Abstract
This review article compiles 1H and 13C NMR signals and specific rotations of reported eremophilane-type sesquiterpenoids. The data presented here are intended to assist chemists in natural products chemistry, synthetic chemistry, and computational chemistry. This review focuses on 71 furanoeremophilanes that contain a C-10 hydroxy group with oxygen functionalities in the skeleton, collected from 40 references. NMR data were collected from the published papers using SciFinder. Eremophilanes featuring 15,6-lactones, 6,15-ethers, and 1-en-10-ols are not included in this chapter. Dihydrofurans, lactols, dimers, nor- and seco-compounds, along with other related structures, will be addressed in separate reviews.
Introduction
Isolation of numerous eremophilane type sesquiterpenoids has been reported,1–3 and the number of new compounds continues to increase. They are biosynthesized through cyclization, rearrangement, functionalization, and elimination from simple small compounds. In the course of our research work, we have encountered many sesquiterpenoids, such as eremophilane, eudesmane, bisabolane, bakkane, and other related sesquiterpenoids. 3 The NMR data of known compounds are sometimes not completely listed in the paper. Supporting information is highly useful to identify the compound. Particularly 13C NMR data are important for comparison with known compounds. 4 Recently reviews of eremophilane sesquiterpenoids from Ligularia were published.2,3 As a part of an ongoing series of reviews,5–8 data of 71 furanoeremophilane sesquiterpenoids are listed in Tables 1-13. This review focuses on furanoeremophilan-10-ols bearing oxygenated functionalities in the skeleton. NMR data have been compiled from the published papers using SciFinder covering the period from the 1960s to the present; however, due to the inherent limitation of not being able to review all available literature, it does not necessarily encompass all existing data. Compounds and the numberings mentioned in this review are shown in Figures 1-6. 10-Hydroxyfuranoeremophil-1-en-3-ones are not included in this review, but in the following review of alkene part. Dihydrofurans, lactols, dimers, nor- and seco-compounds, and other related compounds will be discussed in separate reviews in the future. In the case of compounds having more than two acyloxy groups, the one substituted in the lower numbered position is numbered with a prime, the next with a double prime, and so on. The coupling constants are sorted from the largest to the smallest; the order of the shapes, d, t, or q, must correspond to the value of the coupling constant. In the case of apparent situation, original data were fixed to the proper order, or the problem with the data pointed out in the footnotes. The data not assigned in the original papers were tentatively listed in the tables without any supporting evidence. The names of skeletons and the numbering system used in this review should refer to the ones used in the previous paper.5–8 Abbreviations used in this review are as follows5–8: solvents; A: acetone or acetone-d6, B: benzene or benzene-d6, C: chloroform or chloroform-d, D: dimethyl sulfoxide or dimethyl sulfoxide-d6, E: ethanol, F: dichloromethane, H: hexane, M: methanol or methanol-d4, P: pyridine or pyridine-d5, T: carbon tetrachloride. Source of compounds; Ca: Cacalia (current Parasenecio, however original description has been remained), Cr: Cremanthodium, E: Eremophila, F: Farfugium, L: Ligularia, P: Petasites, S: Senecio, V: Valeriana.

Tetradymol (

10-OH Compounds with Oxygenated Functionalities at C-1 and C-6,

10-OH Compounds with Oxygenated Functionalities at C-3 and C-6,

10-OH Compounds with Oxygenated Functionalities at C-3 and C-9,
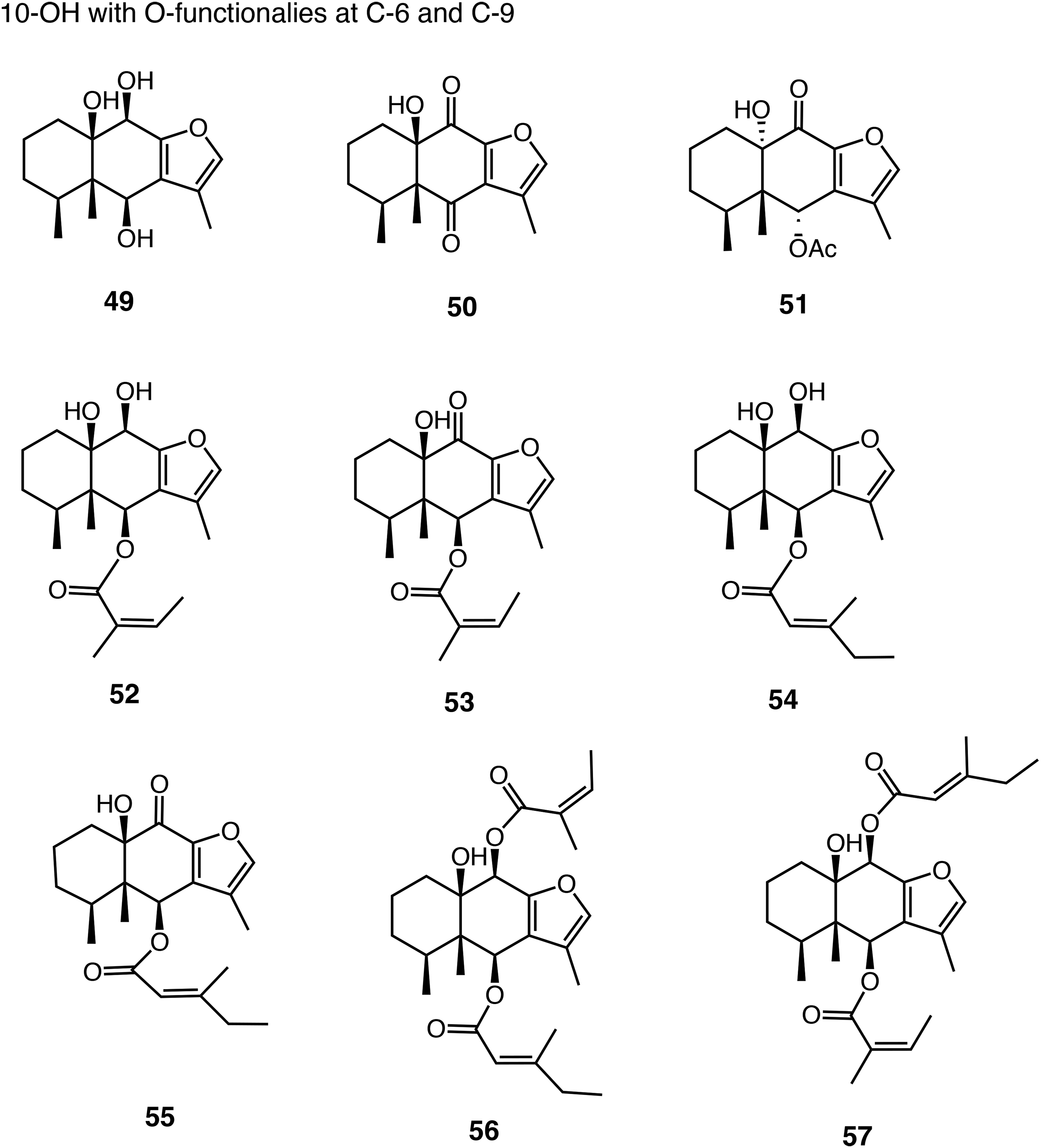
10-OH Compounds with Oxygenated Functionalities at C-6 and C-9,

10-OH Compounds with Oxygenated Functionalities at C-1, C-3, C-4, C-6, and C-9,
NMR Chemical shifts (solvent, δ, multiplicity, and J (Hz)), Specific Rotations, Source of Compounds, and References of Compounds

Abbreviation: nd, not displayed.
X-ray.
Solvent only described as either CDCl3 or CCl4.
Measured at 60 °C.
Multiplicity and J not shown.
J = 9 Hz seems to be unusual.
NMR Chemical Shifts (Solvent, d, Multiplicity, and J (Hz)), Specific Rotations, Source of Compounds, and References of Compounds

Abbreviation: nd, not displayed.
The data of specific rotation was revised to +33 (E) in ref. 15
Tentative.
Obtained after the deuterium exchange experiment.
NMR Chemical Shifts (Solvent, d, Multiplicity, and J (Hz)), Specific Rotations, Source of Compounds, and References of Compounds

Abbreviation: nd, not displayed.
Tentative.
H-6 and OH not shown in ref. 17
NMR Chemical Shifts (Solvent, d, Multiplicity, and J (Hz)), Specific Rotations, Source of Compounds, and References of Compounds

Abbreviation: nd, not displayed.
Multiplicity not shown, but must be “s”.
Multiplicity shown as “q”, but must be “d” (AB quartet).
NMR Chemical Shifts (Solvent, d, Multiplicity, and J (Hz)), Specific Rotations, Source of Compounds, and References of Compounds

Abbreviation: nd, not displayed.
X-ray.
Compounds
Tentative.
Solvent C or T, not shown.
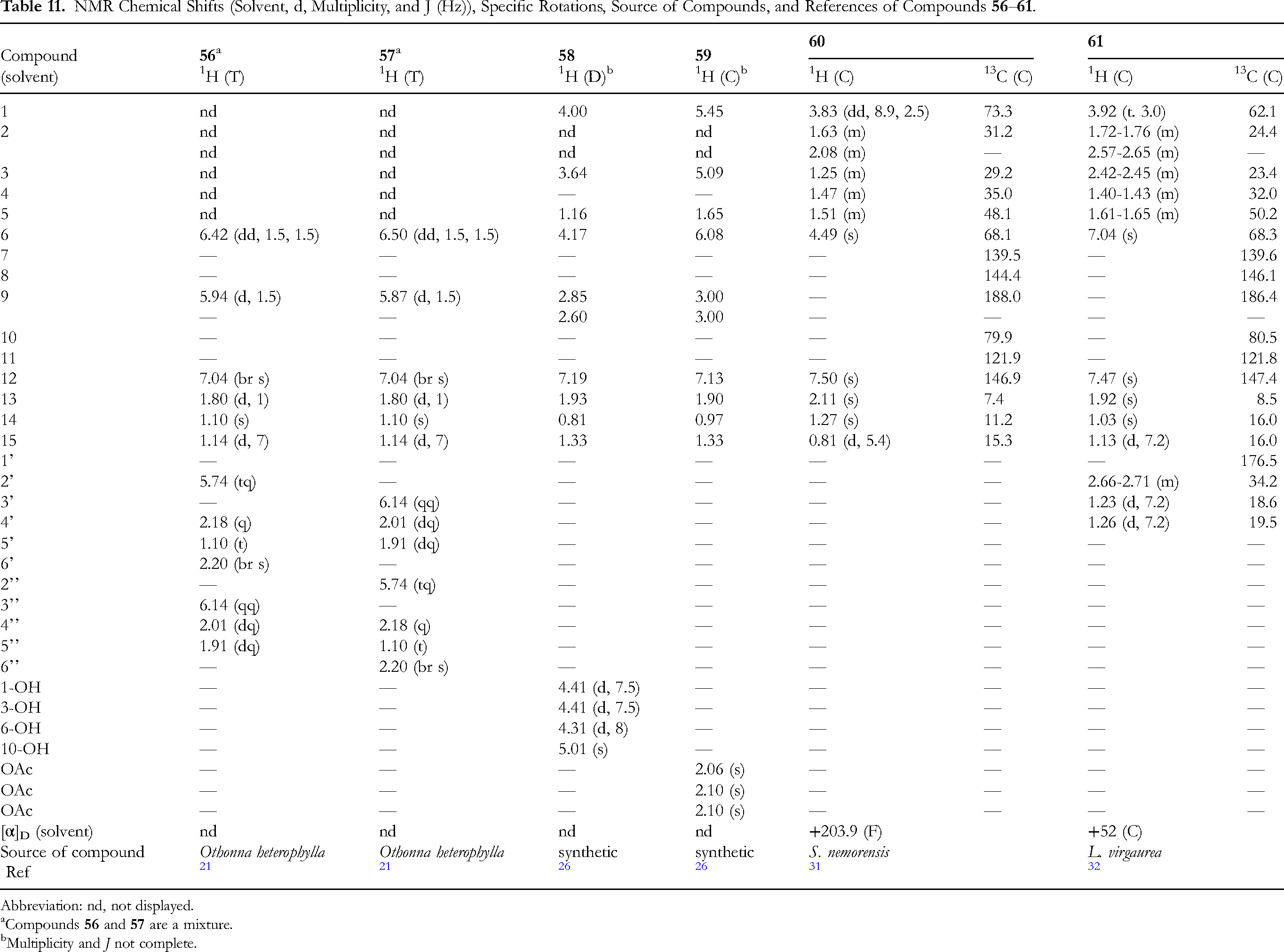
Multiplicity and J not complete.
Measured at 80 °C.
May be interchanged.
May be interchanged.
NMR Chemical Shifts (Solvent, d, Multiplicity, and J (Hz)), Specific Rotations, Source of Compounds, and References of Compounds

Abbreviation: nd, not displayed.
Multiplicity and J not shown.
NMR Chemical Shifts (Solvent, d, Multiplicity, and J (Hz)), Specific Rotations, Source of Compounds, and References of Compounds

Abbreviation: nd, not displayed.
Compounds
Compouonds
Measured at 75˚C.
Shown only as “13”, but most probably “6.13”.
Measured at 436 nm.
NMR Chemical Shifts (Solvent, d, Multiplicity, and J (Hz)), Specific Rotations, Source of Compounds, and References of Compounds

Abbreviation: nd, not displayed.
CD measured.
NMR Chemical Shifts (Solvent, d, Multiplicity, and J (Hz)), Specific Rotations, Source of Compounds, and References of Compounds

Abbreviation: nd, not displayed.
CD measured.
Only [α]D shown.
NMR Chemical Shifts (Solvent, d, Multiplicity, and J (Hz)), Specific Rotations, Source of Compounds, and References of Compounds

Abbreviation: nd, not displayed.
Compounds
Multiplicity and J not complete.
NMR Chemical Shifts (Solvent, d, Multiplicity, and J (Hz)), Specific Rotations, Source of Compounds, and References of Compounds
Abbreviation: nd, not displayed.
Compounds
Multiplicity and J not complete.
NMR Chemical Shifts (Solvent, d, Multiplicity, and J (Hz)), Specific Rotations, Source of Compounds, and References of Compounds

Abbreviation: nd, not displayed.
CD shown in ref. 35
Solvent not shown, but may be CDCl3 by analogy.
Measured at 70˚C.
Multiplicity must be “dd”.
One J missing.
J not shown.
NMR Chemical Shifts (Solvent, d, Multiplicity, and J (Hz)), Specific Rotations, Source of Compounds, and References of Compounds

Abbreviation: nd, not displayed.
Numbering changed.
X-ray.
Assigned to H-2 in ref, 35 but must be H-12.
Not assigned, but must be H-5’ (“q” should be “quint”).
The data of tetradymol (
Table 2 shows the data of 4αH-10β-hydroxyfuranoeremophilan-3-one (
The data of 6β-methoxyfuranoeremophilan-10β-ol (
Table 4 illustrates the data of 6β-acetoxyfuranoeremophilan-10β-ol (
The data of four 6β-acyloxyfuranoeremophilan-10β-ols (
Table 6 shows the data of 10β-hydroxyfuranoeremophilane-1,6-dione (
The data of four 3β-acetoxy-6β-acyloxyfuranoeremophilan-10β-ols,
Table 8 provides the data of four 10β-hydroxyfuranoeremophilan-3-ones,
The data of two 6β-acyloxy-10β-hydroxyfuranoeremophilan-3-ones,
Table 10 presents the data of two 3β-acyloxyfuranoeremophilan-10β-ols,
The data of two 6β,9β-diacyloxyfuranoeremophilan-10β-ols,
Table 12 illustrates the data of two 1,6-diacyloxy-10β-hydroxyfuranoeremophilan-9-ones,
The data of bezoylated compound
Conclusion
This review focuses on 71 furanoeremophilanes that contain a C-10 hydroxy group with oxygen functionalities in the skeleton. The simplest compound is tetradymol (
Footnotes
Acknowledgements
The author thanks laboratory staff members engaged in these projects acquiring NMR data. Special thanks are due to Dr Yasuko Okamoto and Prof. Masakazu Sono, Tokushima Bunri University, Prof. Yoshinosuke Usuki, Osaka Metropolitan University, and Emeritus Prof. Chiaki Kuroda, Rikkyo University, for their help to check the references appeared in this review.
Ethical Considerations
Ethical approval is not applicable for this review.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
