Abstract
As part of our continuous studies on dietary supplements for diabetes, 1 new phenolic diterpene, 7-butoxyrosmanol (
Introduction
Obesity is now a global public concern that exerts adverse effects on health and imposes a huge economic burden on society. Pharmacotherapy is the most common treatment option for obesity. Pancreatic lipase is well known to play an important role in lipid digestion. 1 Thereby, the inhibition of pancreatic lipase is thought to be an effective therapy for obesity. Orlistat is a specific pancreatic lipase inhibitor and is clinically used as an anti-obesity drug in many countries. 2 However, orlistat also causes gastrointestinal side effects. 3 Therefore, it is necessary to find diet-derived anti-obesity compounds with excellent bioefficacy and long-term safety. 4
Rosemary (Rosmarinus officinalis L.) is widely used as a food ingredient and culinary spice. Its extracts have been used for the treatment of Alzheimer's disease, cancer, cardiovascular disease, obesity, and diabetes.5‐9 The major constituents of rosemary have been reported to be phenolic acids, terpenoids, and flavonoids.5,10 In the present work, we report the isolation and pancreatic lipase inhibition of phenolic diterpenes from rosemary.
Results and Discussion
The methanol extract of rosemary was subjected to repeated column chromatography to yield one new phenolic abietane diterpene, 7-butoxyrosmanol (
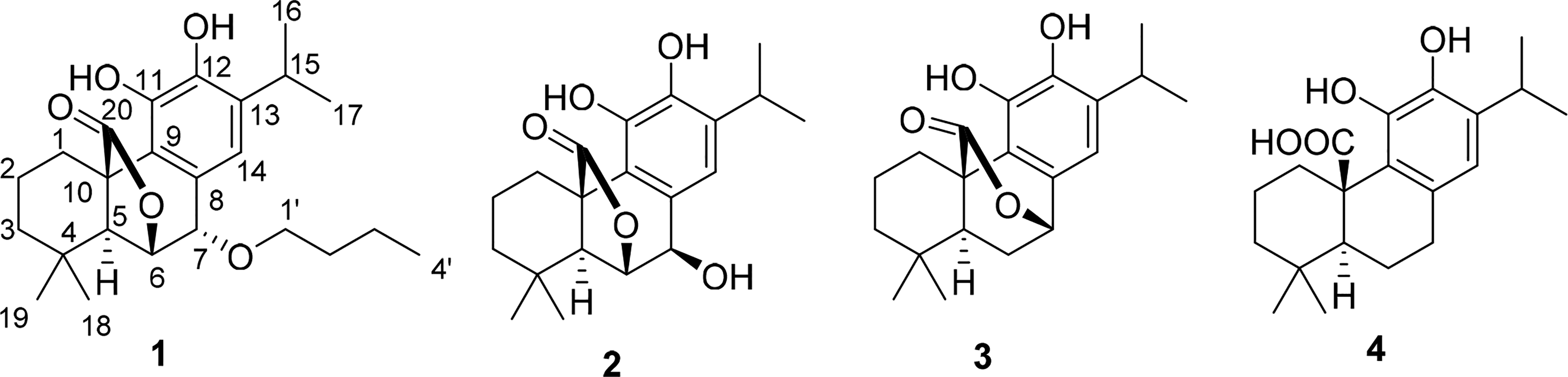
Structures of compounds
Compound

Tandem mass spectrometry (MS/MS) of compound

H-H COSY (blue bold lines) and key heteronuclear multiple bond correlations (HMBC) (red arrows) of
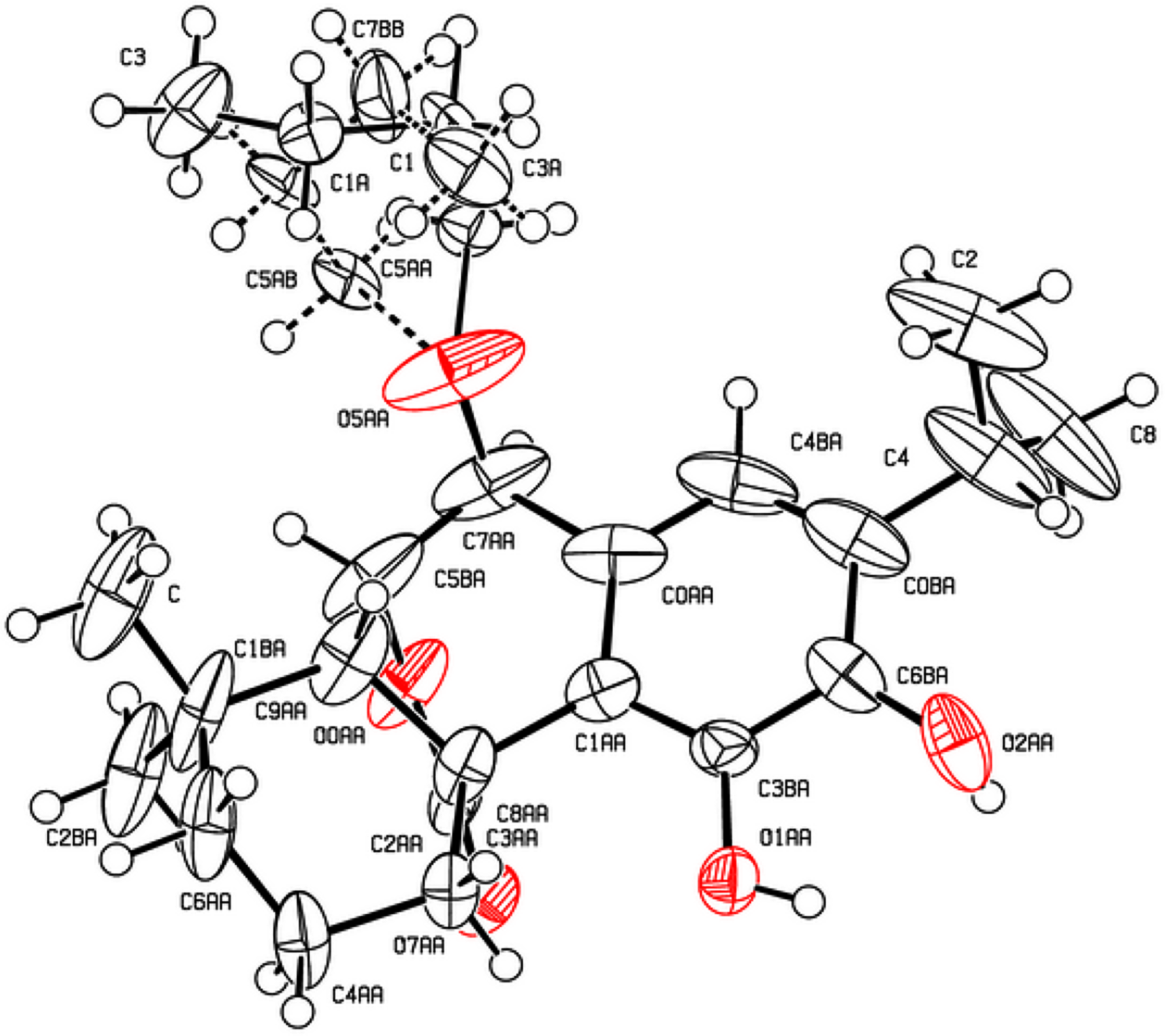
The x-ray crystal structure of
1H NMR (500 MHz) and 13C NMR (125 MHz) Spectroscopic Data of Compound

Abbreviation: NMR, nuclear magnetic resonance.
Compound
Experimental
General
The NMR data were acquired with Bruker Advance 500 MHz spectrometers (Bruker AVANCE NEO 500). The HRESIMS were measured on a Thermo Exactive Orbitrap mass spectrometry (Thermo Fisher Scientific), the IR spectra on a Bruker VERTEX 70 FT-IR spectrometer, the ultraviolet (UV) spectra on a Shimadzu 160 UV/VIS Spectrometer, the optical rotations on an Anton Paar MCP 300 polarimeter with 1 dm cell, and the x-ray diffraction data on a dual-source Rigaku Oxford Diffraction Supernova diffractometer (using graphite-monochromated Cu K α radiation). The structure was solved with the Superflip program using charge flipping and refined with the ShelXL program. Sephadex LH-20 (Amersham Pharmacia Biotech), D101 macroporous resin (Sinopharm Chemical Reagent Co., Ltd) and YMC Gel (ODS-A, 12 nm, S-50 μm, YMC Co.) were used for column chromatography. Preparative high-performance liquid chromatography was carried out on a Waters 1525 instrument with a 2598 detector (Waters), using an YMC-Pack ODS-A column (250 mm × 20 mm, 5 μm). TLC was conducted on Merck silica gel 60 F 254 plates (Merck KGaA).
Plant Material
The leaves of rosemary were purchased from Yulin, in August 2015, and were authenticated by Professor Yin Li, School of Pharmacy, Southwest Minzu University, China. A voucher specimen (ID 20150803) has been deposited in the Herbarium of Materia Medica, School of Biotechnology and Health Sciences, Wuyi University, Jiangmen, China.
Extraction and Isolation
The dried leaves of R officinalis (12.0 kg) were soaked (extracted) with EtOH (3 × 25 L) at room temperature. The solvent was evaporated under reduced pressure to yield 561.5 g of extract. The crude extract was suspended in water (2.5 L) and then successively solvent partitioned with n-hexane, acetate, and n-butanol. Four layers with increasing polarity were obtained: hexane-soluble (170.5 g), EtOAc-soluble (77.0 g), BuOH-soluble (85.0 g), and H2O-soluble (114.6 g) fractions. The hexane soluble was subjected to D101 resin and eluted with water-EtOH (1:0, 7:3, 1:1, 3:7, 1: 9, and 0:1, vol/vol) to afford 12 fractions (H1-H12). Fraction H1 (1.61 g) was further separated by CC using silica gel (4:1, light petroleum-EtOAc) to give compound
7-Butoxyrosmanol (1 )
Colorless needle crystals; mp 232 to 235 °C;
Crystallographic data for
Pancreatic Lipase Assay
The pancreatic lipase activity was measured according to the previously reported method, with a minor modification. 14 The pancreatic lipase (50 U/mL) and 4-methylumbelliferyl oleate (4-MU oleate, 0.1 mM) were dissolved in a buffer consisting of 13 mM Tris-HCl, 150 mM NaCl, and 1.3 mM CaCl2 (pH 8.0). The tested sample (25 μL, dissolved in 10% DMSO) was preincubated with 25 μL of lipase solution (50 U/mL) at 25 °C for 10 min, and then, 50 µL of 0.1 mM 4-MU oleate solution was added. The reaction mixture was incubated at 25 °C for 30 min. Fluorescence was then analyzed with an excitation of 355 nm and an emission wavelength of 460 nm by using a microplate reader (Biotek NEO2).
Conclusions
A new phenolic diterpene, 7-butoxyrosmanol (
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221075854 - Supplemental material for A New Phenolic Diterpene From the Leaves of Rosmarinus officinalis
Supplemental material, sj-docx-1-npx-10.1177_1934578X221075854 for A New Phenolic Diterpene From the Leaves of Rosmarinus officinalis by Tingwen Zhang, Deng-Gao Zhao, Shuting Li, Kun Zhang, Mei-Li Yang, Xuan Huang, Leyi Li and Yan-Yan Ma in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Jiangmen Program for Innovative Research Team (No. 2018630100180019806), the Department of Education of Guangdong Province (No. 2017KSYS010), and the Scientific Research Foundation of Shaanxi Provincial Key Laboratory (No. 18JS008).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
