Discussion
The data of 10αH-furanoeremophilane (1),
3
10βH-furanoeremophilane (2), and furanoeremophilan-1α-ol (3) (Figure 1) are listed in Table 1. Both 10αH- and 10βH-furanoeremophilanes are known and five reports are found for compound 2.3-7 Unfortunately, the detailed assignment for 1 and 2 has not been published. The numbering for C-14 and C-15 was adjusted to our system.1-3 The detailed J values of the oxymethine proton at C-1 of compound 3 resonating at δ 3.88 were not shown.
8
Table 2 shows the data of 6-epi-ligularol (4) (Figure 1), which exists in nature. Synthetic compound is also recorded, but its full assignment of 1H nuclear magnetic resonance (NMR) is not found in the literature.4,9 However, the 13C NMR data are analyzed in detail to find the conformation of this molecule.7,10 Both “steroid-like” (minor) and “non-steroid-like” (major) conformations (vide infra) are found at −70 ˚C.
Table 3 includes the data of petasalbin (=ligularol) (5) (Figure 1).7,9-13 Although this compound has been isolated from many plants, its full assignment has rarely appeared in the literature.
12
Petasalbin itself was isolated by Novotný, Herout, and Šorm in 1964, but its structure was not determined at that time. Later Ishii, Tojo, and Minato isolated ligularol (5), which was identical with petasalbin, and studied in detail to assign its structure as furanoeremophilan-6β-ol. Thus, we decided to use the name “ligularol” in this review. Please note that “petasalbine” is used by the original authors, but “petasalbin” by the recent authors. The conformations of ligularol (5) have been studied using temperature dependent NMR spectra in various solvents.
10
They found the presence of conformational equilibria in various eremophilane sesquiterpenoids, and discussed the predominant conformation by use of temperature dependent NMR and CD spectra.9,10
The data of ligularol methyl ether (=petasalbin methyl ether) (6),
14
its ethyl ether (7),
15
its acetate (8),
16
four furanoeremophilan-9-ols, 9 to 12, and furanoeremophilan-1-one (13) are included in Table 4 (Figure 1). The numbering for C-14 and 15 has been confusing. We use our system as described before.
1
Compounds 9 to 12 are particularly interesting, because four possible isomers are compared. Usually in the case of 10βH-eremophilanes a doublet methyl group (H3-15) resonates at the higher field than a singlet methyl group (H3-14) (see compounds 6-8, and 12), while for compounds, 9, 11, and 13, opposite. However, the data of compound 11 by Novotný et al3,17 and are quite different, although these two reports were published by the same group. The NMR solvent for compound 13 is not described (CDCl3 or CCl4).
The data of ligularone (14) are listed in Table 5 (Figure 1).4,7,9,18-22 Ligularone (14) was isolated from L sibirica in 1965,
8
but its botanical name was later revised to L fischeri.
4
The conformation of this compound is also studied by temperature dependent NMR and CD spectra in different solvents.7,21 However, the detailed data of H2-1, 2, 3, and H-4 still need to be reported.
Table 6 shows the data of five furanoeremophilan-6-ones, 15 to 19,20,23 and furanoremophilan-9-one (20) (Figure 1).6,13,17,20,24-26 Compounds 18 and 20 are 10αH-6-one and 9-one, respectively. Compound 15 has a double bond at C-2/C-3, 16 at C-3/C-4, and 17 at C-4/C-15, respectively. Compound 19 is 4-epi-ligularone
19
and apparently chemical shifts of H3-15 and H3-14 are quite different from those of ligularone (14) (Table 5). The chemical shift of H-12 of compound 20 is at the lower field than that of compound 19.13,26 It is because the carbonyl group of compound 20 is present at the C-9 position. The 13C NMR chemical shifts of C-7 and C-8 of furanoeremophilanes are usually around 116 and 149 ppm, respectively, but they are 135.3 (C-7) and 147.7 (C-8) for compound 20. These differences can be seen in all the tables. Unfortunately, no specific rotation was found for compounds 15 to 20.
Table 7 illustrates the data of 10βH-furanoeremophilan-9-one (21)6,17,27,28 (Figure 1) and two 1β-acyloxyfuranoeremophilanes, 22
29
and 23
30
(Figure 2). Since the carbonyl group of compound 21 exists at the benzylic position of furan, H-12 of compound 21 resonates at the lower field than that of ligularone (14)18,20,22 as discussed in compound 20 (Table 6). The protons at C-1 of compounds 22 and subspicatin C (23) have two big and one small couplings, and thus these molecules adopt the nonsteroid-like conformation (vide infra). Apparently this is due to the 1,3-diaxial repulsion of the H3-14 and the acyloxy moiety at C-1 in the steroid-like conformation. Please note that compound 22 and subspicatin C (23) were measured in C6D6 and H3-14 appeared at the higher field δ 0.74.29,30
The data of two 2β-acyloxyfuranoeremophilanes, furanojaponin (24) and compound 25 (Figure 2) are compiled in Table 8. The acyloxy groups of 24 and 25 are angeloyloxy and senecioyloxy, respectively. The shape of H-2 of furanojaponin (24) is dddd with two 8 Hz and two 3 Hz coupling constants.
31
It seems that this compound adopts the steroid-like conformation and thus H-2α is in the axial position, although 8 Hz (actually 8-10 Hz by Bohlmann and Ziesche
31
) is rather small as axial–axial coupling.
31
The C-10 carbon signals of furanojaponin (24) and 25 are missing.
32
Furanojaponin (24) was first isolated by Naya et al,
14
which is not cited by Bohlmann and Ziesche,
31
unfortunately.
Table 9 illustrates the data of four 6β-acyloxyfuranoeremophilanes, 26 to 29 (Figure 2). The acyloxy groups are, 2-methylpropanoyloxy, 2-methylbutanoyloxy, angeloyloxy, and senecioyloxy, respectively.28,33,34 The NMR spectra of compounds 26 and 27 were measured at 50 ˚C and 60 ˚C, respectively, due to signal broadening at room temperature. The detailed data except for the specific rotation of albopetasin (28) have not been reported.
34
Compound 28 was assigned as ligularol angelate without the detailed discussion.
34
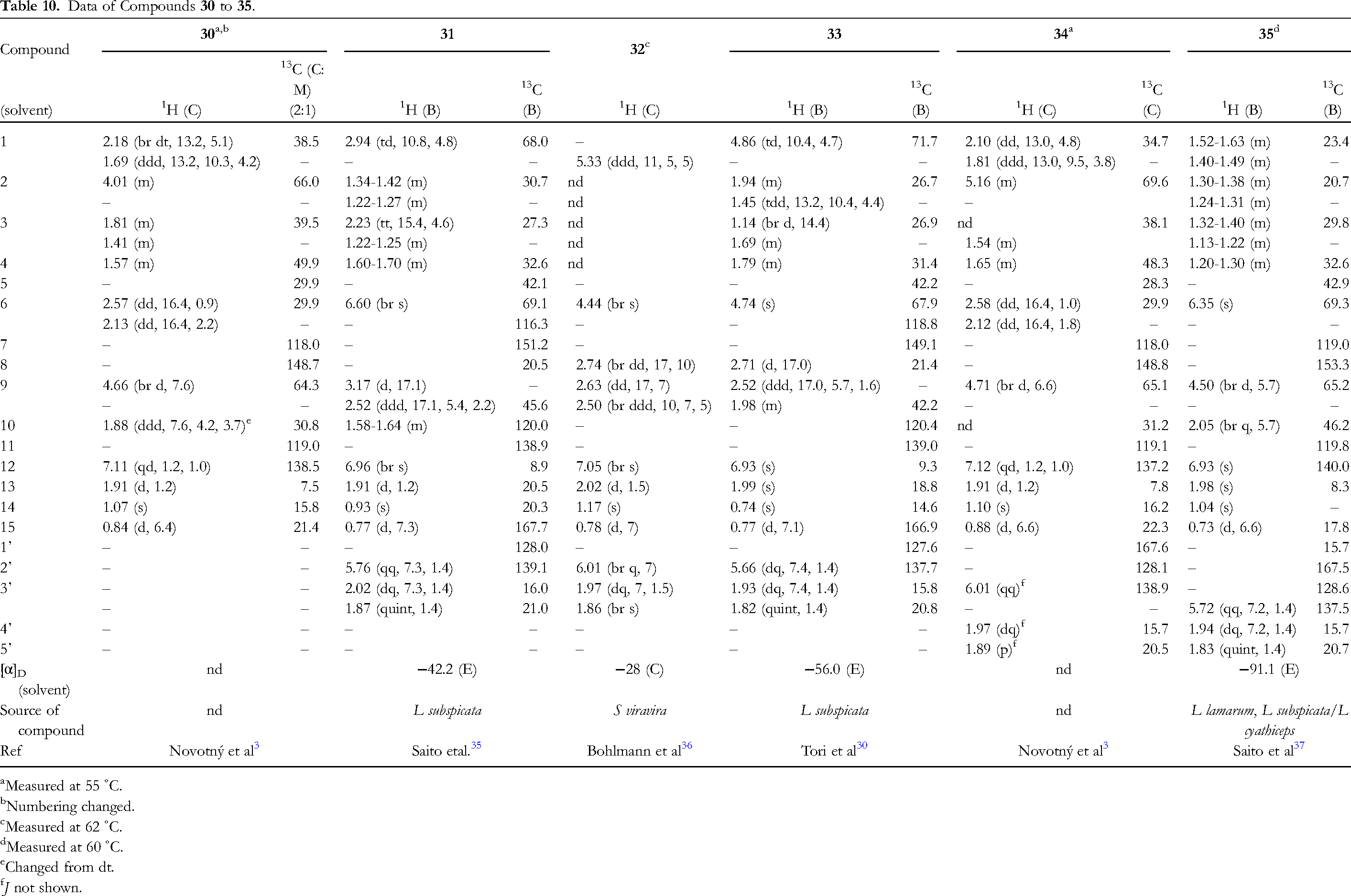
Table 10 shows the data of diol 30, and five diol mono acyloxy compounds, 31 to 35 (Figure 2). Compound 30 is furanoeremophilane-2β,9β-diol,
3
and compound 34 is the 2β-angeloyloxy derivative of 30.
3
Subspicatol A (31) is 6β-angeloyloxyfuranoeremophilan-1β-ol,
35
while compounds 32 and subspicatin B (33) are 1α- and 1β-angeloyloxyfuranoeremophilan-6β-ol, respectively.30,36 Compound 35 is 6β-angeloyloxyfuranoeremophilan-9β-ol.
37
The NMR data of compounds 30 and 34 were measured at 55 ˚C,
3
32 at 62 ˚C,
36
and 35 at 60 ˚C,
37
respectively, presumably due to signal broadening at rt. Compound 30 must be in the steroid-like conformation, while 31 the nonsteroid-like conformation.
The data of 6β-tigloyloxyfuranoeremophilan-9β-ol (36),
38
1β-angeloyloxyfuranoeremophilan-13-ol (subspicatin A) (37) (Figure 2),
30
6β-ethoxy-1α-angeloyloxyfuranoeremophilane (38),
39
and three 6β-acetoxy-1β-acyloxyfuranoeremophilanes, 39 to 41 (Figure 3),
40
are found in Table 11. Subspicatin A (37) has a hydroxy group at C-13, which is relatively rare.
30
Compound 38 has a 1α-acyloxy substituent,
39
while compounds 39 to 41 1β-acyloxy groups. Judging from the coupling constants of H-1α (one big and two small) of compound 38, this molecule adopts the steroid-like conformation, while compounds 36 and 37, and 38 to 40 the nonsteroid-like conformation. Please note that Bohlmann et al
38
cited compound 28 with Naya et al
14
in this review (Tada et al
23
in the original paper), but this is wrong.
Table 12 provides the data of six 1β,6β-diacyloxyfuranoeremophilanes, 42 to 47 (Figure 3). Compounds 42, 44, and 45 have 1β-angeloyloxy groups, while 43, 44, and 46, 6β-angeloyloxy groups. Subspicatin O1 (42) and subspicatin O2 (43), two acyloxy groups being attached at C-1 and C-6 in opposite positions to each other, were analyzed as a mixture.
37
The senecioyloxy group is attached at C-6β of 45 and 47, C-1β of 46 and 47.
40
Table 13 shows the data of four furanoeremophilan-1-ones, 48 to 51 (Figure 4). Compounds 48 and 49 are C-10 epimers, namely, 48 is the 10αH isomer and 49 the 10βH isomer, respectively.
41
Comparing the data of 48 with 49, H-6 and H3-15 of 48 appeared at the lower field than those of 49. The 13C NMR data of C-6 and C-10 appeared at the lower field than those of 49, however, C-15 of 48 significantly at the higher field.
41
Compound 50 has the 6α-acetoxy group.
42
Compound 51 is the 2-methylpropanoyl derivative of compound 48.41,43
The data of five 6-acyloxyfuranoeremophilan-1-ones, 52 to 56, are listed in Table 14 (Figure 4). Compound 53 has 10αH,
41
while compounds 52, and 54 to 56, 10βH.41,42,44 Compounds 52 to 54 have 6β-acyloxy groups, while 55 and 56 6α-acyloxy groups. Both the pair of compounds 51 (Table 13) and 52, and 53 and 54 are C-10 epimers.
41
Comparing the data of 53 with those of 54, H-6 of 53 appeared at the lower field as mentioned in the part of Table 13, however, H3-15 appeared in the same region. The C-6 and C-10 of 53 appeared at the lower field, showing the same tendency mentioned in Table 13.
41
The 1H NMR data of 52 were the same as those of 54, but the 13C NMR data only of 54 were described.
41
It is interesting that the data of 55 and 56 are quite different.42,44
The data of 1β-hydroxyfuranoeremophilan-6-one (57),
41
1α-acetoxyfuranoeremophilan-6-one (58),
45
and three 9-hydroxyfuranoeremophilan-6-ones, 59-61,
20
are compiled in Table 15 (Figure 4). Compounds 60 and 61 have 10αH and a double bond at C-2/C-3. Compounds 60 and 61 are C-9 epimers. The H-1 of compound 57 appeared at δ 3.41 (ddd, 10.5, 9.7, and 51), showing that the hydroxy group should be in a β orientation in the nonsteroid-like conformation.
41
Contrary to 57, compound 58 adopts the steroid-like conformation, because H-1 resonates at δ 5.22 (dt, 11.0, 4.6), with one large and two small couplings.
45
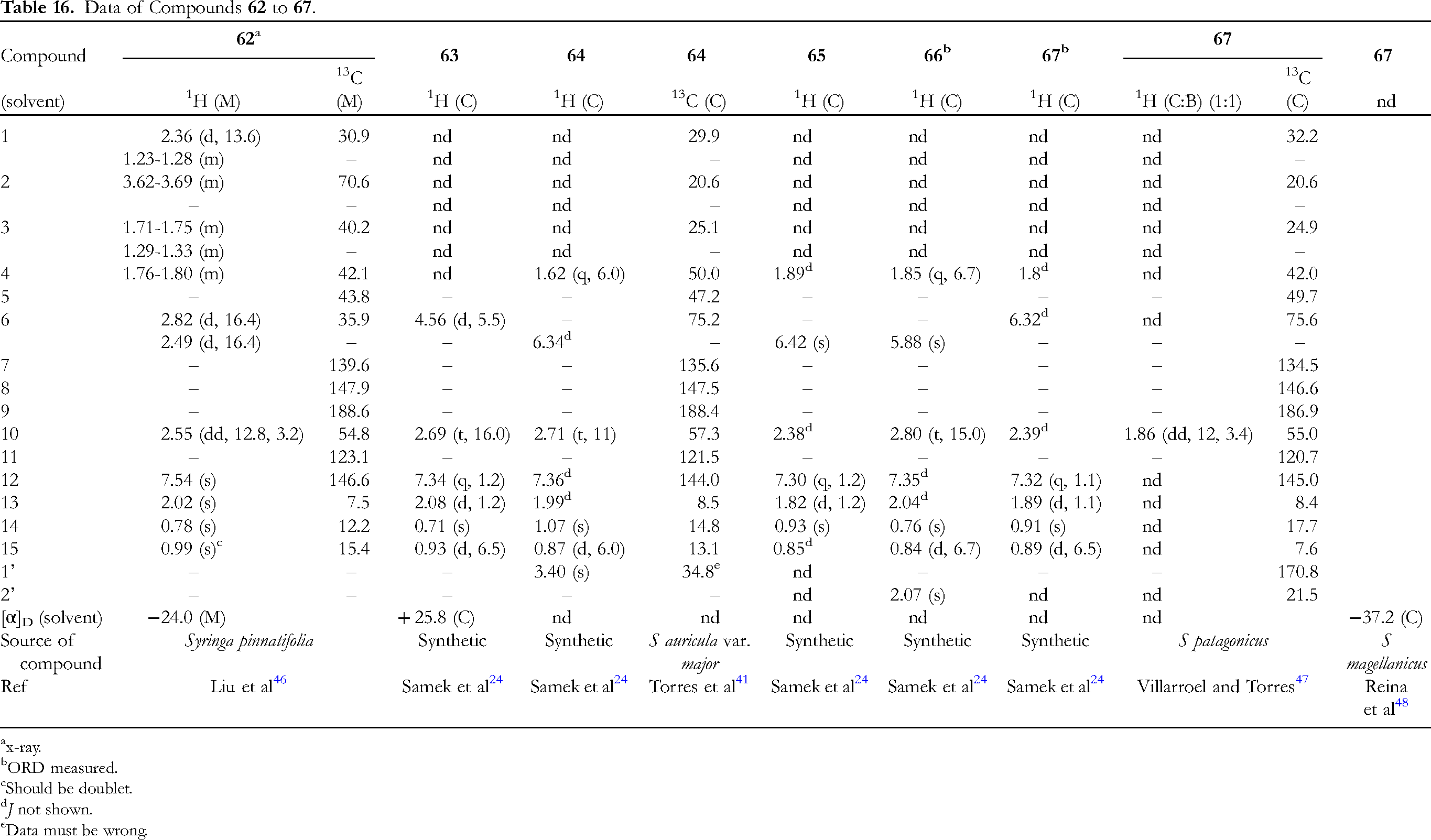
The data of six 10αH-furanoeremophilan-9-ones, 62-67, are provided in Table 16 (Figure 5). Alashanoid Q (62) has 2β-hydroxy,
46
compound 63 6α-hydroxy,
24
64 6α-methoxy,24,41 65 6α-ethoxy,
24
66 6α-acetoxy,
24
and 67 6β-acetoxy substituents.24,47,48 Compound 66 is an acetate of 63.
24
Compounds 66 and 67 are C-6 epimers.
24
The 13C NMR signal of OMe of compound 64 was attributed to 34.8, but this is too high for the methoxy carbon.
41
The structure of alashanoid Q (62) was analyzed by x-ray.
46
The ORD spectra of compounds 66 and 67 were measured.
24
The data of four 10αH-6β-acyloxyfuranoeremophilan-9-ones, 68 to 71, are found in Table 17 (Figure 5). Compound 68 has 2-methylpropanoyloxy,24,47 69 2-methylbutanoyloxy,
49
70 angeloyloxy,24,50 and 71 tigloyloxy substituents.
51
The 1H NMR data of sesquiterpene part of four compounds are almost the same. The ORD spectrum of compound 70 was measured.
24
Table 18 shows the data of seven 10αH-6-acyloxyfuranoeremophilan-9-ones, 72 to 78 (Figure 5). Compound 72 has senecioyloxy,
52
73 2-methylbut-3-enoyloxy,
49
74 3-methylpent-2-enoyloxy,
52
75 2-hydroxymethylpropenoyloxy,
53
76 2-methyl-2,3-epoxypropanoyloxy,
54
and 77 and 78 2-methyl-2,3-epoxybutanoyloxy substituents.51,54 Please note that H-12, H3-13, H3-14, and H3-15 of 75 resonate at the higher field, because they are measured in C6D6.
53
The H-6β of 77 appeared at δ 6.50, slightly at the lower field than H-6α of other compounds.
54
The data of four 10βH-furanoeremophilan-9-ones, 79 to 82, are listed in Table 19 (Figure 6). Compound 79 has the senecioyloxy group at C-2β position.
55
Compounds 80 and 82 has the 6α-hydroxy group, and 82 a double bond at C-2/C-3.
20
Compound 81 is the C-6 epimer of 80. Unfortunately, the data of H-6 of compounds 80 and 82 were not described.
20
Compound 81 was measured at 60 ˚C.
36
The data of four 10βH-6-acyloxyfuranoeremophilan-9-ones, 83 to 86, are compiled in Table 20 (Figure 6). Compounds 83 and 84 are C-6 epimers.20,24 Compound 85 has 6β-(2-methylpropanoyloxy) and 86 6β-(2-methylbutanoyloxy) groups. The 1H NMR spectrum of compound 85 was measured at 75 ˚C in C6D6.
51
The H-4 coupling patterns of compounds 83, 85, and 86 were described as q, indicating the coupling with H3-15, but they should have more couplings with H2-3.
24
The data of four 6β-acyloxyfuranoeremophilan-9-ones, 87 to 90, are presented in Table 21 (Figure 6). The configuration at C-10 of compound 87 has not been determined.
56
However, it is not easy to imagine H10α or H10β. The structure of compound 88 was analyzed by x-ray.
57
The C-1, C-2, and C-9 signals of compound 88, and C-1 and C-2 of 90 were not detected in C6D6 due to signal broadening (temperature was not raised).
13
Table 22 shows the data of seven furanoeremophilane-6,9-diones, 91 to 97 (Figure 6).
20
Compounds 91 to 93 have 10αH, and 94 to 97 10βH, respectively. Compounds 91, 93, 94, and 96 have 4α-methyl groups. Compound 97 has a double bond at C-3/C-4, and 93 and 96 at C-2/C-3. Compounds 91 and 94, and 92 and 95 are C-10 epimers, respectively. Unfortunately no 13C NMR data was described for these compounds.
20
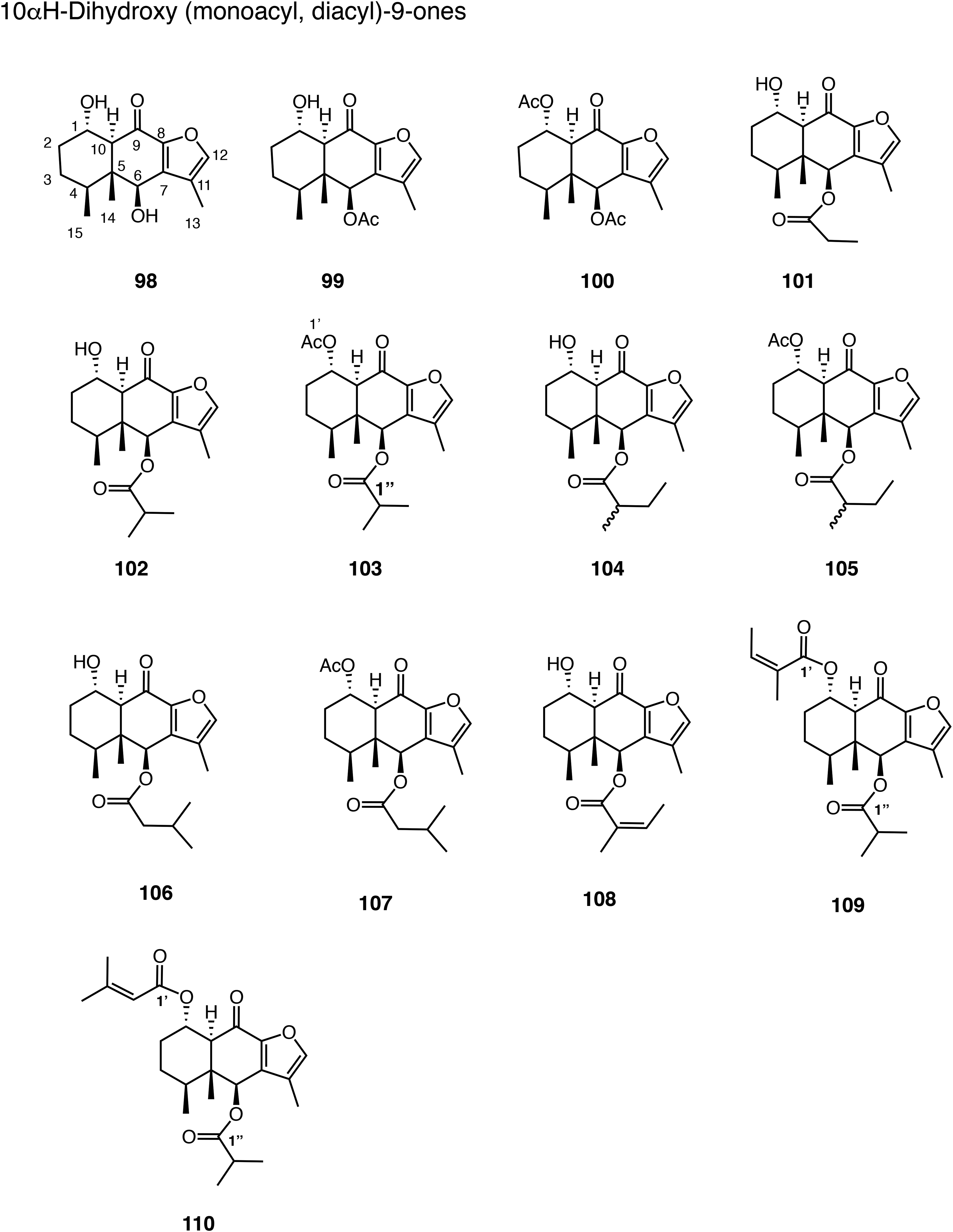
The data of six 10αH-furanoeremophilan-9-ones, 98 to 103 are found in Table 23 (Figure 7). Compound 98 has 1α,6β-dihydroxy,
58
99 6β-acetoxy-1α-hydroxy,59,60 100 1α,6β-diacetoxy,59,60 101 1α-hydroxy-6β-propanoyloxy,
61
102 1α-hydroxy-6β-(2-methylpropanoyloxy),62,63 and 103 1α-acetoxy-6β-(2-methylpropanoyloxy) substituents.
47
The solvent for NMR was not described for compounds 98 and 102, but most probably CDCl3 by analogy.58,62 The numbering used for compound 103 was changed to our system.
47
Two hydroxy resonances of compound 98 were assigned, respectively.
58
Table 24 gives the data of three 1α-hydroxy-6β-acyloxyfuranoeremophilan-9-ones, 104, 106, and 108, two 1α-acetoxy-6β-acyloxyfuranoeremophilan-9-ones, 105 and 107, and two 1α,6β-diacyloxyfuranoeremophilan-9-ones, 109 and 110 (Figure 7). Compounds 105 and 107 are acetyl derivatives of 104 and 106, respectively.
63
The solvent used for 110 was not described, but most probably CDCl3 by analogy.
62
The H-1 of compound 104 at δ 4.17 (m, 5.1) must be strange.
63
This could be just multiplet or could have more couplings. One more coupling should be shown for H-1 and H-4 of compound 109.
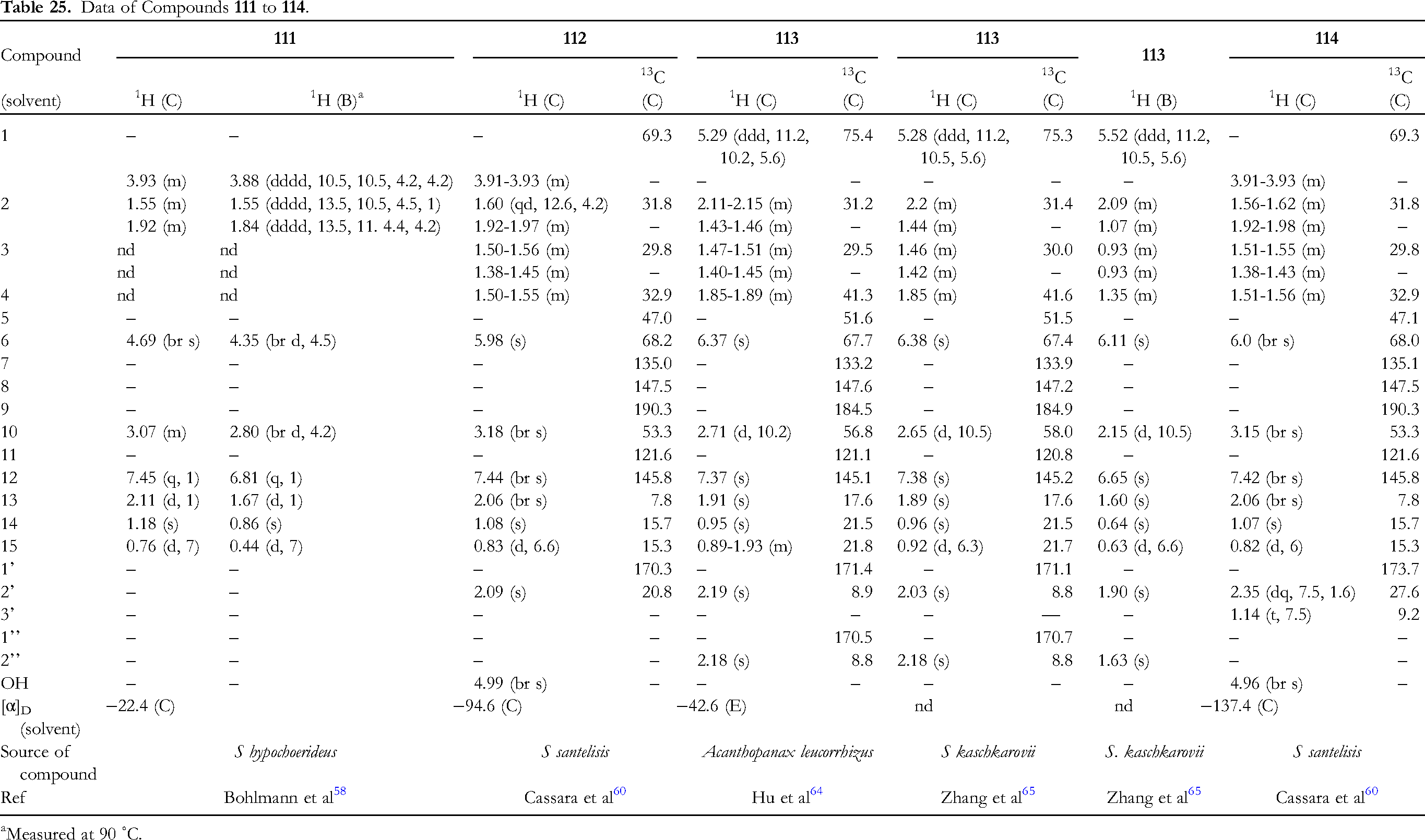
The data of four 10βH-1,6-dioxidized furanoeremophilan-9-ones, 111 to 114, are given in Table 25 (Figure 8). Compounds 111, 112, and 114 have 1α-hydroxy,58,60 and 113 1β-acetoxy groups.64,65 Compounds 98 (Table 23) and 111, 99 (Table 23) and 112, and 101 (Table 23) and 114 are C-10 epimers, respectively. Concerning the configuration of C-1 of compound 111, it is quite doubtful.
58
The H-1 has two big and two small (one due to the coupling with OH) coupling constants (Table 25). This can only be true if the molecule adopts the nonsteroid-like conformation substituted with C-1β oxygen function as seen in compound 113. Since H-1 of 112 and 114 appears multiplet, it is not possible to discuss this situation in these cases.
60
However, compound 38 (Table 11, Figure 3) clearly indicated that H-1β appeared at δ 5.46 (br dt, 11, 5.5), showing one big and two small couplings.
39
This is only possible when the molecule bears the C-1α oxygen function, and adopts the steroid-like conformation.
The data of 1α-hydroxy-6β-(2-methylpropanoyloxy)furanoeremophilan-9-one 115,
63
and three 4α-hydroxy-6β-acyloxyfuranoeremophilan-9-ones, 116 to 118,
66
are shown in Table 26 (Figure 8). The configuration at C-10 of compound 117 has not been determined.
66
The coupling constant of H-1 of 115 was not shown, but described as multiplet.
63
The configurations at C-4 of compounds 116 to 118 were determined by the presence of intramolecular hydrogen bonding with the acyloxy group at C-6 detected in their IR spectra.
66
The data of two 4α-hydroxy-6β-acyloxyfuranoeremophilan-9-ones, 119 and 120 (Figure 8), and three polyoxygenated furanoeremophilanes, 121 to 123 (Figure 9), are found in Table 27. The data of compounds 119 and 120 are almost the same as those of compounds 116 to 118 (Table 26).
66
Subspicatin G (121) has 1β-angeloyloxy and 6β,13-dihydroxy substituents.
33
Velatumin (122) has three hydroxy groups at C-2β, C-6β, and C-13 positions.
67
Compound 123 is 10αH-2β-hydroxy-6β-(2-methylpropanoyloxy)-1α-senecioyloxyfuranoeremophilan-9-one.
62
The NMR data of the 2-methylpropanoyloxy group of 123 is described in the footnote b.
62
Subspicatin G (120) adopts the nonsteroid-like conformation as suggested by two big and one small couplings.
33
The data of two 15-oic acids, 124 and 127, and two methyl esters, 125 and 126, are provided in Table 28 (Figure 9). Compound 125 is the methyl ester of 124.
68
Compound 126 is the 6-one derivative of 125.
69
Compound 127 is the C-6β angeloyloxy derivative of 124.
70
The structure of 126 shown by Shen et al
69
(compound 1 in the original ref) is wrong; methyl ketone (C(O)Me) shown by Shen et al
69
should be methyl ester (CO2Me).
69
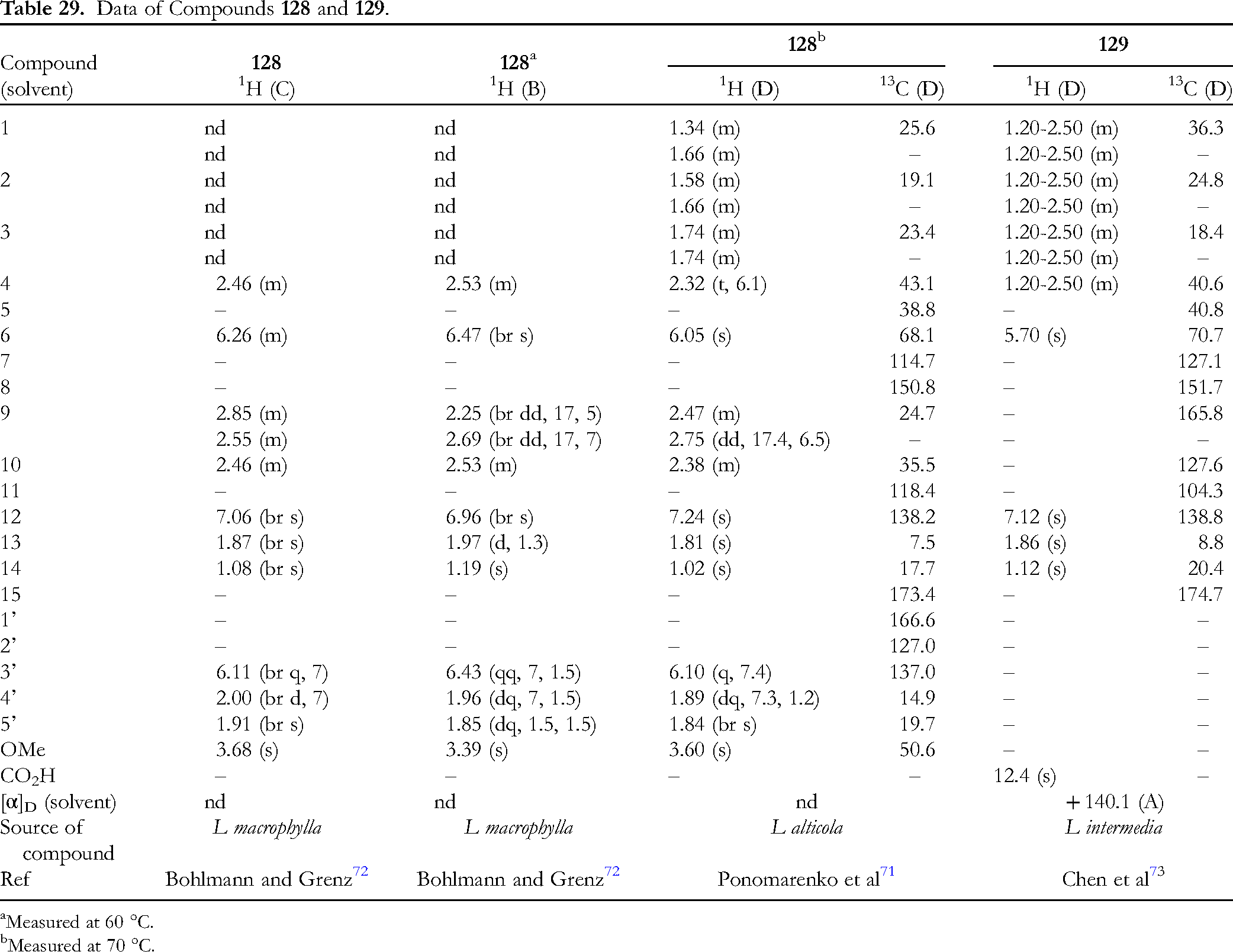
Table 29 lists the data of methyl ester 128 and acid 129. Compound 128 is the corresponding methyl ester of 127 (Figure 9).71,72 Compound 129 is an enol form of 9-one derivative.
73
It was not shown why 9-one takes an enol form only in this case.
73
Another possibility for 129 is a dimer bridging two identical units with an ether oxygen atom, although this author has no evidence.
Table 30 shows the data of 10βH-furanoeremophilan-15,6α-olide (130) (Figure 10). The NMR data in CDCl3,68,73-77 (CD3)2CO,
68
and (CD3)2SO
71
are provided. The chemical shifts of C-1 and C-11 slightly changed to the lower field, and C-3, C-7, and C-9 to the higher field in (CD3)2SO.
71
The ORD spectrum of compound 130 was measured.
68
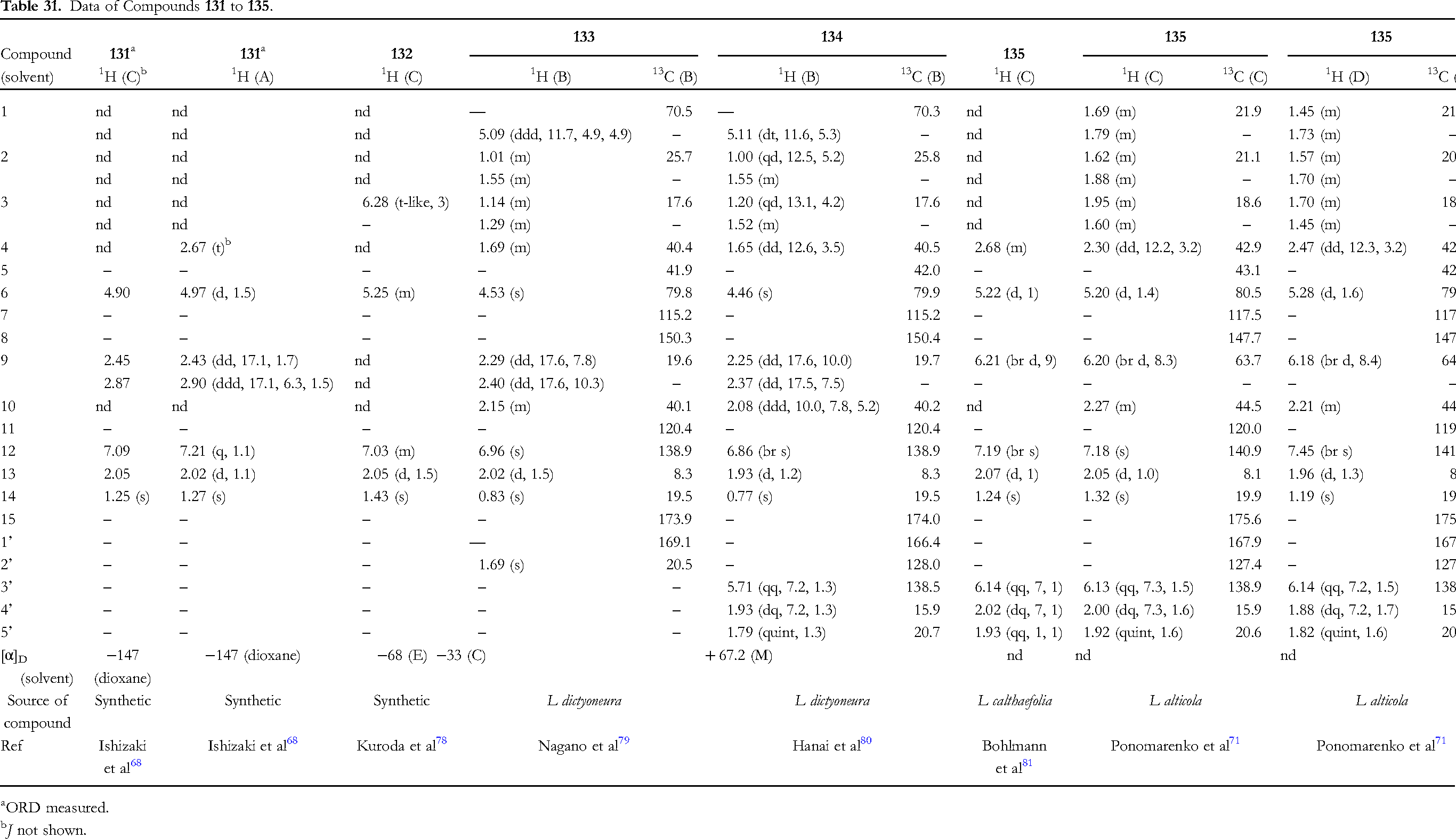
The data of five 10βH-furanoeremophilan-15,6α-olides, 131 to 135, are presented in Table 31 (Figure 10). Compound 132 is a C-3/C-4 dehydro derivative of compound 131.
78
Compounds 133 to 135 have 4αH, while 131 4βH. Compounds 133 and 134 adopt the steroid-like conformation fixed by the five-membered lactone, and C-1α substituents are oriented to the equatorial position.79,80 Thus, H-1β has one big and two small couplings. The configuration of C-4 of compounds 133 and 134 can isomerize to 4βH under basic conditions.
68
It is worth pointing out that the chemical shift values of H-4 of 135 (in CDCl3) is slightly different between Ponomarenko et al
71
and Bohlmann et al.
81
The only comparable figure is δ 2.26 (in CDCl3) of compound 130 (Table 30),
77
and thus δ 2.30 seems more accurate.
The data of two cyclic ether derivatives, 136 and 137, and two methoxy derivatives, 138 and 139 are listed in Table 32 (Figure 10). Compounds 136 and 137 are C-4 epimers.68,75 Compound 137 has been found in nature
82
and also synthesized from 130.
68
Compounds 138 and 139 are C-15 epimers.
83
The configurations at C-15 of compounds 138 and 139 were determined by the NOE experiment.
83
The H-6 of compound 137 resonates in the lower field than that of 136. It is interesting to note that there are significant differences in the chemical shifts of H-15 and OMe between compounds 138 and 139.
83
The data of four nonlinear type 10βH-furanoeremophilanes, 140 to 143, are given in Table 33 (Figure 10). Isoligularone (141), the 9-oxo derivative of compound 140,
84
was synthesized by thermal isomerization of ligularone (14).
85
The NMR data of isoligularone (141) were measured in CDCl3 and a mixture of CDCl3:(CD3)2CO (1:2) at different temperatures to study the preferred conformation.9,21 The CD spectra of isoligularone (141) were also measured at different temperatures.9,21 One more proton of 141 was recorded in ref.
84
Two hydroxy protons of compound 142 were detected in (CD3)2SO.
86
The H-12 of compound 143 appeared at the lower field than other furanoeremophilanes, due to the presence of a carboxy group attached to C-11.
87