Abstract
Background:
Exposure to ionizing radiation has undesirable effects leading to adverse physiological changes; therefore this study aims to investigate the radioprotective effects of Ashwagandha (Ag) on single and fractionated dose of gamma radiation induced-physiological alterations.
Method:
The study included 48 rats divided into six equal groups as follows (n = 8/group); normal control, Ag-treated group (300 mg/kg b.w orally), Single dose of radiation group (8Gy), group of rats treated with Ag and exposed to single dose of radiation, Rats exposed to fractionated radiation dose (2Gy every other day for 4 time), and group of rats treated with Ag and exposed to fractionated radiation. The gene expression of NFKB and MAPK in blood, the inflammatory mediators and apoptotic biomarkers were measured in serum of animals using ELISA method.
Results:
obtained data has shown that radiation exposure induces increased gene expression of NFKB and MAPK, levels of proinflammatory cytokines, including interleukin-1β (IL-1β) and IL-17 along with a significant reduction in the levels of anti-inflammatory cytokines; IL-4 and IL-10. Also, exposure either single or fractionated dose significantly stimulate apoptotic markers (caspase-3, Bax) accompanied by significant increase anti-apoptotic markers; BCL2 and survivin. However, administration of ashwagandha markedly suppressed inflammatory mediators and apoptotic biomarkers.
Conclusion:
The obtained data indicates that Ashwagandha might be a potent anti-inflammatory and anti-apoptotic agent, which can aid the prevention of chronic inflammatory conditions. Further trials are necessary to investigate its wider impact effects and elucidate the underlying mechanisms.
Introduction
Ionizing radiation generates free radicals, including reactive oxygen species (ROS) and reactive nitrogen species (RNS). High levels of radiation exposure can cause severe acute effects and lead to chronic inflammation, which may contribute to the development of various diseases. 1 Inflammation is a primary response to ionizing radiation and can damage multiple organs following exposure. This complex process involves vascular injury, leukocyte migration to the irradiated site, and the release of various immune mediators.2,3
Understanding how biological responses vary with different radiation exposure patterns is essential for refining therapeutic approaches. Many studies demonstrated the differing effects of acute radiation (a single high dose) and fractionated radiation (multiple lower doses) on apoptotic markers and inflammatory mediators in the blood. Acute radiation exposure is known to cause a rapid and substantial increase in caspase 3 activation, signifying the swift initiation of the apoptotic pathway and leading to extensive cell death in the affected tissue. 4
In contrast, fractionated radiation generally results in a more gradual rise in caspase-3 levels. The time intervals between doses allow for partial cellular repair and recovery, which may contribute to a lower overall rate of apoptosis throughout the treatment process. 5 Additionally, acute radiation exposure is typically associated with a significant upregulation of the pro-apoptotic protein Bax alongside a reduction in the anti-apoptotic protein BCL2. This imbalance favors apoptosis and increases cell mortality. 6
Fractionated radiation may not lead to drastic changes in Bax and BCL2 expression. Instead, it fosters a controlled response, enabling surviving cells to retain anti-apoptotic signals that support adaptation and persistence. 7
Previous studies suggest that acute radiation exposure reduces anti-inflammatory cytokines like IL-4 and IL-10, potentially weakening the body's ability to regulate inflammation. 8 However, fractionated radiation may help preserve some level of anti-inflammatory signaling, resulting in relatively higher IL-10 and IL-4 levels post-exposure, as the body has more time for recovery and regulatory adjustments between doses. 4 Fractionated treatments are typically used in clinical radiotherapy to maximize tumor control while minimizing damage to surrounding healthy tissue.
The response of normal tissue to ionizing radiation depends on the exposure dose. As radiation dosage increases, the likelihood of vascular injury, hypoxia, and necrosis also rises. These changes are closely linked to alterations in cytokine profiles. Higher doses of ionizing radiation (>1 Gy) typically result in necrosis rather than apoptosis, triggering a cascade of inflammatory responses. In contrast, lower doses (<1 Gy) may have anti-inflammatory effects by increasing apoptosis rates compared to necrosis.9,10
High-dose radiation exposure has been shown to cause extensive DNA damage and cellular apoptosis. It also leads to the release of danger-associated molecular patterns (DAMPs), including HMGB1, uric acid, and heat shock proteins (HSPs). 1 ionizing radiation activates inflammatory pathways, upregulating key mediators such as MAPKs, NF-κB, and COX-2. Consequently, inflammatory cytokines including IL-1, IL-6, IL-8, TNF, IL-33, and IFN-γ are released in greater quantities, creating a positive feedback loop that amplifies inflammation. 11 Additionally, persistent ROS and nitric oxide production exacerbate radiation-induced inflammation. If anti-inflammatory mechanisms fail to counteract this response, prolonged inflammation and oxidative stress can impair organ.11,12
Given these harmful biological effects, it is crucial to protect against radiation-induced. Antioxidants and free radical scavengers have been identified as potential radioprotectors, helping to prevent physiological harm before it occurs. 13 Studies suggest that radioprotective drugs limit radionuclide absorption by activating free radical scavenging pathways. 14 Numerous herbs, such as ashwagandha (Withania somnifera), have been documented to possess radio protective effects through mechanisms including free radical scavenging, inflammation reduction, DNA repair promotion, and hematopoietic cell regeneration. 15
Ashwagandha (Withania somnifera), a prominent herb in Ayurvedic medicine, is a natural bioactive compound isolated from Withania somnifera, has been identified as a promising anti-cancer and anti-inflammatory compound, and is renowned for its adaptogenic properties, aiding the body in managing stress and restoring balance. Its bioactive compounds, notably withanolides like withaferin A, contribute to its therapeutic effects. Ashwagandha and its active compounds, particularly withaferin A, offer a range of health benefits, including stress reduction, cognitive enhancement, hormonal balance, and anti-inflammatory effects. Withaferin A, a key withanolide in ashwagandha, exhibits significant anti-inflammatory and antioxidant properties. It affects various inflammatory signaling pathways, including NF-κB and Nrf2, and may reduce plasma C-reactive protein levels, contributing to its therapeutic potential. 16
Ashwagandha has been investigated as a potential remedy for various symptoms linked to inflammation, such as diabetes, cancer, neurological disorders, autoimmune diseases, pulmonary diseases, and cardiovascular ailments. 17 Prior clinical investigations have demonstrated that this plant suppresses inflammatory indicators, including cytokines (TNF-αand IL-6), nitric oxide, and ROS, thereby diminishing inflammation and modulating mitochondrial function and apoptosis. 18
So, the primary aim of this study is to investigate the radioprotective effects of Ashwagandha (Ag) on physiological alterations induced by single and fractionated doses of gamma radiation in a rat model. This study will specifically focus on 1- Evaluating the impact of Ashwagandha on blood inflammatory mediators, including: Pro-inflammatory cytokines: interleukin-1β (IL-1β) and interleukin-17 (IL-17). And Anti-inflammatory cytokines: interleukin-4 (IL-4) and interleukin-10 (IL-10). 2-Assessing the effects of Ashwagandha on blood apoptotic biomarkers, which include: Apoptotic markers: caspase-3 and Bax. Anti-apoptotic markers: Bcl-2 and survivin.
By measuring these mediators, the study aims to elucidate the potential therapeutic role of Ashwagandha in mitigating the adverse effects of ionizing radiation, thereby contributing to the understanding of its anti-inflammatory and anti-apoptotic properties. The outcomes of this research could provide insights into using Ashwagandha as a protective agent against chronic inflammatory conditions associated with radiation exposure.
Materials and Methods
Materials
Ashwagandha Root Extract (withanolides as active ingredient 1.5%) is a standardized herbal extract produced by GNC Herbal Plus® and distributed by General Nutrition Corporation Pittsburgh, PA 15,222. This herbal supplement comes in capsules, each containing 470 mg of the extract from these roots.
Radiation Facility
Rats were exposed to whole-body gamma irradiation using Canadian γ-cell-40 (137Cs) at NCRRT (Cairo, Egypt), and the dose rate during the experiment was 0.621 Gy/sec/min at the time of the investigation.
Animals
48 adult female Swiss albino rats (120-150 g) used in this study was obtained from the Experimental Animal House Laboratory of the National Center for Radiation Research and Technology. The animal care was consistent with the guidelines of Ethics by the Public Health Guide for the Care and Use of Laboratory Animals (National Research Council, 1996) and ARRIVE 2.0 guidelines 19 under the proper care and use of laboratory animals approved by the animal care committee of the National Center for Radiation Research and Technology, Cairo, Egypt.
Ethics Approval Statement
The care and use of experimental animals in this study were carried out in accordance with recommendations of the National Institute of Health (NIH no 85:23, revised 1996) and with guidelines adopted by the NCRRT Ethics Committee (Ref No: F 39 A/21).
Experimental Design
Forty-eight adult rats were divided into six equal groups (n = 8/group) as follows:
Control group (C): Normal rats received saline. Ashwagandha group (Ag): The animals were treated orally with Ag (300 mg/kg b.wt.) daily
20
for 15 consecutive days. Single radiation dose Group (Rs): The animals were exposed to a single dose of γ-radiation (8 Gy)
21
on the 15th day from the start of the experiment. Group (Ag + Rs): The animals were treated orally with Ag (300 mg/kg b.wt.) daily for 15 consecutive days and exposed to a single dose of 8 Gy. Fractionated radiation dose Group (Rf): The rats were exposed to a fractionated dose of γ-radiation (2 Gy every other day for four times)
22
starting from day 9. Group (Ag + Rf): The animals were treated orally with Ag (300 mg/kg b.wt. per day for 15 consecutive days) and exposed to a fractionated dose of γ-radiation (2 Gy every other day for four times) starting on day 9.
At the end of the experiment, the rats were anaesthetized and then sacrificed. Serum was collected for investigations.
RNA Extraction and Real-Time Quantitative Polymerase Chain Reaction
Total cellular RNA was extracted from blood using RNeasy® Mini kit according to manufacturer's instructions (Qiagen, Hilden, Germany). The concentration and purity of total RNA were assessed by measuring absorbance at 260 and 280 nm, respectively, in a spectrophotometer (Nano Drop 2000; Thermo Fisher Scientific, USA). First strand complementary DNA (cDNA) was synthesized using Thermo Scientific™ RevertAid™ First-Strand cDNA Synthesis Kit (Fermentus, Thermo Fisher Scientific Inc, Runcorn, UK). Real-time polymerase chain reaction (PCR) amplification and analysis were performed in an optical 96-well plate in ABI PRISM 7500 Fast Sequence Detection System Thermal Cycler (Applied Biosystems, Foster City, CA, USA) using Power SYBR® Green PCR Master Mix (Applied Biosystems). The amplification protocol consisted of 40 cycles (denaturation at 95 °C for 15 s, annealing at 55 °C for 20 s, and extension at 72 °C for 20 s). Primer used for STAT3, TNF-α and glyceraldehyde-3-phosphate dehydrogenase (GAPDH, endogenous reference gene) is represented in Table 1. The relative expression of selected genes was determined by the ΔΔCT method. 23
NFKB, MAPK & GAPDH Primers for RT-PCR.

Biochemical Assay
In serum, ELISA was measured using enzyme-linked immunosorbent assays both Caspase-3 (CASP3) (Rat) ELISA kit (catalog. number: E4592-100 | abID, Abcam, Cambridge, United Kingdom); Rat Survivin (SUR) ELISA kit (competitive ELISA)-catalog. Number: MBS728167, MyBioSource, San Diego, USA; Rat Bcl-2 (B-cell lymphoma/leukaemia 2) ELISA kit catalog. Number: MBS2515143,MyBioSource,SanDiego, USA; Rat TNF alpha ELISA kit (ab100785) Abcam, Cambridge, UK; Rat IL-10 ELISA kit (ab214566) Abcam, Cambridge, United Kingdom; Rat BAX (Bcl-2 Associated X Protein) ELISA kit catalog. number: MB S2512405, MyBioSource, San Diego, USA; Rat CRP (C-reactive protein) ELISA kit (ab256398), Abcam, Cambridge, UK; Rat IL-17A ELISA kit (ab214028) Abcam, Cambridge, United Kingdom; Rat IL-4 ELISA kit (ab100770) Abcam, Cambridge, UK; and Rat IL-1 beta ELISA kit (ab100768) Abcam, Cambridge, UK; according to the manufacturer's instructions via a microplate reader (DV990 BV 416; Gio.DE VITA and CO., Rome, Italy).
Annexin V Detection of Apoptosis by Flow Cytometry Analysis
For flow cytometry, blood cells were washed with cell staining buffer, centrifuged at 400 xg for 5 min at 4 °C, and the supernatant was discarded. The pellet was then resuspended in cell staining buffer, a cell count and viability analysis were performed using trypan blue and the bright line haemocytometer where the suspension was adjusted to a concentration of 1 × 106 cells/mL. Phosphatidylserine exposure on the outer leaflet of the plasma membrane was detected using the Annexin V-FITC/PI Apoptosis Detection Kit (BD pharmingin TM, BD Biosciences Co., USA; Number # 51-66121E), where cells were then resuspended in 1X Binding Buffer at a concentration of 1 × 106 cells/ml. To a 5 ml culture tube, 5 µl of FITC Annexin V and 5 µl PI were added to 100 µl of the solution (1 × 105 cells). The cells were gently vortexed and incubated for 15 min at 25 °C in the dark. Finally, 400 µl of 1X Binding Buffer was added to each tube and 10,000 cells were analyzed by flow cytometry within an hour on a FACSC-LSR (Becton and Dickinson Company) equipped with Cell Quest software. Three specimens were analyzed from each group. 24
Statistical Analysis
Statistics were performed using one-way analysis of variance (ANOVA) followed by Duncan's Multiple Range tests using the Statistical Package for the Social Sciences (SPSS) version 17.0 for Windows. The significance level was considered at P ˂ 0.05. G-power statistical program was used to calculate the sample size.
Results
Effect of radiation and/or Ashwagandha on the gene expression of NFKB and MAPK
A significant up regulation was demonstrated in the expression of NFKB and MAPK gene in irradiated group respectively, compared to not irradiated one (Control), where the results of the Rs and Rf groups showed a significant increase (p < 0.05) in NFKB, MAPK gene expression compared to the control value as shown in Figure 1 (a and b). In contrast, Ag treatment showed apronounced decrease (p < 0.05) in NFKB, MAPK gene expression in the Ag + Rs group and Ag + Rf group compared to the groups of Rs and Rf, respectively (Figure 1 a, b)

Effect of Ashwagandha and/or Radiation on Gene Expression of NFKB, MAPK. Each Value Represents Mean ± SE (n = 5). Columns Denoted with “a” Significant from C, “b”: Significant from Ag, “c” Significant from Rs, “d” Significant from Ag + Rs, “e” Significant from Rf and “f” Significant from Ag + Rf, at (P < 0.05).
Effect of Radiation and/or Ashwagandha on Inflammatory Mediators
The possible role of pro-inflammatory factors was evaluated in this study, as shown in Figure 2 (a, b and c), where the results of the Rs and Rf groups showed a significant increase (p < 0.05) in levels of IL-IB, IL-17 and TNF compared to the control value, In contrast, Ag treatment showed a significant decrease (p < 0.05) in the Ag + Rs group and Ag + Rf group compared to the groups of Rs and Rf, respectively (Figure 1a, b and e), in addition to a pronounced reduction in the inflammatory index CRP (d). In addition, no significant change was observed in Ag + Rs and Ag + Rf groups When compared to each other.
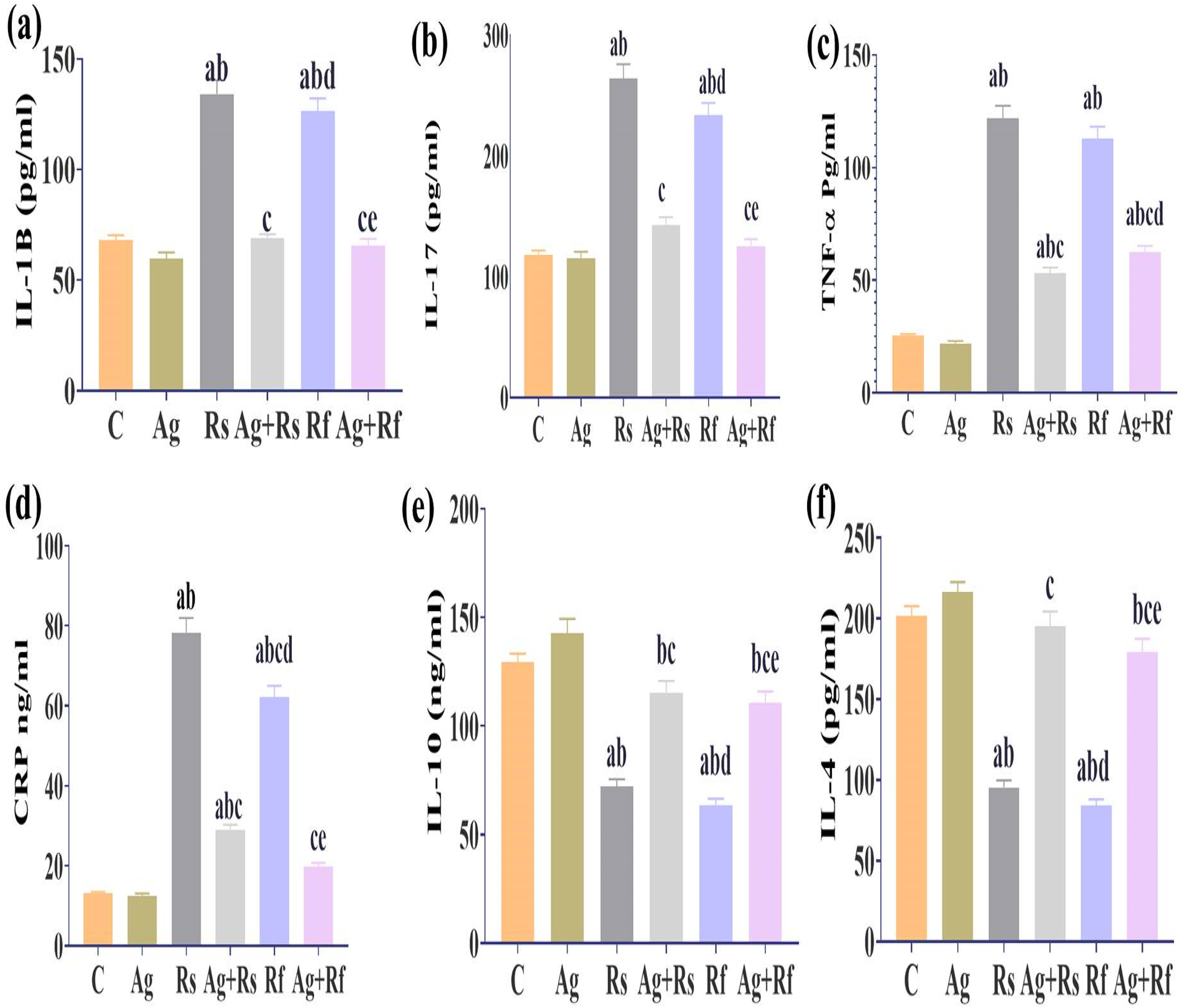
Effect of Ashwagandha and/or Radiation on Serum Inflammatory Mediators. Each Value Represents Mean ± SE (n = 5). Columns Denoted with “a” Significant from C, “b״: Significant from Ag, “c” Significant from Rs, “d” Significant from Ag + Rs, “e” Significant from Rf and “f” Significant from Ag + Rf, at (P < 0.05).
Whereas, the results of the irradiated groups showed significant changes in anti-inflammatory cytokines, evidenced by a significant decrease (P < 0.05) in IL-10 and IL-4 levels in the Rs and Rf groups compared to the control group. Moreover, Ag treatment significantly attenuated the decrease (P < 0.05) in IL-10 and IL-4 levels in the Ag + Rs and Ag + Rf groups compared to the Rs and Rf groups, respectively. However, when compared to each other, there were no significant changes in IL-10 and IL-4 levels in the Ag + Rs and Ag + Rf groups (Figure c, d).
Effect of Radiation and/or Ashwagandha on Apoptotic Mediators
Figure 3 revealed that no statistically significant change was observed in the apoptosis level of the treated group (Ag) compared to the control group. However, the irradiated groups exhibited a significant (p < 0.05) increase in the levels of both caspase-3 and Bax, accompanied by a decrease in BCL-2 levels and survivin rates in Rs and Rf groups compared to the control group. Further, a significant difference was observed between the Rs and Rf groups in caspase and Bax levels when compared to each other Figure 3 (g, h). On the other hand, While, Ashwagandha treatment showed significant modifications in the results, which reveal a considerable reduction in apoptotic markers (caspase-3, Bax) accompanied by an increase of anti-apoptotic markers (Bcl-2) compared to the Rs and Rf group., as well as improved survival rates (Figure 3g, h, i, and j).

Effect of Ashwagandha and/or Irradiation on the Apoptotic Mediator in Serum. Each Value Represents Mean ± SEM (n = 5). Columns Denoted with “a” Significant from C, “b”: Significant from Ag, “c” Significant from Rs, “d” Significant from Ag + Rs, “e” Significant from Rf, and “f” Significant from Ag + Rf, at (P < 0.05).

(A): A Representative from Each Group, (C): Control Group, (Ag): Ashwagandha Group, (Rs): Group Exposed to Single Radiation Dose (8 Gy), (Rf): Group Exposed to Fractionated Dose (2Gy/Time: to Total 8 Gy). The Upper Right and Upper Left Quadrants Represent the Late Apoptotic and Necrotic Cells, Respectively. the Lower Left and Lower Right Quadrants Represent the Viable and Early Apoptotic Cells, Respectively in Percent of the Total Gated Cells.
Apoptosis detection by flow cytometry with Annexin V-FITC conjugated with PI staining.
Apoptosis was detected by initially staining the cells with Annexin V and propidium Iodide (PI) solution followed by flow cytometry analysis. 22 Annexin V FLUOS staining kit (F. Hoffmann-La Roche, catalogue number: 11858777001). The flow cytometric dot plot chart illustrates the different stages of the apoptotic cascade, helping to differentiate between necrotic and apoptotic cells based on their binding to annexin V (x-axis) and propidium iodide (PI) (y-axis). Cells that have undergone necrosis are indicated by double positive staining in the upper left quadrant, while apoptotic cells are identified as annexin V positive in the lower right quadrant.
Using this methodology, the Rf group's apoptosis rate was significantly higher than all other groups. Figure 4 (A) displays representative results from each group, and Figure 4 (B) presents the mean results from three samples per group. The proportion of cells in late-stage apoptosis increased from 20.7% in the Rs group to 26.4% in the Rf group. Conversely, concurrent treatment with ashwagandha reduced this percentage to 13% in the Ag + Rs group and 16.3% in the Ag + Rf group. Additionally, the rate of necrotic cells increased following radiation exposure, rising from 5.9% in the Rs group to 19% in the Rf group. However, simultaneous treatment with ashwagandha lowered the percentage of necrotic cells to 3% in the Ag + Rs group and 0.8% in the Ag + Rf group, as shown in Figure 4 (a, b). Notably, the greatest percentage of viable cells and the lowest rates of late-stage apoptosis and necrosis were observed in the ashwagandha-treated group for both single and fractionated radiation doses single and fractionated radiation doses.

(B): The Means of Three Samples from Each Group, (C): Control Group, (Ag): Ashwagandha Group, (Rs): Group Exposed to Single Radiation Dose (8 Gy), (Rf): Group Exposed to Fractionated Dose (2Gy/time: to Total 8 Gy). Values are the mean ± SEM (n = 3).
Discussion
Radiotherapy is involved in 50% of all cancer treatments and 40% of cancer cures. Most of these treatments are delivered in fractions of equal doses of radiation (Fractional Equivalent Dosing (FED)) in days to weeks. 25 The concept of fractionation regimens originally comes from a series of extensive radiobiological studies conducted in France in 1910s1930s. As one of the pioneering studies, Claudius Regaud reported in 1911 that sterilization of testes can be achieved with minimal skin damage when fractionated. 26 This fractionation schedule is commonly used in the treatment of early-stage non-small cell lung cancer (NSCLC), breast and prostate cancer to improve outcomes while minimizing side effects. The fractionated approach allows for the repair of normal tissues between treatments while still delivering a sufficiently high dose to the tumor. Normal cells generally have better recovery mechanisms than cancer cells, which allows for increased tumor control probability (TCP) while reducing the probability of normal tissue complications (NTCP). In addition, fractionated regimens can help achieve gradual and sustained pain relief for patients with bone metastases, improving quality of life. Moreover, fractionation minimizes acute side effects compared to single high doses, making it more tolerable for patients, especially those with multiple comorbidities. In the study by Peters et al the use of 2 Gy × 4 demonstrated efficacy in improving local control rates for NSCLC patients, enhancing the therapeutic outcome while maintaining patient quality of life. This supplementation of ongoing supportive care is vital in comprehensive cancer management. 27
A prominent example of a single 8 Gray (Gy) dose used in human radiotherapy is in the context of certain palliative treatments for metastatic bone pain or spinal cord compression. In the manuscript puplished in Radiotherapy oncology journal at 1999 found that a single fraction of 8 Gy is as safe and effective as a multifraction regimen for the palliation of metastatic bone pain for at least 12 months. 28 The greater convenience and lower cost make 8 Gy single fractions the treatment of choice for the majority of patients. The study of Shuja et al was to determine the efficacy of single fraction palliative radiotherapy for painful bone metastases. 29 Worldwide different radiotherapy schedules are being used for BM. A single administration of 8 Gy, often delivered in a single session. Single fractions are designed for fast symptom relief, particularly effective for patients with metastatic bone pain. The use of a single 8 Gy dose can lead to quicker clinical benefits, reduces the number of hospital visits required from patients, which is particularly beneficial for those with limited life expectancy or mobility issues. Palliative care often focuses on easing suffering, and fewer visits can reduce treatment burden.The study by Hartsell et al illustrates the effectiveness of single 8 Gy fractions in providing pain relief for patients with bone metastases. 30 The results indicated comparable outcomes in pain management with minimal side effects, demonstrating the role of such regimens in palliative care. In conclusion, both fractionated (2 Gy × 4) and single dose (8 Gy) radiotherapy play critical roles in the management of cancer, depending on treatment intent and patient needs. Fractionated doses are generally more favorable for curative treatments due to enhanced therapeutic benefits and safety profiles, while single doses can provide quick relief in palliative contexts. The choice of regimen underscores the importance of individualized patient care in oncology, balancing efficacy, safety, and quality of life.
Ionizing radiation (IR) poses numerous dangers that impact the body's physiology and disrupt the equilibrium of inflammatory mediators, resulting in a cascade of inflammatory reactions, that regulate cell cycle arrest, programed cell death, and ultimately, immunological abnormalities. Additionally, free radicals generated by ionizing radiation impair biological components, disrupt the antioxidant system and may induce genetic mutations that result in cancer, necrosis, or apoptosis.31,32 In this study, we investigated the impact of radiation and Ashwagandha (Withania somnifera) on the gene expression of NFKB and MAPK, two critical signaling pathways involved in inflammatory responses and cellular stress. Our results indicate a significant upregulation of both NFKB and MAPK gene expression in the irradiated groups (Rs and Rf) compared to the control group (Figure 1a and b). This finding is consistent with previous literature that has reported radiation exposure as a potent activator of these pathways, leading to increased cellular stress responses.33,34 The substantial rise in NFKB and MAPK expression following exposure to single and fractionated doses of radiation underscores the role of these genes in mediating the biological effects of ionizing radiation. NFKB, in particular, is crucial for regulating inflammatory responses, and its activation serves as a cellular mechanism to combat the consequences of damage incurred from radiation. 35 Similarly, the MAPK pathway is recognized for its involvement in cellular proliferation and apoptosis, suggesting that the observed upregulation in these genes may reflect an adaptive response to mitigate radiation-induced damage. 36
Interestingly, our results demonstrated that Ashwagandha treatment significantly decreased the expression of NFKB and MAPK genes in the Ag + Rs and Ag + Rf groups compared to the corresponding irradiated groups. This suggests that Ashwagandha may exert a protective effect against radiation-induced gene expression changes. Previous studies have highlighted the potential of Ashwagandha to exhibit anti-inflammatory and radioprotective properties through various mechanisms, including the modulation of oxidative stress and inflammatory cytokine production.37,38 The observed downregulation of NFKB and MAPK in the presence of Ashwagandha may indicate its ability to inhibit the pathways activated by radiation, thereby promoting cellular homeostasis. The phytochemicals present in Ashwagandha, such as withanolides, might play a significant role in this modulation by interfering with the signaling cascades associated with these pathways. 39 Overall, our findings suggest that the incorporation of Ashwagandha could serve as a therapeutic strategy to mitigate the adverse effects of radiation by downregulating crucial signaling pathways like NFKB and MAPK.
The current findings indicate that exposure to ionizing radiation disrupts the balance of pro-inflammatory and anti-inflammatory cytokines, evidenced by elevated levels of pro-inflammatory cytokines (IL-1B, IL-17, and TNF) (depending on the different exposure patterns) Figure 2 (a, b, c) respectively, and reduced levels of anti-inflammatory cytokines (IL-10, IL-4) Figure 1 (e, f) respectively. This aligns with the studies by40,41 and may be attributed to excessive ROS generation induced by free radicals, resulting in oxidative modification of biomolecules and impairment of the antioxidant defense system. 1 Additionally, it may be ascribed to the release of inflammatory mediators (MAPKs, NF-κB, and COX-2) that initiate the production of inflammatory cytokines (IL-1B, TNF, and IFN-γ).11,12
Furthermore, 42 shows that ionizing radiation impairs cell junctions by activating the inflammatory NF-κB signaling pathway, resulting in heightened oxidative stress and elevated levels of inflammatory cytokines. When anti-inflammatory systems are ineffective in inhibiting the inflammatory response, free radicals impair normal organ function, resulting in chronic inflammation. We note that the results of our study are consistent with other research indicating the suppression of pro-inflammatory cytokine release, specifically IL-1β, IL-17, and TNF-α levels. 43
The data revealed that both acute radiation (Rs) and fractionated radiation (Rf) significantly increase the levels of pro-inflammatory cytokines IL-1β, IL-17, and TNF-α compared to the control group (p < 0.05). This suggests that radiation exposure triggers a strong inflammatory response. The observed increase in these cytokines aligns with previous studies, 44 which have reported that radiation can create an inflammatory microenvironment due to oxidative stress and cellular damage. The rapid activation of pro-inflammatory pathways serves as a biological mechanism for tissue repair; however, if not properly regulated, it may also lead to detrimental effects.
Fractionated radiation appears to maintain certain anti-inflammatory signals, as the recovery period between exposures allows for relatively elevated levels of IL-10 and IL-4. 4 This aligns with the findings of the present study, which observed a notable reduction in the anti-inflammatory cytokines IL-10 and IL-4, as shown in Figure 1(c) and (b), in both radiation groups (Rs and Rf) compared to control levels (p < 0.05). Since IL-10 plays a crucial role in regulating inflammation, its decline indicates compromised inflammatory control. Likewise, the downregulation of IL-4, which facilitates anti-inflammatory pathways and aids in Th2 cell differentiation, suggests an overall shift towards heightened pro-inflammatory activity in response to radiation-induced stress.
Exposure to various radiation doses was also found to increase pro-apoptotic markers (caspase-3 and Bax) while decreasing anti-apoptotic markers (Bcl-2). This finding is in line with research by, 45 which demonstrated that microwave radiation elevates ROS levels and triggers oxidative stress, causing neuroinflammation in the hippocampus via oxidative stress-induced apoptosis. Given these harmful effects, the use of natural compounds to mitigate radiation-induced damage has gained attention.
Many studies have highlighted the protective role of Ashwagandha in enhancing antioxidant levels. The pharmacological properties of withanolides in Ashwagandha have been shown to suppress neutrophil infiltration and reduce inflammatory cytokine release.46,47
The impact of ashwagandha on radiation-induced apoptotic alterations was examined. It was found that it increased pro-apoptotic markers (caspase-3 and Bax) while downregulating the anti-apoptotic protein Bcl-2 in groups exposed to radiation. Also, Ashwagandha administration significantly reduced caspase activity and altered the expression of Bcl-2 and Bax compared to irradiated groups, whether single or fractionated doses.21,48
Radiation exposure triggers an increase in pro-inflammatory cytokines such as tumor necrosis factor-alpha (TNF-α), interleukin beta (IL-1β), and interleukin 6 (IL-6), leading to severe tissue damage. Research suggests that ashwagandha, particularly its bioactive compound withaferin A, can mitigate this effect by regulating the NF-κB signaling pathway. By inhibiting NF-κB translocation to the nucleus, ashwagandha reduces cytokine gene expression, thereby exerting anti-inflammatory effects. 49
Ashwagandha belongs to a group of herbs known as adaptogens, which are natural substances that help the body adapt to physical and mental stress. Adaptogens work by (a) stabilizing physiological functions, (b) enhancing the body's resilience to future stressors, and (c) improving overall performance and well-being. This category of herbs includes several well-known plants, such as Rhodiola, ginseng, Schisandra, and maca. 50
Traditionally, adaptogens have been used to support stress management, cognitive health, and adrenal function, particularly in regulating the hypothalamic-pituitary-adrenal (HPA) axis. In recent years, they have also gained attention in the field of sports nutrition, where they are incorporated into supplements aimed at enhancing physical endurance and recovery. While some studies suggest promising benefits,51,52 the findings remain inconsistent. As a result, further research is necessary to fully understand their effectiveness as ergogenic aids.
Ashwagandha treatment also inhibited the mRNA expression of inflammatory cytokines (TNF-α, IL-1β, and IL-12) while enhancing the expression of anti-inflammatory cytokines in human keratinocyte (HaCaT) cells. This effect is likely due to Ashwagandha's ability to suppress the NF-κB and mitogen-activated protein kinase (MAPK) pathways, regulating cytokine expression 53 our results are consistent with these study.
WA's (Withania somnifera) anti-inflammatory effects may also be linked to its interaction with peroxisome proliferator-activated receptors (PPARs), which are essential for lipid metabolism and belong to the nuclear receptor family. These transcription factors regulate gene expression upon ligand activation. 54 PPARs help mitigate NF-κB-driven inflammation by inhibiting both NF-κB and activator protein-1 (AP-1). Research indicates that WA influences PPAR expression; however, findings on the direction of this regulation have been inconsistent. An in vitro study using 3T3-L1 adipocytes showed that WA reduces PPAR expression through MAPK phosphorylation, whereas an in vivo study on mice with high-fat diet-induced obesity observed increased hepatic PPAR mRNA expression.16,55
Significant, Ashwagandha treatment demonstrates significant enhancements in inflammatory responses relative to the irradiated group, as evidenced by a marked reduction in pro-inflammatory cytokine levels Figure 2 (a, b, c,), an elevation in anti-inflammatory cytokine levels Figure 1 (e, f), and diminished activity of the inflammatory marker C-reactive protein Figure 1 (d). This aligns with the findings of56,57 and can be attributed to the antioxidant and anti-inflammatory properties of Ashwagandha, which mitigates the formation of ROS, prevents the accumulation of cellular molecules, and reduces oxidative stress, thereby alleviating inflammation. 34
A study by 58 has shown that Ashwagandha reduces the expression of pro-inflammatory cytokines TNF-α and NF-κB while increasing levels of the anti-inflammatory cytokine IL-10, supporting our findings. Additionally, W. somnifera extracts have been reported to influence multiple immune system pathways in rats. 59 Several studies have linked Ashwagandha's anti-inflammatory effects to its ability to inhibit key components of inflammatory signaling pathways, including NF-κB, kinase signaling, and heat shock proteins (HSPs). The destabilization of HSPs is associated with reduced oxidative stress and inflammation.60–63
Several studies have linked Ashwagandha's anti-inflammatory effects to its ability to inhibit key components of inflammatory signaling pathways, including NF-κB, kinase signaling, and heat shock proteins (HSPs). The destabilization of HSPs is associated with reduced oxidative stress and inflammation. 64 It can also modulates apoptosis and reduces inflammation by inhibiting inflammatory markers, including cytokines (TNF-α, IL-6), nitric oxide (NO), and ROS. These studies support the results of the current study, demonstrating the role of ashwagandha in inhibiting indicators of apoptosis as shown in the data obtained.
Ashwagandha has also been found to significantly reduce cardiac damage caused by isoproterenol, likely due to its anti-apoptotic and anti-inflammatory properties, as well as its ability to restore oxidative balance.65,66 Furthermore, 31 demonstrated that Ashwagandha reduces inflammation in hippocampal neurons by decreasing the expression of caspase-3 and caspase-7. These findings are in agreement with our study's results, further supporting Ashwagandha's role in mitigating radiation-induced inflammation and apoptosis.
Therefore, radiation countermeasure agents should be used to reduce the hazardous effects of IR. Radioprotectors are various agents that act via different mechanisms involving: scavenging of free radicals and ROS; improvement of the DNA repair process; synchronizing of cells; enhancing antioxidant and redox-sensitive genes; modulating cytokines and growth factors; inhibiting apoptosis; repurposing of drugs; and tissue regeneration. Scavenging of free radicals is the most common mechanism of radioprotection, whereas the alteration of growth factors, cytokines and redox genes appears to be an effective strategy. 67 Our previous work stated that fucoxanthine have a noticeable radioprotective action mediated through its regulatory effect on the apelin-13/APJ/NF-κB signaling pathway which attributed to its antioxidant and anti-inflammatory activity that was reflected in different physiological processes. It could be recommended to use FX in cases of radiation exposure to protect normal tissues. 68 What is more, Sirtuin (ultra pure trans-Resveratrol; t-Res) had a radio-protective effect via its antioxidant and anti-inflammatory effects on liver and kidney of rats subjected to a harmful whole body single or a fractionated radiation at a dose of 6 Gy. This is attributed to its ability to down-regulate the hypoxia factor (HIF-1α) and increase Nrf-2 gene expression, which is associated with a decline in lipid peroxidation (MDA) and enhancement in antioxidant enzyme (CAT), which supports its ability to curb inflammatory responses in vulnerable tissues. 40 Moreover, Pomegranate peel extract sensitizes hepatocellular carcinoma cells to ionizing radiation, induces apoptosis and inhibits MAPK, JAK/STAT3, β-Catenin/NOTCH and SOCS3 signaling. 69
Ashwagandha has garnered attention for its radioprotective properties, particularly in mitigating the adverse effects of radiation exposure. The following recent studies provide a stronger theoretical foundation for its mechanisms of action. Where Verma and shukla, 70 highlights Ashwagandha's ability to scavenge free radicals and enhance the expression of antioxidant enzymes like superoxide dismutase (SOD), catalase, and glutathione peroxidase. These mechanisms play a vital role in protecting cellular components from oxidative damage caused by radiation. And the study of Bhattacharya et al, 71 demonstrates that Ashwagandha significantly reduces the levels of pro-inflammatory cytokines (IL-1β, TNF-α) and promotes anti-inflammatory cytokines (IL-10) following radiation exposure. This modulation of the inflammatory response helps in minimizing tissue damage and promoting recovery. More over the study of Gupta et al, concludes that Ashwagandha modulates apoptotic pathways by downregulating pro-apoptotic proteins (Bax, caspase-3) and upregulating anti-apoptotic proteins (Bcl-2, survivin). 72 This regulatory effect enhances cell survival in healthy tissues during radiation therapy. In addition Azab et al found that Withania somnifera (Ashwagandha) root extract counteract acute and chronic impact of γ-radiation on liver and spleen of rats, Ashwagandha could exerts radio-protective influences because of its antioxidants and anti-inflammatory capabilities. 21 These studies collectively underscore the multifaceted radioprotective mechanisms of Ashwagandha, including its roles in antioxidant defense, modulation of inflammatory responses, regulation of apoptosis, enhancement of DNA repair mechanisms, and activation of cellular stress responses. Incorporating these insights into our understanding of Ashwagandha's protective effects provides a robust theoretical foundation for our research.
Study Limitations
While this study presents valuable insights into the radioprotective effects of Ashwagandha, several limitations should be acknowledged: In vitro versus In vivo Results: The study primarily employed in vitro experimental models, which may not fully replicate the complex biological interactions present in live organisms. Future research including in vivo studies will be necessary to validate the findings and ascertain the clinical relevance of Ashwagandha's protective effects against radiation. Further research is necessary to elucidate the precise molecular mechanisms underlying these effects and to explore the potential clinical applications of Ashwagandha in radiation therapy contexts. Dosage and Administration: The specific dosages and administration routes of Ashwagandha used in this study may not perfectly align with those used in human clinical settings. The rationale for the 300 mg/kg dosage of Ashwagandha used in our study lacks pharmacokinetic data. This absence of quantitative data may limit the ability to establish direct correlations between Withaferin A concentrations and the observed biological effects. Limited Sample Size: The experiments conducted in this study utilized a relatively limited number of cell types and concentrations. Expanding the variety of cell lines and treatment concentrations in future studies could provide a more comprehensive understanding of the herb's potential effects and mechanisms. Absence of Long-Term Follow-Up: The study assessed the immediate effects of Ashwagandha on radiation-induced cell damage without considering long-term outcomes. Additional research examining the long-term impacts and potential cumulative effects of Ashwagandha exposure after radiation treatment would enhance our understanding of its overall efficacy. Control Group Limitations: Although radiation groups serve as positive controls, the absence of a direct comparison with other known radioprotective agents limits the ability to evaluate Ashwagandha's effectiveness relative to these alternatives. Including additional control groups in future studies could provide context for interpreting the results.
By acknowledging these limitations, we hope to pave the way for future research that can address these issues and further elucidate the role of Ashwagandha in radiation protection.
Conclusion
Acute and fractionated radiation exposure triggers distinct cellular death and inflammatory pathways. Acute radiation often leads to a sharp rise in cell death and pro-inflammatory mediators, whereas fractionated radiation allows partial recovery and modulation of these responses. Recognizing these differences is crucial for refining radiation therapy and minimizing its adverse effects.
Ashwagandha has potent anti-inflammatory and anti-apoptotic properties which help counteract radiation-induced cellular damage. As well as data obtained suggest that ashwagandha may enhance specific biomarkers, regulate certain physiological functions, and its ability to modulate NFKB and MAPK gene expression, cytokine levels and suppress cell death highlighting its potential as a radioprotective agent. However, additional clinical studies are needed to evaluate its efficacy and optimize its therapeutic application for radiation-exposed individuals.
Footnotes
Acknowledgements
I would like to express my gratitude to Dr Monda Badawy, National center for radiation Research and Technology, Egyptian Atomic Energy Authority, for the performance of Flow Cytometer investigation, which enriching this study.
Ethical Considerations
This study was done in accordance with guidelines adopted by the NCRRT Ethics Committee (Ref No: 39 A/21).
Author Contributions
M.K.A, N.M.T designed the study. N.M.E and R.E.M carried out the practical work. G.R.A performed the data analysis and wrote the draft of the manuscript. K.S.A, M.K.A, N.M.T critically read and revised the manuscript. All authors approved the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable
