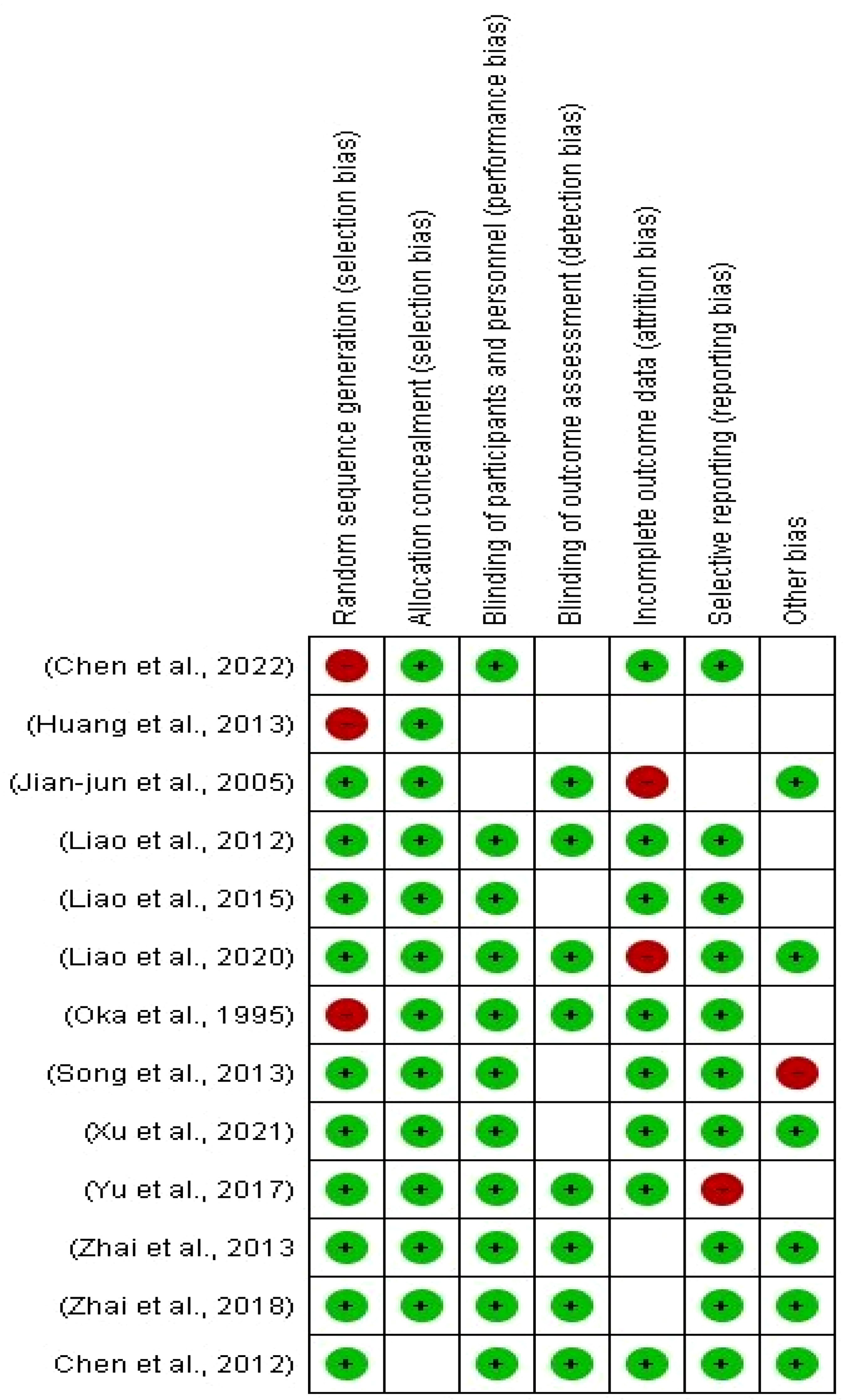
Risk of Bias for Included Studies Summary.
Data Analysis
Data analysis was performed using Cochrane Collaboration Review Manage software (RevMan 5.2) Continuous data were pooled as mean difference with 95% confidence interval (CI) and dichotomous outcomes were in the ratio of risks. Heterogeneity among the included studies was assessed using the I² statistic. When substantial heterogeneity was observed (I² > 50%), a random-effects model was applied to account for variability across studies. In cases of low heterogeneity, a fixed-effect model was used. A P-value of less than .05 was considered indicative of statistical significance in comparisons between treatment and control groups.
Results
Characteristics of the Included Studies
A total of 280 studies were obtained. There were 28 studies duplicated, 217 studies that did not look into outcomes of interest (eg, only looked into Chinese medicine syndrome scales), 75 studies that reviewed the literature, 17, abstract and conferences, 6 involved animals, 119 irrelevant studies (eg, looked into CHM for lymphedema following mastectomy), and 22 studies that were not RCTs. Thus, only 13 studies met the requirements for selection. In this meta-analysis, 13 RCTs were considered (Figure 3). Almost all of the included studies have a low risk of bias since all of the patients enrolled in the 13 trials were postoperative liver cancer patients. Figures 1 and 2 displayed the bias risk of each included study. In the included trials, the time frame of treatment ranged from 2 to 38 weeks. Table 1 included the therapy modalities and outcome measures used in each research. Every study asserted that age, TNM (tumor node metastasis) stage, or histology were equivalent baseline data.

Flow Diagram of Literature Selection.
A Summary of Each Study's Therapy Approach and Outcome Measures.

AFP: Alpha-Fetoprotein; ALT: Alanine Aminotransferase; AST: Aspartate Aminotransferase; BST: Bovine Serum Transferrin; FZHY: Fuzheng Huayu (a traditional Chinese medicine formula); TACE: Transcatheter arterial chemoembolization; TCM: traditional Chinese medicine.
Tumor Response
Three trials’ worth of data demonstrated that patients receiving chemotherapy in addition to Chinese herbal medicine had either a full or partial response, (Figure 4).

Forest Plot and Funnel Plot of Tumor Response.
Survival Rate
The survival rate of patients who had chemotherapy in addition to CHM was reported in seven trials’ worth of data, suggesting that Chinese herbal medicine with chemotherapy was more successful therapy for liver cancer (chi2 = 15.95: 95% CI = −0.03; P < .001) (Figure 5).

Forest Plot and Funnel Plot of Survival Rate.
Child Pugh Score
Findings from four studies indicated that patients receiving Chinese Herbal Medicine (CHM) alongside chemotherapy demonstrated significantly improved outcomes in terms of survival, supporting the benefit of the combined treatment approach for liver cancer (Chi² = 6.82; 95% CI = 0.02; P < .001) (Figure 6).

Forest Plot of Child Pugh Score.
Discussion
According to recent studies, people with cancer—especially those with liver cancer—have a significant prevalence of using CHM. Chinese herbal medicine (CHM) is particularly well-liked among cancer patients. However, because many studies were published in Chinese, it is unclear how well CHM paired with chemotherapy works for individuals with liver cancer. The objective of this study was to integrate the findings using a meta-analysis, which involved statistical examination of the findings from separate investigations. The study's pooled data indicates that the tumor response and performance status of patients with liver cancer were considerably enhanced by CHM in addition to chemotherapy. It was also observed that the combined therapy considerably reduces side effects associated with chemotherapeutic interventions when compared to chemotherapy alone. Adverse effects commonly observed with chemotherapy, such as grade III–IV nausea and vomiting, significant reductions in white blood cell (WBC) counts, and platelet decline (ranging from grades I–IV), were notably alleviated when CHM was administered concurrently. This meta-analysis suggests that incorporating Chinese Herbal Medicine (CHM) with chemotherapy enhances tumor response and overall clinical outcomes, while also mitigating toxic side effects like leukopenia, thrombocytopenia, and gastrointestinal distress. Similar benefits of CHM as an adjunct to chemotherapy have been reported in studies involving other cancers, including advanced non-small-cell lung cancer, colorectal cancer, nasopharyngeal carcinoma, and hepatocellular carcinoma. Collectively, these results reinforce the role of CHM as a promising supportive therapy in the management of liver cancer.24,25 TCM theory states that illness results from an unbalanced Yin and Yang; healing illness requires reestablishing this equilibrium. When the Yin and Yang of the human body are balanced, people can have a strong self-healing potential to eliminate pathogenic causes and regain health. Additionally, the importance of the human body's capacity for self-healing in the treatment of cancer. 26
The substantial heterogeneity observed in tumor response outcomes (I² = 94%) may be attributed to multiple factors. First, the composition of Chinese herbal medicine varied considerably across studies—some utilized single herbs, while others employed complex multi-herb formulations, leading to differences in bioactivity and synergistic effects. Second, the chemotherapy regimens combined with CHM were not standardized across studies, potentially influencing outcomes. Third, patient populations differed in terms of disease stage, baseline liver function, and geographic setting. Finally, methodological inconsistencies such as dosage, duration of treatment, and outcome measurement techniques may also contribute to this variability. Studies on the most commonly used herbs have shown that they are effective in promoting macrophage and natural killer cell activity while suppressing T-helper cell type 2 cytokines, hence promoting immunoregulation, immunological restoration, and anticancer effects.27,28 Additionally, Radix Astragalus helps lessen the toxicity that cyclophosphamide, a commonly used chemotherapy drug, causes. 29 The ability of CHMs to boost the immune system and lessen the toxicity caused by chemotherapy may be the two main benefits of using CHMs as an adjuvant therapy for liver cancer, even though the mechanism underlying their anticancer actions is not entirely understood. The study included four key outcome measurements: immunoregulation, toxicity produced by treatment, performance status, and tumor response. All included studies did not, however, release all results at the same time. As an illustration, Li and Han 30 solely documented chemotoxicity and tumor response. In spite of this, no subjective selection was used in the analysis of any of the accessible data in these trials. Measurements such as CD3/CD4T cell levels, CD8T cell levels, and CD4/CD8 ratios are used to assess immunoregulation in patients with liver cancer. With the exception of the fact that various chemotherapy regimens are employed in various research, the variation in the heterogeneity test in these studies may be caused by differences in treatment length and therapeutic dose. Under the registration number CRD42024558228, the study protocol was registered in the International Prospective Register of Systematic Reviews, or PROSPERO. There were a few limitations in the study even though all of the reviewers had excellent experience in systematic review and we closely adhered to the Cochrane Collaboration's review protocol. The majority of the included studies, to start, lacked a clear description of allocation concealment and blinding, which could lead to an overestimation of the treatment group's effect and the introduction of bias. Second, as practically all of the included studies reported favorable outcomes, there may be publication bias in this analysis. It is possible that some studies with negative outcomes were selectively unpublished or excluded, resulting in potential publication bias in this review. Additionally, many of the included trials exhibited methodological limitations—such as insufficient reporting on random sequence generation, lack of intention-to-treat analyses, incomplete follow-up details, and omission of dropout rates—which may have introduced bias into the findings. Furthermore, variability across the studies in terms of chemotherapy protocols, CHM formulations (single herbs vs combinations), routes of administration (oral vs intravenous), treatment durations, and dosages likely contributed to the observed heterogeneity.
Footnotes
Authors’ Contribution
Y.W and J.X the concept and design of the study and data acquisition; Q.Z statistical analysis; H.Y analyzed the data and drafted the manuscript. All authors critically revised the manuscript, approved the final version to be published, and agree to be accountable for all aspects of the work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
