Abstract
The inhibitory mechanism of engeletin against α-glucosidase was investigated for the first time by fluorescence spectroscopy and molecular docking. The results showed that engeletin could inhibit α-glucosidase in a noncompetitive inhibition mode with a half-maximal inhibitory concentration value of 48.5 ± 6.0 µg/mL (0.11 ± 0.014 mmol/L). It was found that engeletin could cause static fluorescence quenching of α-glucosidase by forming a complex with α-glucosidase. The thermodynamic parameters indicated that the combination of engeletin and α-glucosidase was driven by hydrophobic force. The molecular docking results confirmed that some amino acid residues of α-glucosidase (Trp391, Arg428, Glu429, Gly566, Trp710, Glu771) could interact with engeletin by hydrogen bonding.
α-Glucosidase (EC 3.2.1.20), mainly present in the small intestinal villi mucosal cells, is an important digestive enzyme in human metabolism, 1 accounting for maintaining the normal physiological functions of the human body. However, for diabetic patients, large amounts of saccharides are catalyzed by α-glucosidase to form glucose after a meal, which leads to postprandial hyperglycemia and causes damage to the patient’s health. 2 Therefore, in clinical practice, α-glucosidase inhibitors are usually used to reduce the activity of the enzyme in order to effectively delay the rise of postprandial blood glucose in diabetic patients, hinder the progression of the disease, and control the incidence of complications. 3 At present, common α-glucosidase inhibitor drugs include voglibose and acarbose, 4 but these have toxic side effects. Plants could be a rich and safe source of α-glucosidase inhibitors. 5,6 Recent studies have shown that many natural compounds, such as flavonoids, phenolic acids, and saponins, possess α-glucosidase inhibitory activity. 7,8
Engeletin (Figure 1), a flavanone rhamnoside, mainly derived from
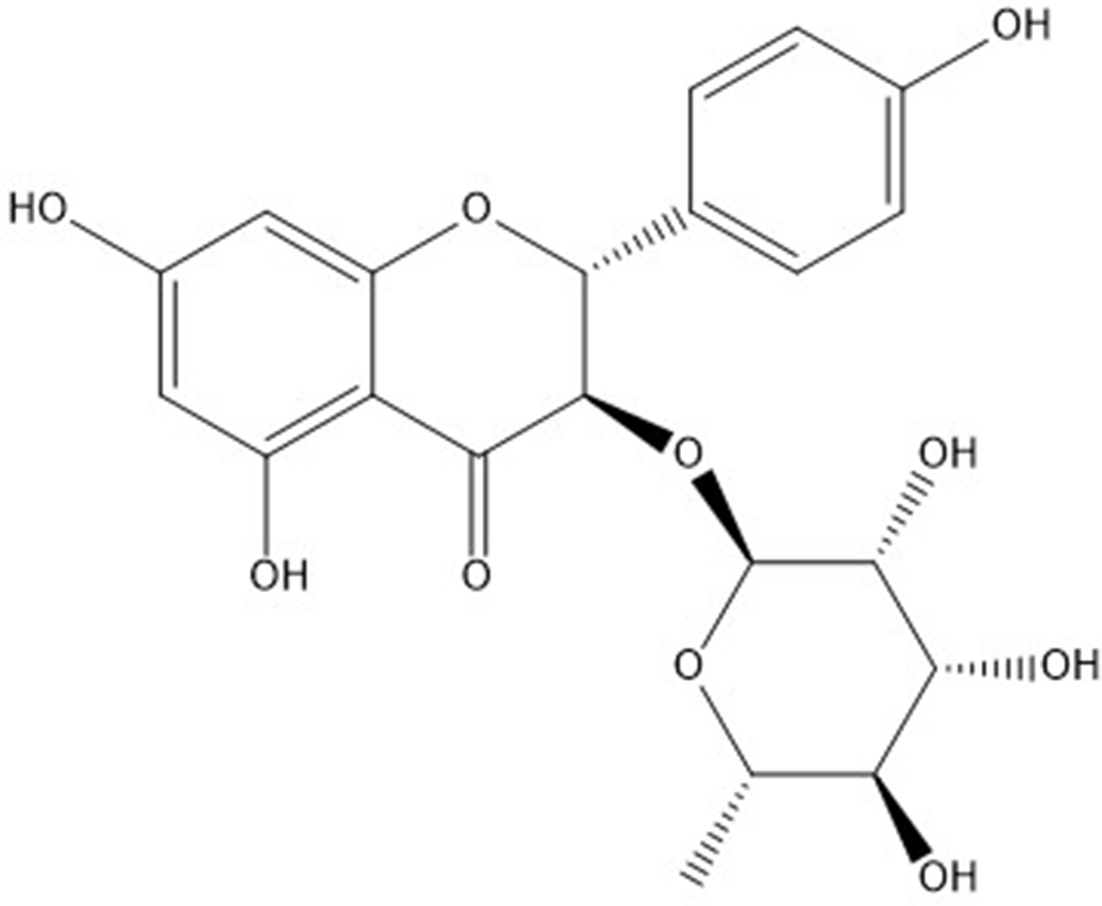
Chemical structure of engeletin.
Results and Discussion
Figure 2 shows the inhibitory activities of engeletin and acarbose on α-glucosidase at different concentrations. With an increase in concentration, their inhibitory effect also gradually increased. When the concentration of engeletin was 100 µg/mL, its α-glucosidase inhibition ratio was 65.6%. At a concentration of 3 mg/mL, the inhibition ratio of acarbose was only 61.2%. The inhibitory performance of engeletin (half-maximal inhibitory concentration [IC50] 0.11 ± 0.014 mmol/L) was significantly higher than that of acarbose (IC50 3.11 ± 0.22 mmol/L). Thus, it could be concluded that engeletin might be a strong α-glucosidase inhibitor with potential application in functional foods and medicines.

Inhibitory performance of engeletin and acarbose on α-glucosidase.
The reaction rates of enzymatic hydrolysis of

Lineweaver-Burk plot for the inhibition of engeletin on α-glucosidase.
By comparing the fluorescence spectra before and after the binding of biological macromolecules to other molecules, it is possible to infer the binding constant (


Effects of engeletin on the fluorescence spectra of α-glucosidase at 30 ℃ (A) and 37 ℃ (B).
Stern-Volmer Quenching Constants Between Engeletin and α-Glucosidase.

For the static fluorescence quenching process,

Binding Parameters Between Engeletin and α-Glucosidase.

In order to reveal further the binding mechanism of engeletin and α-glucosidase, the thermodynamic constants (Table 3) of their interaction were obtained by using the Van’t Hoff equation (3) and Gibbs free energy equation (4).


Thermodynamic Parameters Between Engeletin and α-Glucosidase.

The free energy change (Δ
The interaction between polyphenols and enzymes involves complicated structural changes, in which the enzyme may undergo orderly high-level structural reorganization, accompanied by new changes such as energy transfer and signal molecule transmission. 15 In recent years, molecular docking, quantum chemical calculation, and molecular dynamics have been applied to investigate the binding mechanism of polyphenols and enzymes. 16 The molecular docking simulation result could clarify the binding mode of engeletin and α-glucosidase (Figure 5).

Molecular docking result between engeletin and α-glucosidase: (A) Binding mode of engeletin and α-glucosidase, (B) hydrogen bonding between engeletin and α-glucosidase, and (C) hydrophobic interaction between engeletin and α-glucosidase.
As shown in Figure 5(A) and (B), engeletin formed hydrogen bonds with some amino acid residues of α-glucosidase (Trp391, Arg428, Glu429, Gly566, Trp710, and Glu771), among which Gly566 formed 2 hydrogen bonds with α-glucosidase, and the hydrogen bond lengths were 2.1 and 2.0 Å, respectively. However, GLu429, Arg428, Trp391, Trp710, and GLu771 formed 1 hydrogen bond with engeletin; their hydrogen bond lengths were 2.1, 2.3, 2.3, 1.9, 2.1 Å, respectively. The hydrophobic interaction is also summarized in Figure 5(C). Engeletin had hydrophobic interaction with many surrounding hydrophobic residues (Tyr709, Trp391, Gly566, Trp710, Glu771, Arg428, Glu429, Phe385, Phe786, Phe444, Arg387, Phe389, Val440, Trp789, Asp392, Trp715), which was consistent with the results of fluorescence spectroscopy.
Conclusions
Engeletin inhibits α-glucosidase in a noncompetitive inhibition mode, and its inhibitory capacity is significantly higher than that of acarbose. Fluorescence quenching analysis suggested that under the drive of hydrophobic force, engeletin could combine with α-glucosidase at a molar ratio of 1:1. Molecular docking confirmed that engeletin formed hydrogen bonds and hydrophobic interaction with some amino acid residues of α-glucosidase. The obtained results could promote the application of engeletin in foods and medicines.
Materials and Methods
Chemicals
Engeletin, acarbose, and
α-Glucosidase Inhibitory Assay

The inhibitory type of engeletin was analyzed from the Lineweaver-Burk plot. At different
Fluorescence Spectra
According to the previous method, 3 1 mL of sample solution of different concentrations was uniformly mixed with 4 mL of α-glucosidase solution. The mixture was incubated at either 30 °C or 37 °C for 10 minutes, and then the fluorescence spectrum was collected. The excitation wavelength was fixed at 295 nm, and the emission wavelength range was 320‐380 nm. Both the slit width values were set at 10 nm.
Molecular Docking
The binding mode of engeletin and α-glucosidase docking was obtained using the AUTODOCK 4.2 software. The 3-dimensional structure of engeletin was optimized by the density functional method (B3LYP/6‐31g+) with Gaussian 09 software. The crystal structure of α-glucosidase was from the RCSB PDB database (http://www.rcsb.org/, PDB number 4J5T). Docking was based on the genetic algorithm method.
Statistical analysis
Each measurement was repeated 3 times and expressed as mean ± SD.
Footnotes
Acknowledgments
The authors are very thankful to all the authors whose work has been cited in this paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Key Scientific and Technology Project in Henan Province of China (No. 202102110135).
