Abstract
Objectives
This study aimed to investigate the modulatory effects of Tetracarpidium conophorum (African walnut) nuts on lipogenesis in adipocytes and hepatocytes by targeting the SREBP-1/ACCA-1/FASN signaling axis, which plays a pivotal role in obesity-associated lipid metabolism.
Methods
Obesity was induced in rats over 36 weeks, after which groups were treated with whole extract (WE), ethyl acetate fraction (EA), residue (RES) of T. conophorum nuts, or Orlistat (5.14 mg/kg). Post-treatment, hepatic and adipose tissues were collected and analyzed for gene expression levels of SREBP-1, ACCA-1, and FASN using PCR. Additionally, GC-MS-identified compounds from the nut extracts were subjected to molecular docking against the target proteins, and their drug-likeness and ADMET profiles were evaluated.
Results
Treatment with WE, EA, and RES significantly downregulated the expression of SREBP-1 and FASN in both hepatic and adipose tissues compared to the obese control. ACCA-1 expression in the adipose tissue was not significantly affected, but hepatic overexpression was significantly attenuated by WE (p < 0.05), EA (p < 0.01), and RES (p < 0.01). Molecular docking revealed strong binding affinities and stable interactions between bioactive compounds and the target proteins. Notably, 6-Isopropenyl-4,8a-dimethyl-4a,5,6,7,8,8a-hexahydro-1H-naphthalen-2-one exhibited the strongest binding affinities to SREBP-1 (−7.7 kcal/mol), ACCA-1 (−7.2 kcal/mol), and FASN (−7.8 kcal/mol), outperforming Orlistat (−5.9 to −6.2 kcal/mol). Drug-likeness assessments and ADMET predictions supported the therapeutic potential of the compounds.
Conclusion
T. conophorum nuts demonstrate significant anti-obesity potential through the inhibition of hepatic and adipose lipogenesis via downregulation of the SREBP-1/ACCA-1/FASN signaling pathway, with specific bioactive compounds showing promising molecular interactions and pharmacokinetic properties.
This is a visual representation of the abstract.
Keywords
Introduction
African black walnut (Tetracarpidium conophorum or Plukenetia conopohor), is a tree nut produced by the plant family, Euphorbiaceae.1,2 This plant is cultivated mainly for its nuts which are usually cooked, 3 roasted or dried 4 and consumed as snacks 3 as they are a rich source of minerals, particularly manganese and the B vitamins. 5 The nuts also possess impressive medicinal properties. 5 Studies have shown that African walnut (Tetracarpidium conophorum) possess antioxidant and anti-inflammatory properties which could be beneficial in managing obesity and related metabolic conditions. 6 In our just concluded study, it was shown that the nuts exerted positive modulation at the molecular level, on the low-grade inflammatory process associated with obesity. 7 Preclinical studies suggest that T. conophorum nuts may reduce lipid accumulation and improve lipid profiles in animal models of metabolic disease. Nwaichi et al reported in 2017 that African walnuts reduced weight gain, plasma triglycerides, total cholesterol, LDL-cholesterol, VLDL-cholesterol, and atherogenic indices in hyperlipidemic rat models in a dose-dependent manner. 8 Similarly, Uti et al 9 demonstrated that T. conophorum nuts lowered total lipid, triglyceride, and cholesterol levels in obese rat models, preventing hepatic lipid accumulation and indicating potential lipid-lowering properties that could mitigate obesity-related metabolic effects.
In addition, it has been shown to reduce hyperglycemia and protect against diabetes-induced oxidative liver damage.6–10 The African walnuts have been strongly implicated in the promotion of weight loss,2,9,11 In an earlier study by Uti and colleagues, the African walnuts were clearly shown to attenuate body weight gain and fat storage, both in the adipose and ectopic regions, in monosodium glutamate obese rat models,9,11 hence a potential natural resource for sourcing anti-obesity therapeutics. However, the molecular mechanism(s) underlying this reported anti-obesity action is yet unknown. Thus, the current study sought to know the molecular actions of the nuts: How the nuts are able to attenuate fat accumulation both in the primary fat depot, the adipose, and in an ectopic depot like the liver. In doing that, we studied the interaction of extracts from the nuts with key molecular targets of the lipogenic pathway in vivo and in silico.
Obesity characterized by a BMI greater than or equal to 25 kg/m2 12 is one of the most serious public health problems in the world. According to reports by the World Health Organization (WHO) in 2024 the occurrence of obesity since 1990 has almost tripled in adults and quadrupled in adolescents, contributing substantially to overall illness and mortality. 13 In addition to its impact on public health, obesity is a major risk factor for the development of metabolic syndrome and its components. Obesity is also associated with increased oxidative stress, 14 and oxidative stress plays a role in the pathogenesis of numerous diseases, such as diabetes mellitus and cancer. 15 Hypertension, cardiovascular diseases, stroke etc are among the common comorbidities linked to obesity, contributing further to adverse health outcomes.16,17 Moreover, obesity may contribute to the modification of plasma lipoproteins, enhancing their atherogenic potential. 18
Given these risks, understanding the underlying causes of obesity is critical. The development of obesity is engendered by an imbalance between energy intake and energy expenditure.19,20 This delicate balance is tightly regulated and maintained by a complex process where the feeding regulatory centers of the hypothalamus exert control on peripheral fat storage and mobilization. Therefore, any dys-regulation in the expression or function of the genes that encode the molecular components of this control system or dysfunction in their gene products can contribute to the onset of obesity and its related disorders.21,22
Consequently, key genes involved in this complex regulatory process such as the transcription factor, SREBP-1 (Sterol regulatory element-binding protein – 1) were studied. SREBP-1 is a transcription protein that directly regulates the expression of pacesetter enzymes of fatty acid synthesis 23 and storage, namely fatty acid synthase (FASN) and acetyl CoA carboxylase (ACCA-1). Lipid metabolism, the body's process of synthesizing, storing, and breaking down fats and lipids, depends on SREBP-1, FASN, and ACCA-1. 24 The transcription factor SREBP-1 stimulates the expression of enzymes involved in the synthesis of fatty acids and cholesterol. 25 FASN is a multi-enzyme complex which catalyzes the de novo synthesis of fatty acids 26 whereas ACCA-1 catalyzes the committed step in the synthesis of fatty acids. 27 FASN is considered a candidate gene for determining body fat. 28 Its high expression in lipogenic tissues and its role in adipocyte differentiation and lipid accumulation make it a promising target for therapeutic applications. 29 Over-expression of this gene has been observed in the adipose tissue of obese rats and is associated with obesity and type II diabetes mellitus (T2DM). 30 Furthermore, inhibiting FASN activity has been shown to block adipocyte differentiation and reduce the number of adipocytes. 31 High expression of ACCA-1 has been linked to obesity. As a key regulator of lipogenesis, increased ACCA-1 activity contributes to obesity in mice. Conversely, inhibiting ACCA-1 has been shown to counteract obesity. 32 As a key regulator of lipogenesis and a rate-limiting enzyme in fatty acid biosynthesis, ACCA-1 has been targeted in obesity research. Its role in limiting the synthesis of fatty acids makes it a promising candidate for anti-obesity drug development. Notably, reduced hepatic ACCA-1 mRNA expression has been associated with lower hypertriglyceridemia, decreased hepatic triacylglycerol content, and improved insulin sensitivity. 32 Accordingly, the expressions of these genes were evaluated after a 6-week exposure of obese rats to extracts and fractions of the African walnuts and correlated with in silico interaction of their protein products with GC-MS ligands in the African walnut extracts. As a conventional nomenclature, the genes encoding the transcription factors shall be referred to as upstream genes and those whose expression they regulate, as the downstream genes.
This study aimed to investigate the potential anti-obesity effects of Tetracarpidium conophorum nuts by modulating key lipogenic pathways in monosodium glutamate (MSG)-induced obese rats. By assessing the influence of the nut extracts on the SREBP-1/ACCA-1/FASN signaling axis, the study sought to elucidate their role in lipid metabolism regulation. These study seeks to highlight the therapeutic potential of T. conophorum nuts in mitigating obesity-associated dyslipidemia and metabolic disorders by targeting key regulators of lipogenesis.
Materials and Methods
Chemicals and Reagents
Easy Script One-Step RT-PCR SuperMix (Beijing TransGen Biotech Co., Ltd, Beijing, China), Primers (Integrated Data Technologies, 802 Old Dixie Hwy #2, North Palm Beach, FL 33408, United States), TRI quick reagent (Solarbio Life Sciences) and RNase free water were used in this study. All other reagents and chemicals used but not listed were of analytical grade and were purchased from Sigma-Aldrich, USA, unless otherwise stated.
Plant Collection and Extraction
The African walnuts were purchased from Sagamu, Ogun State, brought to Biochemistry laboratory of Evangel University, Akaeze, Ebonyi State, Nigeria. They were authenticated by Dr Bruno Aduo, a botanist of the Department of Biological Sciences, Evangel University. The nuts were de-shelled chopped into bits and dried at room temperature (25 °C - 29 °C), after which they were pulverized. The pulverized sample was suspended in absolute ethanol (BDH) in the ratio 1: 2, sample to solvent and the resulting suspension thoroughly agitated in an electric blender and allowed to stand for a period of 48 h at room temperature. It was consequently filtered first, with a cheese material and afterwards with Whatman No. 2 filter paper and the filtrate concentrated in a rotary evaporator (45 °C – 50 °C) to about 1/10th of the original volume. Afterwards, the concentrate was allowed to evaporate to complete dryness in a water bath (45-50 °C).
Liquid-Liquid Fractionation of the Extract
The liquid-liquid fractionation of the whole ethanol extract was carried out as described in our earlier study. 9 Briefly, a measured quantity of the crude extract was sequentially separated in different solvents of graded polarity, using separating funnel. Ten (10) g of the whole ethanol extract was re-dissolved in 10 mL of absolute ethanol (the solvent used for the extraction), followed by stepwise addition of a portion of the fractionating solvent ie ethyl-acetate, and shaken vigorously, then allowed to stand for 5–10 min to separate into two visible layers. The denser lower layer was carefully collected into a labeled beaker, separated from the less dense (ethyl acetate soluble) layer. This was repeated several times until presumably all the components of the whole extract, soluble in the ethyl acetate were collected. Using a rotary evaporator, the fractionated portion was evaporated to dryness and labeled ethyl acetate fraction (EAF). The ethyl acetate non-soluble portion was also concentrated and the dried concentrate labeled residue (RES). Hence, the whole extract (EWE), EAF and residue (RES) constituted the test substances in this study.
Induction of Experimental Obesity
Adult female and male Wistar rats obtained from the Animal House, College of Medical Sciences University of Calabar, were co-habited in the proportion of 3 females to 1 male. These mated and produced offspring (pups) which were used for the production of obese animal models according to a procedure described in our earlier study.9,10 The pups were swiftly divided into two groups on the day of delivery; one group (Group 1) was injected i.p. 4 mg/g of monosodium glutamate (MSG) reconstituted in normal saline on postnatal days 2, 4, 6, 8 and 10 while the other group (Group 2) was treated similarly, but with normal saline instead. The rats were weaned on the 21st day, raised normally and thereafter studied at the age of 12 weeks. Lee's Index and BMI were measured as indicators of obesity; rats with Lee's indices 0.3 and above were considered obese, or with BMI 20% greater than controls were considered obese 9 and included in the study.
Experimental Design and Procedures

The extracts and standard drug were treatment administered via oral gavage for 6 weeks, using doses determined and used previously in our laboratory.9,10 The rats were fed rat pellets with its nutrient composition given in Table 1, and tap water ad libitum and maintained in a standard animal facility of room temperature 25 °C ± 3 °C. After 6 weeks of treatment, the animals were euthanized and sacrificed by one of the most commonly used and acceptable pharmacologic methods - injectable anesthesia overdose, followed by bleeding out via cardiac or heart puncture. That is, ketamine (2 µl/kg b.w.) was injected intra-peritoneal, and the rats soon loss consciousness, after which the rats were bled out completely via heart puncture, using hypodermic needles and syringes. Thereafter, the animals were surgically dissected and the livers and epididymal adipose tissues excised under aseptic condition into sample bottles containing RNA later and stored frozen at −80 °C until used for analysis. The animal handling protocol was in line with the guidelines of the National Institute of Health (NIH) publication (1985) for laboratory animal care and use. Moreover, ethical approval for the use of animals in this study was obtained from the Faculty of Basic Medical Sciences Animal Research Ethics Committee (FAREC- PA, Approval number: UC/049,), University of Calabar, Nigeria. The protocols subscribe to the global conventional use of animal models in research. This study was conducted and reported in accordance with Animal Research: Reporting In Vivo Experiments (ARRIVE) guidelines. 12
Binding Energies of Hit Compounds with the Respective Targets.

BE: Binding Energy
RNA Isolation, Polymerase Chain Reaction (PCR) and Quantification of SREBP1, ACCA1 and FASN
Total RNA from the liver and adipose tissues were isolated and purified following the TRIzol-based protocol (Life Technologies) as described in an earlier study by Umoru et al, 2024. 7 The purity and yield (concentration) of the extracted RNA were determined using a Nano drop 2000 spectrophotometer at 260 nm and 280 nm. 33 Five hundred nanograms (500 ng) of total RNA was reverse-transcribed (RT) using a Transgen Easy Script® one-step RT-PCR supermix (Beijing Trans Gen Biotech Co., Ltd, Beijing, China) according to the manufacturer's instructions, and the PCR was performed using a Bio-rad c1000 touch harmac-cycler. The primers and reaction condition used for the quantitative PCR experiments are listed in supplementary Data (Table S1 and Table S2). The Reverse Transcriptase- Polymerase Chain Reaction (RT-PCR) amplicons were subjected to agarose gel electrophoresis for detection and band was quantified using the image J software. 34
Molecular Docking Analysis
The compounds herein called ligands detected by Gas chromatography-mass spectroscopy analysis of the T. conophorum (supplementary table 3) in an earlier study9,10 were docked with SREBP-1, ACCA-1 and FASN using PyRx 0.8 software, in order to determine the binding energies.
Preparation of Target Protein
The crystal structures of SREBP-1 (Protein Data Bank –PDB ID: 5GPD), ACCA1 (3PGQ) and FASN (2PX6) were retrieved from the PDB's structural base (https://www.rcb.org/). 35 The active sites were determined using Computed Atlas of Surface Topography of Proteins (CASTP) and separated from their ligands. Also, all hetero atoms and crystal water molecules were removed while hydrogen atoms and gasteiger charges were added to the proteins. Then, the structures were minimized using the University of California San Francisco (UCSF) chimera1.4 software and saved in PDB, Partial charge and Atom type (PDBQT) format for further analysis.
Ligand Preparation
The crystal structures of chemical compounds (ligands) detected by Gas chromatography-mass spectroscopy analysis of the T. conophorum (supplementary table 3) fractions were retrieved from the PubChem database (pubchem.ncbi.nlm.nih.gov/compound) in structure data file (sdf) format, imported onto the PyRx 0.8 software and subjected to minimization. Energy minimization was performed with the universal force field (UFF), using conjugate gradient algorithm 36 ; the minimized compounds were converted into PDBQT format and further studied.
Virtual Screening
With the 5GPD crystal structure as a receptor and the crystal structures of the chemical compounds of T. conophorum nuts as ligands, the binding energies were calculated using PyRx0.8. A grid box was set to span the active site of the receptor with the dimensions shown in supplementary file (Table S4). An exhaustiveness of 8 was used and the top three hit compounds were selected for screening. The top three hit compounds were passed through a second screening using AutoDock 4.236,37 and analysis of the findings were performed using Discovery studio and PyMOL programs. 38
ADMET/Pharmacokinetics Predictions Analysis
Absorption, Distribution, Metabolism, Excretion and Toxicity (ADMET) analysis constitute the pharmacokinetics of a drug molecule.39–41 In broader terms, it encompasses the molecular physicochemical properties typically found in chemicals referred to as drugs. 42 In this study, the molecular structures of the top three hit ligands were submitted to Swissadme (http://www.swissadme.ch) 43 and admetSAR server (http://lmmd.Ecust.edu.cn:8000) to examine their drug likeliness using the Lipinski rule of five. The rule states that a molecule or an inhibitor can be orally absorbed/active if two (2) or more of the threshold properties are not violated:(i) its molecular weight (Mw) of is < 500, (ii) the octanol/water partition coefficient (Ilogp) is ≤ 5, (iii) the number of hydrogen bond acceptors (Nhba) is ≤ 10, (iv) the number of hydrogen bond donors (Nhbd) is ≤ 5 and (v) the topological polar surface area (TPSA) is < 40 Å2). 44 Different pharmacokinetic and pharmacodynamics parameters including mutagenicity, blood brain barrier penetration, human intestinal absorption (HIA), cytochrome P450 solubility, and cytochrome P (CYP) inhibitory promiscuity and Ames toxicity were also analyzed.
Statistical Analysis
The results were expressed as the Mean ± Standard Deviation (SD), and the differences between treated and control groups were statistically assessed using one-way ANOVA and paired with one sample T-test. All statistical calculations were performed using GRAPHPAD prism7.0 (GraphPad Software, CA, USA). The results were considered significant at * p < 0.05, ** p < 0.01, *** p < 0.001.
Results
SREBP-1 Gene Expressions in the Adipose and Liver Tissues
The effects of the extract and fractions of T. conophorum nuts on the expressions of SREBP-1c, an upstream lipogenesis regulatory gene, in the adipose and liver of MSG obese rats are shown in Figure 1. Higher expression levels of SREBP1 were observed in the adipose tissue and liver of MSG induced obese rats. However, 6-week oral administration of the whole extract (p < 0.05), ethyl-acetate (p < 0.01) and residue (p < 0.01) fractions of the African walnut significantly attenuated the expression levels of SREBP-1c in the adipose tissue when compared to the obese control. Similarly, the liver expression levels of SREBP-1c was significantly attenuated by treatment with the whole extract (p < 0.01), ethyl-acetate (p < 0.001) and residue (p < 0.001) fractions T. conophorum nuts in relation to obese control.

Relative Expression of SREBP-1c in the (A) Epididymal Adipose Tissue, and (B) Liver of Monosodium Glutamate Induced Obese Rats Administered Tetracarpidium conophorum Nut Extracts and Fractions. Values are Expressed as mean ± SD, n = 3. NC: Normal Control, OC: Obese Control, STD: Standard Control, WE: Whole Extract, ET: Ethyl-Acetate Fraction, RES: Residue Fraction. * p < 0.05, ** p < 0.01, *** p < 0.001.
Expressions of the Downstream Lipogenesis Target Genes (ACCA-1 and FASN) in the Adipose and Liver Tissues
Figure 2 shows the relative expressions of ACCA-1 in the adipose and liver tissues of MSG obese rats administered T. conophorum nut extracts and fractions for 6 weeks. There were no significant differences in the adipose tissue expression levels of ACCA-1 in MSG induced obese rats when compared with rats treated with the ethyl-acetate and the residue fractions of T. conophorum nut. However, rats treated with the whole extract exhibited high (p < 0.05) adipose tissue expression levels of ACCA-1 when compared with the control and other treatment groups. On the contrary, high expression levels of ACCA-1 was observed in the liver of MSG-induced obese rats and was significantly attenuated by 6 weeks’ oral administration of the whole extract, ethyl-acetate fraction and the residue from the T. conophorum nut.
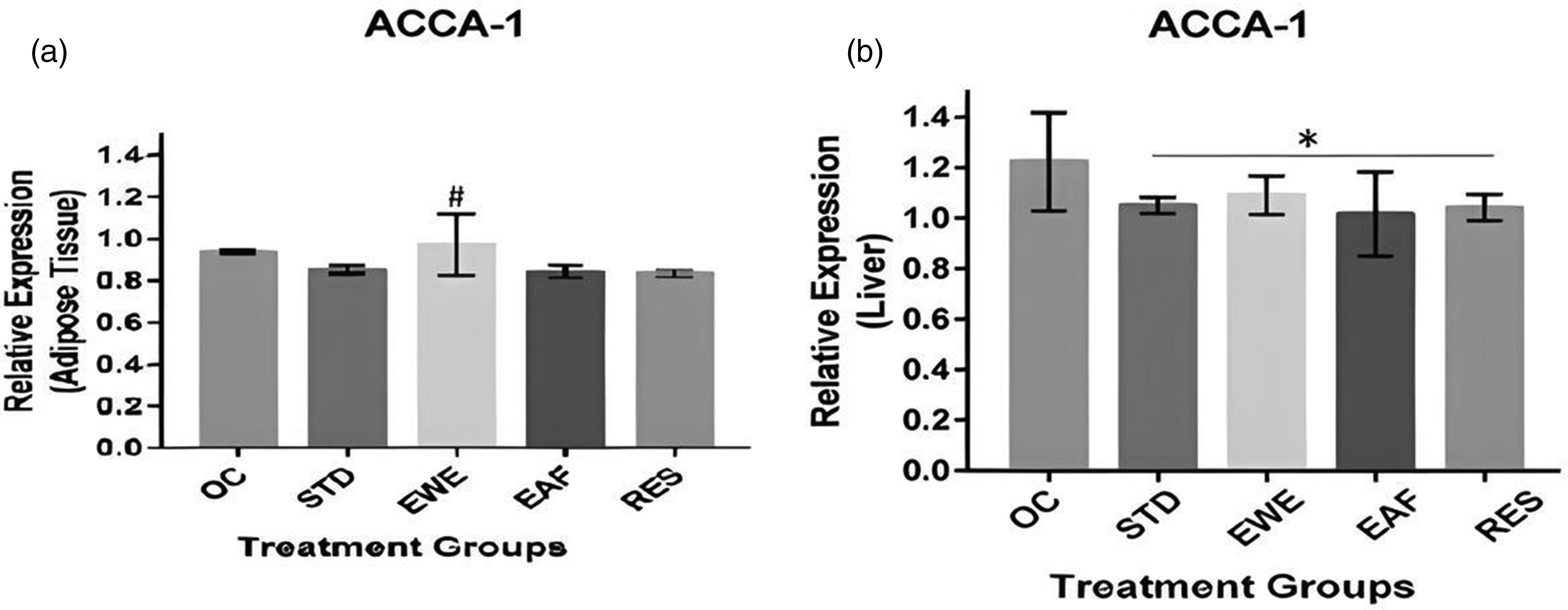
Relative Expression of ACCA-1 in the (A) Epididymal Adipose Tissue, and (B) Liver of Monosodium Glutamate Induced Obese Rats Administered Tetracarpidium conophorum Nut Extracts and Fractions. Values are Expressed as mean ± SD, n = 3. NC: Normal Control, OC: Obese Control, STD: Standard Control, WE: Whole Extract, ET: Ethyl-Acetate Fraction, RES: Residue Fraction. # p < 0.05, * p < 0.05.
The gene of the complex enzyme, FASN complex, was studied in the adipose and the liver; data obtained are shown in Figure 3. The results indicate a significant elevation of adipose and liver tissues expression levels of FASN. Interestingly the ethyl acetate and residue fractions significantly (p < 0.05) suppressed the expression levels of FASN in the liver as well as the adipose tissue when compared with the non-treated control. The whole extract, however, attenuated the deregulatory expression of FASN in adipose tissue but not in the liver tissue. The activities demonstrated by the ethyl acetate and residue fractions was comparable to that of the standard drug.

Relative Expression of FASN in the (A) Epididymal Adipose Tissue, and (B) Liver of Monosodium Glutamate Induced Obese Rats Administered Tetracarpidium conophorum Nut Extracts and Fractions. Values are Expressed as mean ± SD, n = 3. NC: Normal Control, OC: Obese Control, STD: Standard Control, WE: Whole Extract, ET: Ethyl-Acetate Fraction, RES: Residue Fraction. * p < 0.05, ** p < 0.01.
In Silico Studies of Lipogenesis Regulatory Target (Upstream) Gene, SREBP-1
The GC-MS detected ligands of T. conophorum nuts were docked with the protein, SREBP-1. The interaction of the ligands with the protein is shown in Table 1. The top three hit compounds having the highest binding affinity compared to the standard drug Orlistat (Figure 4a) were found to be: 6-Isopropenyl-4,8a-dimethyl-4a, 5, 6, 7, 8, 8a-hexahydro-1H-naphthalen-2-one (Figure 4b) having a high SREBP-1 binding affinity of −7.7 Kcal/mol, one (1) carbon-hydrogen bond, seven (7) van der Waal forces, one (1) alkyl residue, one (1) Pi-alkyl bond and interactions of the ligand with the SREBP-1 protein was via His 794, Leu 769 and Tyr 765 residues. The second hit compound was 9, 12, 15- octadecatrienoic methyl ester (Z, Z, Z) (Figure 4c), the docking characteristics were binding energy of −6.6 Kcal/mol,one (1) conventional hydrogen bond, three (3) van der Waal forces, two (2) covalent bonds and interactions with the protein via Asp 770 and Asp 775 residues. The molecule with the lowest binding affinity of the 3 hit compounds detected was ethyl 9, 12, 15-octadecatrienoate (Figure 4d) with binding energy −6.5 Kcal/mol, one (1) conventional hydrogen bond, one (1) carbon-hydrogen bond, eight (8) van der Waal forces, one (1) Pi-sigma interaction, two (2) alkyl bonds, three (3) Pi-alkyl bonds and interactions via Lys 789, Ile 785, Tyr 765, Leu 793, Leu 783, His 741, Thr 792, Leu 769 and Ser830 residues.

In Silico Studies of Lipogenesis Target Gene in the Adipose and Liver: ACCA-1
The interaction of the ligands from T. conophorum nut with ACCA-1, a fat-specific target protein ACCA-1 is shown in Table 1 and the pose for the top three hit compounds as well as the standard drug orlistat in (Figure 5a). Of the 29 compounds docked, 6-Isopropenyl-4,8a-dimethyl-4a,5,67,8,8a-hexahydro-1H-naphthalen-2-one (Figure 5b) had the highest binding affinity ie −7.2 Kcal/mol, one (1) conventional hydrogen bond, seven (7) van der Waal bonds and two (2) alkyl and one (1) pi-alkyl bonds compared to the standard drug orlistat (−6.2 kcal/mol). These interactions were via the Arg 2120, Leu 1532, Trp 2124 and Phe 2121 residues. Closely following in its wake is ethyl octadecatrienoate (−6.2 kcal/mol) (Figure 5c) with one (1) conventional hydrogen bond, eleven (11) van der Waal forces, one (1) carbon-hydrogen bond, three (3) alkyl bonds and two (2) pi-alkyl bonds and interacting amino acid residues were Thr 1504, His 1501, Pro 2160, Leu 154, Val 2163, Trp 2174 and Leu 1797. Lastly, 9, 12,-Octadecadienoic acid ethyl ester (−6.1 kcal/mol) (Figure 5d), with two (2) conventional hydrogen bonds, eight (8) van der Waal forces, 2 alkyl bonds and two (2) pi-alkyls bonds. It interacts with the protein were via Pro 2160, Leu 1797, Leu 1514 and Thr 1505 residues.

In Silico Studies of Lipogenesis Target (Downstream) Genes in the Adipose and Liver: FASN (Fat-Specific Genes)
The ligands were also docked with another fat specific target protein, FASN. The top three HIT compounds having the highest binding affinities compared to the standard drug orlistat (Figure 6a) are 6-Isopropenyl-4,8a-dimethyl-4a,5,6,7,8,8a-hexahydro-1H-naphthalen-2-one (Figure 6b) with low binding energy and high binding affinity of −7.8Kcal/mol, one (1) conventional hydrogen bond, eight (8) van der Waal forces, one (1) Pi-Sigma bond and one (1) alkyl bond while interacting with the protein via the residues Tyr 2425 and Leu 2279. Followed closely was 9, 12, 15-Octadecatrienoic acid methyl ester (Z, Z, Z) (Figure 6c). It had binding energy of −6.3 kcal/mol with 2 conventional hydrogen bonds, ten (10) van der Waal forces, one (1) alkyl bond, three (3) Pi-alkyl bonds and interacting residues Arg 2421, Ser 2417, Tyr 2425, Leu 2279, Arg 2275 and Phe 2418. The molecule with the lowest binding affinity of the 3 hit compounds was 2, 3-Dihydroxypropylelaidate (Figure 6d) with low binding energy and high binding affinity of −6.2 Kcal/mol, four (4) conventional hydrogen bonds, five (5) van der Waals forces, two (2) carbon-hydrogen bonds, three (3) alkyl and two (2) pi-alkyl bonds. The interaction with the protein was via Leu 2279, Tyr 2425, Asp 2280, Arg 2421, Ser 2281, Arg 2275, Phe 2418, Arg 2421 and Arg 2428 residues.

Absorption Distribution Metabolism Excretion & Toxicity (ADMET)/Pharmacokinetics Predictions Analysis
Drug Likeness
The potential ADMET profiles of the five candidate compounds were predicted using AdmetSAR server, 9,12-Octadecadienoic acid, ethyl ester and ethyl- 9,12,15- octadecatrienoate violated only one of the five Lipinski rules while 6-Isopropenyl-4,8a-dimethyl-4a,5,67,8,8a-hexahydro-1H-naphthalen-2-one, 9,12,15-Octadecatrienoic methyl ester (Z,Z,Z) and 2,3-Dihydroxypropyl elaidate violated no rule at all (Table 2).
Absorption Distribution Metabolism Excretion & Toxicity (ADMET)/Pharmacokinetics Properties of the Compounds.

HBD - H-bond Donors, HBA - H-bond Acceptors, Mol. Wt – molecular weight, HIA - Human Intestinal Absorption, BBB - Blood Brain Barrier
Absorption
All five compounds were positive for human intestinal and blood-brain barrier absorption. Four compounds were negative as P-glycoprotein substrates and only 9, 12, 15-Octadecatrienoic methyl ester (Z, Z, Z) was positive as P-glycoprotein inhibitor. Three of the compounds namely 9, 12-octadecadienoic acid, ethyl ester and ethyl- 9, 12, 15-octadecatrienoate were positive for Caco-2 permeability (Table 2).
Metabolism
All five compounds were negative for CYP inhibition and also failed to act as CYP substrates. However, 6-Isopropenyl-4,8a-dimethyl-4a,5,67,8,8a-hexahydro-1H-naphthalen-2-one and 9,12,15-Octadecatrienoic methyl ester (Z,Z,Z) were found to act as CYP3A4 substrates while 9,12-Octadecadienoic acid and ethyl ester are positive for CYP1A2 inhibitor (Table 1).
Toxicity
The Toxicity prediction showed that all 5 compounds are negative for carcinogenicity and hepatotoxicity. But for ethyl- 9, 12, 15-octadecatrienoate and 9, 12, 15-octadecatrienoic methyl ester (Z, Z, Z), the other compounds were negative for mutagenicity. Moreover, the majority of these active compounds were shown to be biodegradable in the ADMET evaluation (Table 2).
Discussion
The present study evaluated in part, the molecular mechanism of the previously reported anti-obesity effect of Tetracarpidium conophorum nuts otherwise called African walnut, in MSG-induced obesity in Wistar rats by studying its impact on the lipogenesis regulatory target (upstream) gene - Sterol Regulatory Element Binding Protein- 1 (SREBP-1) of the adipose tissue and liver tissues.
In both adipose tissue and liver of obese rats, the SREBP-1gene expression was up-regulated suggesting increased adipogenesis and lipogenesis. However, a 6-week oral intervention with whole ethanol extract, EAFs and the fractionation residue of T. conophorum nuts was found to significantly down-regulate the expression of this transcription factor for liver. This observed effect suggests decreased adipogenesis and lipogenesis upon treatment both in the adipose and the liver. SREBP-1 is a transcription factor responsible for the regulation of the FASN gene, a key lipogenic gene45,46 as such, a decrease in expression, suggests repression of fatty acid synthesis, ultimately decreasing fatty acid storage which usually leads to obesity.45,47 Studies have also shown that over-expression of SREBP-1 induces lipogenic enzymes resulting in triacylglycerol accumulation 46 and vice versa. Thus, SREBP-1 is one of the plausible molecular targets of the bio-actives of African walnut and its modulation a likely molecular approach by which African walnut exerts anti-obesity activity.
The effect of our intervention on the expressions of lipogenesis target (downstream) genes namely Acetyl-CoACarboxylase-1 (ACCA-1) and FASN genes of the adipose and the liver was also studied. The results obtained showed that induction of obesity up-regulated the expression of both genes in the liver and the adipose tissues signifying increased fatty acids biosynthesis. However, a 6-week oral intervention with the extract and fractions of T. conophorum was found to cause a significant down-regulation in ACCA-1and FASN gene expressions both in the liver and the adipose tissues suggesting a positively attenuated lipogenesis. An earlier study had also shown that down-regulation of ACCA-1 gene led to reduction in hypertriglyceridemia and hepatic glyceride content,48–50 hence obesity. Also, another study showed that FASN is over-expressed in adipocytes of obese rats, 48 but upon inhibition, induces profound weight loss indicating its involvement in body weight regulation via alterations in feeding behaviour and energy homeostasis in animals. 51 Hence, it is reasonable to state that ACCA-1 and FASN are among the molecular targets modulated by the African walnuts to achieve body weight control.
Earlier studies have established that over-expression of FASN and ACCA-1 in obese rats is dependent on high transcriptionally active SREBP-1 activity. 52 Hence one of the major findings of this study is the dual approach - cascade and direct regulations of fatty acid biosynthesis in the liver and adipose tissues by the African walnuts: Up-regulated expression of the upstream SREBP-1 gene in the adipose and liver resulting in a corresponding up-regulation in the expressions of FASN and ACCA genes in obese rats; and contrariwise, a down-regulation in SREBP-1 expression upon oral treatment with African walnut which produced a corresponding down-regulation in the expressions of the downstream FASN and ACCA-1 genes. Aside, this cascade effect, it is also found that the downstream genes - FASN and ACCA-1, were directly repressed or modulated by the African walnuts, providing a dual molecular approach in controlling fatty acid biosynthesis in obesity.
The potential mechanisms by which Tetracarpidium conophorum (African walnut) nut extracts inhibit the SREBP-1/ACCA-1/FASN signalling network involve multiple pathways. The extracts significantly attenuated SREBP-1c expression in the adipose tissue and liver of MSG-induced obese rats, indicating an interference with this key transcription factor that regulates lipogenesis. Additionally, while ACCA-1 expression remained unchanged in adipose tissue, it was significantly downregulated in the liver by all treatment groups, whereas FASN expression was notably suppressed in both tissues by the ethyl-acetate and residue fractions, suggesting a broad inhibition of fatty acid biosynthesis. Further, in silico molecular docking revealed that GC-MS-identified bioactive compounds from T. conophorum formed strong interactions with SREBP-1, ACCA-1, and FASN through hydrogen bonding, van der Waals forces, alkyl, and π-alkyl interactions, demonstrating a direct inhibitory potential at the molecular level. Notably, the strongest binding compound, 6-Isopropenyl-4,8a-dimethyl-4a,5,6,7,8,8a-hexahydro-1H-naphthalen-2-one, exhibited higher affinity (−7.7 to −7.8 kcal/mol) than Orlistat (−5.9 to −6.2 kcal/mol)
Given these impressive molecular data obtained, molecular docking studies were carried out to determine the best in silico interaction of the T. conophorum nut derived ligands (active compounds) with the products of the studied genes.
The result obtained showed that five active compounds (ligands) namely (i) 6-Isopropenyl-4,8a-dimethyl-4a,5,6,7,8,8a-hexahydro-1H-naphthalen-2-one, (ii) 9,12-Octadecadienoic acid ethyl ester, (iii) Ethyl- 9,12,15- Octadecatrienoate, (iv) 9,12,15-Octadecatrienoic methyl ester (Z,Z,Z), and (v) 2,3-Dihydroxypropyl elaidate, showed recurring binding affinities with the target proteins. These ligands are the likely compounds responsible for the observed molecular effects, hence may be the active agents in African walnut accounting for the anti-obesity action. Of particular note, is the ligand, 6-Isopropenyl-4,8a-dimethyl-4a,5,6,7,8,8a-hexahydro-1H-naphthalen-2-one, identified as a sesquiterpene lactone, 53 a derivative of the secondary metabolite terpenoids found in plants 54 which showed high interactions with all the target proteins studied, making it a very promising candidate for obesity treatment. 52 According to previous reports, diterpenoids and their derivatives play a role in anti-obesity. 55 For instance, Teucerin A isolated from Teucerinchae modrys reduced body weight in Sprague Dawley rats. 56 Similarly, terpenoid fractions of Ilex aquifolium leaves reduced hepatc lipid accumulation in obese Zucker rats. 57 In fact, in a particular European patent application, a sesquiterpene cynaropicrin is claimed to be responsible for significant weight loss given its effects on energy metabolism. 56 Other studies have also identified 9,12,15-Octadecatrienoic methyl ester (Z,Z,Z), and Ethyl- 9,12,15- Octadecatrienoate, as derivatives of alpha linolenic acids.58–60 Alpha linolenic acid (ALA) is an essential n-3 PUFA with cardio-protective, anti-inflammatory and anti-oxidative effects.61,62 Also, ALA or ALA-enriched diets alter body composition, improve glucose tolerance, attenuate insulin resistance 60 and show significant lipid-lowering activity, including reducing liver fat accumulation, alleviating liver steatosis, and lowering blood lipid levels. 63 9,12-Octadecadienoic acid ethyl ester on the other hand is a linoleic acid, 64 they are essential n-6 PUFAs which are responsible for modulating vital functions such as inflammatory response. 65 They have also been shown to improve lipid profile and glycemic index. 66 The adequate consumption of Linoleic acids have been linked to reduction in plasma low-density lipiprotein cholecterol (LDL-c) followwed by a consequent reduction in risk of cardiovascular disease. 67 The strong binding affinity of the characterized compound to SREBP-1, ACCA-1, and FASN suggests its potential role in modulating lipid metabolism. SREBP-1 regulates lipogenic gene expression, while ACCA-1 and FASN are key enzymes in fatty acid synthesis. Inhibiting these targets may reduce lipid accumulation, offering therapeutic potential for obesity and dyslipidemia. The compound's high affinity indicates possible competitive inhibition, disrupting lipid biosynthesis pathways. Taken together, the results of these previous studies are basically consistent with our findings suggesting that the ligands may be responsible for the observed molecular actions, hence lead compound for further anti-obesity research.
The Tetracarpidium conophorum (African walnut) extracts exhibited notable effects on gene expression and docking affinity, surpassing Orlistat in certain aspects. The extracts significantly attenuated SREBP-1c expression in the adipose and liver tissues of obese rats, with the ethyl-acetate and residue fractions demonstrating effects comparable to Orlistat. Additionally, these extracts suppressed FASN expression in both tissues, whereas Orlistat's effect was primarily observed in adipose tissue. In molecular docking, the top ligand from T. conophorum exhibited a binding affinity of −7.8 kcal/mol for FASN, surpassing Orlistat (−6.2 kcal/mol). Similarly, for SREBP-1
Regardless of how well a ligand fits its receptor, it is ineffective if it is poorly absorbed, metabolized, or excreted from the body.68,69 To test for this, the ADMET (absorption, distribution, metabolism, excretion and toxicity) evaluation was conducted to predict the pharmacokinetic (drug-likeness) profiles of the potential lead compounds. In this study, the ligands not only exhibited high binding affinities but also complied with the Lipinski rule of five, which assesses a compound's likelihood of being absorbed through the gastrointestinal tract through passive diffusion70,71 Consequently, these compounds possess high permeability profile and are readily absorbed.
To further support this, all hit compounds tested positive for HIA, indicating their potential for effective absorption from the intestinal tract following oral administration. Additionally, all five hit compounds demonstrated the ability to penetrate the blood-brain barrier. Notably, none of the ligands are P-glycoprotein substrates, unlike the standard drug, making them less likely to be expelled from cells by P-glycoprotein. The primary isoforms involved in drug metabolism P2D6 cytochrome (CYP2D6) and P3A4 cytochrome (CYP3A4).72–76 From the in silico data, it can be seen that almost all the ligands did not affect or inhibit the CYP2D6 and CYP3A4 enzymes, except the 6-Isopropenyl-4,8a-dimethyl-4a,5,67,8,8a-hexahydro-1H-naphthalen-2-one and 9,12,15-Octadecatrienoic methyl ester (Z,Z,Z) compounds, which inhibited the CYP3A4 enzyme. It is therefore reasonable to predict that the derivatives may undergo metabolism by the P450 enzymes in the body.
The Ames toxicity test revealed that all test ligands, except Ethyl-9,15-Octadecatrienoate and 9,12,15-Octadecatrienoic methyl ester (Z,Z,Z), showed negative results, similar to the standard drug. Additionally, the hepatotoxicity test indicated that none of the compounds exhibited hepatotoxicity, unlike the standard drug. These findings suggest that the ligands are non-mutagenic and safe for the liver, supporting their potential as promising anti-obesity drug candidates for further investigation.
Earlier research has demonstrated that T. conophorum (African walnut) possesses beneficial effects against metabolic disorders, including obesity and dyslipidemia. For example, Nwaichi et al 8 reported that T. conophorum reduced weight gain, plasma triglycerides, total cholesterol, LDL-cholesterol, VLDL-cholesterol, and atherogenic indices in hyperlipidemic rat models in a dose-dependent manner. Similarly, Uti et al9,11 showed that the nuts significantly lowered total lipid, triglyceride, and cholesterol levels in obese rats while preventing hepatic lipid accumulation. Additional studies highlighted its potential to modulate obesity-related inflammation, hyperglycemia, and oxidative liver damage. 11 This study builds upon previous research by introducing key advancements in understanding the anti-obesity mechanisms of T. conophorum. Unlike earlier studies that primarily reported metabolic benefits, this research provides mechanistic insights by demonstrating that T. conophorum modulates key lipogenic regulatory genes, including SREBP-1, ACCA-1, and FASN, which are crucial for lipid biosynthesis. Furthermore, in silico docking analysis revealed that bioactive compounds from T. conophorum exhibited stronger interactions with these lipogenic regulators than the standard anti-obesity drug, Orlistat. The study also demonstrated favourable pharmacokinetics of these compounds, showing good intestinal absorption, blood-brain barrier penetration, and low toxicity, reinforcing their potential as anti-obesity agents. Overall, this study not only confirms prior research on the anti-obesity effects of T. conophorum but also introduces novel mechanistic insights and strengthens the case for its therapeutic potential. The findings suggest that T. conophorum may serve as a viable natural alternative for obesity management, warranting further clinical studies to validate its effects in humans.
However, the study's limitations include generalizability, sample size due to cost implications, single species, duration of study, dose-response relationship, ethical considerations, and translation to human consumption. Addressing these limitations through further research, clinical trials, longer-term studies, and rigorous study designs would strengthen the evidence base for the potential benefits of Tetracarpidium conophorum nuts in modulating lipogenesis and combating obesity-related metabolic disorders. However, this study could serve as a baseline study for translation to clinical and human experiments.
Conclusion
In conclusion, the results of this investigation clearly show that African walnuts have strong anti-obesity effects via modulating important lipogenic regulatory genes, such as SREBP-1, as well as important downstream lipogenic genes, ACCA-1 and FASN. The mechanism that is suggested, as illustrated in Figure 7, provides substantial evidence in favour of the claim that the molecular actions that are observed are caused by the combined actions of the bioactive chemicals that are found in the nuts. This strong evidence show that African walnuts’ may be potentially viable therapeutic agents in the fight against obesity. However, further studies are required to confirm these findings specifically, studies involving the proteins of the genes.

Schematic Summary of the Anti-Obesity Action of African Walnut (Tetracarpidium conophorum).
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251344035 - Supplemental material for Modulation of Lipogenesis by Tetracarpidium conophorum Nuts via SREBP-1/ACCA-1/FASN Inhibition in Monosodium-Glutamate-Induced Obesity in Rats
Supplemental material, sj-docx-1-npx-10.1177_1934578X251344035 for Modulation of Lipogenesis by Tetracarpidium conophorum Nuts via SREBP-1/ACCA-1/FASN Inhibition in Monosodium-Glutamate-Induced Obesity in Rats by Grace U Umoru, Item Justin Atangwho, Essien David-Oku, Daniel Ejim Uti, Opeyemi C De Campos, Precious A Udeozor, Samuel O Nfona, Bashir Lawal and Esther Ugo Alum in Natural Product Communications
Footnotes
Acknowledgements
The authors wish to acknowledge Humphrey Chukwudi Omeoga, of Thomas BehLey Laboratory, Department of Biological Sciences, State University of New York, at Albany, New York USA for using his licensed accessed BioRender for the diagram sketching.
Author's Contribution
Conceptualization, IJA & ED conceived the study; Analysis GUU, DEU, OCD, and SON carried out the animal experiments and laboratory analysis; Interpretation, GUU, PAU, and BL analyzed the data; Writing-original draft preparation was carried out by GUU & PAU;, IJA, BL EUA, & ED did the writing-review and editing for intellectual content, All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analysed during this study are available from the corresponding author on request.
Institutional Review Board Statement
The study was conducted in accordance with the approval of the Ethics Committee of the Faculty of Basic Medical Sciences, University of Calabar, Cross River State. (Approval number: 032BCH3319). The protocols subscribe to the global conventional use of animal models in research. This study was conducted and reported in accordance with Animal Research: Reporting In Vivo Experiments (ARRIVE) guidelines.
Statement of Human and Animal Rights
The ethical approval for the use of animals in this study was obtained from the Faculty of Basic Medical Sciences Animal Research Ethics Committee (FAREC- P.A, Approval number: UC/049,), University of Calabar, Nigeria.
Statement of Informed Consent
NA
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
