Abstract
Objective
Hemoglobin (Hb) glycation occurs due to persistent hyperglycemia caused by insulin secretion abnormalities. Methylglyoxal (MG) causes post-translational modifications of proteins, including hemoglobin, generates cross-links and some advanced glycation end products (AGEs), but is not responsible for the formation of glycated hemoglobin.
Methods
The current study has sought to investigate the ability of the aqueous extract of Nigella sativa L. seeds (NS) and its bioactive constituent, thymoquinone (TQ), to prevent glycation and aggregation in the MG-derived Hb-AGE production. Molecular docking was utilized to study the interaction between the hemoglobin (Hb) active site residues and potential inhibitors.
Results
Our data demonstrated that NS and TQ inhibited the fructosamine content by 32.53 ± 0.12% and 20.72 ± 0.62%, fluorescent AGEs by 62.34% and 38.69%, and scavenge carbonyl content by 26.89 ± 0.011% and 17.08 ± 0.007% respectively. Furthermore, the inhibition of Hb aggregation induced by MG in the presence of NS and TQ by 42.89 ± 0.60% and 33.33 ± 0.79%. The findings showed that NS and TQ effectively inhibit MG-derived Hb-AGE formation. Both NS and TQ exhibited a significant antioxidant effect, effectively preventing the glycoxidation of DNA.
Conclusion
The study revealed a significant positive correlation between the antiglycation and anti-aggregation potential. Through their antioxidant potential, NS and TQ may reduce the production of MG-derived Hb-AGE formation. These results have significant implications for the development of potential herbal treatments aimed at preventing or treating complications associated with (AGEs) in diabetes.
Introduction
The Maillard reaction, or non-enzymatic glycation, is the spontaneous post-translational alteration of proteins/amino acids by reducing sugars. The molecules formed from this exposure are referred to as advanced glycation end products (AGEs). 1 AGEs are implicated in the progression of several chronic diseases, including diabetes, cardiovascular diseases, neurodegenerative disorders, and renal diseases. 2 Studies have also investigated AGE effects on mitochondrial dysfunction and cellular aging, particularly in neurodegenerative diseases like Alzheimer's, where AGEs’ accumulation has been linked to amyloid plaque formation. 3 When AGEs interact with their receptors of advanced glycation end products (RAGE) on the plasma membrane, it causes changes in gene expression, production of free radicals, intracellular signaling, and the release of pro-inflammatory chemicals. 4 Compounds that can disrupt the interaction between AGEs and their receptor RAGE have sparked a lot of attention. For example, RAGE Antagonists directly inhibit the binding of AGEs to RAGE, thereby preventing the downstream signaling that leads to inflammation and oxidative stress. For instance, Azeliragon (TTP488) is a small molecule RAGE inhibitor that has been investigated for its potential in treating Alzheimer's disease. 5 Natural compounds like curcumin and resveratrol have demonstrated potential in inhibiting the formation of Advanced Glycation End-products (AGEs), thereby reducing their accumulation and subsequent interaction with the receptor RAGE. 6 Alagebrium (ALT-711) is a compound investigated for its ability to break existing AGE-induced crosslinks in the vasculature, potentially improving arterial stiffness and function. 7
Glycation has been identified as a primary underlying cause of various problems such as diabetes, cardiovascular disease, neuropathy, retinopathy, and nephropathy.8,9 Long-term diabetes can result in a rise in glycated hemoglobin levels (HbA1c). HbA1c is glycated hemoglobin in which blood glucose has altered the valine residue at the β-subunit's N-terminus. It forms in the body when the oxygen-transporting protein hemoglobin interacts with sugar molecules and becomes glycated. 10 Diabetes produces oxidative stress, leading to a faster progression of the disease consequences. Antioxidants can help lessen these negative effects by chelating free radicals and donating electrons and protons, which stabilize oxidative stress products that might otherwise be harmful. 11
One of the most effective AGE precursors is methylglyoxal (MG) (Figure 1A), a reactive dicarbonyl compound formed by glycolysis. 12 The rate at which MG reacts with proteins is dramatically 50,000 times higher than glucose, but has a plasma concentration of around 25,000 times lower than glucose. 13 Thus, MG rapidly forms glycation adducts on extracellular as well as cellular proteins, 14 lipids, 15 and DNA, 16 potentially changing how they function. The MG-derived hydroimidazoline 1 (MG-H1) is a significant MG-derived AGE, 17 and N2-carboxyethyl-2′-deoxyguanosine (CeDG) is the predominant MG-DNA adduct. 18 MG is a highly reactive compound that primarily reacts with arginine residues in proteins. The cross-linking of cellular and extracellular proteins and cell malfunctioning is caused by MG modification to proteins. As a result of its link to genomic instability, MG-mediated genomic DNA modification poses serious risks. 19 Although hemoglobin glycation is well known and has been extensively examined, there have been very few reports on its alteration by MG. It is well known that glycation and AGE-induced toxicity have been associated with increased production of free radicals. Therefore, agents with high antioxidant activity that mop up free radicals can prevent the development of AGEs. Some plant extracts can trap or scavenge methylglyoxal, reducing its harmful effects on the body. This can help prevent the formation of AGEs and reduce the risk of diabetic complications.

2D Structure of Methylglyoxal and Thymoquinone. A) The 2D Structure of MG (PubChem ID: 880) and B) the 2D Structure of TQ (PubChem ID: 10281) Were Retrieved From https://pubchem.ncbi.nlm.nih.gov/.
Nigella sativa, a highly valued nutraceutical plant with a wide range of health advantages, is gaining popularity among researchers and pharmaceutical companies. 20 Herbal medicines to treat chronic illnesses, such as diabetes, have become a global trend in recent years. Since the beginning of time, cultures in the Mediterranean region have employed Nigella. sativa. This medicinal herb has black seeds famous for their terpenoid content, particularly Thymoquinone (Figure 1B), and fatty acids like linoleic acid, oleic acid, etc 21 Current research on N. sativa highlights its significant therapeutic potential in managing diabetes, cardiovascular diseases, cancer, and neurodegenerative disorders due to its antioxidant, anti-inflammatory, and immunomodulatory properties, primarily attributed to thymoquinone. 22 Studies show promising results in reducing oxidative stress, inflammation, and AGE formation, though further clinical trials are needed to confirm its efficacy and safety. 23 In the present study, Nigella sativa L. seed extract (NS) and its bioactive constituent, Thymoquinone (TQ; Figure 1B) have been tested for antiglycation and antioxidant capabilities. The specific focus on how NS and TQ affect BSA-MG glycation is novel and could lead to new insights into their bioactivity. This novelty underscores the potential for groundbreaking findings that have not been previously reported.
Material and Methods
Materials
The acquisition of the specified chemicals and reagents for the current research was carried out through commercial suppliers. A list of the key chemicals and reagents used has been mentioned here. Aminoguanidine (AG), hemoglobin (Hb), methylglyoxal, sodium phosphate dibasic, sodium phosphate monobasic, 5,5’-dithiobis (2-nitrobenzoic acid) (DTNB), guanidine hydrochloride, trichloroacetic acid (TCA), and thymoquinone (TQ), were sourced from Sigma Aldrich. pBR322 plasmid DNA was procured from ThermoFisher Scientific. Furthermore, Congo red, sodium azide, nitrogen blue tetrazolium (NBT), and 2,4-dinitrophenylhydrazine (DNPH) were procured from HI Media, Mumbai. All other chemicals used in the research were of high analytical grade.
Collection of Plant Material
The seeds of Nigella sativa L. were obtained from Dr A. Sattar at the Royal College in Mumbai. The plant's taxonomic identification and classification were officially verified by the Institute of Herbal Science, which is associated with the Plant Anatomy Research Centre in Chennai, India. The authenticity and taxonomic recognition of the plant were documented with an official certificate (Certificate Number - PARC/2019/3911) issued by the research center. This certificate ensures that the seeds were properly identified and classified, adhering to scientific standards for plant taxonomy.
Preparation of Nigella sativa L. Seed Extract (NS) and Thymoquinone (TQ)
A Soxhlet apparatus was used to extract the air-dried, powdered seeds. The resulting seed powder was properly cleaned in distilled water before 50 g of it was packed in a Soxhlet apparatus with 500 ml of distilled water. To produce the crude extract, all the filtrate was pre-concentrated using a thermostat at 50–60 °C. The extract was then diluted in water to the desired concentration and kept at −20 °C. The yield was calculated as follows: % yield = (Dry weight of extract ÷ Dry weight of plant material) × 100. For every 100 grams of N. sativa seeds processed, 16.8 grams of the extractable substances were obtained in the form of the aqueous extract. The commercially available thymoquinone was dissolved in Dimethyl Sulfoxide (DMSO).
Characterization of NS and TQ Using FTIR Spectroscopy
Using an FTIR RFPC 5301 (Perkin Elmer) spectrometer, Fourier transform infrared spectroscopy (FTIR) was performed on Nigella sativa L. seed powder and TQ powder. The resulting peaks were evaluated using a standard IR spectra database. 24
In Vitro Glycation of Hemoglobin (Hb) Induced by Methylglyoxal (MG)
In vitro glycation of Hb was carried out using an earlier approach described by Rubab et al (2021). 25 Briefly, 0.1 M phosphate buffer (pH 7.4), 1 mg/ml Hb, 5 mM MG, and 0.02% sodium azide were incubated in the presence/absence of NS (100 µg/ml) and TQ (100 µM) dissolved in DMSO for 4 weeks at 37 °C. Aminoguanidine (10 mM) was used as a standard.
Determination of Soret Band/Peak
A Soret peak, also known as a Soret band, is a strong peak in the visible spectrum blue wavelength range. The red band shift was measured over a wavelength range of 380–450 nm using a Shimadzu UV 1800 spectrophotometer. 26
Measurement of Browning
To assess the formation of glycated products, the browning was measured using a Shimadzu UV 1800 spectrophotometer at a specific wavelength, 420 nm.. 27 The inhibition percentage was calculated with the following formula: Inhibition (%) = (A0-A1)/A0 × 100; where A0 refers to the absorbance of the control, and A1 refers to the absorbance of the sample.
Estimation of Fructosamine Content Formation
The Nitroblue tetrazolium (NBT) assay was used to measure the level of fructosamine, the Amadori product, using an earlier approach described by Kumar et al (2022). 28 The glycated samples (10 μl) of BSA in the presence of plant extract were incubated with 500 μmol/litre NBT (100 μl) at 37 °C for 15 min. The intensity was measured at 530 nm and an extinction coefficient of 1-deoxy-1-morpholino-fructose (DMF) (12,640 cm−1mol−1) was used for the calculation of the amount of fructosamine content.
Estimation of Carbonyl Groups
To estimate the protein oxidation level in glycated Hb, the formation of carbonyl groups was measured using a slightly modified version of a method developed by Levine et al (1990). 29 In this method, 0.4 ml of 10 mmol/liter DNPH was added to glycated samples (0.1 ml) and incubated in the dark for an hour. Then, 20% (w/v) trichloroacetic acid (0.5 ml) was added and placed in ice for protein precipitation for 5 min. Centrifugation at 10,000 rpm for 10 min at 4 °C was carried out, following the washing of the protein pellet with 0.5 ml of an ethanol/ethyl acetate (1:1) mixture three times. Lastly, the pellet was resuspended in 0.1 ml of 6 M guanidine hydrochloride. The intensity was recorded at 370 nm using a Shimadzu UV-1800 spectrophotometer, and an extinction coefficient of 22,000 cm−1mol−1 was used to calculate carbonyl content.
Estimation of Thiol Groups
The free thiol group of native and modified Hb was measured using an earlier approach described by Ellman (1959). 30 70 µl of glycated samples were incubated for 15 min with 5 mM DTNB (5,5-dithio-bis in 0.1 M PBS, pH 7.4, at 25 °C. At 410 nm, the absorbance of the samples was measured. The L-cysteine (0-1.5 mM) was used as a standard, and the concentration of free thiols was determined and expressed as nmol/mg protein.
Estimation of Total Fluorescent AGE Formation
The formation of fluorescent AGEs is assessed by monitoring the fluorescence emission at 440 nm after exciting the sample at 370 nm using a spectrofluorometer (Varian, Cary Eclipse model). 31 The glycated sample was diluted to 1 mg protein content and excited at 370 nm wavelength. The emission was recorded in the range of 380–600 nm. % inhibition was determined from the observed fluorescence: Fluorescent AGEs inhibition (%) = [1-(F1/F0)] × 100; where F1 refers to the fluorescence intensity of the sample and F0 refers to the fluorescence intensity of the control.
Estimation of Individual AGEs Formation
In the context of AGEs, certain compounds like argpyrimidine, pentosidine, vesperlysine, and crossline are known to exhibit auto-fluorescence at specific excitation and emission wavelengths. The glycated sample was diluted to 1 mg protein content and was analyzed for different fluorescent AGEs at marked excitation and emission wavelengths 32 represented in Table 1. % inhibition was determined from the observed fluorescence: Fluorescent AGEs inhibition (%) = [1-(F1/F0)] × 100; where F1 refers to the fluorescence intensity of the sample and F0 refers to the fluorescence intensity of the control.
Excitation and Emission Wavelengths for Measurement of Individual AGE)

Determination of Inhibition of Hb Aggregation Induced by MG
Estimation of Amyloid β-Structures Using Congo red Assay
Congo red staining is a qualitative approach to identifying amyloids in vitro. 33 Glycated samples (50 µl) were incubated for 20 min with 100 µM Congo red dye (50 µl) and then the absorbance was measured at 530 nm. The inhibition percentage has been calculated using the same formula as mentioned in browning.
Estimation of Amyloid β-Structures Using Thioflavin T Assay
Thioflavin T (ThT) selectively binds to amyloid fibrils, which are typically rich in β-sheet structures, causing a distinct shift in its fluorescence properties. 34 In summary, 20 µM ThT (dissolved in 0.1 M PBS; pH 7.4) was introduced to the glycated samples (100 µl), and the mixture was incubated for 30 min at 25 °C. The samples were excited at a wavelength of 450 nm and the resulting fluorescence emission was measured at 600 nm. The % inhibition was determined from the observed fluorescence: Inhibition (%) = [1-(F1/F0)] × 100, where F1 refers to the fluorescence intensity of the sample and F0 refers to the fluorescence intensity of the control.
Antioxidant Activity Using the DPPH Method
The antioxidant activity of the samples was determined by testing their ability to scavenge free radicals using the DPPH technique as described previously with slight modifications. 35 1 ml of test samples (NS and TQ) at different concentrations have been added to 0.2 mM DPPH (500 µl) in methanol and incubated at 37 °C for 30 min in the dark and finally absorbance was measured at 517 nm. Ascorbic acid was used as a positive control. The scavenging property is measured spectrophotometrically and calculated with the following formula: % Inhibition = (A0–A1 /A0) × 100%, where A0 refers to the absorbance of the control and A1 refers to the absorbance of the test sample.
In Vitro Glycation of pBR322 Plasmid DNA Induced by MG
The glycation of pBR322 plasmid DNA induced by MG was studied by Ali et al, 2022. 36 DNA (0.25 μg) was incubated with lysine (20 mM), MG (200 mM), and FeCl3 (100 μM) with/without ie AG, NS, and TQ at a concentration of 10 mM, 100 µg/ml and 100 µM, respectively at 37 °C for 3 h. Following incubation, samples were separated on a 1% agarose gel electrophoresis and visualized using Gel-Doc. For agarose gel electrophoresis, 20 μl of samples were mixed with 4 μl gel loading dye (6X) and loaded onto 0.8% agarose gel (1% dissolved in 1X TAE buffer and 30 μl of ethidium bromide added). Agarose gel electrophoresis was done at 70–80 V until the dye band ran 2/third of the gel length. Subsequently, the gel was visualized under UV in the Gel-Doc, and the bands were analyzed.
Molecular Docking
Template Search and Energy Minimization
The human hemoglobin protein sequence was used for a BLAST search, 37 and the template with the highest identity and query coverage was selected for structure validation and docking. We downloaded the chosen template with the best resolution from the Protein Data Bank 38 and performed energy minimization using GROMACS software. 39 Thymoquinone, with a PDB ID of 7SJG, 40 was downloaded from the Protein Data Bank and processed using the FROG2 online server for conformer generation. 41 Additionally, methylglyoxal was sourced from the PubChem database 42 and its stable conformers were generated using the FROG2 server.
Protein-Ligand Docking
We docked the energy-minimized hemoglobin individually with thymoquinone and methylglyoxal using AutoDock 1.5.6 software. 43 Additionally, we applied Kollman charges to the protein and Gasteiger charges to the ligand before docking. The grid for thymoquinone was positioned based on the active site information gathered from the literature review. The grid dimensions for thymoquinone were set to 28 Å, 46 Å, and 32 Å along the x, y, and z axes, respectively, with the grid box centered at −4.66 Å, 4.22 Å, and 2.38 Å for the x, y, and z coordinates. Grid maps were generated using AutoGrid 4.0 and AutoDock 4.0, and 10 conformers were produced using the Lamarckian Genetic Algorithm (LGA) for the ligand. For methylglyoxal, the grid size was set to 24 Å, 34 Å, and 26 Å along the x, y, and z axes, respectively, based on information from the literature.44, 45 We maintained the grid size for thymoquinone at 28, 46, and 32 for x, y, and z, respectively. The grid box was set to −4.66 Å, 4.22 Å, and 2.38 Å for x, y, and z, respectively. The AutoGrid 4.0 and AutoDock 4.0 programs were used to generate grid maps, and 10 conformers were generated using a LGA for the compound. Here to the grid box size is set to −9.69 Å, 13.00 Å, and 213.16 Å for x, y, and z, respectively. Similar steps were followed for the docking of the ligand and docked complexes were visualized using Discovery Studio software 2024. 46
Statistical Analysis
Statistical significance was determined using a one-way analysis of variance (ANOVA) conducted in GraphPad Prism software version 8.0, with sets of experiments performed in triplicate (n = 6). p ˂ 0.05 was considered for the significance of the probability of occurrence. The results are presented with the standard error, and Tukey's multiple comparison test was applied to assess differences between treatments.
Results
FTIR Spectrum Analysis
The FTIR spectrum analysis was performed to identify the biomolecules present in the Nigella sativa L. seed powder (Figure 2A) and the commercially available TQ powder (Figure 2B). The intense band present at 2924.09 cm−1 of NS and 2962.66 cm−1 of TQ corresponds to the C-H stretching of aliphatic groups, which is slightly shifted from the previously reported value of 2924 cm−1 for TQ. A weaker band observed at a higher wavenumber (∼3040 cm−1) was attributed to the stretching of the vinylic C-H group in the C = C-H structure, which has been previously reported at 3041 cm−1 according to AIST: Spectral Database for Organic Compounds, SDBS, 2003. 47 The characteristic strong stretching band of the carbonyl group of cyclohexadiene is observed at the wavenumber at 1631.78 cm−1 in TQ.

A) FTIR Spectral Analysis of the Nigella sativa L. Seeds Powder and B) FTIR Spectral Analysis of Thymoquinone (TQ) Powder. The X-axis Represents the Wavenumber (cm−1), and Y-axis Represents the % Transmittance.
Inhibitory Effect of NS and TQ on Glycation of Hemoglobin (Hb) Induced by Methylglyoxal (MG)
Determination of Band Shift of Heme Proteins
A comparison of the band shifts in the Soret band of hemoglobin has been shown in Figure 3. Maximum variations have been observed in the case of the glycated system (Hb + MG). The test sample ie, Hb + MG + AG, Hb + MG + NS, and Hb + MG + TQ do not affect the absorptivity of heme proteins.

Soret Band Statue of Heme Proteins in Hemoglobin Induced by MG in Vitro.
Measurement of Browning
A simple and fast method to assess the progress of Maillard reactions is to measure the degree of browning. Browning value increased as heating duration increased and was notably higher in the Hb + MG (control) compared to the others. The absorbance of the samples after the 28-day incubation is shown in Table 2. The inhibition (%) was calculated using the following mathematical formula: Inhibition (%) = [(Acontrol−Asample)/Acontrol)] × 100. The browning value increased as the duration of incubation increased and was impressively high in the glycation system as compared to native Hb. For the glycated system, the percentages of inhibition in browning by AG, NS, and TQ were 29.14 ± 0.073, 25.87 ± 0.086%, and 17.60 ± 0.092%, respectively. This indicates that thymoquinone, an active constituent of N. sativa L. seed extract, plays a major role in inhibiting browning. These results agreed with the findings of Rubab et al (2021), who reported that the presence of NS significantly reduced the amount of browning in the glucose-induced glycation system. 25
The effect of AG (10 mM), NS (100 µg/ml), and TQ (100 µM) on Browning in Terms of Absorbance.

Hb: hemoglobin; MG: methylglyoxal; AG: aminoguanidine; NS: Nigella sativa L. seed extract; and TQ: Thymoquinone. Results are represented as mean ± SD.
Inhibitory Effect of NS and TQ on Fructosamine Production
Fructosamines are considered to be early-stage glycation adducts. The most common assay available for fructosamine measurement in serum is the colorimetric-based assay. This assay utilizes the reduction of the dye nitroblue tetrazolium (NBT) to formazan. The rate of formazan formation is directly proportional to the fructosamine concentration and is measured with the spectrophotometric technique. 48 Following Hb incubation with MG, fructosamine levels rise but the formation of fructosamine content in the native Hb was found to be negligible (Figure 4). After 28 days of incubation, the amount of fructosamine levels decreased by 36.81 ± 0.42% in the presence of AG (10.71 ± 0.18 µmol/mg protein), 32.53 ± 0.12% in case of NS (11.43 ± 0.27 µmol/mg protein) and 20.72 ± 0.62% in the presence of TQ (13.41 ± 0.40 µmol/mg protein), respectively in comparison to the control, ie, Hb + MG considered as 100% (16.94 ± 0.013 µmol/mg protein). Researchers have shown that natural compounds, such as polyphenols, polysaccharides, terpenoids, vitamins, and alkaloids, are good candidates for discovering new drugs to inhibit AGE formation. 5

The Effect of AG, NS and TQ on NBT Reduction in Hb Glycation Induced by MG. Hb: Hemoglobin; MG: Methylglyoxal; AG: Aminoguanidine; NS: Nigella sativa L. Seed Extract; and TQ: Thymoquinone. The Data Represented the Mean ± SD of Three Independent Experiments. Compared to the Control (Hb + MG), all the Results are Statistically Significant at p < 0.05.
Inhibitory Effect of NS and TQ on Carbonyl Content Production
Protein carbonyls are one of the most common markers of protein oxidation formed during glycation, and accumulation of protein carbonyls has been observed in several human diseases, including Alzheimer's disease, diabetes, inflammatory bowel disease, and arthritis, just to name a few. 49 When compared to Hb alone as a negative control, Hb that had been glycated for 28 days had more protein-bound carbonyl groups (Figure 5). AG, NS, and TQ prevented the development of carbonyl groups linked to proteins, as compared to the control (Hb + MG). In comparison to the glycated Hb (13.46 ± 0.42 µmoles/mg protein), the amount of free carbonyl groups in Hb drastically decreased by 28.60 ± 0.39% in the presence of AG (9.61 ± 0.36 µmoles/mg protein), 26.89 ± 0.011% in case of NS (9.84 ± 0.70 µmoles/mg protein) and by 17.08 ± 0.007% in the presence of TQ (11.16 ± 0.41 µmoles/mg protein). Chayaratanasin et al (2021) reported that Clitoria ternatea L. flower extract (0.25-1 mg/ml) reduced protein carbonyl content in glycated BSA (p < 0.05). A 22% reduction of protein carbonyl content was detected in the BSA/MG system with flower extract. 50

The Effect of NS and TQ on Carbonyl Group Formation in Hb Glycation Induced by MG. Hb: Hemoglobin; MG: Methylglyoxal; AG: Aminoguanidine; NS: Nigella sativa L. Seed Extract; and TQ: Thymoquinone. The Data Represented the Mean ± SD of Three Independent Experiments. Compared to the Control (Hb + MG), all the Results are Statistically Significant at p < 0.05.
Inhibitory Effects of NS and TQ on Thiol Group Formation
The DTNB reagent was used to assess the free thiol groups in the MG-induced Hb glycation system. In comparison to Hb, the amount of free thiol group formation dramatically declined in the glycated system (Hb + MG), while the presence of AG, NS, and TQ in the system improved thiol groups. The amount of thiol groups found in native Hb, glycated system (Hb + MG), glycated system in the presence of NS (Hb + MG + Ext) and TQ (Hb + MG + TQ) are 0.248 ± 0.001 mM, 0.096 ± 0.0006 mM, 0.198 ± 0.003 mM, and 0.239 ± 0.0012 mM respectively (Figure 6).

The Effect of NS and TQ on Thiol Group Formation in Hb Glycation Induced by MG. Hb: Hemoglobin; MG: Methylglyoxal; AG: Aminoguanidine; NS: Nigella sativa L. Seed Extract; and TQ: Thymoquinone. The Data Represented the Mean ± SD of Three Independent Experiments. In Comparison to the Control (Hb + MG), all the Results are Statistically Significant at p < 0.05.
Inhibitory Effect of NS and TQ on Fluorescent AGEs Formation
AGEs are brown substances formed in the late stage of the glycation reaction. These AGEs can alter the normal functions of proteins and disturb the balance within cells. The proteins modified by AGEs are further linked to angiogenesis, cell proliferation, chemotaxis, and oxidative stress. 51 In the present research, it has been discovered that the Nigella sativa L. seed extract (NS) and its bioactive constituent, TQ, had beneficial effects on the suppression of AGEs, as measured by the Hb-MG glycation system (Figure 7). The Hb-MG model reveals a considerable production of fluorescent AGEs. It is interesting to note that TQ at a concentration of 100 µM inhibits AGEs effectively when compared to NS (100 µg/ml). After 28 days of incubation, glycated Hb showed considerably higher fluorescence intensities than native hemoglobin, demonstrating the development of glycated AGEs over time. The level of fluorescent AGE formation in the Hb-glycated system induced by MG dramatically decreased by 62.34% in the presence of NS and by 38.69% in the presence of TQ. Furthermore, AG reduced the formation of fluorescent AGEs by 69.95%. Jariyapamornkoon et al (2013) demonstrated that in week 4 of incubation, red grape skin extract (0.031-0.500 mg/ml) inhibited AGE formation by 55.23% to 63.52%, respectively. 52 A significant inhibition of AGEs formation (73.3%) was observed in fructose-induced glycated BSA when treated with AG (0.5 mg/ml). These findings align with the growing body of evidence supporting the anti-glycation potential of natural extracts.

The effect of NS and TQ on fluorescent AGEs formation in Hb glycation induced by MG in terms of fluorescence intensity. Hb: hemoglobin; MG: methylglyoxal; AG: Aminoguanidine; NS: Nigella sativa L. Seed Extract; and TQ: Thymoquinone.
Inhibitory Effects of NS and TQ on Individual AGE Formation
The Fluorescence spectra Showed That the Presence of AG, NS, and TQ Caused the Amounts of Individual AGEs (Argpyrimidine, Pentosidine, Vesperlysine as Well as Crossline) in the Glycated Sample to Decrease (Figure 8). The concentration of these four was raised in the glycated system (considered as 100%). After completion of incubation, it was discovered that AG, NS, and TQ caused a decrease in fluorescence intensity of argpyrimidine in the range of 23.98 ± 1.93% to 38,78 ± 2.18%, pentosidine in the range of 19.02 ± 1.16% to 34.02 ± 1.12%, vesperlysine in the range of 21.13 ± 2.14% to 33.21 ± 2.13%, crossline in the range of 14.28 ± 1.32% to 29.16 ± 3.23%, to drop.
Inhibitory Effect of NS and TQ on Hb Aggregation Induced by MG
Congo red Assay
The study commenced with the presumption that the aggregation of β-amyloid structures within the Hb and MG glycation system had reached 100%. Subsequently, a comparative analysis was conducted, examining the aggregation of glycated BSA in the presence of an aqueous extract of N. sativa seeds and TQ using a Congo red dye assay. In comparison to glycated systems, AG, NS, and TQ greatly reduced secondary structural modifications in Hb. When incubated with Hb and MG, the AG and NS showed the prevention of β-amyloid formation by 45.44 ± 0.82% and 42.89 ± 0.60%, whereas TQ showed inhibition by 33.33 ± 0.79% (Figure 9).

The Effect of AG, NS, and TQ on Individual AGEs Formation in Hb Glycation Induced by MG in Terms of Fluorescence Intensity. Hb: Hemoglobin; MG: Methylglyoxal; AG: Aminoguanidine; NS: Nigella sativa L. Seed Extract; and TQ: Thymoquinone.

The Effect of AG, NS and TQ on Protein Aggregation Formation in Hb Glycation Induced by MG in Vitro. Hb: Hemoglobin; MG: Methylglyoxal; AG: Aminoguanidine; NS: Nigella sativa L. Seed Extract; and TQ: Thymoquinone. The Data Represented the Mean ± SD of Three Independent Experiments. In Comparison to the Control (Hb + MG), all the Results are Statistically Significant at p < 0.05.
Thioflavin T Assay
The aggregation of proteins in the presence of N. sativa seed extract in the glycated system was also checked fluorometrically with thioflavin-T dye. Glycation of protein may have gradually triggered the formation of amyloid structures in Hb because the fluorescence intensity of thioflavin T was much higher in hemoglobin incubated with MG compared to the negative control (Hb). The results of this investigation for glycated Hb demonstrated that the AG, NS, and TQ suppressed the aggregation of thioflavin T-labelled amyloid by 68.30%, 61.23%, and 30.38%, respectively. Figure 10 displays the outcomes.

The Effect of NS and TQ on the Formation of Amyloid-β Products in Hb Glycation Induced by MG in Terms of Fluorescence Intensity. Hb: Hemoglobin; MG: Methylglyoxal; AG: Aminoguanidine; NS: Nigella sativa L. Seed Extract; and TQ: Thymoquinone.
Antioxidant Activity Using the DPPH Method
Table 3 demonstrates the results of the DPPH radical scavenging activity of NS and TQ in comparison to the well-known antioxidant ascorbic acid. They both demonstrated the donating capacity of protons. The NS had a stronger scavenging activity with an IC50 value of 0.64 mg/ml against the DPPH radical than TQ (IC50 value of 97.49 µM) comparable to ascorbic acid (IC50 value of 0.19 mg/ml).
Free Radical Scavenging Activities of Ascorbic Acid, NS, and TQ.

Inhibitory Effect of NS and TQ on Glycoxidation of DNA
Glycoxidation is a process that combines both oxidation and glycation, resulting in the formation of AGEs. This process is implicated in diabetes and its secondary complications, such as cardiovascular diseases, Alzheimer's disease, and nephropathy. Under oxidative stress, lipid peroxidation generates methylglyoxal (MG) and glyoxal (GO), which then participate in glycoxidation, ultimately leading to the formation of AGEs. 53 Figure 11 depicts how AG, NS, and TQ affect DNA cleavage caused by MG-mediated lysine glycation in the presence of Fe+3. The addition of MG and lysine to plasmid DNA enhanced the intensity of the open circular form of the bands, indicating an increase in strand breakage. However, adding lysine, MG, FeCl3, and AG/NS/TQ to plasmid DNA did not cause DNA cleavage; instead, the plasmid DNA persisted in its supercoiled shape.
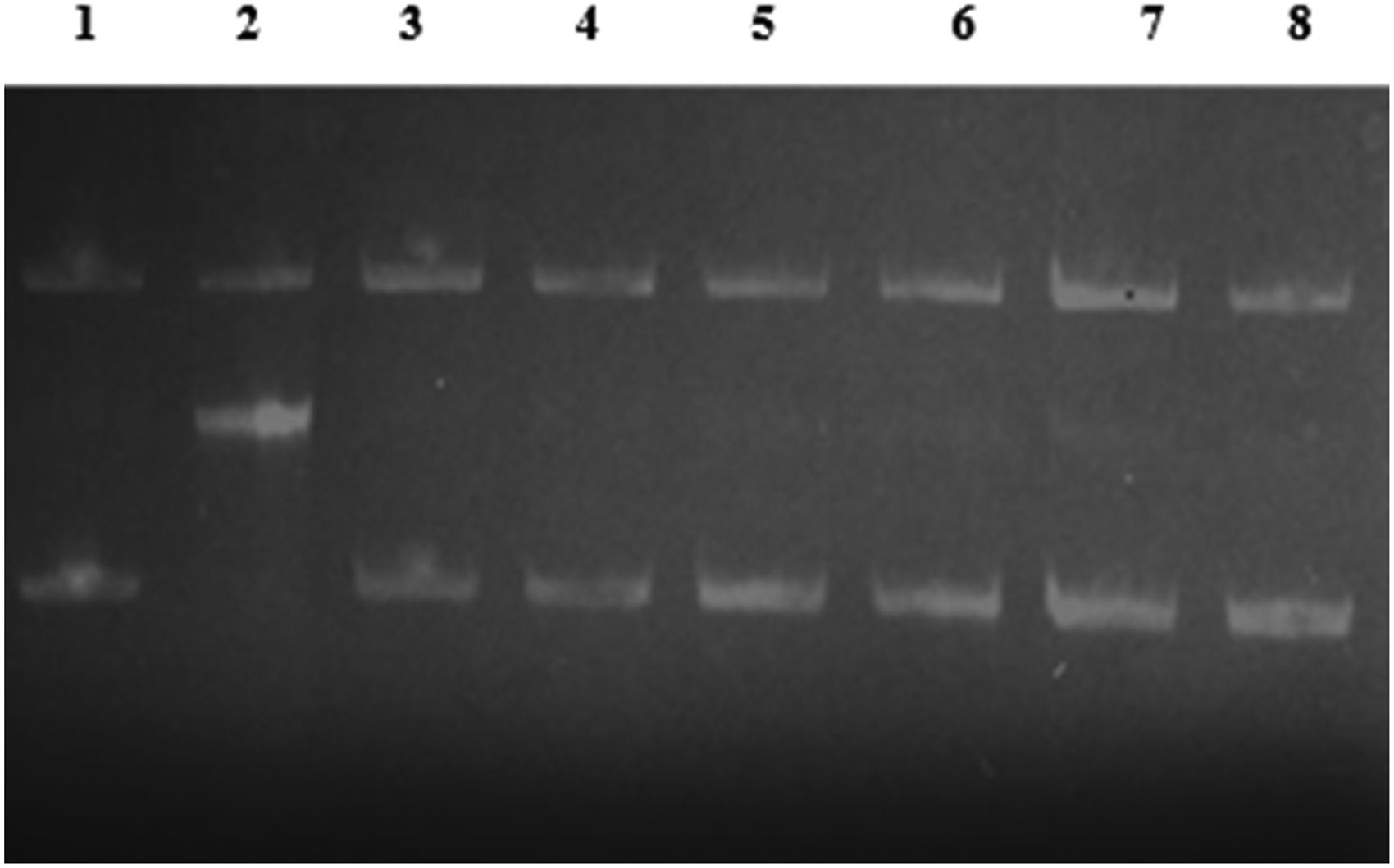
In Vitro Glycation of pBR322 Plasmid DNA Induced by MG. The Effects of NS and TQ on pBR322 Cleavage-Mediated by Glycation of Lysine with MG in the Presence of Fe+3. Lane 1: pBR322 Alone; Lane 2: pBR322 + Lysine + MG + FeCl3 (GS); Lane 3; pBR322 + AG; Lane 4: pBR322 + GS + AG; Lane 5; pBR322 + NS; Lane 6: pBR322 + GS + NS; Lane 7; pBR322 +TQ; Lane 8: pBR322 + GS + TQ.
Molecular Docking
Template Selection and Protein-Ligand Docking
The BLAST search identified PDB ID: 3B75 54 of glycated human hemoglobin as the potential hit with a resolution of 2.30 Å. We subjected this 3D structure to energy minimization and considered docking with the ligands thymoquinone and methylglyoxal separately into their respective binding sites, based on a literature review. According to the docking study, thymoquinone has a binding affinity of −5.8 kcal/mol and an inhibitory constant value of 51.08 µM. The docked complex with a higher binding affinity was visualized, confirming a single hydrogen bond with His58. However, there were four alkyl interactions with Phe43, His83, Leu91, and Val93. Concerning methylglyoxal, the binding affinity was −2.72 kcal/mol, with an inhibitory constant value of 10.17 micromolar. Structural visualization confirmed two hydrogen bonds (Arg92 and Arg141) with the ligand molecule (Figure 12).

Molecular Interaction between A) Hemoglobin and Methylglyoxal B) Hemoglobin and Thymoquinone.
Correlation Analysis among Antiglycation Assay of NS and TQ
The correlation analysis was used to investigate the relationship between the independent analytical methodologies for antiglycation and the anti-aggregation potential of NS and TQ. Total fluorescent AGE production was highly related to fructosamine and carbonyl content inhibition (Table 4). A significant linear correlation was found between ThT values with the inhibitory activity of MG-derived AGEs (R2 = 0.8724) as well as between ThT values with fructosamine and carbonyl content reduction (R2 = 0.9528, R2 = 0.9578). Thus, analytical criteria for protein glycation and aggregation revealed that NS and TQ both suppressed the MG-mediated glycation and aggregation process.
Pearson Correlation Analysis among Browning, Fructosamine Content, Carbonyl Content, Total Fluorescent AGEs, and β-Amyloid Aggregation by ThT.

Discussion
The capacity of the studied Nigella sativa L. seed extract (NS) and its bioactive TQ to suppress glycation was investigated by examining it at three levels: a) glycation reaction (early and late stage), b) hemoglobin oxidation, and c) β-amyloid aggregation. The study has allowed us to improve our understanding of Nigella sativa L. extract (NS) and the role of TQ as potent antioxidants and inhibitors of protein glycation and glycoxidation, which prevent methylglyoxal from modifying hemoglobin in vitro. It appears that both of them may operate at several stages of the glycation process and the AGEs production.
One of the ways to prevent glycation is through the inhibition of free radicals generated by protein glycation, which therefore inhibits protein modification. It is shown that the scavenging impact of polyphenols on free radicals produced by the glycoxidation process is closely related to their inhibitory activity against protein glycation. Analyzing the carbonyl content of proteins is a typical indicator of protein oxidation. 55 Disulfide bonds may develop when a protein's sulfhydryl group is oxidized. Conditions with a higher carbonyl content and a lower free sulfhydryl group stimulate the production of free radicals and oxidative stress, which can accelerate the progression of diabetes and its accompanying problem. 56 For patients who are at risk of developing diabetic problems, the positive benefits of Nigella sativa L. may be used to prevent or control AGE-mediated diseases.
In this study, the content of total fluorescent AGEs and individual AGEs (argpyrimidine, vesperlysine, pentosidine, and crossline) were determined. It has been demonstrated that NS and TQ inhibit the formation of AGEs throughout the glycation process. TQ appears to have significant antiglycation efficacy against MG-derived hemoglobin advanced glycation end products which indicates that it has a major role in the pharmacological properties of Nigella sativa L. Our data partially agrees with Losso et al (2011) findings. 57 They showed that the primary quinone from Nigella sativa L., thymoquinone, has an inhibitory effect on the glycation process. TQ reduced hemoglobin glycation by 39% at 20 μM. Wolffenbuttel et al (1996) proposed that the long-term modification of hemoglobin by advanced glycosylation end products (Hb-AGEs) could serve as a more reliable indicator of long-term glycemia in patients with diabetes. 58 In another study, it was shown that non-enzymatic glycation was considerably decreased at higher NS doses, indicating that NS has a dose-dependent inhibitory potential. 59 Researchers found that Nigella sativa L. seed extracts at 1 mg/ml concentration have shown significant preventive effects against the generation of early glycation products. 25 In particular, the extracts demonstrated a reduction of up to 30.57% (BSA-Rib) and 64.90% (Hb-Glu) in the formation of early glycation products. Researchers have observed that glycation can accelerate the formation of β-amyloid aggregates, which may contribute to metabolic and neurodegenerative disorders. 60 The Congo red assay and ThT-based fluorometric assay revealed that the presence of NS and TQ significantly reduced aggregation in the MG-mediated glycation system. Nigella sativa L. Nigella sativa L. has been identified as a promising drug candidate for reducing amyloid β-structures in Alzheimer's disease. 61 The low IC50 of DPPH free radical scavenging ability of NS, as well as TQ, showed that Nigella sativa L. seems to be active against DPPH radicals. Besides protein glycation, MG contributes to oxidative DNA damage and cell cytotoxicity, resulting in conditions like cell apoptosis. 62 Glycation and AGE-induced toxicity are known to be linked to increased free radical production. Therefore, agents with strong antioxidant properties, capable of neutralizing free radicals, can also inhibit the formation of advanced glycation end-products (AGEs). 63 In this regard, polyphenols from Nigella sativa L. can effectively serve as antioxidants and antiglycation agents in the diets of diabetics. 64 Generic nucleotide adducts are produced as a result of DNA glycation, some of which have been reported to exacerbate oxidative stress. When oxidized bases are removed, glycosylase enzymes can directly produce aldehydic apurinic/apyrimidinic (AP) sites in DNA. In addition to being linked to mutagenesis, carcinogenesis, and diabetes mellitus, DNA damage brought on by glycation is also thought to accelerate the disease. It has been hypothesized that the glycation of DNA, which produces nucleotide AGEs, is linked to a rise in mutation frequency and cytotoxicity. 65 The findings of the current investigation demonstrated their protective effects against ROS and glycation-induced structural changes in Hb. As per the docking report, thymoquinone displayed a stronger binding with the alpha subunit while interacting with positive and hydrophobic amino acids within the drug binding site. Thus, four alkyls and a single hydrogen bond were observed. However, methylglyoxal is preferred to bind to the terminal end of the alpha subunit along with positive amino acids like arginine.
Conclusion
Nigella sativa L. seed extract and thymoquinone demonstrate significant potential in inhibiting both early and advanced glycation end products, offering promising prospects for mitigating glycation-related damage. The antioxidants and phytoconstituents present in the extract play a pivotal role in blocking protein aggregation, countering MG-mediated glycation, and preventing glycoxidation in DNA. However, it is imperative to conduct further rigorous and extensive studies to fully assess the therapeutic efficacy of Nigella sativa L. seed extract and thymoquinone in the management of glycation-related diseases such as diabetes, cardiovascular diseases, and neurodegenerative disorders. Given the complexity of the pathophysiological mechanisms underlying these diseases, it is crucial to elucidate the precise molecular targets and mechanisms through which Nigella sativa L. and thymoquinone exert their protective effects. Comprehensive scientific investigations, including clinical trials, are essential to establish their full therapeutic potential, validate their mechanisms of action, and translate their benefits into clinical applications for these challenging and prevalent diseases.
Footnotes
Acknowledgements
The authors would like to extend their sincere appreciation to the Ongoing Research Funding Program, (ORF-2025-694), King Saud University, Riyadh, Saudi Arabia.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ongoing Research Funding Program, (ORF-2025-694), King Saud University, Riyadh, Saudi Arabia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
