Abstract
Introduction
Diabetes mellitus, a metabolic disease characterized by chronic high blood sugar,1,2 Its global prevalence is rising rapidly, making it one of the most significant public health challenges of the twenty-first century.3,4 According to the IDF Diabetes Atlas, 10th edition, it is estimated that by 2030, approximately 643 million individuals worldwide will be living with diabetes, a figure projected to rise to around 783 million by 2045.5,6 At present, the treatment of diabetes predominantly depends on oral hypoglycaemic medications and insulin replacement therapy. Nevertheless, these methods frequently entail side effects, exhibit limited effectiveness, and incur substantial costs, thereby presenting considerable challenges for patients and healthcare systems alike.7-10 In severe cases, it can lead to a series of complications,including cardiovascular disease, renal failure, and vision impairment, 11 not only diminish patient quality of life but also impose considerable economic burdens on healthcare systems globally.12,13 In the current context of medical research, patients not only focus on the effectiveness of treatments but also place great importance on their safety and potential adverse reactions. Consequently, the investigation of safe, effective, and cost-effective alternative therapeutic strategies remains crucial.
Traditional Chinese Medicine (TCM) is a common naturopathic medicine that promotes the balance of two opposing yet complementary forces: Yin and Yang, and the maintenance of Qi, 14 which is uniquely advantageous in the treatment of disease.. 15 Huanglian-Wumei serve as a classic herb pair in the Wu Mei Pill from Shang Han Lun, which is widely used in the treatment of various complex diseases across different clinical specialties. Both domestic and international studies have documented their efficacy in combating diabetes. 16 CR, noted for its bitter profile, berberine exerts its effects by activating the AMPK and PI3K/Akt signaling pathways, resulting in marked reductions in blood glucose levels and enhanced insulin sensitivity,17,18 It can also be through the TAS2R-IL-25 signaling pathway 19 and Inhibiting NLRP3 inflammasome activation exerts anti-diabetic effect. 20 MF, characterized by its sour and astringent taste, is abundant in organic acids and other bioactive compounds. Some organic acids exhibit antioxidant and anti-inflammatory effects, which can synergistically mitigate chronic inflammation associated with diabetes. Furthermore, when combined with berberine, these organic acids can exert synergistic hypoglycemic effects through multiple mechanisms.21,22 Further modulate glucose levels by improving lipid metabolism and supporting liver function.23,24 Although studies have shown that Huanglian and Wumei individually exhibit significant hypoglycemic effects, the mechanisms and clinical efficacy of their combined use still require further investigation. Bitter products are chosen to clear away internal heat, and acidic products are chosen to astringe qi and yin. This synergistic pharmacological interaction enables CRMF to regulate glucose metabolism and its associated pathological mechanisms holistically. This will provide a scientific foundation for developing novel treatment strategies for diabetes based on traditional Chinese herbal formulations.
CRMF exhibits therapeutic potential by modulating diverse metabolic pathways and targets, though fully elucidating its mechanisms remains a challenge. LC-MS/MS offers a highly sensitive and precise approach for profiling chemical constituents, particularly in TCM, enabling robust data acquisition for active substance investigations. Network pharmacology leverages big data analytics to map active compounds to disease-related targets, uncovering their multi-target regulatory mechanisms. Concurrently, molecular docking simulates drug-protein interactions at the molecular level, providing insights into binding dynamics.25,26 Collectively, these methodologies not only highlight the regulatory role of traditional compounds within metabolic networks but also establish a framework for innovating multi-target drug development.
The study draws on the classical Wu Mei Pill formula, 27 which reflects the modern innovative application of the classic formula. And based on the theory of “Ku Suan Zhi Tian”, 28 is the inheritance and innovation of traditional Chinese medicine. Employing LC-MS/MS, network pharmacology, and molecular docking techniques, alongside animal experiments, the research comprehensively investigated the anti-diabetic properties of CRMF. By examining its chemical profile, target-specific interactions, and associated biological mechanisms, the findings provide innovative insights into its potential role in diabetes management.
Primary Experimental Materials
The study utilized the following instruments and reagents: High-Performance Liquid Chromatography (HPLC) (SHIMADZU Nexera X2), Quadrupole-TOF Mass Spectrometer (Applied Biosystems 4500 QTRAP), Glucose Meter (Roche Diagnostics, Batch 404577), ELISA Analyzer (Hangzhou Allsheng Instruments Co., Ltd, FlexA-200), and Tissue Floating-out Machine (Shanghai Leica Instruments Co., Ltd, RM2016). The chemicals employed included Streptozotocin (STZ) (Beijing Boaotoda Technology Co., Ltd, S 6060), Metformin Hydrochloride Tablets (Merck Pharmaceuticals, ACG 4579), and Sodium Chloride Injection Solution (Xi'an Jingxi Shuanghe Pharmaceutical Co., Ltd, 211223 4B). Biochemical assay kits for Total Cholesterol (TC), Triglyceride (TG), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), malondialdehyde (MDA), superoxide dismutase (SOD), and glutathione peroxidase (GSH) were procured from Nanjing Jiancheng Bioengineering Institute, while assays for Serum insulin (INS), C-reactive protein (CRP), tumor necrosis factor (TNF), and interleukin-6 (IL-6) were supplied by Shanghai Enzyme-linked Biotechnology Co., Ltd
Experimental Methods
Preparation of CRMF
Professor Chongbo Zhao from the Department of Chinese Medicine Authentication at the School of Pharmacy, Shaanxi University of Chinese Medicine, authenticated the Huanglian and Wumei used in this study. CR denotes the rhizome of Coptis chinensis Franch, while MF represents the dried, near-mature fruit of Prunus mume (Sieb. & Zucc.). A combined total of 77 g of CRMF was precisely measured, followed by reflux extraction using water at 10-fold and 8-fold volumes, respectively, for 1 h. The resulting filtrates were combined and concentrated to achieve a final crude drug concentration of 1 g/mL and subsequently stored at 4 °C for future experiments.
Detection of CRMF Chemical Components
Sample Preparation
The samples underwent centrifugation at 12 000 rpm for 3 min, after which the supernatant was retrieved and filtered using a 0.22 μm microporous membrane. The filtrate was then transferred into sample vials for UPLC-MS-based qualitative analysis.
UPLC-MS/MS Analysis
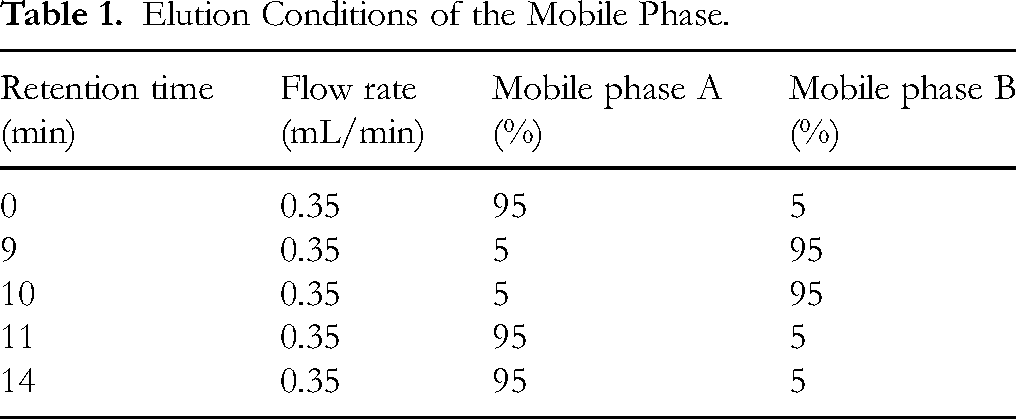
The SHIMADZU Nexera X2 UHPLC system, equipped with an Agilent SB C18 column (1.8 µm, 2.1 mm × 100 mm), was utilized for sample separation. The mobile phases consisted of ultrapure water containing 0.1% formic acid (Phase A) and acetonitrile containing 0.1% formic acid (Phase B). A gradient elution was employed in accordance with the parameters outlined in Table 1. The injection volume was 4 μL, and the column temperature was consistently maintained at 40 °C.
Elution Conditions of the Mobile Phase.
Mass spectrometry conditions
A QTRAP 4500 triple quadrupole mass spectrometer was used for signal acquisition under both positive and negative ion detection modes, operating within a voltage range of −4000 to 5000 V. Data collection was performed in multiple reaction monitoring (MRM) mode. Electrospray ionization (ESI) was employed as the ion source, maintaining a temperature of 550 °C. High collision energy was applied, with nitrogen serving as the collision gas to enhance ion fragmentation and detection efficiency.
Qualitative of Chemical Component Analysis
Raw mass spectrometry data were processed using Analyst software (version 1.6.3) to identify chemical components in CRMF samples. A proprietary database provided by Shanghai MetWare Metabolomics Co., Ltd facilitated qualitative analysis, enabling peak area extraction, integration adjustments, and relative quantification of mass spectrometry peaks. Chromatographic peaks were further refined based on retention time and peak shape parameters to enhance the precision and reliability of both qualitative and quantitative assessments.
Screening of Active Components in CRMF
Using the qualitative and quantitative data obtained from UPLC-MS/MS, the 50 most abundant chemical components were selected for network pharmacology analysis. Lipinski's Rule of Five was applied to assess oral drug-likeness, streamlining the screening process and reducing associated drug development costs. The selection criteria included molecular weight <500, logP < 5, fewer than 10 hydrogen bond acceptors, and fewer than 5 hydrogen bond donors. Components satisfying at least two of these criteria were deemed to exhibit favorable pharmacokinetic characteristics and bioavailability, qualifying them as active compounds in CRMF.
Target Prediction of Active Components
The active ingredient targets of CRMF were identified through the Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP, https://tcmsp-e.com/) and the Swiss Target Prediction database (http://www.swisstargetprediction.ch/index.php). The 2D structures of the active components were retrieved from the PubChem database in SDF format. These structures were subsequently analyzed using the Swiss Target Prediction platform, with species designated as Homo sapiens and a gene probability threshold set to >0, enabling the selection of potential targets. The resulting targets were aggregated and refined, with duplicate entries removed. The Uniport module in Perl software was employed to standardize target names to official human gene symbols, ensuring consistency and accuracy in downstream analysis.
Identification of Anti-Diabetic Targets of CRMF
The term “Diabetes” served as the primary keyword for identifying diabetes-related targets across the OMIM (https://omim.org/), DisGeNet (https://www.disgenet.org/), and GeneCards (https://www.genecards.org/) databases. In GeneCards, targets with a relevance score exceeding 10 were prioritized, while all targets from OMIM were included, and DisGeNet with a score greater than 0.1 were considered for analysis. The collected targets underwent consolidation, with duplicate entries eliminated to produce a refined list of diabetes-related targets. The intersection of drug and disease targets was determined using the Venny_2.1 platform (https://bioinfogp.cnb.csic.es/tools/venny/index.html), identifying the shared targets as the anti-diabetic targets of CRMF.
Construction and Topological Analysis of the PPI Network
The anti-diabetic targets of CRMF were analyzed using the STRING database, with the species restricted to Homo sapiens and a confidence threshold set at 0.4, excluding non-interacting proteins. The resulting interaction data, saved as “string_interactions.tsv,” was processed in Cytoscape to develop the PPI network. Key targets within the network were determined using the CytoHubba plugin, employing the “degree” algorithm for target prioritization.
GO and KEGG Analysis
The molecular mechanisms underlying CRMF's anti-diabetic effects were investigated through GO functional and KEGG pathway enrichment analyses, focusing on key targets identified in the PPI network with degree values exceeding the average threshold in the DAVID database (https://david.ncifcrf.gov/). GO terms and KEGG pathways with P < .05 were deemed significantly enriched and subjected to detailed examination. Visual representations, including bar charts for GO terms and bubble charts for KEGG pathways, were generated using the Bioinformatics Online Platform (https://www.bioinformatics.com.cn). Pathways irrelevant to the study objectives were excluded, and the top 10 GO terms along with the top 20 KEGG pathways, ranked by P-value, were prioritized for analysis.
Construction and Topological Analysis of the “Compound-Anti-Diabetic Target-Significant Pathway” Network
A “compound-target-pathway” network integrating CRMF's bioactive components, anti-diabetic targets, and associated pathways was established using Cytoscape software (version 3.9.1). In this model, nodes symbolize active compounds, molecular targets, or relevant pathways, visually mapping their interconnections. Network centrality was quantified through the Network Analyzer plugin, with node degree representing the extent of connectivity to other targets. Nodes with elevated degree values were identified as central elements within the network structure.
Molecular Docking
The 2D structures of core components within the compound-target network were obtained from the PubChem database and stored in SDF format. For key targets in the PPI network, the highest-resolution 3D structures specific to Homo sapiens were retrieved from the RCSB PDB database (https://www.rcsb.org/) and saved in PDB format. Using PyMOL (version 2.4.1), proteins were isolated from their ligands, water molecules were removed, and the structures were saved individually. Preprocessing of key targets and core components was conducted with AutoDockTools (version 1.5.6). Molecular docking was performed using AutoDock Vina (version 1.1.2) at docking sites corresponding to the original ligand-binding positions of each protein, with docking results presented for core components and key targets. Binding energy of the lowest-scoring conformation was recorded, with values <−5.0 kcal/mol denoting strong receptor-ligand affinity. Optimal docking results based on minimal binding energy for each core target were analyzed visually in PyMOL.
Establishment of a Diabetic Mouse Model
Sixty male mice (clean grade, 20 ± 2 g) were selected and acclimatized under standard feeding conditions for one week (Ethical Number: SUCMDL20240712004). A total of 10 mice were randomly assigned to the NC group, while the experimental group received an intraperitoneal injection of 1% STZ-citrate buffer at a dose of 150 mg/kg, prepared fresh and maintained on ice. The NC group was administered plain citrate buffer as a control. Blood samples were collected from the tail tip after three days, following a 12-h fasting period, to measure glucose levels. A blood glucose concentration of ≥11.1 mmol/L was used to confirm successful modeling; for mice that failed to reach this threshold, the procedure was repeated.
Grouping and Drug Administration
Following successful model establishment, mice were stratified into six groups (10 per group) based on body weight and blood glucose levels. The groups included the CRMF groups, Mod group, NC group, and Met group. The Met group received 0.25 g/kg metformin hydrochloride solution, while the Mod and NC groups were administered an equivalent volume of saline. The remaining treatment groups were dosed with the herbal extract at 5, 10, and 15 g/kg, respectively. All administrations were performed via gavage at a consistent daily schedule for 21 days.
Sample Preparation
Blood Sample Preparation
Following 21-day drug treatment, mice were subjected to a 12-h fasting period with unrestricted access to water. Blood samples were collected via retro-orbital puncture into EP centrifuge tubes and centrifuged at 10 000 r/min for 15 min. The resulting supernatant was carefully transferred to EP tubes and stored at −80 °C for subsequent analysis.
Tissue Sample Preparation
Tissue sampling was performed on ice to preserve sample integrity. Blood was collected via retro-orbital puncture, followed by the surgical opening of the abdominal cavity for immediate dissection of the pancreas, liver, and kidneys. The entire pancreas, liver, and kidneys from each mouse were excised, rinsed with saline, blotted dry, and weighed. Organ-to-body weight ratios were determined as organ weight (g)/body weight (g) for the liver, kidneys, and pancreas. Subsequently, portions of the liver and pancreas were fixed in 4% paraformaldehyde, while additional sections were stored at −80 °C for subsequent analysis.
Observation Indicators
General Condition of Mice
Body weight, hair condition, food and water intake, and general activity of the mice were monitored every three days, with body weight recorded during each observation.
Fasting Blood Glucose (FBG)
Fasting blood glucose levels were assessed on days 0, 7, 14, and 21 for all groups. Mice were fasted for 12 h with access to water before each test, and measurements were consistently performed at the same time each day. Blood samples were collected from the tail tip using a Roche automatic glucometer. Prior to fasting, bedding was replaced to maintain hygiene. To ensure accuracy, blood was allowed to flow freely from the tail vein without excessive squeezing, thereby minimizing contamination from tissue fluid.
Oral Glucose Tolerance Test (OGTT)
After 21 days of treatment, mice underwent overnight fasting for 12 h. Following the fasting blood glucose measurement, a glucose solution (2 g/kg) was administered. Blood glucose levels were measured at 0, 0.5, 1, and 2 h post-administration, and the corresponding OGTT curve was generated.
Biochemical index Measurement
Serum levels of INS, CRP, TNF, and IL-6 were quantified using ELISA, while the HOMA-IR index was calculated based on fasting blood glucose (FBG, mmol/L). Levels of LDL-C, HDL-C, TG, TC, MDA, SOD, and GSH were determined using assay kits from Nanjing Jiancheng Bioengineering Institute.
Pancreatic HE Staining
Pancreatic tissue was fixed in 4% paraformaldehyde for 24 h, then dehydrated in an ethanol Pancreatic tissues were fixed in 4% paraformaldehyde for 24 h, followed by ethanol gradient dehydration at room temperature, xylene clearing, and dual wax immersion before paraffin embedding. Sections of approximately 5 μm thickness were prepared and stained with hematoxylin and eosin (HE). Morphological features were observed using a optical microscope, and images were captured for damage assessment. Following image acquisition, the islet area within each 400× field was measured using Image-Pro Plus 6.0 software (Media Cybernetics, USA). All measurements were standardized and reported in millimeters (mm2) as the unit of measurement.
Statistical Analysis
Data were analyzed using SPSS 25.0 and GraphPad Prism 8.3.0, with results expressed as mean ± standard deviation (x̅ ± s). The data were normally distributed (P > .05) and exhibited homogeneity of variance (P > .05); Group differences were evaluated using one-way ANOVA, with statistical significance defined as P < .05.
Results of the Experiment
Identification of the Chemical Composition of CRMF
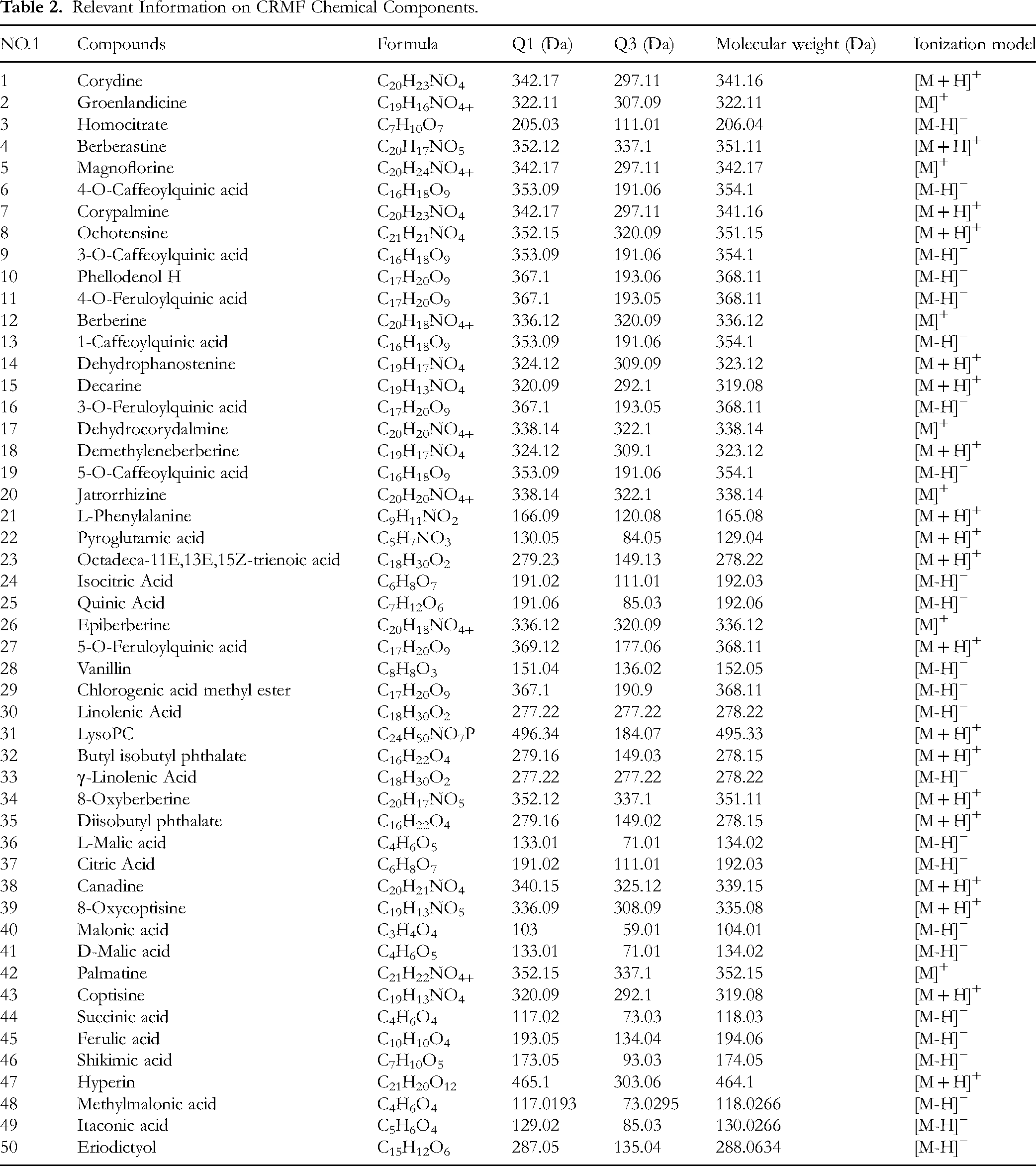
Figure 1 displayed the total ion chromatograms of CRMF samples analyzed in both positive and negative ion modes. A comprehensive qualitative and quantitative evaluation of the original mass spectrometry data identified 1169 chemical constituents in the CRMF samples. From this dataset, the top 50 compounds, ranked by relative content and informed by literature review, were selected as candidate compounds for subsequent network pharmacological analysis related to diabetes treatment. Detailed data were summarized in Table 2.

The total ion chromatogram obtained from CRMF samples in negative ion mode (A) and positive ion mode (B).
Relevant Information on CRMF Chemical Components.
Results of Screening Active Components and Target Prediction for CRMF
All identified chemical components were confirmed as active compounds following an assessment of drug-likeness using Lipinski's Rule of Five. Detailed absorption properties were summarized in Table 3. A comprehensive search across the TCMSP and Swiss Target Prediction databases identified 664 potential targets for the components.
Absorption Properties of Active Components in CRMF.

Determination of Anti-Diabetic Targets in CRMF
Relevant disease targets were identified using predefined criteria across DisGeNET, OMIM, and GeneCard databases. Following the elimination of duplicate entries, a total of 1462 diabetes-related targets were obtained (Figure 2). By intersecting diabetes-related targets with drug targets, 208 shared targets were pinpointed as potential anti-diabetic targets of CRMF.

Venn diagram displaying the overlap between CRMF active component targets and diabetes-related targets.
PPI Network Construction and Analysis
The 208 overlapping targets identified between the drug and the disease were analyzed for protein-protein interactions (PPI) using the String database, yielding a network comprising 208 nodes and 3615 edges, as illustrated in Figure 3. Core targets were determined through the Degree unDir filter in the Centiscape 2.2 plugin of Cytoscape, highlighting GAPDH, TNF, AKT1, IL6, and ALB as the top five targets based on degree value (Figure 4). These targets were suggested as key components of CRMF's anti-diabetic mechanism.
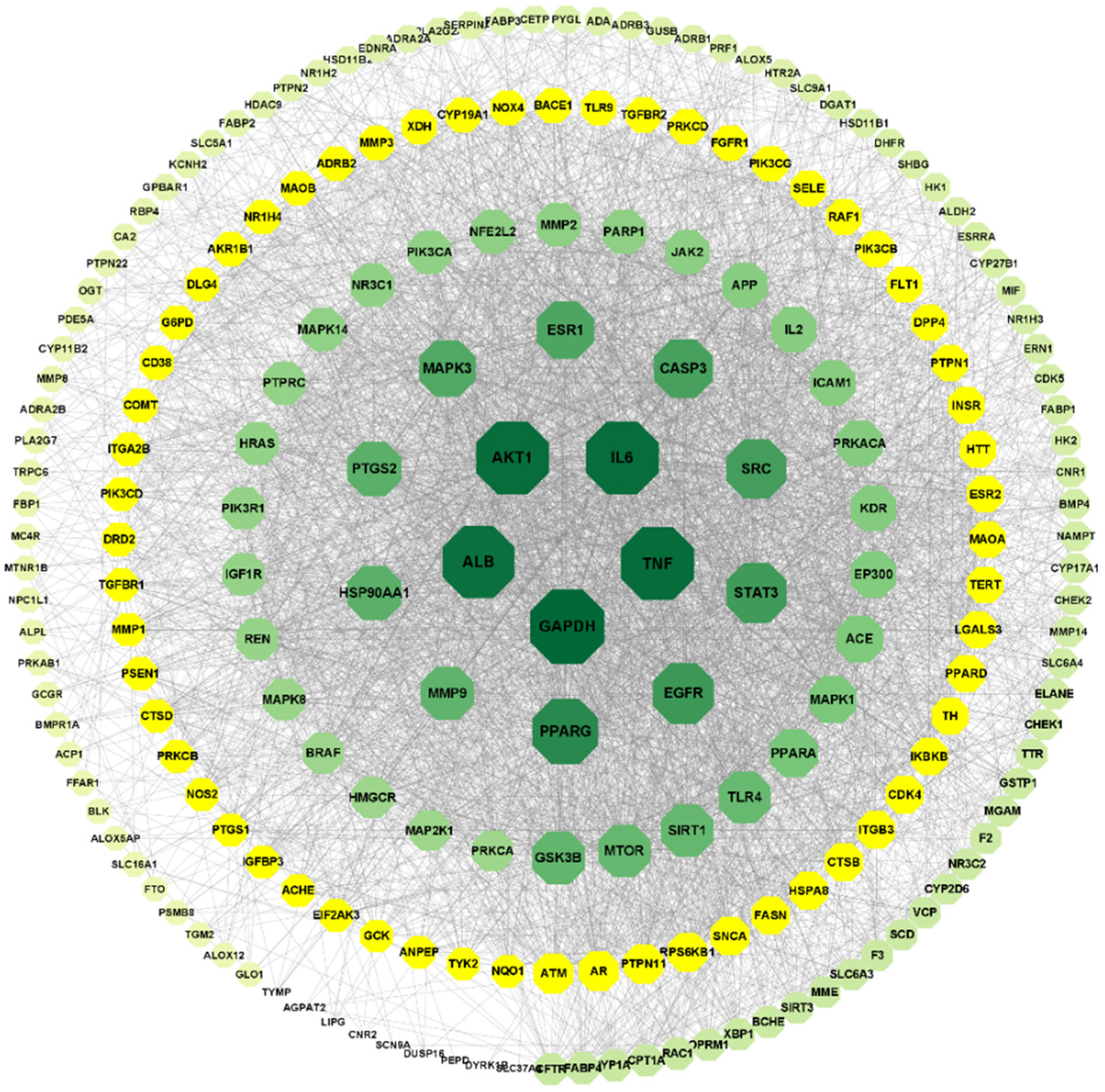
PPI network of the core anti-diabetic targets of CRMF.

The top 5 core targets ranked by degree value.
Analysis of Enrichment
GO functional enrichment analysis revealed 822 significant GO terms (P < .05), comprising 608 related to Biological Process (BP), 83 to Cellular Component (CC), and 131 to Molecular Function (MF). The top-ranked GO terms in each category, based on P-value, were illustrated in Figure 5. The analysis indicated that BPs associated with CRMF in diabetes prevention and treatment predominantly involved glucose homeostasis, regulation of gene expression (positive), apoptosis inhibition, MAPK cascade activation, and responses to xenobiotic stimuli and lipopolysaccharides. MFs were primarily linked to enzyme binding, nuclear receptor activity, steroid and ATP binding, protein tyrosine kinase activity, transcription factor binding, and diverse enzyme activities. CCs were largely associated with the plasma membrane, receptor complexes, cytoplasm, and extracellular space.
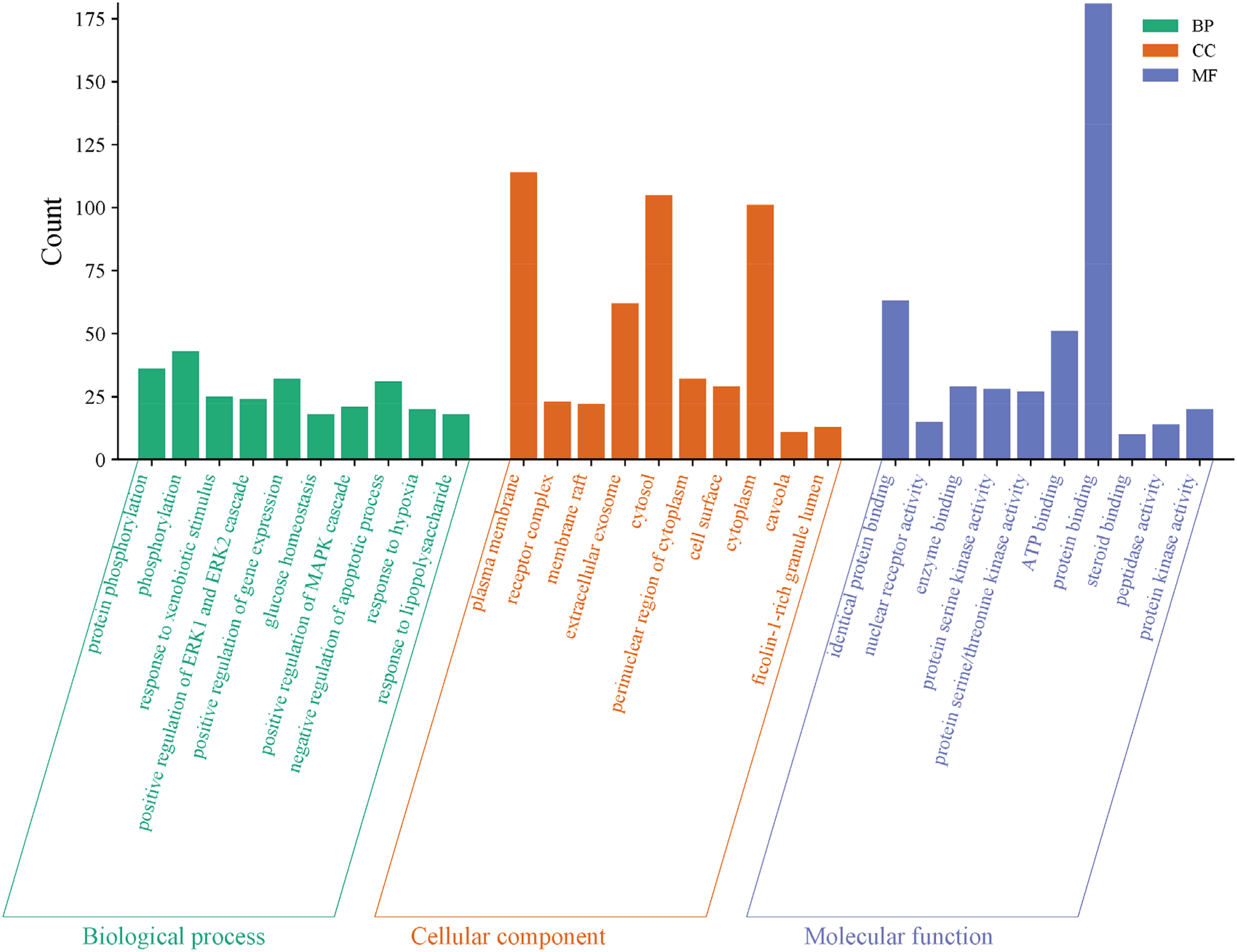
Gene ontology (GO) functional enrichment analysis.
KEGG enrichment analysis identified 208 intersecting targets significantly associated with 177 pathways (P < .05). Figure 6 displayed a bubble chart highlighting the top 20 pathways ranked by P-value. The anti-diabetic effects of CRMF were associated with pathways including AGE-RAGE signaling, endocrine resistance, cancer-related mechanisms, lipid metabolism, atherosclerosis, insulin resistance, EGFR tyrosine kinase inhibitor resistance, and several signaling cascades (eg, FoxO, HIF-1, VEGF, TNF, PI3K-Akt, AMPK, mTOR, MAPK, and JAK-STAT), along with human cytomegalovirus and papillomavirus infections. Among these, the AGE-RAGE signaling pathway was particularly significant, directly contributing to diabetic complications and remaining a prominent focus in diabetes research.

Enrichment analysis of KEGG pathways.
Construction and Topological Analysis of the “Active Ingredients-Anti-Diabetic Targets-Significant Pathways” Network
A total of 44 active constituents were identified from 208 CRMF anti-diabetic targets. Based on the interactions among constituents, targets, and significant pathways, an “active constituent-anti-diabetic target” network was constructed using Cytoscape software, as depicted in Figure 7. Topological analysis of the network quantified degree values for constituent nodes, identifying Corydine, Magnoflorine, Ochotensine, Palmatine, Dehydrocorydalmine, Linolenic acid, Itaconic acid, Jatrorrhizine, 8-Oxyberberine, Epiberberine, Linolenic Acid, Berberine, Quinic acid, 3-O-Feruloylquinic acid, Ferulic acid, and Citric acid as the most prominent constituents. These compounds, characterized by their relatively high concentrations in CRMF, were posited as the primary pharmacologically active agents underlying CRMF's anti-diabetic effects, serving as key bioactive components.

Active component-anti-diabetic target-significant pathway network.
Molecular Docking Between Core Targets and Principal Active Components
Interactions between CRMF diabetes core targets (GAPDH, TNF, AKT1, IL6, and ALB) and principal active constituents (eg, Corydine, Magnoflorine, Ochotensine, Palmatine, Dehydrocorydalmine, Citric acid, Itaconic acid, Jatrorrhizine, 8-Oxyberberine, Epiberberine, Berberine, Quinic acid, 3-O-Feruloylquinic acid, Ferulic acid) were analyzed through molecular docking simulations. Binding energy calculations revealed that most active constituents exhibited values below −5 kcal·mol−1 for the five core targets. Notable interactions included 8-Oxycoptisine with ALB (−11.6 kcal·mol−1), Jatrorrhizine with ALB (−10.1 kcal·mol−1), Epiberberine with GAPDH (−9.4 kcal·mol−1), and 3-O-Feruloylquinic acid with ALB (−8.9 kcal·mol−1). Detailed results were summarized in Table 4 and Figure 8, corroborating network pharmacology predictions. Pymol software facilitated visualization of the strongest binding affinities between active constituents and core targets. Hydrogen bonding and hydrophobic interactions predominantly mediated these molecular interactions, as illustrated in Figure 9.

Molecular docking outcomes between key active components and five core targets.
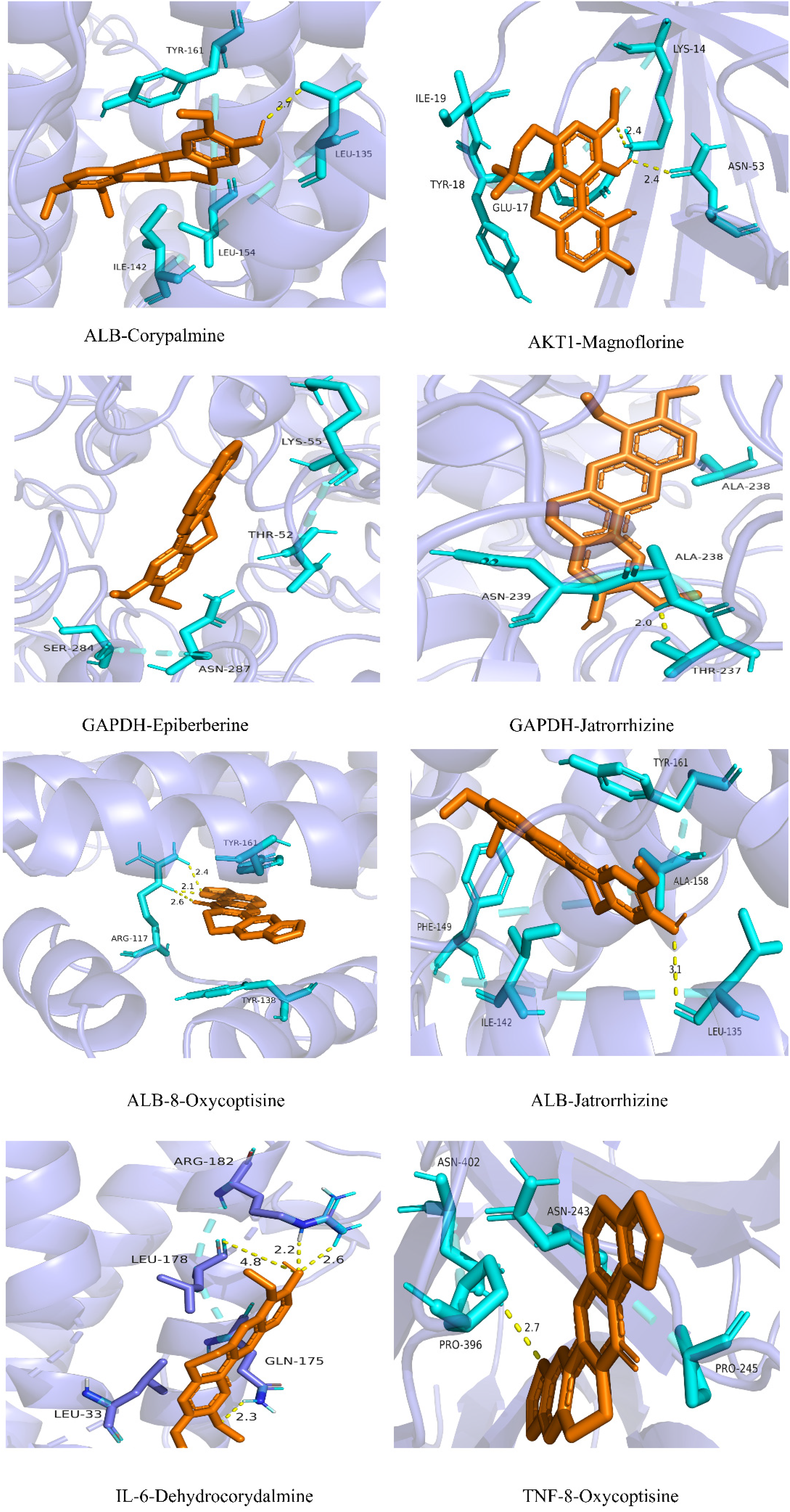
Schematic of visualization analysis for core targets and key active components.
Molecular Docking Results Between key Active Components and Five Core Targets.
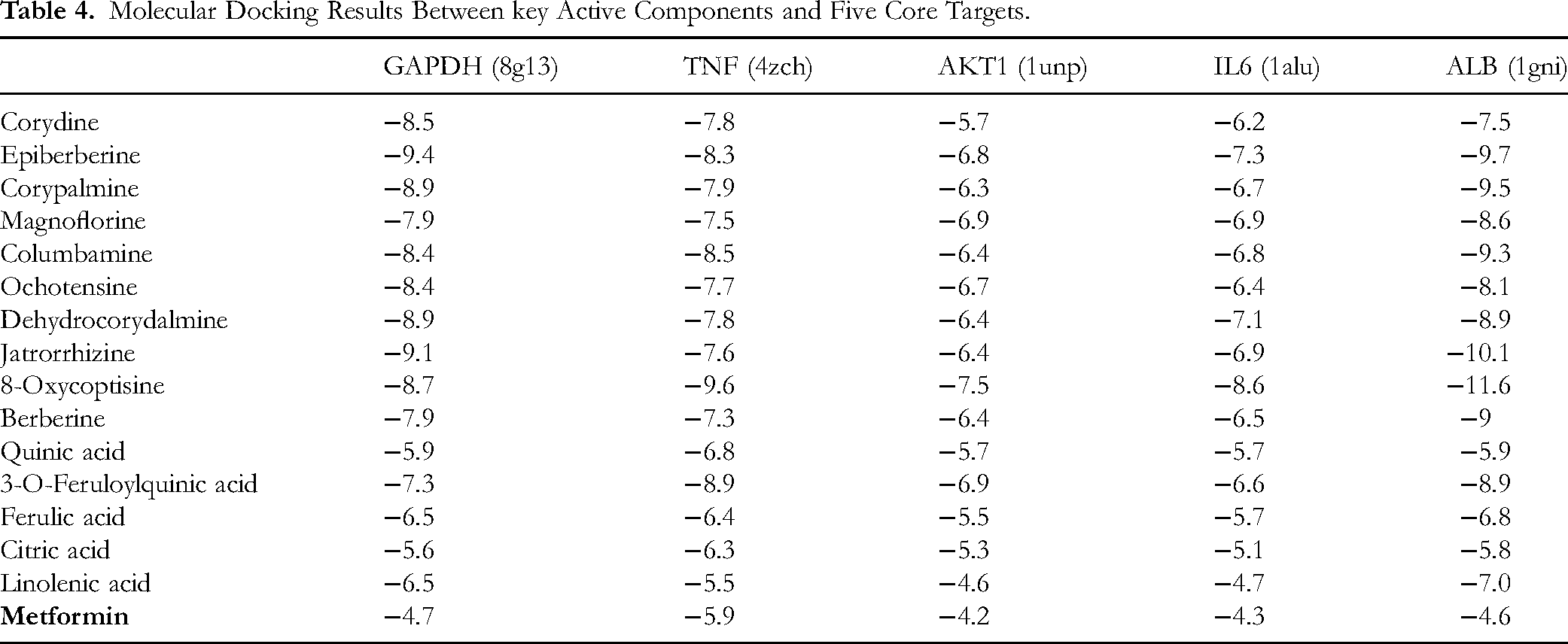
General Conditions, Body Weight, Blood Glucose, and OGTT Changes
Over a 21-day pharmacological intervention study, the physical condition, coat appearance, body weight, fasting blood glucose, and glucose tolerance were assessed in a diabetic mouse model. Mice in the NC group remained in good health, while those in the Mod group exhibited typical diabetic symptoms. Intervention groups, particularly the high-dose cohort, showed substantial improvement. Weight analysis (Figure 10a) confirmed the model's successful establishment, though post-treatment differences among groups were not statistically significant (P > .05). Fasting blood glucose measurements (Figure 10b) indicated a significant decline in glucose levels across all treatment groups after intervention (P < .05). The glucose tolerance test (Figure 10c) provided additional evidence of treatment efficacy, with varying degrees of blood glucose reduction observed in the treatment groups at 60 and 120 min post-challenge. These results highlight the intervention's ability to improve physiological parameters in the diabetic mouse model, offering robust experimental support for its potential in diabetes therapy.

Effects of CRMF on changes in body weight, blood glucose, and OGTT in mice.
The Effects of CRMF on Organ Weight and Organ index in Groups of Mice
Organ weights and indices for each mouse group were presented in Figure 11. The analysis revealed that liver weight in the Mod group was significantly reduced compared to the NC group, while the liver, kidney, and pancreas indices were notably elevated (P < .01 or P < .05). Following drug intervention, all measured organs exhibited varying degrees of improvement relative to the Mod group.

Organ weights and organ indices across mouse groups (mean ± SD, n = 6). Data were expressed as means ± SD. (n = 6). #P < .05, ##P < .01vs.NC group; ∗P < .05, ∗∗P < .01 versus Mod group.
The Effects of CRMF on INS and HOMA-IR in Groups of Mice
Fasting insulin levels and the insulin resistance index (HOMA-IR) were evaluated in mice. The Mod group displayed significantly elevated fasting insulin levels compared to the NC group (P < .01). Treatment with Metformin (Met) and CRMF at high, medium, and low doses (CRMF-Hig, CRMF-Mid, and CRMF-Low) resulted in significantly reduced fasting insulin levels compared to the Mod group (P < .05). HOMA-IR values in the Mod group were markedly increased (P < .01), indicating heightened insulin resistance. In contrast, HOMA-IR was significantly reduced in the Met and CRMF treatment groups (P < .01), as illustrated in Figure 12.

Effects of CRMF on INS and HOMA-IR in groups of mice. Data were expressed as means ± SD. (n = 6). #P < .05, ##P < .01vs.NC group; ∗P < .05, ∗∗P < .01 versus Mod group.
The Effects of CRMF on TC, TG, LDL-C, and HDL-C Levels in Groups of Mice
Compared to the NC group, the Mod group exhibited significantly higher serum levels of TC, TG, and LDL-C (P < .01), alongside a notable reduction in HDL-C levels (P < .01). Relative to the Mod group, the Met group and all CRMF treatment groups (CRMF-High, CRMF-Mid, and CRMF-Low) showed marked decreases in TC, TG, and LDL-C levels (P < .01) accompanied by significant increases in HDL-C levels (P < .01, P < .05). Among the treatment groups, CRMF-High demonstrated the most pronounced effects, as depicted in Figure 13.

Effects of CRMF on TC, TG, LDL-C, and HDL-C levels in groups of mice. Data were expressed as means ± SD (n = 6). #P < .05, ##P < .01vs.NC group; ∗P < .05, ∗∗P < .01 versus Mod group.
The Effects of CRMF Intervention on Oxidative Stress and Inflammatory Factor Levels in Mice
This study assessed oxidative stress and inflammation markers in various mouse groups to evaluate the effects of model induction and therapeutic interventions. Compared to the NC group, the Mod group demonstrated significantly reduced SOD and GSH levels (P < .01) alongside markedly elevated MDA concentrations (P < .01). Furthermore, CRP, IL-6, and TNF-α levels were significantly higher (P < .01), confirming the successful induction of oxidative stress and inflammation. Following drug intervention, MDA levels in the Met group and all CRMF dose groups were significantly decreased (P < .01) relative to the Mod group, while SOD and GSH levels showed significant improvement (P < .01 or P < .05). IL-6, CRP, and TNF-α levels were notably reduced (P < .01) across all CRMF dose groups, indicating strong antioxidant and anti-inflammatory properties of CRMF, as shown in Figure 14A-B.
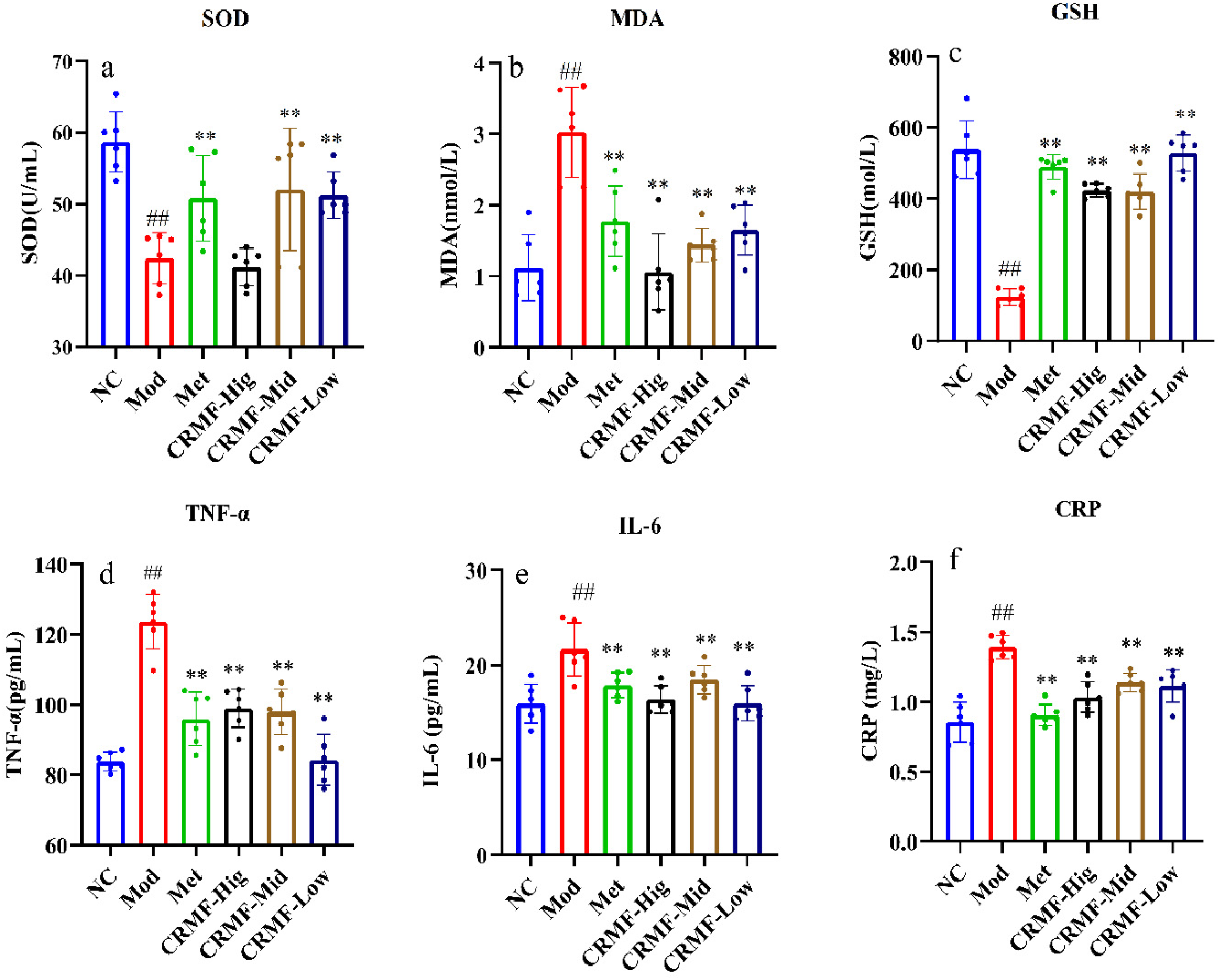
Effects of CRMF intervention on SOD, MDA, GSH, CRP, IL-6, and TNF-α in groups of mice. Data were expressed as means ± SD. (n = 6). #P < .05, ##P < .01vs.NC group; ∗P < .05, ∗∗P < .01 versus Mod group.
The Effects of CRMF Intervention on Mice Tissue Morphology
Histopathological analysis of pancreatic tissue revealed distinct morphological variations among the groups. In the NC group, the parenchyma of the pancreas in the field of view consisted of the exocrine and endocrine parts. The exocrine part was a plasma complex tubular vesicular gland with normal cell morphology. The endocrine part was the pancreatic islets, which had a regular shape, a regular arrangement of the cells within the islets, and a normal morphology. No obvious abnormality was seen. In the Mod group, vacuolar degeneration of follicular cells was observed in the exocrine division, with round vacuoles in the cytoplasm (black arrows). The endocrine division consisted of pancreatic islets, which were significantly reduced in size and irregular in shape (Figure 15(B)). The cells within the islets were irregularly arranged, and occasionally exhibited hydropic degeneration (blue arrows). Capillary dilatation was frequently observed in the pancreatic islets (yellow arrows). Following drug administration, the exocrine part of the pancreas in the treatment group returned to a plasma complex tubular vesicular gland with normal cell morphology. The endocrine part consisted of pancreatic islets, which showed a slight decrease in volume but a more regular shape. The cell arrangement within the islets improved, although occasional small amounts of cell watery degeneration were still present. Cell swelling was reduced, and the cytoplasm remained sparse and light-stained, as depicted in Figure 15(A).

Effect of CRMF on histopathology of pancreas in mice of all groups (HE, 400 ×) (A). The measurement of Islet Area in mice of all groups(B). Data were expressed as means ± SD. (n = 3). #P < .05, ##P < .01vs.NC group; ∗P < .05, ∗∗P < .01 versus Mod group.
Discussion
Diabetes poses a substantial threat to human health by causing both acute and chronic complications, often resulting in multi-organ damage29,30 As a multifaceted metabolic disorder, 31 its pathogenesis is driven by intricate interactions among numerous molecular targets and signaling pathways. 32 The Huanglian-Wumei medicinal pair is a commonly utilized combination for the prevention and treatment of type 2 diabetes mellitus, derived from the Wu Mei Pill, which has been extensively employed in clinical practice. This pair of drugs is a good example of the “Ku Suan Zhi Tian”’ approach to preventing and treating type 2 diabetes mellitus, and this study provides a reference for the clinical use of bitter and sour herbs in the prevention and treatment of diabetes mellitus. The findings of this study suggest that the primary active chemical constituents responsible for the anti-diabetic effects of the CRMF herb pair include berberine, 8-oxycoptisine, dehydrocorydalmine, corydine, epiberberine, corypalmine, magnoflorine, ochotensine, palmatine, citric acid, itaconic acid, jatrorrhizine, quinic acid, 3-O-feruloylquinic acid, ferulic acid, among others. Study shows that berberine, one of the key bioactive components, upregulates AMPK protein expression, 33 thereby inhibiting hepatic glucose production. Furthermore, it accelerates channel closure, reduces KCNH6 current, and promotes insulin secretion. 34 Epiberberine and magnoflorine exhibit significant anti-diabetic potential, functioning as both PTP1B inhibitors and ONOO(-)-mediated protein tyrosine nitration inhibitors. 35 Citric acid. itaconic acid, quinic acid, 3-O-feruloylquinic acid, and ferulic acid serve as crucial intermediates that enhance ATP production in the citric acid cycle, thereby facilitating optimal insulin secretion.36,37
We identified five core targets for the anti-diabetic effects of the CRMF through PPI network analysis. As a crucial enzyme in glycolysis, GAPDH not only modulates insulin signaling pathways but also plays a significant role in the pathogenesis of diabetic complications, particularly retinopathy and vascular endothelial dysfunction.38,39 TNF-α and IL-6 promote insulin resistance and chronic inflammation through activation of the NF-κB and JNK pathways. 40 These cytokines exacerbate insulin resistance, directly impair pancreatic β-cell function, and drive diabetes-related vascular complications.41,42 AKT plays a central role in cell survival, growth, metabolism, and migration, and its dysfunction is closely associated with various diseases, including cancer and diabetes.43,44 Elevated ALB levels have been correlated with insulin resistance, further substantiating its relevance in diabetes pathology 45 In animal models, CRMF intervention markedly increases oxidative stress markers, including SOD and GSH levels, while reducing MDA concentrations, suggesting an enhancement of antioxidant capacity potentially mediated. Additionally, significant reductions in pro-inflammatory mediators, such as CRP, IL-6, and TNF-α. And improved insulin levels. Studies have indicated that activation of the AGE-RAGE signaling pathway induces extensive disruptions in metabolic and inflammatory processes, elevates oxidative stress, and impairs insulin and lipid metabolism, potentially contributing to chronic inflammation, cardiovascular diseases, and metabolic disorders.46,47 Therefore, we predict that CRMF may have been introduced via the AGE-RAGE signalling pathway, insulin pathway. Thus, it exerts an anti-diabetic effect.
However, there are some limitations to this study, the animal model and dosing regimen employed in this study may not adequately recapitulate the chronic progression of diabetes in human patients. Furthermore, the limitations of current online pharmacological databases could potentially compromise the accuracy of predicted outcomes. In order to advance this research, future studies will clarify the material basis of the pharmacological effect and establish a model of type 2 diabetes by combining high-fat feed with STZ to determine the optimal dose to be administered and investigate the mechanism of its antidiabetic action. Further provide theoretical basis and experimental basis for the treatment of diabetes mellitus.
Conclusion
Huanglian – Wumei herb pair has some therapeutic effects on diabetes mellitus, and it is predicted that its active ingredients may act mainly through the AGE-RAGE signalling pathway, insulin pathway, etc, and provide new insights into diabetes mellitus treatment.
Footnotes
Abbreviations
Acknowledgements
Key Laboratory of Pharmacodynamics and Material Basis of Chinese Medicine, Shaanxi Administration of Traditional Chinese Medicine is gratefully acknowledged. The authors would like to thank Wuhan Sevier Biotechnology Co. for the experimental results of histopathological examination, and Wuhan Maiwei Metabolic Bio-Technology Co., Ltd for determining the chemical composition. We thank Bullet Edits Limited for the linguistic editing and proofreading of the manuscript.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Statement of Human and Animal Rights
The protocol for animal experimentation was executed by internationally recognized principles for the use and care of laboratory animals. This article does not any studies with human.
ORCID iDs
Ethical Considerations
All experimental procedures conducted in this study adhered strictly to international and national ethical guidelines for the use of animals in research. The protocol was approved by the Experimental Animal Ethics Committee of Shaanxi University of Chinese Medicine, located in Xianyang, China (Approval No.: SUCMDL20240712004; Approval Date: July 12, 2024). All procedures were carried out by trained personnel to ensure both animal welfare and the scientific rigor of the study. We adhere to all established ethics standards in authorship, ensuring the paper's originality, proper acknowledgement of funding sources, and disclosure of any conflicts of interest.
Author contributions/CRediT
Huili Kang: Data curation, Formal analysis, Investigation, Methodology, Resources, Software, Validation, Visualization, Roles/Writing – original draft. Panpan Zhang: Validation, Data curation. Yi Jiang: Investigation, Project administration. Dongdong Zhang: Validation. Xiaomei Song: Supervision. Yuze Li: Formal analysis. Wenli Huang: Data curation. Yuhong Duan: Supervision, Formal analysis. Huawei Zhang: Conceptualization, Visualization, Funding acquisition, Project administration, Writing-reviev. Chong Deng: Conceptualization, Methodology, Project administration, Supervision, Resources, Writing-reviev & editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the State Administration of Traditional Chinese Medicine high-level key discipline construction project (zyyzdxk-2023202); the State Administration of Traditional Chinese Medicine and the Sci-Tech Innovation Talent System Construction Programme of Shaanxi University of Chinese Medicine (2023-CXTD-05).
Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data will be made available on request.
