Abstract
The incidence of ulcerative colitis (UC), an inflammatory bowel disease (IBD), is drastically increasing globally, leading to greater economic hardship and patient morbidity. This study evaluated the effects of chelerythrine on acetic acid (AA)-induced UC in a mouse model. Mice received chelerythrine (25, 50, and 100 mg/kg, i.g.) and sulfasalazine (SASP) (500 mg/kg, i.g.) for 7 consecutive days following the induction of colitis via the intrarectal administration of AA (5% v/v). Disease activity index (DAI), tissue morphology, tissue damage, oxidative stress (OS), and inflammatory markers were then evaluated. The results revealed that the colonic levels of total antioxidant capacity (T-AOC), glutathione (GSH), superoxide dismutase (SOD), and catalase (CAT) had decreased, while colonic myeloperoxidase (MPO), malondialdehyde (MDA), lipid peroxide (LPO) activity, colonic and serum interleukin 6 (IL-6), tumor necrosis factor alpha (TNF-α) levels, and serum lactate dehydrogenase (LDH) had all increased significantly in the colitis untreated group compared to that of the normal control. Treatment with chelerythrine considerably improved most of these parameters, and a dose of 100 mg/kg, was found to be comparable to that of 500 mg/kg SASP. This alkaloid also markedly reversed body weight (BW), clinical activity score, gross lesion score, mucosal edema, and colon weight/length in a dose-dependent manner. In conclusion, the protective effect of chelerythrine could be attributed to its anti-inflammatory and antioxidant activities.
Introduction
Ulcerative colitis (UC) is a chronic inflammatory bowel disease (IBD) that affects the colonic mucosa. 1 A variety of potential etiological factors are associated with IBD, including smoking, infective agents, and genetic or immunological factors. However, the UC pathogenic mechanism has not yet been fully elucidated. 2 Recently, UC morbidity has been gradually increasing. 3 Representative UC symptoms include abdominal pain and discomfort, repeated excrement, weight loss, fever, fatigue, and bloody diarrhea. 4 UC is not generally lethal, yet it can develop into toxic colonic dilatation, intestinal tract bleeding, or even colorectal cancer (CRC). 5 At present, the treatment methods for UC mainly focus on reducing inflammatory response, improving the intestinal mucosal barrier, and promoting intestinal mucosal repair. 6 The main drugs available for the treatment of UC include 5-aminosalicylic acid, mesalamine, SASP, corticosteroids, anti-integrin antibodies, and Janus kinase inhibitors. 7 Many of the problems associated with anti-UC drugs remain, and there is still a need for more inexpensive, highly effective drugs with anti-inflammatory and intestinal microbiota regulatory activity.
UC is a chronic nonspecific IBD associated with immunization and characterized by mucosal, colonic, and rectal subserosa inflammation. 8 Free radicals contribute, aggravate, and precipitate pathological changes in the colonic mucosa, and therefore, could be potential therapeutic targets. Oxidative stress (OS) and inflammation have also been identified as critical factors affecting UC pathophysiology. Various stimuli activate inflammation, which in conjunction with aggravated inflammatory cells, can result in OS. 9 The products of OS combined with proinflammatory peculiarity facilitate the destruction of the cellular structure via lipid peroxidation, resulting in the induction of cell apoptosis or necrosis. 10 Furthermore, these dead cells are considered novel stimuli that can initiate a vicious circle. The key proinflammatory factors, including interleukin 6 (IL-6) and tumor necrosis factor alpha (TNF-α), are useful for targeting UC. 11 In other studies, it has been reported that anti-inflammatory agents (glucocorticosteroids or aminosalicylates), along with antioxidants, may ameliorate UC.12–14 In addition, a large number of plant-based ingredients (eg, berberine, sanguinarine, and ginsenoside Rh2) have been shown to have protective effects against UC and will likely become promising new approaches for the treatment of UC in humans.15,16
Acetic acid (AA) has been extensively utilized to induce UC. A UC mouse model simulates intestinal inflammation in the human body by increasing neutrophil infiltration, ulcer production, free radical release, and inflammatory mediator generation. 17 The main objective of an anti-UC agent is to rapidly alleviate UC symptoms. Therefore, an AA-induced UC model was utilized in this experiment.
Isoquinoline alkaloids have a wide range of pharmacological effects and are widely found in plants of the
Materials and Methods
Materials
Chelerythrine (98% purity based on HPLC analysis) was purchased from Chengdu Pufei De Biotech Co., Ltd. SASP, used as a positive control, was obtained from Shanghai Xinyi Jiahua Pharmaceutical Co., Ltd. The ELISA kits for mouse IL-6 and TNF-α were purchased from Beyotime Biotechnology, the biochemical detection kits for lactate dehydrogenase (LDH), catalase (CAT), glutathione (GSH), superoxide dismutase (SOD), myeloperoxidase (MPO), lipid peroxide (LPO), malondialdehyde (MDA) and total antioxidant capacity (T-AOC) from Jiancheng Bioengineering Institute, TRIzol reagent from Sigma-Aldrich, Inc., and the SYBR Green QuantiTect RT-PCR kit and RT-PCR reagent from Thermo Fisher Scientific Inc. All the primers were synthesized by Wuhan GeneCreate Biological Engineering Co., Ltd.
Animals
The 6- to 8-week-old male C57BL/6 mice, weighing between 18 and 22 g, were purchased from the Hubei Provincial Center for Disease Control and Prevention. The animals were housed in cages, and the temperature was maintained at 22 ± 2°C with relative humidity (RH) set at 40%-60%, under a photoperiod of 12 h/12 h. Mice were given standard laboratory chow diet, with ad libitum tap water. The mice were acclimated to their surroundings for 3 days. All animal experiments were conducted in accordance with the Animal Care and Use Committee of the Institute of Materia Medica, China.
AA-Induced UC Mouse Model
C57 mice were used to prepare the UC model in accordance with previously published methods. 28 In brief, the animals were fasted for 24 h and then given ether for light anesthesia. Then, one 3.5-Fr catheter was inserted into the colon with caution, with the tip 4 cm from the anus. Finally, 1 mL of AA solution (5%, v/v) contained within 0.9% saline was injected into the colonic lumen to induce colitis. In order to prevent injection leakage from the colon, each animal was kept in a supine Trendelenburg position for 30 s. In the normal group, only 0.9% saline was injected into each mouse using the identical technique. A total of 72 mice were randomized into 6 groups, including normal, model, SASP (positive control), and 3 chelerythrine groups, with 12 mice in each group. Based on previous research and our pre-experimental results, 20 model groups received 0.9% saline at 50 mL/kg, the positive control group received SASP at 500 mg/kg, and the chelerythrine groups received chelerythrine at 25, 50, and 100 mg/kg, separately. All drugs were suspended in saline and given by intragastric administration once daily. All treatments were maintained for 7 consecutive days.
Disease Activity Index (DAI) and Colon Length Experiments
Throughout the whole experimental period, body weight (BW), gross bleeding, and stool consistency were monitored each day. The DAI was calculated as the score that assessed the percentage of BW loss in comparison with that determined before UC induction, gross bleeding, and stool consistency (Table 1).
Evaluation of Disease Activity Index (DAI).

Histological Evaluation of Colitis Injury
On day 8, ether was injected into each mouse for anesthesia. Clinical activity scores were then measured, and blood samples were obtained via a retro-orbital puncture in order to detect the biochemical indexes. All animals were sacrificed through cervical dislocation. Colon samples were collected, and the adherent adipose tissues were trimmed. Then all samples were rinsed with cooled saline. The colon samples were slightly stretched, and the length between the ileocecal junction and distal rectum was determined. Later, 4% paraformaldehyde (PFA) was used to fix the colon samples for 12 h, followed by gradient ethanol dehydration, paraffin embedding, and sectioning. Afterward, 5-µm sections were placed on slides, followed by clearing, hydration, and H&E staining. Macroscopic inflammation scores were evaluated via a single-blinded method according to the scoring system described. The arbitrary scale (range: 0-4) shown below was utilized, where (0) indicated no macroscopic change, (1) represented mucosal erythema alone, (2) represented mild mucosal edema, small erosion, or mild hemorrhage, (3) suggested moderate edema, bleeding erosion or ulcer, whereas (4) represented severe ulcer, tissue necrosis, or erosion.
Estimation of Serum LDH Activity
When sacrificing the mice, the blood was collected and subjected to centrifugation for 10 min at 3000 rpm to obtain the serum. The supernatants were collected into multiple new tubes and preserved under −80°C prior to subsequent analysis. A commercial detection kit was utilized to measure serum LDH activity according to the manufacturer's instructions.
Estimation of OS Parameters in Colons
The colon tissues were weighed and measured, and then ice-cold normal saline (9 volumes) was added for homogenization. Later, the supernatant was separated following centrifugation (5000 rpm, −4°C, 30 min) to investigate the OS markers, such as MPO, CAT, SOD, GSH, LPO, MDA, and T-AOC via the use of activity assay kits, following the manufacturer's protocols.
RT-PCR Analysis
A total RNA kit was used to extract total tissue RNA according to specific protocols. Then, the RT-PCR reagent was used to generate cDNA, and the 2× SYBR Green QuantiTect RT-PCR kit (Applied Biosystems 7900 Fast Real-Time PCR System, Applied Biosystems) was used for RT-PCR. β-Actin was used as a reference to calculate the overall mRNA quantity. The relative mRNA level was determined by the ΔΔCt approach. Sequences of all PCR primers are presented in Supplementary Table 1.
Statistical Methods
Values are presented as mean ± standard error of the mean (SEM). SPSS 22.0 software was employed for data analysis. ANOVA was used to evaluate significant differences between mean, while Tukey's standardized range test was applied to the post hoc evaluation. A
Results
Chelerythrine Relieved Several Symptoms of AA-Mediated UC Mice
AA was utilized for the construction of the UC mouse model in order to examine the chelerythrine impact. In the present study, the mice from the AA-mediated groups developed severe colitis rapidly, and the typical signs included bloody diarrhea, dramatic BW loss, poor coat quality, and reduced mobility. In addition, gross blood adhesion to the anus was noted in some mice. Figure 1A to E illustrates the BW, DAI score, and colon weight/length, together with the macroscopic evaluation scores that were determined for defining the regulation of chelerythrine on the AA-mediated UC. Compared to the normal group, obvious BW loss and reduced colon weight/length (Supplementary Table 2) were observed in the AA-mediated group, and such results were reversed by chelerythrine treatment in a dose-dependent manner. Meanwhile, the DAI score was significantly increased in AA-mediated mice. However, chelerythrine (25, 50, and 100 mg/kg) and SASP (500 mg/kg) significantly decreased the DAI score. Also, the colons of AA-mediated mice showed marked edema, hyperemia, ulcer, and inflammation, whereas the colons of normal control mice showed either no or slight inflammation. As shown in Figure 1E, administration of chelerythrine (25, 50, and 100 mg/kg) and SASP (500 mg/kg) significantly improved the macroscopic score.

Chelerythrine alleviated acetic acid (AA)-induced ulcerative colitis (UC). (A) Body weight (BW) changes, (B) DAI score, (C) colon weight/length, (D) representative photograph showing the shortening of the colon in mice treated with AA, (E) macroscopic evaluation score and (F) histopathological changes in the colon tissue in different groups (10×).
Chelerythrine Attenuated Histological Injury in AA-Mediated Colitis
As shown in Figure 1F, the colonic epithelial tissues of normal mice were intact, and the tissue structure was normal with regular gland arrangement and no infiltration of inflammatory cells. In comparison, AA-mediated UC displayed excessive epithelial necrosis, submucosal edema, and bleeding areas, along with infiltration of the inflammatory cells. Chelerythrine at a relatively small dose (50 mg/kg) caused the least injury to the mucosa, with mild submucosal edema, as well as mild infiltration of the inflammatory cells. Chelerythrine and SASP at middle and high doses remarkably recovered the colonic mucosa from the AA-mediated UC injury. These results indicated that chelerythrine had a significant protective effect on AA-mediated UC. Importantly, chelerythrine at a low dose (100 mg/kg) produced a more significant protective effect than SASP at a significantly higher dose (500 mg/kg).
Chelerythrine Relieved Inflammation Indicators in Mice
AA treatment significantly increased IL-6 and TNF-α levels in the colonic tissue and serum when compared with normal controls. Administration of chelerythrine significantly reduced these changes (compared with the colitis model group) and the levels of these proteins gradually returned to normal levels (Figure 2A-D). PCR was utilized to detect gene expression in order to verify this conclusion (Figure 2E and F). The activity of chelerythrine at 50 mg/kg was as high as that of standard SASP (500 mg/kg) administration, and chelerythrine at 100 mg/kg was found to be even more potent. As shownin Figure 2G, the release of LDH in the serum increased following AA administration, and chelerythrine and SASP both significantly decreased this change. In the colon samples, the addition of AA led to a significant increase in MPO, but the administration of chelerythrine significantly decreased the MPO level in comparison with the UC model group, returning it to a normal level (Figure 2H).
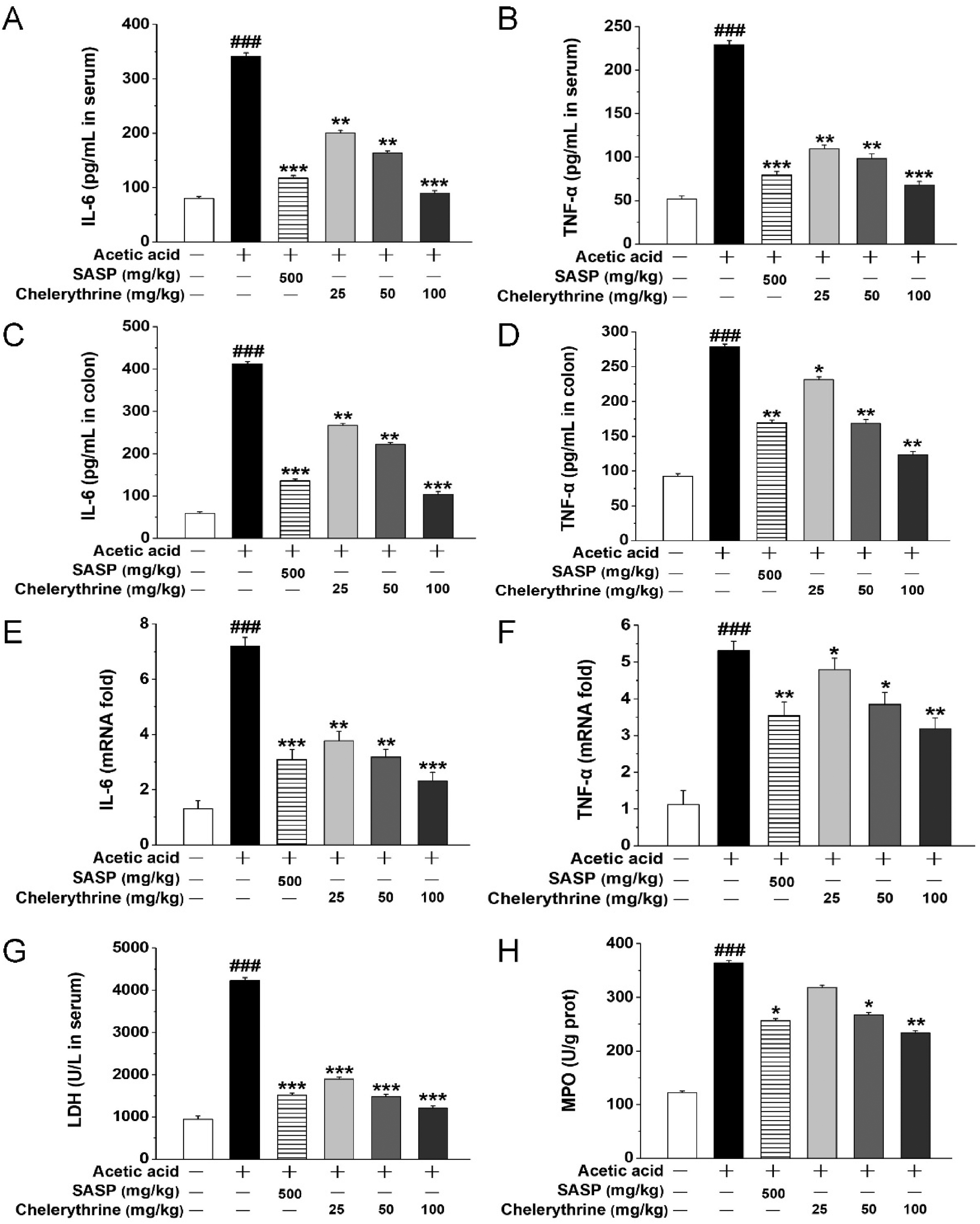
Chelerythrine relieved inflammation in acetic acid (AA)-induced UC mice. (A) The concentration of IL-6 in the serum, (B) the concentration of TNF-α in the serum, (C) the concentration of IL-6 in the colon, (D) the concentration of TNF-α in the colon, (E) the mRNA expression of cytokines IL-6, (F) the mRNA expression of TNF-α, (G) LDH release in the serum, and (H) the activity of MPO in the colon.
Chelerythrine Relieved OS in Mice
As shown in Figure 3A to E, AA led to a significant increase in LPO and MDA, and a significant decrease in CAT, SOD, and GSH in the colons. The administration of chelerythrine significantly reversed such changes in the biochemical parameters in comparison with the UC model group and returned them to more normal levels. Moreover, the T-AOC in the colon also significantly increased following chelerythrine treatment (Figure 3F). Chelerythrine exhibited an effective antioxidant capacity, and even a small dose of chelerythrine (100 mg/kg) was found to be more effective than a higher dose of SASP (500 mg/kg).
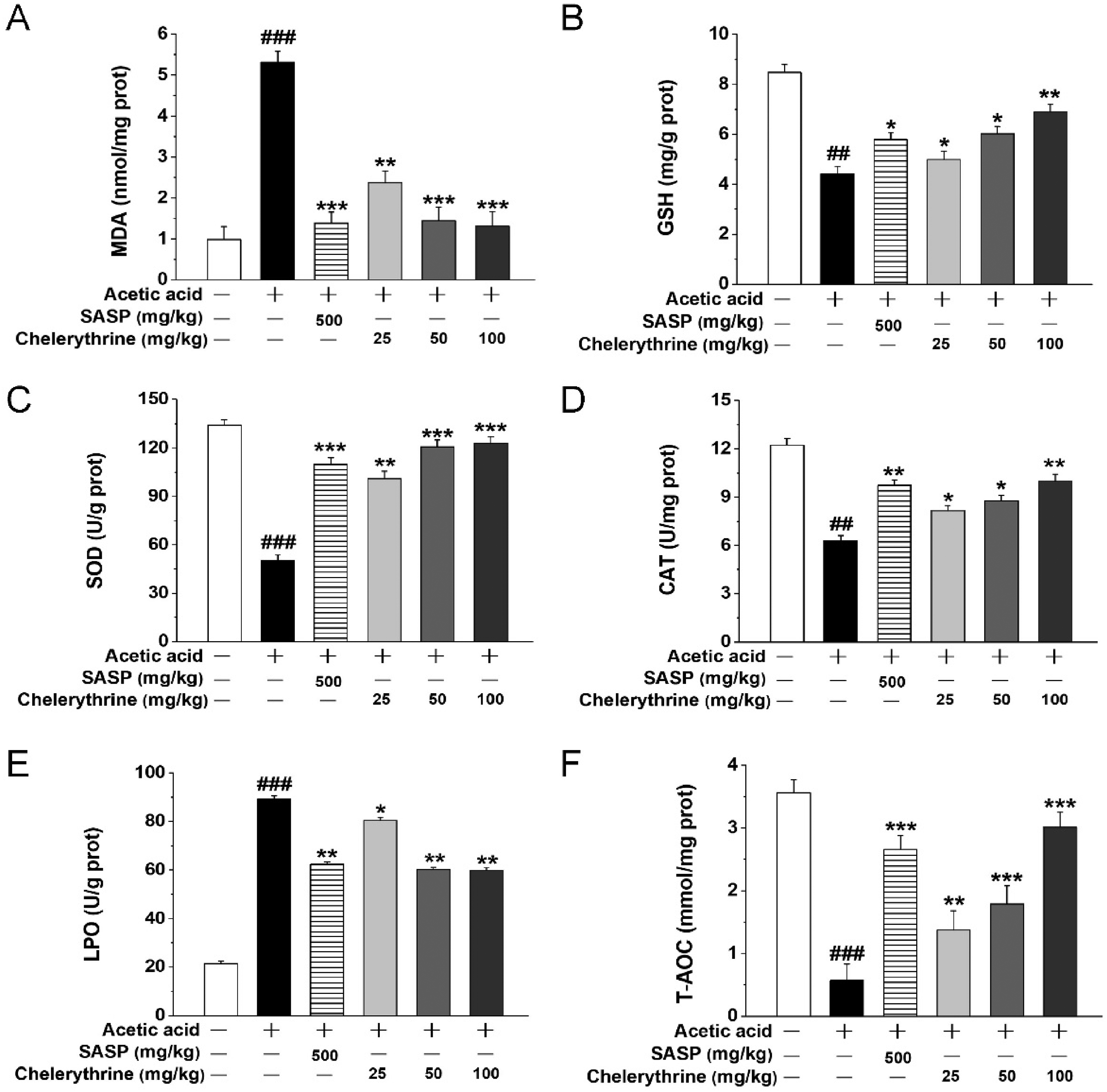
Chelerythrine relieved oxidative stress (OS) in acetic acid (AA)-induced UC mice. (A) The concentration of MDA in the colon, (B) the activity of GSH in the colon, (C) the activity of SOD in the colon, (D) the activity of CAT in the colon, (E) the activity of LPO in the colon, and (F) the total antioxidant capacity (T-AOC) in the colon.
Discussion
UC presents as a chronic, nonspecific, and refractory disorder frequently found in the rectum or the whole colon. Medical treatments for UC cases are primarily focused on the induction and maintenance of symptoms and the alleviation of mucosal inflammation, thus improving the patients’ quality of life when the minimum steroid amount is applied. 24 AA-mediated UC represents an experimental model which simulates human UC in regard to its histopathological alterations and functional and biochemical abnormalities. This model facilitates ulcer production, intestinal inflammation, and up-regulates levels of free radicals and inflammatory factors. 25 In this study, chelerythrine administration remarkably mitigated AA-mediated UC in mice, as reflected by the mitigated diarrhea, weight loss, and bloody stools. In addition, chelerythrine (100 mg/kg) was superior to the positive SASP group for colonic length recovery, and the colonic weight and length ratios were close, suggesting a similarity between the 2 treatments for the improvement of AA-induced UC. Chelerythrine also mitigated AA-mediated morphological damage, inhibited colonic inflammation, and hindered OS.
UC is related to an imbalanced inflammation state. 26 Epithelial cells and immunocytes produce excessive cytokines, resulting in serious mucosal damage or even destruction. 27 TNF-α exerts a vital role in promoting the generation of diverse chemical factors and results in aggravated inflammation by triggering nuclear factor kappa B (NF-κB) generation. 28 IL-6 is also a critical molecule that facilitates the infiltration of neutrophils as well as the apoptosis of cells during colitis. 29 In this experiment, the serum IL-6 and TNF-α contents increased in the UC mice, and their levels decreased following chelerythrine treatment. Reduced IL-6 and TNF-α mRNA expression following chelerythrine administration indicated that this alkaloid suppressed inflammation.
LDH has been recognized as an indicator used to monitor the integrity of the colonic cell membrane, and serum LDH activity reportedly increases in UC. 1 Consistent with the morphology, findings from the current study revealed that the LDH activity in the serum of mice is also enhanced, reflecting the AA-induced damage to the intestinal epithelium. Chelerythrine administration caused a remarkable decline in LDH activity, which indicated that the colonic tissue was damaged. MPO, the enzyme existing in neutrophils, was lowly expressed in the macrophages and monocytes. MPO activity showed a positive correlation with the neutrophil content in the affected tissues. As a result, MPO activity has been recognized as an index used to measure the acute intestinal inflammation quantitatively and sensitively. 30 Chelerythrine significantly decreased the MPO activity compared to the AA-mediated UC model.
OS plays an important role in UC pathophysiology. 4 The recurrent intestinal mucosal damage cycle was shown to result in an increased incidence of colon cancer in patients with UC. The elevated LPO content existing in the colon tissues could trigger a vicious cycle, in which increased amounts of reactive metabolites are generated, finally exhausting the cell antioxidants. MDA is a favorable predictor of lipid peroxidation. 31 GSH takes part in synthesizing and repairing DNA and is considered to block free radical injury, while enhancing vitamin C antioxidation. 32 Moreover, CAT and SOD also serve as oxygen radical inhibitors by eliminating the oxygen radicals.33,34 Chelerythrine treatment recovered changes in MDA, CAT, GSH, and SOD, returning them toward normal. LPO is used as an indicator of OS and reactive metabolites. 35 LPO activity in the current study significantly increased in the AA-mediated group, whereas LPO activity significantly increased in the extract and standard drug-treated groups. Thus, chelerythrine produced a significant antioxidant effect against AA-induced UC injury.
Conclusions
This study suggested that chelerythrine treatment suppressed AA-induced UC in mice, and such protection was partially associated with its antioxidative and anti-inflammatory properties. Nonetheless, more studies are warranted in order to assess whether a comparable therapeutic effect could be attained in other experimental colitis models simulating human IBD using chelerythrine as a treatment for AA-mediated UC.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221132417 - Supplemental material for Chelerythrine Ameliorates Acetic Acid-Induced Ulcerative Colitis via Suppression of Inflammation and Oxidation
Supplemental material, sj-docx-1-npx-10.1177_1934578X221132417 for Chelerythrine Ameliorates Acetic Acid-Induced Ulcerative Colitis via Suppression of Inflammation and Oxidation by Jun-Song Wu, He-Jing Liu, Si-Jie Han, Ning-Feng Mao and Xue-Feng Liu in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The animal study was reviewed and approved by the Ethics Committee of Hubei University of Traditional Chinese Medicine (Hubei, China).
Statement of Human and Animal Rights
All experiments of animals were conducted in conformity with the Animal Care and Use Committee of Institute of Materia Medica, China.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
