Abstract
Introduction
Acute liver injury (ALI) is a serious disease featured on massive hepatocyte death and almost complete loss of liver function, which is the most frequent cause of emergency liver transplantation.1,2 ALI can quickly lead to multiorgan failure, coma and is associated with an extremely high rate of mortality. 3 Diwuyanggan (DWYG) is a traditional chinese medicine (TCM) prescription containing 5 herbal medicines, Schisandra chinensis (Turcz.) Baill., Artemisiacapillaris Thunb., Curcuma longa L., Glycyrrhiza uralensis Fisch., and Rehmannia glutinosa Libosch. It has been proverbially applied to treating liver diseases for more than 30 years in Hubei Provincial Hospital of TCM. 4 There were numerous reports suggesting that DWYG had multiple beneficial pharmacological actions, including protecting liver, regulating liver regeneration, improving liver fibrosis, modulating the immune reaction, ameliorating hepatic injury, killing viruses, and suppressing hepatic cancer.5–11 Meanwhile, previous studies manifested that the active constituents in this formula could offer a role against liver injury. For instance, lignans like Schisandrin B, Schisandrol A, and Schisandrol B were the major active components in Schisandra chinensis (Turcz.) Baill., which could treat drug-induced liver damage by involving in the regulation of inflammatory factors and oxidative stress. 12 Besides, curcumin in Curcuma longa L. was reported to improve survival and minimize hepatocellular injury, necroinflammation, and oxidative stress in rat models of fulminant hepatic failure. 13 However, the underlying mechanisms of the preventive effects of DWYG in moderating ALI remain limited.
Global metabolomics, as a branch of system biology, has been a reliable and efficient approach for systematic analysis of dynamic variations of metabolic profiles when organisms are stimulated or disturbed. 14 Recently, there was accumulating evidences indicating that global metabolomics analysis method was applied to estimate the efficacy of TCM prescriptions and reveal its potential molecular mechanisms. 15 There was a metabolomics study on Xiaoyao San confirming the compatibility effects on depression and the results showed that synergistic effects may be associated with key metabolites in the liver such as glutamate, malate, and taurine. 16 Wang et al 17 applied the same method and found that 20 metabolites altered by insomnia were restored to baseline values after Suanzaoren Decoction treatment, which participated in the regulation of amino acid and fatty acid metabolism disorders. Moreover, ultra-high pressure liquid chromatography together with quadrupole time of flight mass spectrometry (UPLC-QTOF-MS/MS) is an appropriate tool to provide high-throughput information on endogenous metabolite in biological samples. 18 Due to these advantages, global metabolomics analysis based on UPLC-QTOF-MS/MS was expected to assume notable roles in revealing the mechanisms of TCM prescriptions.
Because of the complex components in TCM, it is hard to investigate the molecular targets and action mechanisms, especially in a formula consisting of multiple herb medicines by traditional experimental methods. Network pharmacology is a systematical pattern of drug research aimed at observing how drugs intervene in disease networks and influence them. Network pharmacology, which is based on “disease-gene-target-drug” interactions, focuses comprehensively on the concept of multilevel interaction networks from multiple perspectives.19,20 Thus, network pharmacology can be a forceful tool for discovering the potential mechanism of TCM from the perspective of systems and networks. At present, network pharmacology approaches based on large databases have been used to predict the pathogenesis of diseases or interpret the mechanisms of the TCM formula. 21 Meanwhile, metabolomics analyses could discover potential biomarkers from high-throughput experimental data, which reflect the metabolic changes caused by the intervention of TCM. 22 Therefore, the combination of network pharmacology and metabolomics approaches may help to explore the interaction between organisms and drugs by creating a network of biological and chemical information.
In this study, animal experiments were designed for investigating the treatment of DWYG against thioacetamide (TAA)-induced ALI in mice. Then, a comprehensive analysis combined network pharmacology and metabolomics approaches was employed to elucidate the underlying biomarkers and targets of DWYG against ALI. These results would contribute to further research on pharmacological mechanisms of DWYG for treating ALI.
Methods
Chemicals and Materials
HPLC-grade formic acid was purchased from Mreda Technology Co.,Ltd. Fexofenadine was provided by National Institute for Food and Drug Control. TAA (batch No. BCBX4145) was obtained from Sigma-Aldrich. Alanine aminotransferase (ALT) (batch No. 20210324), aspartate aminotransferase (AST) (batch No. 20210322), superoxide dismutase (SOD) (batch No. 20210325), glutathione (GSH) (batch No. 20210323), and malondialdehyde (MDA) (batch No. 20210326) assay kits were obtained from Nanjing Jiancheng Bioengineering Co. Ltd. Mouse interleukin-6 (IL-6) (batch No. E-EL-M0044c), interleukin-1 beta (IL-1β) (batch No. E-EL-M0037c) enzyme-linked immunosorbent assay (ELISA) kits were purchased from Elabscience Biotechnology Co. Ltd.
Preparation of DWYG Extract
DWYG is a TCM preparation licensed by the Hubei Food and Drug Administration (Grant No. Z20113160). There are 5 Chinese medicinal herbal extracts included in this formula, whose proportions are as follows (w/w): Rehmannia glutinosa Libosch., 20.0%; Artemisiacapillaris Thunb., 33.3%; Glycyrrhiza uralensis Fisch., 13.4%; Schisandra chinensis (Turcz.) Baill., 20.0%; Curcuma longa L., 13.4%. Above 5 herbs were derived from Jiaozuo, Anqing, Ordos, Shenyang, and Yulin in China, respectively. DWYG extract utilized in this study was provided by Hubei Provincial Hospital of TCM. After vacuum seasoning and pulverizing the above-refined concentrations, the DWYG powder was prepared with an extraction yield of 26.32%.
Animals and Treatment
Twenty-four male Kunming mice (20-25 g) were purchased from the Animal Center of Tongji Medical College, Huazhong University of Science and Technology, with certification No. SCXK (e) 2017-0067. The mice were placed in an air-conditioned animal room at 24 ± 2 °C (temperature) and 60% ± 5% (humidity) with a 12 h cycle of light and dark. Following an adaptation period of 3 days, the mice were randomly assigned to the following 4 groups of 6 each: Control group, TAA model group, Control + DWYG group, and TAA + DWYG group. The mice in Control and TAA model groups were given 0.4 mL of distilled water daily by gastric perfusion. The animals in Control + DWYG group and TAA + DWYG group were preadministered with 150 mg/kg of DWYG extract once daily for 14 days. For single gastric perfusion, the samples were dispersed and dissolved homogeneously in 0.4 mL of water. After the last intragastric administration, the animals in model and TAA + DWYG groups were given an intraperitoneal (i.p.) injection of 200 mg/kg TAA for mice to cause ALI.23,24
After 24 h of TAA treatment, blood samples and liver tissues were collected from all the mice under pentobarbitone sodium anesthesia, and then sacrificed by cervical dislocation. By centrifugation at 4000 r/min for 10 min, the corresponding sera samples were immediately separated into aliquots, and stored at −80 ˚C until further analyses. The liver tissues were excised and weighted, and to prepare pathological sections for analysis, a proportion of each liver was soaked in tissue fixation solution, while the others were kept at −80 ˚C for subsequent experiments. All animal experimental procedures were given permission by the Institutional Animal Care and Use Committee of Hubei University of Chinese Medicine (Approval No. 202107002).
Biochemical Analysis and Histological Evaluation
Under the manufacturer's protocol, serum IL-6 and IL-1β levels were measured by ELISA using each of the commercial kits. Other biochemical parameters, such as the activities of serum ALT and AST, the contents of SOD, MDA and GSH in liver tissue were measured by colorimetry using manufactured kits in accordance with the guidelines.
We soaked liver specimens in increasing concentrations of alcohol (75%, 85%, 90%, 95%, and 100% ethanol), then cleared them with xylene and embedded them in paraffin. Hematoxylin and eosin staining was implemented on 4-μm-thick sections. Light microscopy was used to examine the damage and inflammatory infiltration of tissue. Hepatic index was calculated based on the following formula: Hepatic index = (liver weight/body weight) × 100%.
Untargeted Metabolomics Based on UPLC-QTOF-MS/MS
Sample Preparation of Serum
At room temperature, serum samples stored at −80 ˚C were thawed. 40 μL of sample was extracted with 160 μL of acetonitrile solution that contained 100 ng/mL fexofenadine as an internal standard. Particulates and proteins were removed from the extract after vortexing for 2 min, preserving at 4 ˚C for 10 min, and centrifuging at 12000 r/min for 10 min at 4 ˚C. An equal volume of the 150 μL supernatant was transferred into the sample vials. Mix aliquots of all supernatant samples were made into 1 pooled sample to be the quality control (QC) sample.
LC-MS/MS Conditions
With the ACQUITY UPLC M-Class system, Chromatographic separations were carried out at 40 ˚C on a Waters ACQUITY UPLC BEH C18 column (2.1 × 100 mm, 1.7 μm). The flow rate was 0.3 mL/min and the sample injection volume was 2.0 µL. Water/formic acid (1000: 1, v) and methanol were used as mobile phases A and B, respectively. Gradient elution programs for UPLC with linear gradients were as follows: 0 min, 10% B; 15.0 min, 95% B; 20.0 min, 95% B; 21.0 min, 10% B; 25.0 min, 10% B.
Positive ion electrospray data acquisition in the pattern of sensitivity analysis was accomplished using a Waters Xevo G2-XS QTof platform equipped with an electrospray ionization source (Waters). And the following MS operating parameters were used: flow rates of cone gas and desolvation gas were both 50 L/h, temperatures at source and desolvation were 100 and 500 °C respectively, capillary and cone voltages were 3.0 kV and 20 V, respectively. With a scan lasting 1 s, the MS mass ranges for full scanning in MSE mode were set between 50 and 1200 m/z.
Data Processing and Multivariate Data Analysis
In order to obtain raw data files from mass spectrometry, Masslynx V4.1 software (Waters) was used. With the help of Markerlynx XS (Waters), the raw data were extracted, aligned, and matched. In order to reduce the abundance of obvious false positives and noises, the following method parameters were set: (1) the mass tolerance of 0.01 Da for the peak alignment, (2) the noise elimination level was 20, (3) Peaks retention time (RT) from 2 to 20 min were analyzed, (4) peaks with detection rate based on a “70% rule” to reduce the missing value input. After data preprocessing, a data set including information on m/z, RT, and peak intensity of compounds was obtained. The reduced representation of the data table included was transformed by log10 before being analyzed.
For the purpose of multivariate data analysis, SIMCA-P (v13.0, Umetrics) was used to import the data set. Principal component analysis (PCA) was subsequently performed to draw the score plot for display the overall differences. The model was validated and the different metabolites between groups were explored by employing orthogonal projection to latent structure-discriminate analysis (OPLS-DA). For further statistical analysis, metabolites were identified as candidate biomarkers in the light of variable importance in the projection (VIP)˃1.0, fold change (FC) ≥ 1.2or FC ≤ 0.8. Identification of all differential metabolites was performed by accurate fragmentation mass matching with the Human Metabolome Database (https://www.hmdb.ca) and METLIN database (http://metlin.scripps.edu). In order to provide a comprehensive and intuitive picture of the linkages and differences between different groups, a heat map was created to display the relative intensities of differential metabolites in serum using GraphPad Prism 9.0 (GraphPad Software Inc.). Finally, metabolic pathways of metabolites in serum were analyzed by using Kyoto Encyclopedia of Genes and Genome (KEGG) (https://www.kegg.jp) combined with previous literary works.
Network Pharmacology Analysis
Network Construction and Analysis of DWYG on ALI
Collecting chemical compounds of DWYG from the TCM systems pharmacology database (http://lsp.nwu.edu.cn/tcmsp.php; TCMSP). Because of the complex changes in the active chemical composition of the Chinese herbal prescription, the multifarious active constituents were filtered by their pharmacodynamics and pharmacokinetic characteristics, including oral bioavailability (OB) ≥ 30 and drug-likeness (DL) ≥ 0.18. Then, the target proteins of the effective compounds were screened with high credibility of Norm Fit (≥ 0.9) in the Pharmmapper database (http://www.lilab-ecust.cn/pharmmapper). An interaction network for putative targets and active ingredients of DWYG was completed. Visualization of the interactive network was established using the Cytoscape 3.8.2 software (https://cytoscape.org). In order to investigate the targets for preventing ALI, associated targets of ALI with a relevance score ≥ 15 were retrieved from the Genecards (http://www.genecards.org). Searching for gene names and uniform standard protein names for the above-mentioned targets in the Uniprot database (https://www.uniprot.org). Venn analysis was exploited to extract common target genes between the predicted genes of DWYG major active components and ALI-related targets. Overlap was a set of potential targets in the analysis of network pharmacology, which has direct mutual effects between compounds and diseases.
Functional Enrichment Analysis of DWYG on ALI
Based on STRING 11.5 (https://string-db.org/cgi/input.pl), a protein-protein interaction (PPI) network was generated with medium-confidence data. High-throughput genomic data could be analyzed to identify the biological mechanisms using gene ontology (GO) functional categories such as molecular function (MF), biological process (BP), and cytological component (CC). By enriching pathways in the KEGG, the biological function and correlation could be identified according to the candidate targets. Utilizing the Database for Annotation, Visualization, and Integrated Discovery (https://david.ncifcrf.gov), we carried out GO and KEGG pathways analyses in this study. The results were visualized and displayed using Omicshare (https://www.omicshare.com/tools).
Integrated Analysis Involving Metabonomics and Network Pharmacology
The gene targets associated with the differential metabolites were inquired in the STITCH5.0 (http://stitch.embl.de/cgi/input.pl), such that interactions identified with medium confidence were used. Using multiple proteins pattern in the String database, we drew a network of interactions between gene targets related differential metabolites and targets related the main intervention. “Component-Target-metabolite related protein-Metabolite” (C-T-R-M) and “Herb-Component-Target-Metabolite-Pathway” (H-C-T-M-P) networks were visualized by Cytoscape 3.8.2. software.
Real-time Polymerase Chain Reaction Analysis
Liver samples were lysed by RNA extraction (Servicebio, Wuhan, China) to obtain the total RNA. The RNA purity and concentration were measured using a Nanodrop2000 (Thermo Fisher Scientific). SweScript All-in-one First Strand cDNA Synthesis Super Mix for quantitative polymerase chain reaction (qPCR) (Servicebio) was used for reverse transcription reaction. 2 × SYBR Green qPCR Master Mix (Servicebio) and the CFX96 Real-Time polymerase chain reaction System (Bio-Rad) were used for detecting the mRNA expression levels. All genes expression were normalized to Glyceraldehyde-3-phosphate dehydrogenase expression and calculated by the 2−ΔΔCT method. The primer sequences used for PCR were shown in Supplemental Table S1.
Statistical Analysis
Results in the experiment were expressed as means ± standard deviation. Mann-Whitney U test was performed using IBM SPSS 26.0 software (SPSS Inc.) for the statistical analysis. P < .05 and P < .01 were deemed to be statistically significant and highly significant, respectively.
Results
Preventive Effect of DWYG Against ALI
The results on the hepatoprotective effect of DWYG against TAA-induced ALI have been summarized in Table 1. The liver weight and hepatic index of mice with TAA administration (TAA group) prominently increased (P < .01). However, the mice in TAA + DWYG group showed a significant amelioration on hepatic index compared to TAA group after 2 weeks’ pretreatment of DWYG (P < .05). Meanwhile, liver weights of TAA mice treated with DWYG suggested a certain range of reduction, although the difference didn't appear to be statistically noticeable.
Physical and Biochemical Parameters in Mice With Acute Liver Injury (Mean ± SD, n = 6).

Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; IL-6, interleukin-6; IL-1β, interleukin-1β; SOD, superoxide dismutase; MDA, malondialdehyde; GSH, glutathione; TAA, thioacetamide; DWYG, Diwuyanggan; hepatic indexes = (liver weight/body weight) × 100%.
Mann-Whitney U test was used to calculate significant difference.
, P < .05, ##, P < .01, compared with control group.
, P < .05, ∗∗, P < .01, compared with TAA group.
For biochemical parameters, the serum levels of ALT, AST, IL-6, and IL-1β were markedly higher in the TAA group than in the control group. Conversely, DWYG preadministered significantly reduced the elevated ALT, AST, IL-6, and IL-1β by 8.5%, 6.6%, 28.8%, and 42.8%, respectively, in comparison to the TAA group. Meanwhile, the activity of SOD and the content of GSH in TAA-treated group were inferior to those in the control group (P < .01). In contrast, DWYG pretreatment group also showed to inhibit the decrease of SOD and GSH (P < .01). Furthermore, exposure to TAA led to a remarkable increase in liver MDA activity. However, the DWYG preadministration significantly ameliorated the effects on ALI mice, which had abnormally high levels of MDA in liver tissue.
To further investigate the protective effect of DWYG against ALI, the liver morphology and histopathology were performed in Figure 1. As depicted in Figure 1A, TAA obviously induced morphological injury in the liver after 24 h treatment, which manifested as the liver lobuli appeared swollen, congestive, and brittle as well as the tissue color turned into darken. Oppositely, the morphological changes of TAA-induced injury were alleviated by DWYG in mice liver. Moreover, the results of H&E staining (Figure 1B) manifested that the livers in the control group showed normal architecture, whereas the livers in TAA group exhibited loss of cellular boundaries, reduced number of hepatocytes, and no radial arrangement. Similarly, liver sections in TAA + DWYG group showed a reduced extent and progress of multiple degenerative alterations, especially for hepatocellular necrosis. Besides, all mice treated with only DWYG extract had normal livers under the microscope.

Effects of DWYG on TAA-induced histopathological changes in livers. (A) Morphological changes in liver tissues of different groups. (B) Representative light microscopic photographs in the liver sections of mice stained with H&E staining under microscope 100 × . (a) Control group. (b) TAA group. (c) Control + DWYG group. (d) TAA + DWYG group. Abbreviations: DWYG, Diwuyanggan; TAA, thioacetamide; H&E, hematoxylin and eosin.
Untargeted Metabolomics Analysis
As depicted in Figure 2A, an unsupervised model of PCA was first employed to differentiate discrepancies among the control, TAA, TAA + DWYG groups and QC samples in the serum metabolome, which indicated that each group showed an evident clustering trend, and each group was distinctly separated from the others. The PCA score plots in Figure 2(B and C) manifested the significant differences in metabolic profiles between TAA and normal or TAA + DWYG groups. In order to access more valuable information about differences between intergroups, the OPLS-DA models were used and score plots were shown in Supplemental Figure S1 (A and B). The values of R2Y and Q2 were 0.997 and 0.818 for control group and TAA group, and 0.996 and 0.756 for TAA group and TAA + DWYG group of the models, which indicated the excellent fitting and prediction reliability. Permutation tests with 999 iterations showed that the models of OPLS-DA were not over-fitted (Figure S1C and S1D).
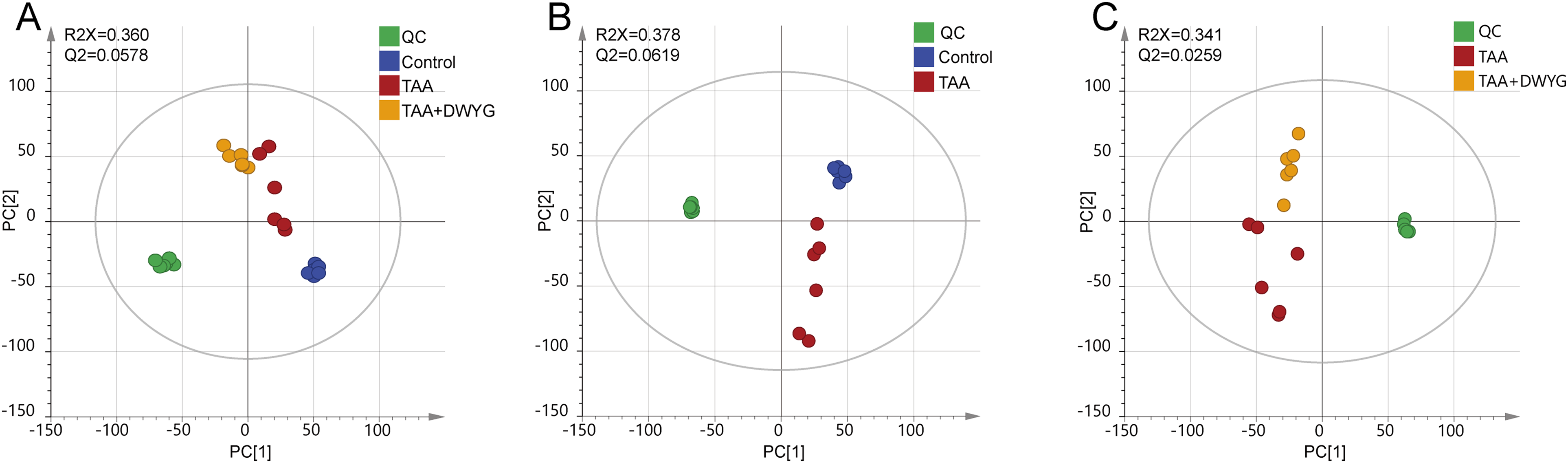
PCA score scatter plots of different groups. (A) PCA score scatter plots obtained from the control, TAA, TAA + DWYG and quality control groups. (B) PCA score scatter plots obtained from the control and TAA group. (C) PCA score scatter plots obtained from the TAA and TAA + DWYG group. Abbreviations: DWYG, Diwuyanggan; TAA, thioacetamide; PCA, principal component analysis.
The raw data consisting of 26 387 features was acquired from the analysis of UPLC-QTOF-MS/MS. Based on the parameters mentioned in the method, data preprocessing was carried out to reduce the interference terms, and then 14 709 features were selected and log10 transformed for multivariate data analysis. In the OPLS-DA model between control and TAA groups, 1539 features with VIP > 1.0 were kept as the important variables. Unretrievable variables were excluded from METLIN and HMDB databases, resulting in 648 variables. There were 294 variables remaining after checking precursor ions in both positive and negative modes. According to FC analysis, 52 candidate metabolites were obtained for further identification. Finally, a total of 28 differential metabolites with reversed trends after DWYG preadministration were retrieved and identified through comparing MS and MS/MS fragment ions pattern with online databases (see Table S2). The homologous formula, as well as, parameters including RT, m/z, VIP values, differences among groups, and the related metabolic pathways of these differential metabolites were shown in Table 2. According to the identification results, there were 27 up-regulated metabolites and 1 down-regulated metabolites in the TAA-induced group, whereas DWYG inhibited the variation in these compounds. To improve the visualization, the distribution patterns and the variations in relative contents of 28 potential metabolites in the 3 groups were displayed by a heatmap (Figure 3). Color changes in the heatmap from blue to red indicated a decrease or increase in the relative content of each metabolite. On the basis of 28 altered metabolites with a reversed trend, a metabolic network was conducted to further analyze the potential pathways of DWYG action in ALI. The enrichment results of metabolic pathways were displayed in Figure 4, which included 6 metabolic pathways regulated by DWYG, tryptophan metabolism, tyrosine metabolism, glycerophospholipid metabolism, fatty acid metabolism, sphingolipid signaling pathway, and bile metabolism.
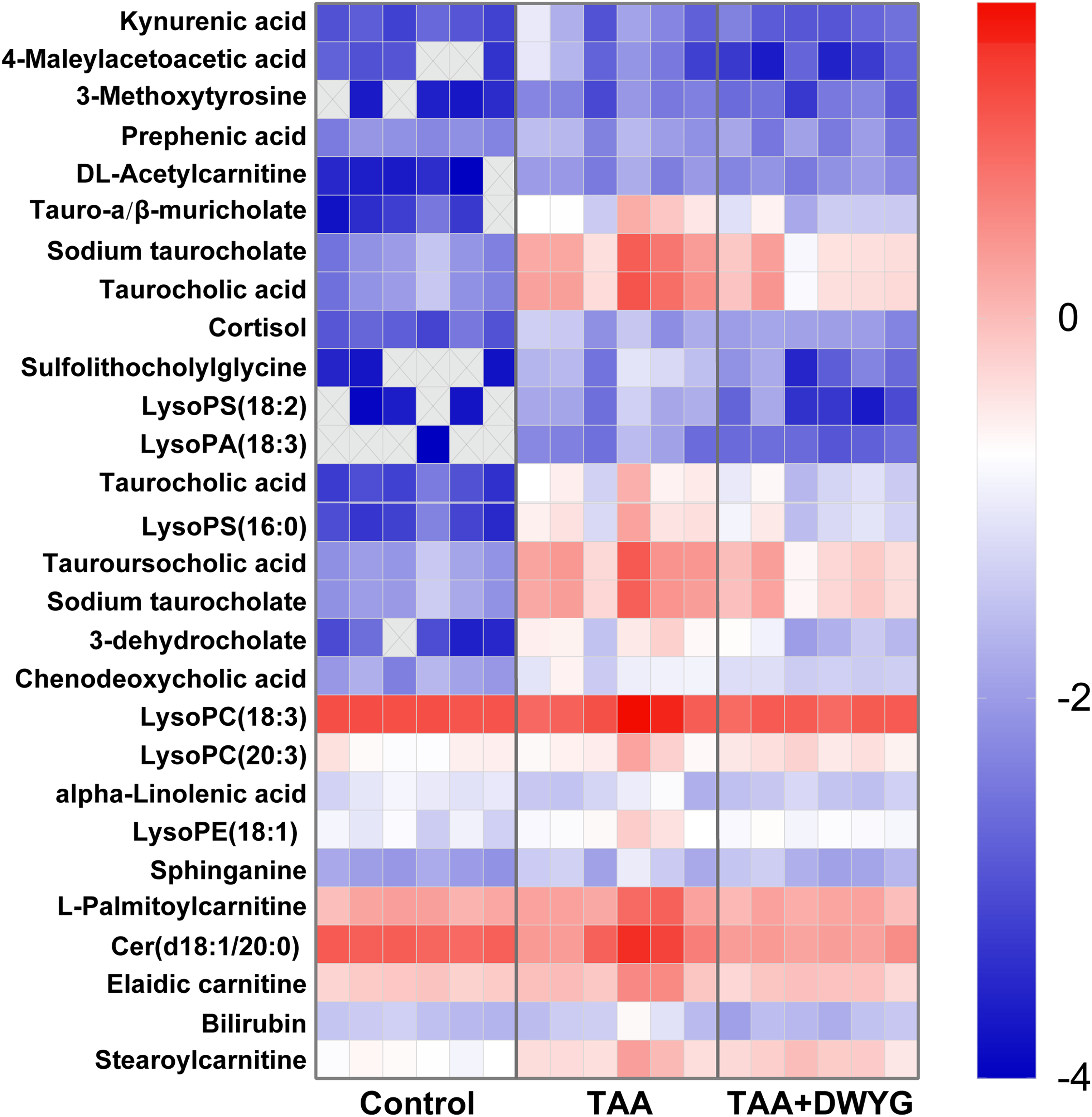
The relative contents of 28 discriminant metabolites in control, TAA, and TAA + DWYG groups. The value of each compound was the log10 transformation of its relative content. The metabolites not detected in the sample were marked as cross in the heatmap. Abbreviations: DWYG, Diwuyanggan; TAA, thioacetamide.

Metabolic pathways of DWYG in the prevention of acute liver injury. The metabolites colored red indicated increasing levels in the TAA model group, compared with in the control group; colored blue indicated declining levels. Abbreviations: DWYG, Diwuyanggan; TAA, thioacetamide.
The 28 Differential Metabolites of TAA Induced Acute Liver Injury Mice Treated by DWYG and Their Related Information.

Abbreviations: DWYG, Diwuyanggan; TAA, thioacetamide; OPLS-DA, orthogonal projection to latent structure-discriminate analysis; VIP, variable importance in the projection.
Metabolite identification was carried out by comparison to the METLIN and HMDB metabolite databases.
Calculated using the OPLS-DA model based on metabolites of the control group and a model group of serum samples.
↑, up-regulated level of the metabolite (the ratio ≤ 0.8); ↓, down-regulated level of the metabolite (the ratio ≥ 1.2), when TAA group compared to control group.
↑, up-regulated level of the metabolite (the ratio ≥ 1.2); ↓, down-regulated level of the metabolite (the ratio ≤ 0.8), when TAA + DWYG group compared to TAA group.
Related metabolic pathway was referred to KEGG and HMDB metabolite databases.
Network Pharmacology Analysis
From the 898 compounds of DWYG, 127 active compounds were selected by the OB and DL filtering criteria from the TCMSP database. Among them, there were 13 active compounds from Artemisiacapillaris Thunb., 3 active compounds from Curcuma longa L., 2 active compounds from Rehmannia glutinosa Libosch., 21 active compounds from Schisandra chinensis (Turcz.) Baill. and 92 active compounds from Glycyrrhiza uralensis Fisch., which shared 4 active compounds (Table S3). Meanwhile, the 110 targets information corresponding to the compounds was collected in Pharmmapper (Supplemental Table S4). For the purpose of identifying the corresponding relationships among the herbs, active constituents and associated targets were guided into Cytoscape to build H-C-T network, as shown in Supplemental Figure S2. The results indicated that H-C-T network contained 5 herbs, 127 components, 110 targets, and 2631 interactions, in which green hexagons, red to orange circles, and purple tetragons corresponded to herbs, active constituents as well as their target proteins, respectively. The above results reflected the features of multiconstituent and multitarget synergy of DWYG.
A total of 665 targets of ALI were discovered in GeneCards according to the Relevance score (Table S5). The result of Venn diagram was established in Figure 5A, which suggested that 26 overlapping genes were obtained by intersecting 110 compound-related genes with 665 disease-related genes. It suggested that DWYG might play a part in the activation of hepatoprotective genes. Therefore, a PPI network of the targets of DWYG to treat ALI was established with the help of STRING, which was displayed in Figure 5B. There were 26 nodes and 118 edges in the interaction network. According to the interaction relationship among targets, the 5 nodes with the largest degree value might be related to the mechanism of DWYG in the prevention of ALI, including albumin (ALB), epidermal growth factor receptor (EGFR), annexin A5 (ANXA5), nonreceptor protein tyrosine kinase (SRC) and mitogen-activated protein kinase 14 (MAPK14).

(A) Venn diagram of drug and disease targets to screen targets of Diwuyanggan (DWYG) for preventing acute liver injury. The blue represented the targets of DWYG; the red represented the targets related to acute liver injury. (B) The interaction network of identified targets. Network nodes represent proteins. Edges represent protein-protein associations.
To elucidate the biological roles of DWYG for treating ALI, we parsed the metabolic pathways by the GO functions and KEGG pathway, on the basis of aforementioned 26 overlapping targets input into DAVID. The results of GO analysis suggested that the BP, CC, MF obtained 112, 18, 34 enrichment results, respectively. As indicated in Figure 6A, the results of enrichment were sequenced with p value, and the top 10 analysis results for 3 sections were plotted using Omicshare, which mainly included positive regulations of reactive oxygen species metabolic process, organ regeneration, and fatty acid oxidation, protein tyrosine kinase activity, MAP kinase activity, and extracellular exosome. Our analysis of KEGG enrichment revealed 65 related signaling pathways, and the top 20 pathways were screened on the basis of that result. As shown in Figure 6B, the primary relevant pathways included the VEGF signaling pathway, PI3K-Akt signaling pathway, and ErbB signaling pathway. Meanwhile, Sphingolipid signaling pathway (hsa04550) with P-value of .008, was significantly enriched. This finding was partly consistent with the results of serum metabolomics analysis by UPLC-QTOF-MS/MS.

(A) Gene ontology terms of the herb-disease targets. The top 10 enrichment terms were identified for each functional category. There were 4 circles in the figure. From outside to inside, the first circle was the classification of enrichment. Different colors represented different classifications. The second circle showed number and P-values of the classification in the background genes. The more the genes, the longer the bars; the smaller the value, redder the color. The third circle was the total number of prospective genes. The fourth circle represented rich factor, which indicated the ratio of genes in the current study versus the total genes in the term. (B) Kyoto Encyclopedia of Genes and Genome (KEGG) pathways of the herb-disease targets. The Y-axis represented the name of the pathway, and the X-axis represented the enrichment factor (Generation, the number of genes belonging to the pathway in the target gene/the number of all the pathway genes in the background gene set). Gene number and -log10 (p) values were indicated by the size of black spots and depth of color, respectively.
Integrated Analysis
As shown in Figure 7(A), the results of network pharmacology and metabolomics analysis were further integrated to construct the C-T-R-M network for the effects of DWYG on ALI. The created networking encompassed 445 connected nodes comprised of 4 parts. The network pharmacology section on the left represented the 127 active components and 26 core targets, respectively. Among the 28 metabolites based on metabolomics analysis on the right, a total of 26 metabolites could find related proteins by means of online databases and be associated with 273 target proteins (see Table S6). Finally, the global view of the H-C-T-M-P regulatory network of DWYG for ALI treatment was shown in Figure 7B. Seven key targets, including mitogen-activated protein kinase 1 (MAPK1), mitogen-activated protein kinase 8 (MAPK8), caspase 3 (CASP3), peroxisome proliferator-activated receptor gamma (PPARG), ALB, carboxylesterase 1 (CES1) and glucocerebrosidase (GBA), were eventually screened, all of which matched the 118 potential active components of DWYG, with a matching rate of 92.91%. The 7 key targets might result in the alteration of 5 metabolites, thus affect tyrosine metabolism, fatty acid metabolism, bile acid biosynthesis, and sphingolipid signaling pathway.

(A) Component-target-metabolite related protein-metabolite interaction network. (B) Herb-component-target-metabolite-pathway interaction network. The red parallelograms represented the effective components of diwuyanggan (DWYG). The green circulars represented the differential metabolites. The purple diamonds represented the metabolite-related proteins. The yellow rectangles represented the potential targets and the orange arrows represented the overlapping target proteins of these components, metabolites, and disease.
Experiment Validation
To evaluate the scientific reliability of the results between metabolomics analysis and network pharmacology, we used real-time polymerase chain reaction (RT-PCR) methods to determine the expression of 7 key targets as validation. As shown in Figure 8, the TAA group suggested significantly elevated MAPK8, CASP3, and GBA expression and reduced MAPK1, PPARG, ALB, and CES1 expression compared to the control group, while the mRNA level of MAPK8, PPARG, ALB, CES1, and GBA significantly changed in a contrary direction after DWYG pretreatment. Meanwhile, DWYG could reverse the abnormal expression of MAPK1 and CASP3 in liver tissue of TAA mice, with a callback rate of 21.29% and 65.38%, respectively, though the difference was not statistically significant.
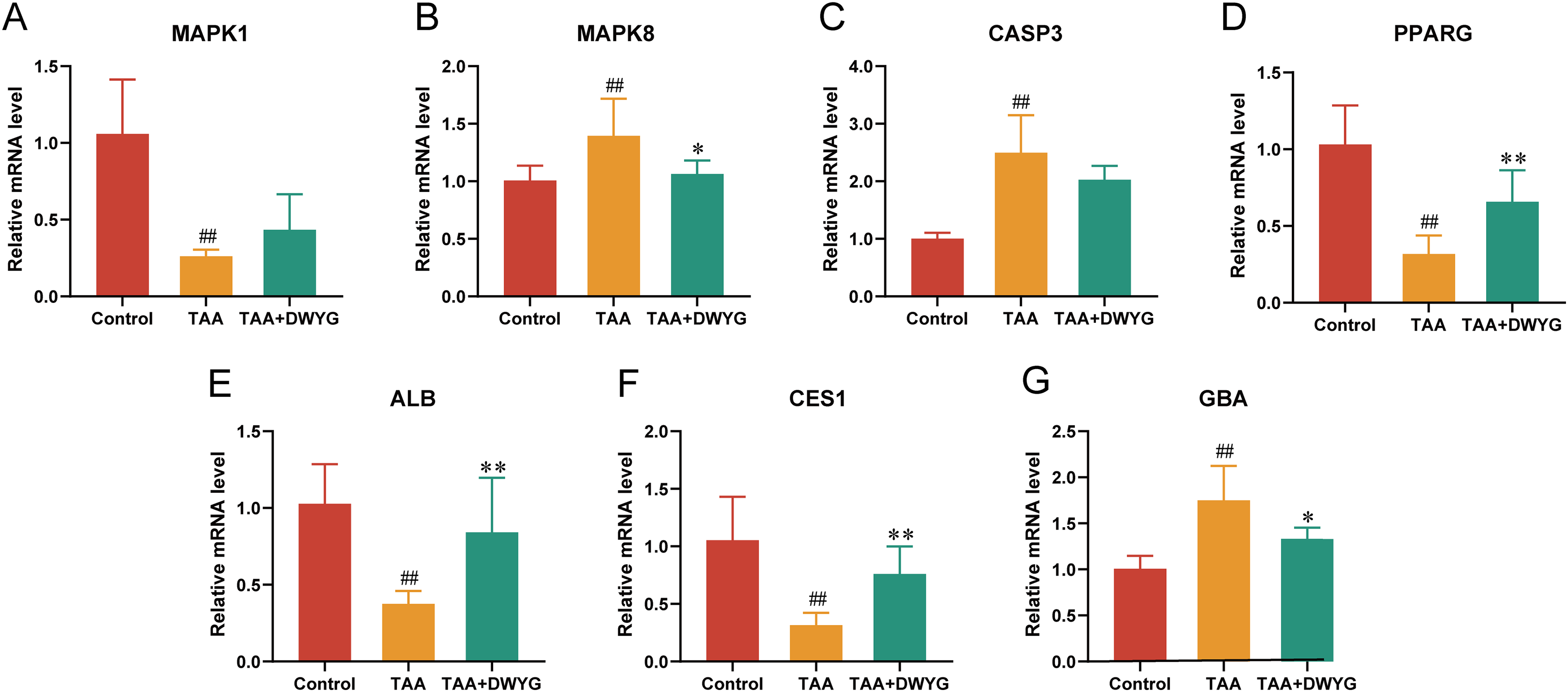
Effect of DWYG extract on the expression of targets in the prevention of acute liver injury detected by RT-PCR (n = 3). (A) Relative mRNA expression of MAPK1; (B) Relative mRNA expression of MAPK8; (C) Relative mRNA expression of CASP3; (D) Relative mRNA expression of PPARG; (E) Relative mRNA expression of ALB; (F) Relative mRNA expression of CES1; (G) Relative mRNA expression of GBA. Mann-Whitney U test was used to calculate significant difference. #, P < .05, ##, P < .01, compared with control group. *, P < .05, **, P < .01, compared with TAA group. Abbreviations: DWYG, Diwuyanggan; TAA, thioacetamide; RT-PCR, real-time polymerase chain reaction; PPARG, peroxisome proliferator-activated receptor gamma; CASP3, caspase 3; ALB, albumin; CES1, carboxylesterase 1; GBA, glucocerebrosidase
Discussion
In this study, an analytical analysis combining network pharmacology and untargeted metabolomics was employed to interpret possible pharmacological mechanism of DWYG on the amelioration for ALI. The results of animal experiments revealed that DWYG could significantly inhibited ALI in TAA model mice. Further, based on the integrated bioinformatics and omics analyses, metabolic pathways related to 28 metabolites and 7 key targets including MAPK1, MAPK8, CASP3, PPARG, ALB, CES1, GBA were revealed to link with the effect of DWYG.
Previous studies showed that DWYG possessed a protective effect on the liver, which was in accord with the results of our study on ALI. DWYG moderated liver damage in embryonic liver L-02 cells and mice caused by injection of CCl4, 8 contributed to the prevention of hepatocellular carcinoma, 9 and had a long-term stable therapeutic effect on treating chronic hepatitis B by improving liver histological response. 10 Accumulating pharmacological experimental studies have demonstrated that herbs in DWYG prescription have hepatoprotective effects. Artemisiacapillaris Thunb. was reported to suppress oxidative stress via the Nrf2 signaling pathway, which could inhibit the expression of NADPH oxidase to prevent TAA-induced liver injury. 25 A previous study indicated that Curcuma longa L. ameliorated lipid dysmetabolism by regulating ER stress and enhancing antioxidant system function. 26 The preventive effects of Schisandra chinensis (Turcz.) Baill. on alcohol-induced liver injury were known to inhibit CYP2E1 activation and activate the antioxidant response element signaling pathway. 27 Glycyrrhiza uralensis Fisch. could alleviate inflammation and enhance the antioxidant capacity. 28 Besides, Rehmannia glutinosa Libosch. was found to have hepatoprotective activity and restore the transaminase activities. 29 In conclusion, the results of our study reconfirmed that DWYG had protective effects on ALI in the following aspects, including pathological tissues, different biochemical indicators such as hepatic function, inflammatory and oxidative stress indicators, and metabolic pathways.
In addition, our findings suggested that DWYG could obviously alleviate metabolic disorders occurred in the progression of TAA-induced ALI, mainly including bile acids, lipids, and amino acids metabolism. When drug-induced liver injury and liver disease caused disruptions in liver synthesis and clearance of bile acids, serum bile acid patterns changed quantitatively. 30 In our metabolomics results, DWYG treatment could protect the liver via bile acid biosynthesis and bile secretion. There were numerous evidences that the marked increase in bile acid levels was one of the main pathophysiology of drug-induced liver injury, which could destruct cholesterol homeostasis, promote liver inflammatory response, and produce toxicity.30–32 There may be a connection between bile acid metabolism and the regulation of DWYG in the inflammatory response.
After 2 weeks’ DWYG extract pretreatment, palmitoylcarnitine, elaidic carnitine, stearoylcarnitine and alpha-Linolenic acid were reversed by comparing with the TAA model group, suggesting that DWYG might contribute to the recovery of the perturbation of mitochondrial fatty acid metabolism. A recent study has proposed that severe inhibition of mitochondrial fatty acid oxidation induced by chemicals could result in an accumulation of long- and medium-chain acylcarnitine and a reduction of free fatty acids in serum, 33 which was consistent with our results.
As for sphingolipid signaling pathway regulated by DWYG, there was evidence that it played critical roles in the progression of liver injury. 34 Ceramide together with sphinganine, as 2 representative products of sphingolipid signaling pathway, were involved in inflammation, inhibiting cell growth, and inducing apoptosis. 35 Studies have shown that liver diseases and drug-induced liver injury often disrupted hepatic metabolism and the levels of sphingolipids were changed notably.33,36 Similar results were acquired in the current study that the levels of serum Cer(d18:1/20:0) and sphinganine were increased in the TAA mice, while attenuated Cer(d18:1/20:0) and sphinganine production might contribute to the preventive effect of DWYG.
In terms of glycerophospholipid metabolism, it was reported that large amounts of lysophospholipids accumulation with potentially cytotoxic led to apoptosis and inflammation in hepatocytes. 37 It might be related to the activation of phospholipase A2 to catalyze the conversion of phospholipids into lysophospholipids, which was crucial in immunity and cellular signal transmission. 38 Furthermore, high levels of lysophospholipids in plasma also demonstrated that there was a disruption in membrane fluidity during the progress of hepatic damage. 39 However, we found that DWYG pretreatment showed favorable modifications of lysophospholipids such as LysoPC, LysoPS, LysoPE, and LysoPA in ALI mice, indicating that the regulation of DWYG on lipid content might inhibit inflammation and promote membrane fluidity and signal transduction in the liver, thereby contribute to its efficacy on ALI.
Alterations of amino acids profiles, especially tryptophan metabolism and tyrosine metabolism, were also observed in the absence of liver regulatory function. Prephenic acid participated in tryptophan metabolism as well as tyrosine biosynthesis. 3-Methoxytyrosine and 4-Maleylacetoacetic acid were intermediates in tyrosine metabolism. Their relative contents in the serum were elevated in mice after TAA exposure in this study, which might be the result of proteolysis and necrosis of liver parenchyma. 40 TAA treatment could reduce the ribosomal RNA of hepatocyte, thus promoting protein degradation and secretion. 41 Kynurenic acid was a natural product metabolized primarily in the liver by tryptophan, which synergistically stimulated the production of IL-6 with inflammatory signaling. 42 A recent animal study indicated an increased level of peripheral kynurenic acid in TAA treatment rats, which was consistent with our results. 43 Our findings of DWYG pretreatment mice, showed a reduction of these amino acid metabolites, suggesting that DWYG could regulate protein synthesis and reduce liver damage by mediating TAA-induced amino acid metabolism disorders.
The results of the integrated metabolomics and network pharmacology analysis indicated that 7 genes including MAPK1, MAPK8, CASP3, PPARG, ALB, CES1, and GBA were considered as potential therapeutic targets. MAPK1, known as extracellular signal-regulated kinase 2 (ERK2), is implicated in regulating cell growth and mitosis. In previous studies, TAA and CCl4-induced hepatotoxicity was shown to reduce ERK2 expression, preventing cells from responding to growth and survival factors.44,45 It has been reported that ERK2 participated in survival signals by phosphorylating tyrosine residues. 46 At the same time, DWYG preadministration increased MAPK1 in the experiment, suggesting that at an early stage in cell regeneration, DWYG might activate tyrosine metabolism to upregulate MAPK1, thus generating regenerative signals and promoting hepatocyte proliferation. MAPK8, as a Ceramide regulatory target in the sphingolipid signaling pathway for apoptosis. It was confirmed to be correlated with the regeneration of inflammation and fatty acid β-oxidation in the liver.47,48 There were many studies indicating that the impaired function of β-cell induced by fatty acid was related to the Ceramide accumulation via a dual mechanism involving the pathways of de novo and rescue.49,50 These evidences suggested that the protective effect of DWYG on mice treated with TAA involved in regulating the levels of Ceramide to inhibit the activation of MAPK8, which could prevent β-cell apoptosis. CASP3, a sensitive marker for liver damage, had a key function in the execution of apoptosis and dramatically upregulated during ALI, 51 hepatocytes could be protected against TAA-induced apoptosis with the ability of DWYG to activate apoptosis protective proteins. PPARG was widely expressed in adipocytes and the liver, which had established an important role in regulating fatty acid storage. 52 The hepatic expression of PPARG was inversely correlated with liver fat and disease severity, 53 which was also observed in our study. Alpha-linolenic acid was a kind of long-chain polyunsaturated fatty acid with a carbon chain binding region, polar carboxyl head, and hydrophobic tail, which was readily oxidized to bind to PPARG. 54 Consequently, we speculated that the beneficial effects of alpha-linolenic acid restoration might explain the activation of PPARG. Furthermore, PPARG might be targeted by the active ingredients in the DWYG formula in order to enhance hepatocyte viability. Two recent reports showed that glabridin and schisandrin B could inhibit inflammation via activation of PPARG, which inhibited carbon tetrachloride-induced liver fibrosis.55,56 ALB was a globulin synthesized by the liver and involved in bile acid biosynthesis. As a strong carrier transport, ALB could transport hormones, bilirubin, and other substances. 57 There was evidence of high mRNA expression of ALB in plasma after TAA treatment, and the increase preceded ALT elevation, therefore ALB might be a predictive biomarker for detecting the early signs of hepatic damage. 58 In the meanwhile, numerous liver diseases showed reduced expression of liver ALB mRNA, which could indicate the degree of liver damage as well. 59 Due to the decline in liver function, the decrease in hepatic ALB levels and its transportation in the liver injury model was closely associated with the reduction of synthesis. 60 Our results suggested that DWYG could upregulate ALB levels and restore its transport function as a carrier, thus restoring liver damage caused by cholestasis. CES1, known as cholesteryl ester hydrolase, was highly expressed in the liver. A report showed that primary liver damage could be directly reflected in CES1 enzyme activity. 61 Meanwhile, CES1 contributed to the detoxification of drugs in liver or serum. 62 It has been demonstrated to promote intracellular cholesterol mobilization and efflux, facilitate bile acid synthesis to protect liver.63,64 Therefore, DWYG influenced bile acid biosynthesis by upregulating the level of CES1 and down-regulating the level of taurine-conjugated primary bile acid in ALI mice. As for GBA, it can contribute to converting glycosylceramide to glucose and ceramide in sphingolipid metabolism. Thus, hyperactivation of GBA may be responsible for the accumulation of ceramide. It was reported that overexpression of GBA could accelerate the progression of hepatocellular carcinoma and affect its poor prognosis. 65 In our study, the level of Cer(d18:1/20:0) in the sphingolipid signaling pathway was reduced after DWYG pretreatment, which might be related to its inhibiting effect on GBA of DWYG. Collectively, our results not only confirmed that these 7 key targets had an essential effect on the protection of ALI by DWYG, but also verified that the results of integrated metabolomics and network pharmacology analysis were credible.
Conclusion
Overall, the study developed an effective strategy combining untargeted metabolomics and network pharmacology to uncover the potential preventive mechanism of DWYG against ALI. As a result, a number of key substances were screened for DWYG to exert preventive effects on ALI, which included 127 bioactive ingredients, 28 differential metabolites, 6 pathways, and 7 key targets. And the action mechanisms of DWYG against ALI might be attributed to the regulation of bile acid, glycerophospholipid, fatty acid, sphingolipid, tryptophan, and tyrosine metabolism pathways. This research provides a theoretical reference for performing an investigation of the mechanism of DWYG and its clinical application.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231180076 - Supplemental material for Integrated Serum Metabolomics and Network Pharmacology Approach to Reveal the Potential Mechanisms of Diwuyanggan Prescription in the Prevention of Acute Liver Injury
Supplemental material, sj-docx-1-npx-10.1177_1934578X231180076 for Integrated Serum Metabolomics and Network Pharmacology Approach to Reveal the Potential Mechanisms of Diwuyanggan Prescription in the Prevention of Acute Liver Injury by Jinlin Xu, Feng Zou, Yuqi Lin, Zhaoxiang Zeng, Yukun Liu, Lijun Zhang, Rongzeng Huang, Xugui Li, Chengwu Song, Yinping Tang and Shuna Jin in Natural Product Communications
Footnotes
Authors’ Contributions
SNJ, and YPT designed the experiments and revised the paper. JLX, and FZ performed the data analyses and wrote the manuscript. YQL, ZXZ, YKL, LJZ, RZH, XGL, and CWS carried out the experiments and collected test data. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Institutional Animal Care and Use Committee of Hubei University of Chinese Medicine, Hubei Province, China (Approval No. 202107002).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science Foundation of the Department of Education of Hubei Province, China (Grant No. Q20212010)
Informed Consent
All animal experimental procedures were conducted in accordance with the Institutional Animal Care guidelines of Hubei University of Chinese Medicine and approved by the Institutional Animal Care and Use Committee of Hubei University of Chinese Medicine, Hubei Province, China (Approval No. 202107002).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
