Abstract
Platelet-derived growth factor-BB (PDGF-BB) plays a central role in smooth muscle cell (SMC) proliferation and inflammation, being involved in atherosclerotic cardiovascular disease. We have previously found that butanolic extract of noni, a tropical plant belonging to the family Rubiaceae, exerts anti-inflammatory effects on endothelial cells exposed to advanced glycation end products (AGEs). Here, we examined the effects of noni extract on oxidative stress production, growth, and inflammatory reactions in PDGF-BB or AGE-exposed SMCs. Reactive oxygen species (ROS) generation, cell proliferation, and adhesion were measured by a fluorescent dye, a colorimetric agent, and labeled THP-1 cells, respectively. Gene and protein expression was evaluated by real-time reverse transcription-polymerase chain reaction and Western blot analysis. Butanolic extract of noni reduced ROS production, monocyte chemoattractant protein-1, intercellular adhesion molecule-1, and proprotein convertase subtilisin kexin type 9 (PCSK9) expression, and proliferation in, and THP-1 cell adhesion to, PDGF-BB-exposed SMCs. Gene expression and protein level of receptor for AGEs (RAGE) were significantly decreased by noni extract in SMCs. Furthermore, AGEs significantly increased PCSK9 mRNA and protein levels in SMCs, which were inhibited by noni extract or an antioxidant N-acetylcysteine. Our present study suggests that butanolic extract of noni not only inhibits the PDGF-BB-induced proliferation and inflammatory reactions in SMCs through its antioxidative properties but also reduces PCSK9 levels in AGE-exposed SMCs via suppression of RAGE expression. Butanolic extract of noni may play a protective role against atherosclerosis.
Atherosclerotic cardiovascular disease is the leading cause of death in diabetic patients. 1 Although endothelial cell damage is an initial step of atherosclerosis, smooth muscle cell (SMC) proliferation, migration, and inflammatory reactions also contribute to arterial stiffness, neointimal thickening, and plaque formation, thereby being involved in the progression of atherosclerosis. 2,3 We, along with others, have shown that platelet-derived growth factor-BB (PDGF-BB) stimulates SMC proliferation, migration, and inflammatory reactions via oxidative stress generation, whose pro-atherosclerotic effects are augmented under diabetic conditions. 2 -5 Moreover, formation and accumulation of advanced glycation end products (AGEs) have progressed under diabetic conditions, and their interaction with the receptor for AGEs (RAGE) has also been shown to evoke proliferative, inflammatory, and osteogenic reactions in SMCs. 6
We have recently found that butanolic extract of noni, a tropical plant widely used in traditional medicine in Pacific Islanders, inhibits the AGE-induced reactive oxygen species (ROS) generation and inflammatory reactions in human umbilical vein endothelial cells. 7,8 However, the effects of butanolic extract of noni on growth and inflammatory reactions in PDGF-BB or AGE-exposed SMCs remain unclear. In this study, we addressed the issue.
Materials and Methods
Materials
Bovine serum albumin (BSA) (essentially fatty acid free), PDGF-BB, and N-acetylcysteine (NAC) were purchased from Sigma (St. Louis, MO, United States). Advanced glycation end product modified BSA and nonglycated control BSA were prepared as described previously. 7 In brief, BSA was incubated with or without 0.1 M glyceraldehyde under sterile conditions at 37°C for 7 days, and then unincorporated sugar was removed by dialysis against phosphate-buffered saline.
Collection and Preparation of n-Butanol Extract of Noni
Noni (Morinda citrifolia) was collected in French Polynesia during January 2004 to December 2006, and immediately freeze-dried and stored in dark place at 4°C. Noni was identified by botanists at Tropical Resources, Inc. (Provo, UT, United States) where voucher specimens were deposited (Part no. 119107, Lot Code 52488, Batch # 23401). Freeze-dried noni was extracted with 50% ethanol, and then the extract was further extracted with ethyl acetate and n-butanol several times as described previously. 7
Cell Culture
Human aortic SMCs (Lonza Group Ltd., Basel, Switzerland) were cultured in basal medium containing 1% fetal bovine serum and 0.1% gentamycin/amphotericin B-1000 with or without 20 ng/mL PDGF-BB, 100 µg/mL AGEs, or 100 µg/mL nonglycated BSA in the presence or absence of 670 ng/mL butanolic extract of noni or 1 mM NAC. After the indicated time periods, ROS generation assay, real-time reverse transcription-polymerase chain reaction (RT-PCR) analysis, Western blot analysis, and cell proliferation and adhesion assays were performed. Cells at passage less than 10 were used for the present experiments. Monocyte chemoattractant protein-1 (MCP-1), intercellular cell adhesion molecule-1 (ICAM-1), and proprotein convertase subtilisin kexin type 9 (PCSK9) gene expression was analyzed by RT-PCR. MCP-1, RAGE, and PCSK9 protein expression was evaluated by Western blot analysis. Platelet-derived growth factor-BB and noni extract was dissolved in basal medium containing 1% fetal bovine serum, 0.1% GA-1000, 4 µM HCl, and 1 µg/mL BSA. So, vehicle (basal medium containing 1% fetal bovine serum, 0.1% GA-1000, 4 µM HCl, and 1 µg/mL BSA)-treated cells were used as a control.
Measurement of ROS Generation
Smooth muscle cells were plated into 96 wells (Corning Japan, Tokyo). Reactive oxygen species generation was evaluated by 5-(and-6)-carboxy-2′,7′-dihydrofluorescein diacetate (carboxy-H2DFFDA) (Thermo Fisher Scientific, Sab Jose, CA, United States) according to the previously described method with some modifications. 9 In brief, SMCs were incubated with or without 10 µM carboxy-H2DFFDA for 30 minutes. Then, the medium was removed, and the cells were treated with or without 20 ng/mL PDGF-BB for 50 minutes in the presence or absence of noni extract. Reactive oxygen species generation was measured by fluorescence intensity.
Reverse Transcription-Polymerase Chain Reaction
Smooth muscle cells were plated into 12 wells (Corning Japan). Total RNA was extracted with NucleoSpin RNA kit (Takara Bio Inc., Shiga, Japan) according to the manufacturer’s instructions. Gene expression of MCP-1, ICAM-1, RAGE, PCSK9, β-actin, and 18S rRNA was evaluated by RT-PCR; IDs of primers for MCP-1, ICAM-1, RAGE, PCSK9, β-actin, and 18S rRNA gene were Hs00234140_m1, Hs00164932_m1, Hs00542592_g1, Hs03037355_m1, Hs01060665_g1, and Hs9999901_s1, respectively. 9
Western Blot Analysis
Smooth muscle cells were plated into 24 wells (Corning Japan). After 24 hours treatment with or without 20 ng/mL PDGF-BB, 100 µg/mL AGEs or 100 µg/mL nonglycated BSA in the presence or absence of 670 ng/mL butanolic extract of noni or 1 mM NAC, 20 µg proteins were extracted from the cells and subjected for Western blot analysis using polyclonal antibody raised against MCP-1 (Abcam, Cambridge, United Kingdom), RAGE (Santa Cruz Biotechnology, Inc., Dallas, TX, United States), or PCSK9 (GeneTex Inc., Irvine, CA, United States) or monoclonal antibody directed against α-tubulin (Abcam) as described previously. 9
Assay of THP-1 Cell Adhesion to SMCs
Human THP-1 monocytic leukemia cells (ATCC, Manassas, VA, United States) were labeled with 3 µM BCECF-AM (Dojindo, Kumamoto, Japan), and their adhesion to SMCs was assayed as described previously. 10 In brief, SMCs were treated with or without 20 ng/mL PDGF-BB in the presence or absence of 670 ng/mL butanolic extract of noni or 1 mM NAC for 24 hours, and then incubated with BCECF-AM-labeled THP-1 cells for 4 hours. After the incubation, nonadherent THP-1 cells were removed, and fluorescent intensities of the adherent THP-1 cells were measured.
Evaluation of Cell Proliferation
Smooth muscle cells were plated into 96 wells (Figure 2(a)) and 24 wells (Figure 2(b)). Cell proliferation was measured with an electron coupling reagent WST-1-based colorimetric assay (Roche Applied Science, Indianapolis, IN, United States).
Statistical Analysis
All values were presented as mean ± standard deviation. Statistical comparisons were performed using ANOVA followed by Tukey-Kramer test (Figures 1(b)-(g) -3(b) and (d)) or Steel-Dwass test (Figure 1(a)), or Student’s t-test (Figure 3 (a) and (c)); P < .05 was considered significant. All statistical analyses were performed with the use of EZR version 1.11 (Jichi Medical University Saitama Medical Center, Saitama, Japan).
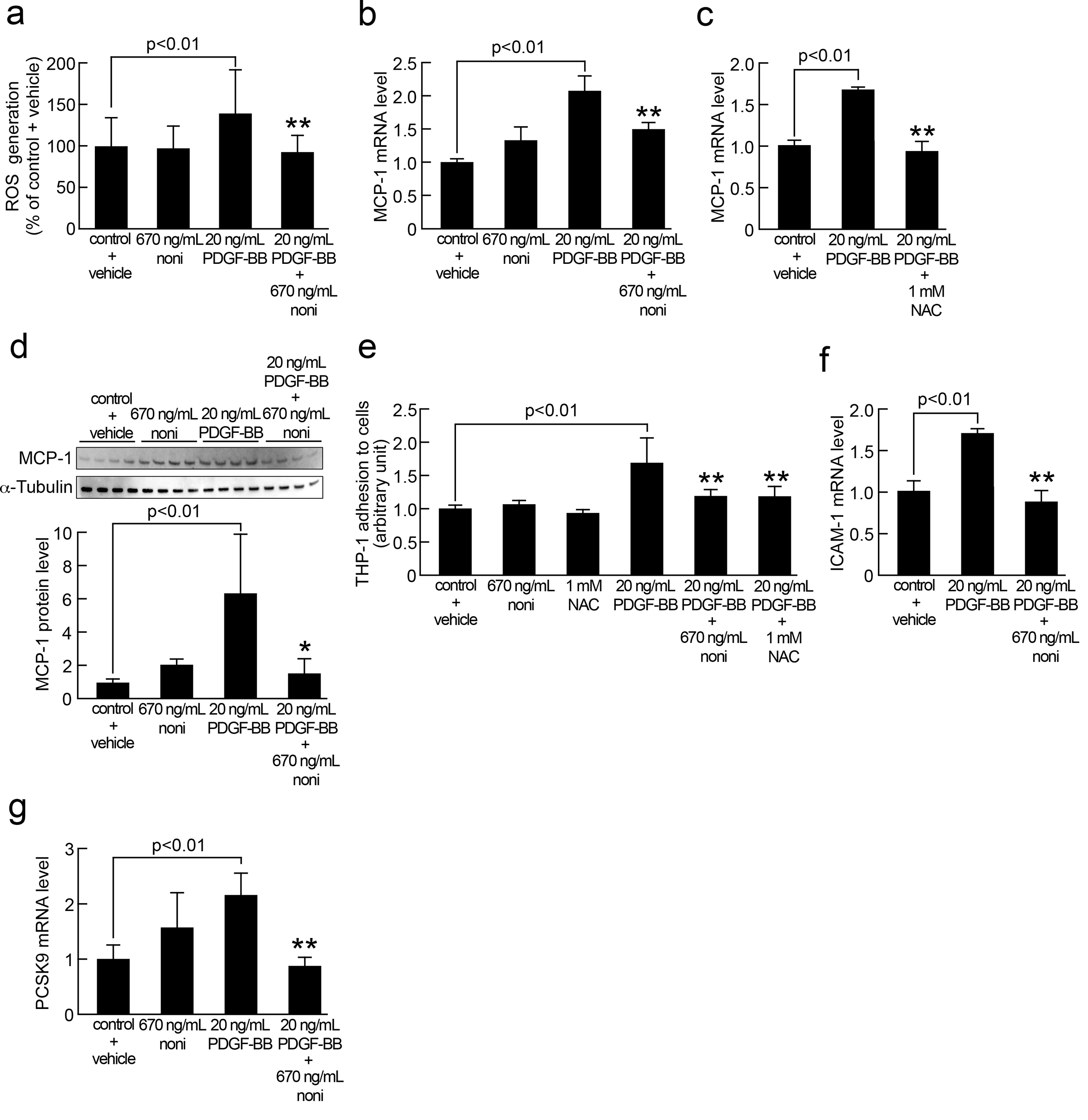
Human aortic smooth muscle cells were cultured in basal medium containing 1% fetal bovine serum with or without 20 ng/mL platelet-derived growth factor-BB in the presence or absence of 670 ng/mL butanolic extract of noni or 1 mM N-acetylcysteine for 50 minutes (a) and 24 hours (b-g). (a) Reactive oxygen species generation was evaluated with 5-(and-6)-carboxy-2′,7′-dihydrofluorescein diacetate. N = 6 wells per group. (b, c, f, g) Total RNAs were transcribed and amplified by real-time polymerase chain reaction. Data were normalized by the intensity of 18S rRNA-derived signals and then related to the control values. (b, g) N = 4 wells per group. (c, f) N = 3 wells per group. (d) Western blot analysis of MCP-1. Data were normalized by the intensity of α-tubulin-derived signals and then related to the control values. Upper panel shows the bands of Western blot analysis. Lower panel shows the quantitative data. N = 4 wells per group. (e) Smooth muscle cells were incubated with BCECF-AM-labeled THP-1 cells for 4 hours. Then, nonadherent THP-1 cells were removed. Fluorescent intensities of the adherent THP-1 cells were measured. N = 8 wells per group. * and ** indicate P < .05 and P < .01 compared with platelet-derived growth factor-BB treated cells, respectively.

Human aortic smooth muscle cells were cultured in basal medium containing 1% fetal bovine serum with or without 20 ng/mL Platelet-derived growth factor-BB in the presence or absence of 670 ng/mL butanolic extract of noni for 24 hours (a, b), or 48 hours (a). Cell proliferation was evaluated by a colorimetric assay. Viable cell number is shown as absorbance at 450 nm (arbitrary units). (a) N = 8 wells per group. (b) N = 6 wells per group. * indicates P < .05 compared with platelet-derived growth factor-BB treated cells.

Human aortic smooth muscle cells were cultured in basal medium containing 1% fetal bovine serum with or without 100 µg/mL Advanced glycation end products or 100 µg/mL nonglycated bovine serum albumin in the presence or absence of 670 ng/mL butanolic extract of noni or 1 mM N-acetylcysteine for 4 hours (a
Results
As shown in Figure 1(a), butanolic extract of noni significantly blocked the increase in ROS generation in SMCs induced by 20 ng/mL PDGF-BB exposure for 50 minutes. Furthermore, noni extract significantly inhibited the PDGF-BB-induced increase in MCP-1 mRNA and protein levels, whereas an antioxidant NAC mimicked the effect of noni extract on MCP-1 gene expression in SMCs (Figure 1(b-d)). Platelet-derived growth factor-BB at 20 ng/mL significantly upregulated ICAM-1 gene expression in, and increased the THP-1 cell adhesion to, SMCs, which were blocked by the treatment with 670 ng/mL butanolic extract of noni or 1 mM NAC (Figure 1 (e and f)). Butanolic extract of noni at 670 ng/mL also inhibited the PDGF-BB-induced upregulation of PCSK9 mRNA levels in SMCs (Figure 1(g)). Butanolic extract of noni alone did not affect ROS generation, MCP-1, and PCSK9 expression in, or THP-1 cell adhesion to, SMCs.
Platelet-derived growth factor-BB at 20 ng/mL stimulated proliferation of SMCs in a time-dependent manner (Figure 2(a)). Since 24 hours treatment with 20 ng/mL PDGF-BB significantly increased SMC proliferation, we chose the 24 hours treatment condition and then examined the effects of noni extract on cell proliferation. As shown in Figure 2(b), butanolic extract of noni completely prevented the increase in viable cell number of SMCs.
As shown in Figure 3(a) and (b), butanolic extract of noni significantly decreased RAGE mRNA and protein levels in SMCs; noni extract reduced the RAGE gene expression by half in vehicle-treated control cells. Compared with nonglycated BSA, AGEs significantly increased PCSK-9 gene and protein expression in SMCs, which were inhibited by 670 ng/mL butanolic extract of noni or 1 mM NAC (Figure 3 (c) and (d)).
Discussion
In this study, we found for the first time that butanolic extract of noni reduced ROS generation, MCP-1 gene and protein expression, proliferation, and ICAM-1 and PCSK9 mRNA levels in, and THP-1 cell adhesion to, PDGF-BB-exposed SMCs. We, along with others, have previously shown that antioxidants, such as NAC and dihydroxyflavonol, inhibited the PDGF-BB-induced proliferation and MCP-1 overexpression in SMCs. 5,11 Furthermore, PDGF-BB has been shown to stimulate SMC proliferation and MCP-1 gene expression in SMCs via NADPH oxidase-induced ROS generation. 5,12 In addition, we found here that NAC as well as noni extract inhibited the ICAM-1 gene expression in, and THP-1 cell adhesion to, SMCs exposed to PDGF-BB. These observations suggest that butanolic extract of noni may block the proliferative and pro-inflammatory effects of PDGF-BB by suppressing NADPH oxidase activity through its antioxidative properties. Since we have previously found that butanolic extract of noni at 670 ng/mL, the same concentration of noni extract used in the present experiments exerts anti-inflammatory effects on AGE-exposed endothelial cells by suppressing oxidative stress generation and that AGEs evoke endothelial cell damage via NADPH oxidase-derived ROS, 7,10 ROS-reducing effects of noni extract may contribute to vascular protection.
Platelet-derived growth factor-BB is a growth factor that is mainly involved in neointimal thickening of atherosclerosis and restenosis after balloon angioplasty. 2 -5 MCP-1 expression is increased in human atherosclerotic plaques, which could play a crucial role in macrophage infiltration to subendothelial areas, thereby promoting foam cell formation and plaque instability within the atherosclerotic lesions. 13 Indeed, MCP-1- or its receptor deficiency has been shown to reduce the atherosclerotic lesions in low-density lipoprotein (LDL) receptor- or apolipoprotein E-knockout mice, respectively. 14,15 Moreover, ICAM-1 stimulates the recruitment and firm adhesion of inflammatory cells to endothelial cells, which could cause endothelial cell damage. 16 Smooth muscle cell proliferation and inflammation could act in concert with endothelial cell damage to promote the progression of atherosclerotic cardiovascular disease. 6,17 -19 Therefore, the present findings suggest that butanolic noni extract may exert an atheroprotective action at least in part by suppressing the deleterious effects of PDGF-BB on MCP-1 and ICAM-1 expression.
PCSK9 is a protease that binds to the LDL receptor and promotes its degradation, thereby being involved in LDL-cholesterol metabolism. 20 The increase in PCSK9 levels contributes to elevation of LDL-cholesterol level, a major risk factor for atherosclerotic cardiovascular disease. 20,21 Furthermore, local expression of PCSK9 by SMCs may evoke inflammatory and thrombotic reactions within the atherosclerotic lesions via ROS generation. 20,21 We demonstrated here that butanolic extract of noni significantly reduced RAGE gene and protein expression in SMCs. In addition, an antioxidant NAC as well as noni extract completely inhibited upregulation of PCSK9 mRNA and protein levels in AGE-exposed SMCs. Receptor for AGEs is a main cell surface receptor that mediates the deleterious effects of AGEs. 6,7,22 -24 Advanced glycation end products have been shown to evoke oxidative stress and inflammatory reactions in SMCs through the interaction with RAGE. 6 Given that RAGE expression is induced by AGE-induced oxidative stress generation, 22 -24 butanolic extract of noni may attenuate the atherogenic effects of AGEs on SMCs by breaking the crosstalk between ROS generation and RAGE expression. Moreover, noni extract has been reported to bind to RAGE and subsequently inhibit the AGE-induced inflammatory reactions in endothelial cells by working as an antagonist for RAGE. 25 This RAGE antagonizing property of noni extract may account for the reduced PCSK9 expression in AGE-exposed SMCs. Butanolic noni extract may play a protective role against atherosclerosis partly by reducing the PCSK9 expression in PDGF-BB- or AGE-exposed SMCs via suppression of ROS generation.
Limitations
We have previously reported that noni extract inhibits the binding of AGEs to RAGE with a dissociation constant of 640 ng/mL 25 and that 670 ng/mL noni extract exhibits anti-inflammatory and antioxidative effects on AGE-exposed endothelial cells. 7 This is a reason why we chose the concentration of 670 ng/mL noni extract in the present experiments. However, many biological agents can exert biphasic effects at low and high concentrations. The single dose in the study may present a biased view. Furthermore, although butanolic extract of noni is lipophilic and could be directly exposed to SMCs, the composition, bioactive stability, and bioavailability of extract will be significantly altered under gastrointestinal conditions if the extract is orally administrated.
Here, we could not fully characterize the active substance of noni extract. Since bioactive compounds that might be responsible to the observed activities in this study remain unknown, it will be challenging for the standardization of crude extract in the future. However, several phytochemicals, including polyphenols and iridoids, were identified in several types of noni extract, which possessed antioxidative and anti-inflammatory properties in both cell culture and animal models. 8,25 -28 Indeed, loganin and morroniside extracted from noni have been shown to inhibit the AGE-induced oxidative stress and inflammatory reactions in mesangial cells, a counterpart of SMCs in the kidney. 28 Therefore, it would be interesting to investigate the effects of loganin and morroniside on ROS generation and inflammatory reactions in PDGF-BB-exposed SMCs. In addition, noni extract may contain glycosaminoglycans capable of binding to and thus sequestering PDGF-BB, 29 which could partly explain the PDGF-BB-inhibitory effects of noni extract in SMCs observed in the present experiments. Anyway, since noni is widely taken as a juice due to its medicinal properties, 27,28 further clinical studies are needed to examine the efficacy and safety of noni extract for preventing atherosclerosis in both animal models and humans.
Conclusion
Our present study demonstrated that butanolic extract of noni inhibited the growth and inflammatory reactions in PDGF-BB or AGE-exposed SMCs, which may exert beneficial effects on atherosclerosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: FI and YA are employees of Morinda Worldwide Inc., Japan. SY received honoraria such as lecture fees from Morinda Worldwide Inc., Japan.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
