Abstract
Background/Objectives
Hypoxia is a severe pathological complication associated with many disorders, as oxygen insufficiency could decline normal cellular homeostasis. Hypoxia causes alteration of the coronary blood flow and reduction of the contractile performance, which eventually leads to myocardial dysfunction. Thus, the current study aims to explore the potential cardioprotective effect of liposomal curcumin (L-CR) against sodium nitrite (NaNO2)-induced hypoxic stress.
Methods
Hypoxia was induced by a single subcutaneous dose of NaNO2 (60 mg/kg), and a prophylactic intraperitoneal dose of either CR or L-CR was given two hours prior to NaNO2 intoxication. Oxidative stress, inflammatory and apoptotic responses in heart tissues were analyzed using molecular, histopathological and immunohistochemical techniques.
Results
NaNO2 caused a significant increase in creatine kinase-MB (CK-MB), lactate dehydrogenase (LDH), and C-reactive protein (CRP), as well as a decline in interleukin-10 (IL-10) levels. Additionally, the hypoxic heart exhibited a significant upregulation of cytochrome c and myocyte enhancer factor-2 (MEF-2), and downregulation of the nuclear factor erythroid-related factor 2 (Nrf-2). Moreover, NaNO2 induced cellular infiltration with a strong immunoreaction towards Apo-1/Fas, as displayed in the histopathological and immunohistochemical examinations. Using either CR or L-CR improved all the altered parameters.
Conclusion
L-CR demonstrated a significant modulation in the expression of some parameters including CK-MB, MEF-2, Nrf-2, and Apo-1/Fas compared to native CR against cardiac stress induced by NaNO2.
Introduction
Hypoxia is a condition that occurs when the amount of oxygen becomes insufficient to maintain normal cellular homeostasis. Depending on the severity and duration of the hypoxic conditions, hypoxia causes changing in coronary blood flow, 1 increasing heart size, 2 and decreasing contractile performance, which eventually cause myocardial dysfunction. 3 Severe oxygen deficiency can interfere with several cellular pathways, such as oxidative mechanisms of respiration, metabolic pathway, cell proliferation, 4 gene expression, 5 and ATP synthesis. 3 These changes occur in an endless cycle that can trigger each other and overwhelm the endogenous antioxidant capacity due to continuous reactive oxygen species (ROS) formation, eventually leading to oxidative myocardial damage and heart failure. 6
In the case of normal physiological conditions, there is a balance between ROS production and elimination processes. Hypoxia alters the electron transport chain by changing the activity of the mitochondrial oxidative phosphorylation signaling pathway, leading to a reduction in ATP synthesis and an increase in ROS generation, 6 as well as a lowering the activity of antioxidant pathways such as nuclear factor erythroid 2–related factor 2 (Nrf-2)-antioxidant response element signaling pathway.7,8 Moreover, chronic hypoxia can initiate oxidative stress that induces inflammation and triggers apoptosis.9,10 It has been shown that hypoxia can drive cardiac inflammation by upregulating interleukin-6 (IL-6). 9 Likewise, following hypoxia, mitochondria release pro-apoptotic factors such as cytochrome-c and apoptosis-inducing factor (AIF) into the cytosol, which activates the caspases signaling pathway, causing apoptosis, DNA fragmentation, and consequently leading to irreversible myocardial damage. 11
Notably, the most efficient treatment for irreversible myocardial damage is heart transplantation. However, this surgical approach is limited due to the small number of donors and the increased risk of transplant rejection. 12 Therefore, developing a pharmacological intervention with antioxidant activity is crucial to prevent irreversible myocardial damage associated with hypoxia. Curcumin (CR) exhibited a promising therapeutic potential due to its antioxidant and anti-inflammatory activities. CR is a natural yellow phenolic compound extracted from Curcuma longa (turmeric plant). Recent studies have reported positive pharmacological effects of CR as an antioxidant, anti-inflammatory, 13 antimicrobial, 14 and anti-apoptotic effect. 15 However, the exact mechanism by which CR can modify the signaling pathway associated with hypoxia is still unknown.
Because CR is a phenolic compound, it has low water solubility properties and rapid metabolism, which limit its bioactivity. 16 Alternatively, liposomal-curcumin (L-CR), a solubilized formula of CR surrounded by the phospholipidic bilayer, can overcome its limitations and significantly improve its bioactivity. 17 Therefore, this study aimed to determine L-CR's prophylactic activity on myocardial damage associated with sodium nitrite (NaNO2)-induced hypoxia.
Materials and Methods
Chemicals
CR and NaNO2 were obtained from Sigma-Aldrich Chemical Co., St. Louis, MO, USA. L-CR (particle size = 200 nm) was purchased from Lipolife Company, Drake Lane, Boreham, UK. NaNO2 was dissolved in normal saline, while CR and L-CR were suspended in 1% carboxy methyl cellulose (CMC) in normal saline. The primary antibodies were purchased from Abcam, and the conjugated anti-rabbit secondary antibody was obtained from Cell Signaling Technology.
Animals and Experimental Design
24 adult male Wistar albino rats weighing 200 ± 10 g were obtained from the Animal Resource Unit, College of Pharmacy, King Saud University, Saudi Arabia. Animals were allowed to acclimate to the lab environment with free access to food and water for at least one week. They were allocated into four groups (6 rats/group). Group 1: normal control (normoxic) rats received 1% CMC, Group 2: the hypoxic group, in which rats was injected with a single subcutaneous dose of NaNO2 (60 mg/kg), 18 Group 3 and 4: rats were injected with CR and L-CR (100 mg/kg, i.p.), 19 respectively, two hours before administration of NaNO2.
One-hour post NaNO2 injection, all rats were euthanized by CO2 and humanely killed by decapitation. Blood samples were collected for serum separation, and the heart tissues were excised and rinsed in ice-cold phosphate-buffered saline (PBS). Parts of heart tissue were fixed in 10% formalin for histopathological and immunohistochemical studies. The other parts were immediately frozen in liquid nitrogen and stored at −80 °C for molecular analyses.
Determination of the serum Levels of CK-MB and LDH
Serum levels of creatine kinase MB (CK-MB) and lactate dehydrogenase (LDH) were estimated using diagnostic kits obtained from Randox (UK) according to the manufacturer's instructions.
Enzyme-Linked Immunosorbent Assay for Evaluating the Inflammatory and Apoptotic Markers in Heart Tissues
To assess the involvement of CR and L-CR in the anti-inflammatory and antiapoptotic responses against NaNO2-induced cardiac hypoxia, we assessed the levels of interleukin-10 (IL-10) and c-reactive protein (CRP), and cytochrome-c. The expression levels of IL-10, CRP, and cytochrome-c in heart homogenates were measured using highly sensitive enzyme-linked immunosorbent assay (ELISA) kits obtained from Immuno-Biological Laboratories (Takasaki-Shi, Gunma, Japan), and Abcam (USA).
RT-PCR for Determination of Nrf-2 and MEF-2 mRNA in Heart Tissues
Because nuclear factor erythroid 2–related factor 2 (Nrf-2) and myocyte enhancer factor-2 (MEF-2) are involved in counteracting oxidative stress and cardiac development/remodeling, respectively, we employed RT-PCR to measure their expression levels. The primer pairs are listed in Table 1.
Forward and Reverse Primer Sequence List.

Histopathological Examination
Deparaffinized heart sections of 5 μm of heart tissues were fixed in formalin, embedded in paraffin, and then stained with hematoxylin and eosin (H&E) to visualize any morphological alterations using light microscopy.
Immunohistochemical Analysis
Immunostaining of the heart paraffin sections for detection of the abnormal immune reaction of primary antibody (Apo-1/Fas) was performed using the streptavidin-biotinylated horseradish peroxidase method (Novalink Max Polymer detection system, Novocastra).
Assessment of DNA Fragmentation in Heart Tissues
DNA fragmentation was assessed using agarose gel electrophoresis which provides a quantitative method for cell death according to the method of Kasibhatla group. DNA leaders were visualized by a UV light source and documented by photography.
Statistical Analysis
Data were expressed as means ± SEM. Statistical analysis was performed using one-way analysis of variance (ANOVA) followed by Tukey's post hoc test to determine the differences between means of different groups using GraphPad Prism. 9 The significance level was set at p ≤ 0.05.
Results
CR and L-CR Prevented NaNO2-Induced Cardiac Injury in Hypoxic Rats
We first confirmed the hypoxic effect of NaNO2 on cardiac tissue by histopathological examination. Figure 1 shows the normal morphological appearance of cardiomyocytes in the control group (Figure 1A). In contrast, NaNO2 administration induced degeneration of cardiomyocytes and inflammatory cell infiltration as shown in Figure 1B. Using either CR or L-CR ameliorated the morphological changes induced by NaNO2 as shown in Figures 1C and D.
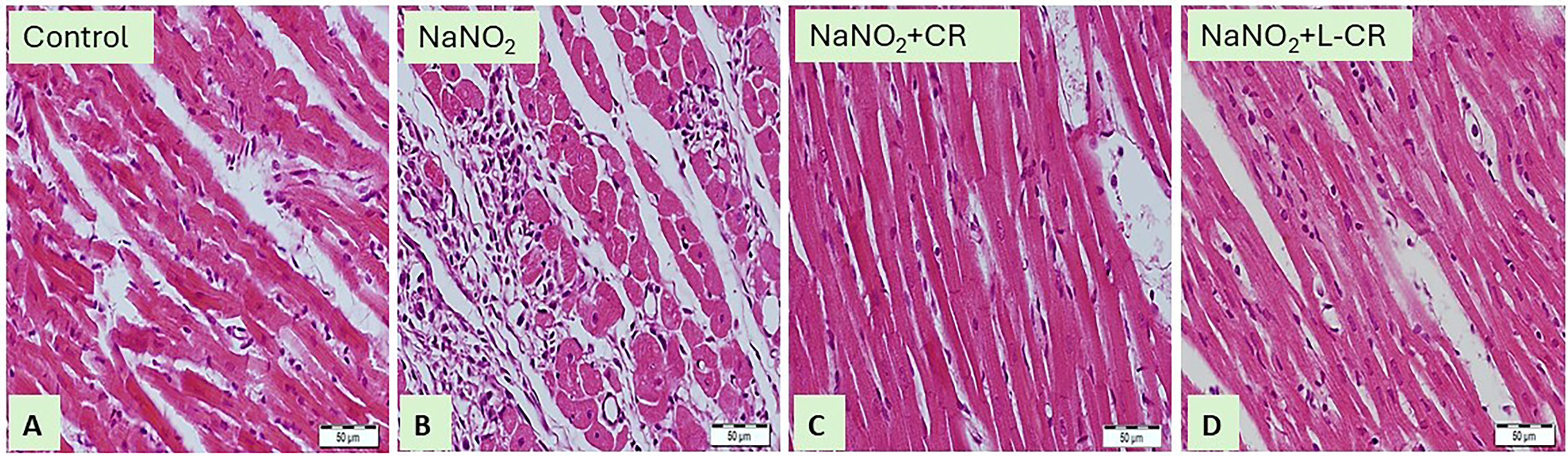
Photomicrographs of H&E-stained Sections (400×) in the Heart Tissues of (A) Control Rats Showing the Normal Histological Structure of the Cardiomyocytes with Typical Myocardial Muscle Fibers. (B) NaNO2-intoxicated Rats Showing Blood Vessel Congestion, Inflammatory Cell Infiltration. (C) NaNO2-intoxicated Rats Treated with CR. (D) NaNO2-intoxicated Rats Treated with L-CR Exhibit Normal Morphological Structure.
Moreover, the cardioprotective effect of CR and L-CR against NaNO2 damage was evaluated by assessing cardiac function-associated markers (CK-MB and LDH). NaNO2-exposed rats showed a significant elevation in serum CK-MB and LDH (Figure 2) compared to control rats (P < 0.001). Notably, treatment of the NaNO2-exposed rats with CR or L-CR markedly restored the levels of CK-MB and LDH (P < 0.001). Furthermore, the effect of L-CR on serum CK-MB was clearly superior to CR (P < 0.05).

CR and L-CR Attenuated NaNO2-Induced Cardiac Biomarkers. Levels of CK-MB and LDH were Measured in Heart Homogenates Using Highly Sensitive ELISA Kits. Treatment with CR and L-CR Decreased CK-MB and LDH in the Heart of NaNO2- Exposed Rats. Data are Expressed as Mean ± SEM (n = 6). * p < 0.05, and *** p < 0.001.
Additionally, we measured the DNA integrity in heart tissues to evaluate the impact of the CR and L-CR treatments on myocardial cell death. As expected, the NaNO2-hypoxic group showed a significant DNA fragmentation level and compromised DNA integrity compared to the control group (p < 0.001). On the contrary, treatment with CR or L-CR resulted in a significant reduction of NaNO2-induced DNA fragmentation (p < 0.001) (Figure 3).

CR and L-CR Prevented DNA Fragmentation in NaNO2-Intoxicated Rats. Agarose Gel Electrophoresis was Used to Analyze DNA Fragmentation. Quantification of DNA Fragmentation as Percentages of the Control. Data are Expressed as Mean ± SEM (n = 6). *** p < 0.001.
CR and L-CR Attenuated NaNO2-Induced Cardiac Inflammation and Apoptosis in Hypoxic Rats
Cardiac IL-10 was significantly decreased in NaNO2-intoxicated rats compared to the healthy control group (P < 0.001); on the other hand, the content of the cellular CRP was markedly increased in the heart of NaNO2-intoxicated rats as a response to inflammation (P < 0.001) (Figure 4). Additionally, NaNO2 markedly upregulated the expression level of cytochrome-c as an indication of apoptosis compared to normal rats (P < 0.001) (Figure 4). CR and L-CR markedly increased the levels of IL-10 and decreased the levels of CRP and cytochrome-c in the heart tissues of NaNO2-treated rats (P < 0.001) (Figure 4), suggesting anti-iflammatory and antiapoptotic properties of CR and L-CR with an obvious effect of L-CR.

CR and L-CR Mitigated NaNO2-Induced Cardiac Inflammation and Apoptosis. Levels of IL-10, CRP, and Cytochrome-c were Measured in Heart Homogenates using Highly Sensitive ELISA Kit. Treatment with CR and L-CR Increased IL-10 and Decreased CRP and Cytochrome-c in the Heart of NaNO2-Exposed Rats. Data are Expressed as Mean ± SEM (n = 6). * p < 0.05, and *** p < 0.001.
CR and L-CR Restored the Cardiac Expression Level of Nrf-2 and MEF-2 mRNA in Hypoxic Rats
Exposure of the rats to NaNO2 markedly downregulated Nrf-2 mRNA in the heart tissues (P < 0.001) compared to the respective control rats. On the contrary, the expression level of MEF-2 mRNA was increased (P < 0.001) in NaNO2-exposed rats, as represented in Figure 5. Interestingly, treatment of the NaNO2-exposed rats with CR or L-CR upregulated cardiac Nrf-2 mRNA and downregulated MEF-2 mRNA (P < 0.001) compared to NaNO2-exposed rats. L-CR showed a pronounced effect in restoring the expression levels of both Nrf-2 and MEF-2 mRNA.

CR and L-CR Elevated Cardiac Gene Expression of Nrf-2 mRNA and Suppressed MEF-2 mRNA Signaling in NaNO2-Intoxicated Rats. The PCR Products were Loaded in 1.5% Agarose Gel, Electrophoresed, and the Bands were Visualized Using UV Transilluminator. Levels of Nrf-2 and MEF-2 Transcripts were Examined by RT-qPCR and Presented as Fold Change Relative to Untreated Control and Normalized to Internal Control, β-Actin. Data are Mean ± SEM (n = 6). *** p < 0.001.
CR and L-CR Attenuated Cardiac Apo-1/Fas Immunoreactivity in Hypoxic Rats
In comparison to healthy control rats, tissue specimens obtained from hypoxic rats showed a strong immunoreaction towards Apo-1/Fas as presented in Figure 6A and B. Treatment with CR or L-CR produced moderate or mild immunoreactivity toward Apo-1/Fas, respectively, as shown in Figures 6 C and D.
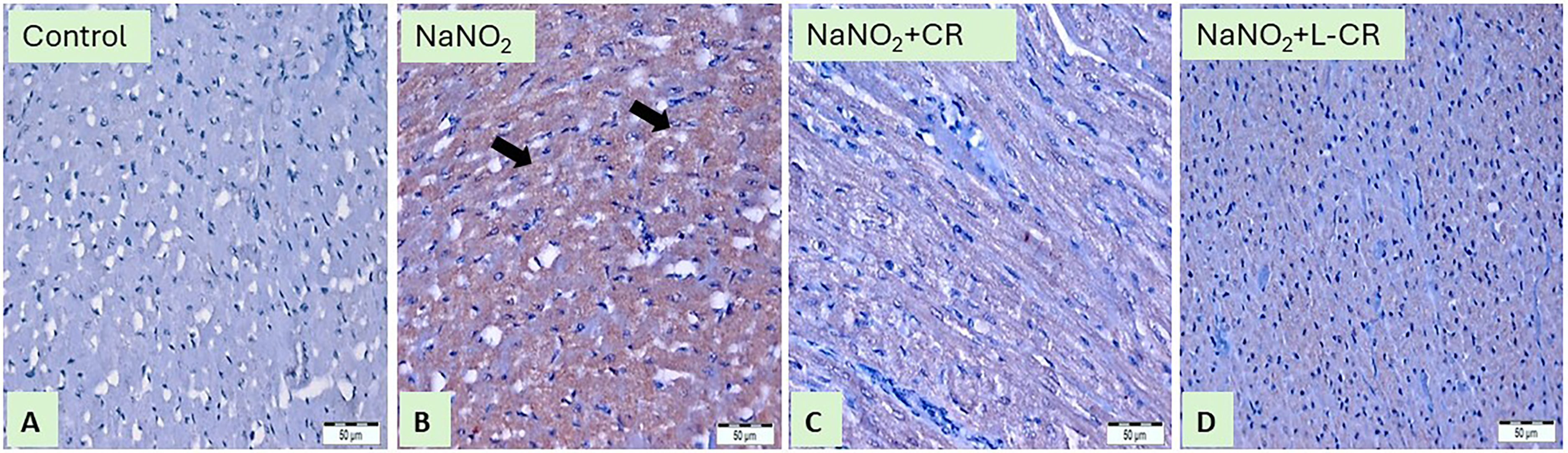
Anti-Apo-1/Fas-Immunostained Sections (400x) of Heart Tissues From Rat: (A) Section From Control Rat Showing Immune Staining in the Endothelium of the Cardiac Sinusoids and Blood Vessels While Cardiomyocyte is Negative in Staining, (B) Cardiac Section From Rat Received NaNO2 is Showing Marked Expression of Apo-1/Fas, the Brownish Color of the Cytoplasm of Muscle Cells (black Arrows), (C) Heart Section From CR Group Showed Moderate Expression of Apo-Fas Antibody as the Brownish Color of the Cytoplasm of Muscle Cells, (D) Heart Section From L-CR Group Showed Mild Expression of Apo-1/Fas as the Brownish Color of the Cytoplasm of Muscle Cells.
Discussion
Hypoxia, oxidative stress, inflammation and apoptosis are closely related terms that are discussed together. Hypoxic is an insult that leads to the generation of ROS, and the accumulation of these species results in oxidative stress, leading to cell injury and tissue inflammation, which is followed by apoptosis. This is the pathophysiology of several disorders, including ischemic heart disease (IHD), which is the most common cause of congestive heart failure (CHF). 20 In the current study, we aimed to investigate L-CR's protective effects compared with CR against myocardial damage associated with hypoxia induced by NaNO2. In the current study, the administration of NaNO2 led to an increase in the cardiac oxidative stress, inflammation, and apoptosis; while, pretreatment with either CR or L-CR significantly modulated the altered parameters.
NaNO2 is an inorganic compound commonly used as a preservative in the food industry. Chronic exposure to NaNO2 causes hypoxia, which may lead to oxidative stress and eventually cause myocardial cell damage. 21 In cardiomyocytes, oxidative stress triggers several cellular and metabolic changes, such as inhibiting oxidative phosphorylation and switching to anaerobic glycolysis utilizing less oxygen to produce ATP. 22 Excessive accumulation of ROS under the stimulation of various physicochemical factors upregulates critical signaling pathways, such as nuclear factor-κB (NF-κB), JNK activator protein-1 (JNK-AP-1), and the mitogen-activated protein kinase (MAPK) pathway. 23
CR is a turmeric yellow spice known for its promising therapeutic potential as an antioxidant, anti-inflammatory, and antiapoptotic activities. 24 L-CR has many advantages over its native drug; it is encapsulated in tiny vesicles called liposomes to enhance its stability and bioavailability, eventually leading to better, potentially promoted therapeutic effects. 25 The liposome exhibits hydrophilic and hydrophobic characteristics that promote drug absorption and enhance its bioavailability. 26 Moreover, the liposome provides a barrier to protect the drug from intestinal digestion and oxidation. 26
In the current study, injury of cardiomyocytes induced by NaNO2 was evidenced by the elevated levels of serum CK-MB and LDH. Clinically, CK-MB and LDH are commonly used as early diagnostic assays for myocardial dysfunction. 27 The elevated levels of serum CK-MB and LDH reflect an impairment of cardiac muscle. 27 It has been shown that myocardial ischemic mice exhibited high levels of CK-MB and LDH, which further increased myocardial infarction. 28 Our study showed that the administration of either CR or L-CR significantly restored the levels of serum CK-MB and LDH. Our results were parallel with a previous study that illustrated the effect of CR in the amelioration of the levels of serum CK-MB and LDH, implying a role of CR in the protection of the heart tissue from cardiac hypoxia. 29
Additionally, our data provided insight into the involvement of CRP and IL-10 as inflammatory mediators in the NaNO2-induced hypoxic cardiomyocytes and the cardioprotective mechanisms of L-CR. The cardiac inflammation was confirmed by the upregulation of cardiac CRP and downregulation of IL-10 in response to NaNO2 administration. CRP is highly expressed in hypoxic conditions; therefore, it is a risk factor for the long-term development of heart failure and death in acute myocardial infarction patients. 30 IL-10 is a major anti-inflammatory mediator that is necessary for maintaining homeostasis in the heart in response to injury. Likewise, it is an important regulatory factor in the natural immune processes, which can inhibit macrophages from producing TNF-α, IL-1β and other inflammatory factors. 31 In our study, the administration of either CR or L-CR significantly downregulated the cardiac expression of CRP and upregulated the expression of IL-10. Therefore, one possible mechanism by which CR or L-CR could prevent myocardial inflammation is reducing the endogenous release of the pro-inflammatory cytokines and mediators. Recent work revealed the anti-inflammatory effects of CR and L-CR through inhibition of the inducible nitric oxide synthase (iNOS), TNF-α, and the transcription factors like NF-ΚB. 29
Since the inflammatory cells can generate free radicals, an additional molecular mechanism was investigated to validate the antioxidant effect of CR and L-CR against NaNO2-induced cardiomyocyte hypoxia by measuring Nrf-2 expression. Our data showed that, compared to the control rats, NaNO2-exposed rats exhibited low expression levels of Nrf-2 mRNA in the heart tissues. In agreement with our study, in vitro and in vivo studies were previously conducted to assess the effect of CR on the expression of Nrf-2 using lung carcinoma epithelial cell line and Dawley rats, respectively. 32 The findings illustrated that CR effectively restored the levels of Nrf-2 that were downregulated during hypoxia. 32 Treatment with either CR or L-CR significantly reversed the effect of hypoxia and increased scavenging activity against ROS through increasing the expression level of Nrf-2 mRNA. Nrf-2 is a promising regulator of physiological and pathological consequences of oxidative stress. 33 This indicates that CR and L-CR may have a role as antioxidants via the modulation of Nrf-2 expression.
Our work examined the involvement of apoptotic mechanism in the NaNO2-induced hypoxia to the heart tissue. As a subsequent clearance of inflammatory cells, apoptosis can be initiated. The current study showed that the expressions of cytochrome-c and MEF-2 were strongly upregulated in the heart tissues post NaNO2 administration. Treatment with either CR or L-CR intervention significantly downregulated the expression of cytochrome-c and MEF-2. It was revealed that the release of cytochrome-c from the mitochondria to the cytosol induces apoptosis through activation of caspase-3 in ischemic/reperfused cardiomyocytes. 34 MEF-2 is a transcription factor contributing to cardiomyocyte differentiation, survival, and development. 35 Therefore, our data suggested the role of CR and L-CR in cardiac remodeling, probably through modulating the expression of some of the evolutionarily conserved cardiac gene regulatory networks, such as MEF-2, considering its contribution to cell survival and differentiation.
Besides the intrinsic mitochondria-dependent apoptotic pathway, there is another major apoptotic pathway that triggers cardiac apoptosis, which is the “extrinsic” Fas receptor-dependent (type I) apoptotic pathway. 36 Accordingly, we investigated the expression of the Apo-1/Fas receptor via immunostaining and found that hypoxic heart sections exhibited a marked overexpression of Apo-1/Fas, which is in agreement with a recent study that demonstrated the enhancement of apoptosis by triggering of Apo-1/Fas protein in hypoxic brain. 37 Likewise, a previous study performed on hypoxia-induced brain edema confirmed an elevation in Fas expression during hypoxia, whereas using CR caused effective inhibition in Fas-mediated apoptosis. 38 Parallel to this study, our results revealed the administration of either CR or L-CR caused a reduction in the immunostaining of Apo-1/Fas. These findings imply that CR and L-CR may contribute to cell survival by inhibiting Apo-1/Fas expression, which induces cell death.
The potential crosstalk between Nrf2, MEF-2, and Apo-1/Fas in hypoxia is complex and not fully elucidated. However, this crosstalk can be elaborated through their roles in oxidative stress and mitochondrial dysfunction. Nrf2 is a key transcription factor in the cellular antioxidant response. Its activation protects cardiomyocytes from hypoxic damage by regulating mitochondrial function, which indirectly influences MEF-2 and Apo-1/Fas signaling by improving cellular health and stress responses. 39 Conversely, Nrf2 downregulation exacerbates oxidative stress, fibrosis, and inflammation in cardiomyopathy models, highlighting its essential role in mitigating hypoxia-induced damage. MEF-2 is involved in cardiac development, stress response and hypertrophy. It may interact with mitochondrial function, potentially influencing the cellular response to hypoxia. 35 The Apo-1/Fas pathway is involved in apoptosis signaling that can be triggered during hypoxia and oxidative stress as a result of mitochondrial dysfunction and excessive ROS production. 40
Regarding the limitations, the sample size was thought to be a major limitation in our study, however, we determined the total number of animals required and effectively implemented a sample size that meets the scientific objectives. Additionally, the small size of rat's heart is a problem, which limits the ability to evaluate other parameters. Thus, more studies are required to elucidate additional signaling pathways involved in the modulatory activity of L-CR including antioxidant, anti-inflammatory, and antiapoptotic impact. Lastly, it is important to note that in our study, we used a single high dose of NaNO2 to induce hypoxic heart, and a single prophylactic dose of L-CR. Multiple doses of NaNO2 could initiate other pathological responses; accordingly, further experiments using different doses and different animal species are needed to explore the toxicities induced by NaNO2, and the ameliorative activity of L-CR.
Conclusion
To sum up, Although both CR and L-CR showed a protective activity against NaNO2-induced cardiotoxicity, L-CR demonstrated a significant cardioprotection on nearly all tested parameters in our study. The potential cardioprotective effect of L-CR was demonstrated through its ability to restore antioxidant activity and reduce inflammatory and apoptotic mechanisms. The protective effect of L-CR could be attributed to the ability of the liposome to adhere to the cell membrane and facilitate the release of its content within the cell. This can be used to support the potential use of L-CR as a cardioprotective supplement against hypoxia-induced myocardial injury.
Footnotes
Acknowledgment
The authors express their gratitude to King Saud University for their support.
Statement of Human and Animal Rights
All procedures in this study were carried out in accordance with the recommendations of the Research Ethics Committee which approved the protocol (KSU-SE-19-60).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Ethical Considerations
The study protocol was approved by the Scientific Research Ethics Committee, King Saud University, Riyadh, Saudi Arabia (KSU-SE-19-60) on 16/4/2019.
Author Contributions/CRediT
AMA considered and designed the study; AMA and WSS carried out the animal experimentation; AMA, WSS, and IHH conducted the biochemical analysis; AMA and WSS performed gene and protein analyses, IHH analyzed the data; ABA, HMA, JFA, and TWM drafted the manuscript, and all authors read and approved the final version of the manuscript.
Funding
This research was funded by the Researchers Supporting Project number (RSPD2025R822), King Saud University, Riyadh, Saudi Arabia.
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data generated during this study are available upon request from the corresponding author.
