Abstract
Introduction
The use of indigenous expertise, especially that of ethnobotanists, and the use of plants as sources of potential medicines have received considerable attention in recent years. 1 Also, herbs have greater economic importance as an essential ingredient in food due to their preservative effects caused by the presence of antioxidants and antimicrobial activity, in perfumery and cosmetics products.2,3 Phytochemical compounds and their chemical analogs have provided abundant clinically useful drugs in the treatment of chronic and acute diseases. Still, research is continuing to search for newer therapeutic agents from medicinal plants. 4 The World Health Organization (WHO) estimates that 80% of the population living in developing countries depends exclusively on traditional medicine for their primary health care and more than half of the world's population still relies entirely on plants for medicines. 5 Various parts of Azadirachta indica (Mim or Neem) (called in India: divine tree) such as flowers, leaves, seeds, and bark have been used to treat both acute and chronic human diseases like pyrexia, headache, ulcers, respiratory disorders, cancer, diabetes, leprosy, malaria, dengue, chicken pox, and dermal complications. 5 The tree is popular for its pharmacological attributes while the extracts are reported to be used as antioxidant, hypolipidemic, microbicidal, anti-inflammatory, hepatoprotective, antipyretic, hypoglycemic, insecticidal, antifertility, nematicide, antiulcer, neuroprotective, cardioprotective, and anti-leishmaniasis properties. 6
The pharmacological activity of a plant can be predicted by the identification of the phytochemicals. Currently, phytochemicals are determined by various modern techniques, but the conventional qualitative tests are still popular for the preliminary phytochemical screening of plants. 7
Characterization and evaluation of plants and their phytoconstituents can explore the evidences to support therapeutic claims of those plants against various ailments. Advanced techniques like High-Performance Liquid Chromatography or Gas Chromatography coupled with mass spectrometry detection (HPLC or GC -MS) are very helpful for detection of phytoconstituents both qualitatively as well as quantitatively. 7
Traditionally, A. indica holds significant importance in Chadian culture as a medicinal plant used to treat various ailments. It serves as one of the primary sources for many therapeutic agents. In Chadian traditional medicine, the leaves of A. indica are utilized for treating common conditions such as fever, headaches, respiratory issues, and malaria. Additionally, a paste made from its kernels is applied as a poultice directly to treat external hemorrhoids (This information was gathered from Chadian healers during an ethnobotanical survey conducted by the author in 2022).
Such is the Republic of Chad, the use of local medicinal plants that could control mosquito larvae and insects carrying larvae causing malaria is highly needed. Knowing the bioactive compounds of A. indica leaves or oil and their formulation as drugs will effectively contribute to the fight against malaria and reduce its propagation. Moreover, A. indica is well-known in Chad traditionally for its medical value in treating fever, Malaria, hemorrhoids, stomach aches, renal problems, and respiratory affection but no studies on its biological activity and chemical compounds were locally conducted in Chad.
The study has the potential to contribute to the development of bioactive substances with therapeutic potential, derived from the local plant A. indica grown in Chad. This plant is used to treat ailments caused by various pathogenic bacteria, including Staphylococcus saprophyticus, Methicillin-resistant Staphylococcus sp., and Staphylococcus aureus. Additionally, the research involves characterizing the chemical profile of A. indica leaf extracts using high-performance liquid chromatography-mass spectrometry (HPLC-MS). This powerful technique allows for a detailed analysis of the active compounds. The goal is to prepare leaf extracts of A. indica for conducting qualitative screening tests, characterizing the chemical composition through HPLC-MS analysis, and determining antibacterial activity.
Material and Methods
Sample Collection
The leaf sample of A. indica (Figure 1) was collected in September 2021 from N'Djamena, Republic of Chad. The plant was identified by the herbarium at Toumaï University in N'Djamena. Assessments were conducted by foresters from the National Federation of Associations of Healers and Practitioners of Medicine (Fédération Nationale des Associations des Praticiens de la Medicine au Tchad) (FENAPMT). The sample has been deposited in the Toumaï University herbarium as well as in the Biology, Ecology and Health Laboratory (UAE/U08FS) herbarium in Morocco, with the voucher specimen code GA-LBES/Ech02.

(a) Neem tree. (b) Leaves. (c) Seeds (Photos from the author, Republic of Chad).
Sample Preparation
The dry leave samples (150 g) of A. indica were ground using a grinder. Once the leaves were well dried, they were powdered and stocked in a glass bottle in the dark for further extraction. The dry crude extracts of ethanol, methanol and water were obtained respectively by evaporating the organic solvents and by freeze-drying the water.
Extraction Procedure
The dried powder sample leaves were extracted by Soxhlet extractor using solvents namely ethanol, methanol and water (distilled water). Briefly, 50 g of dried leaves of A. indica were rigorously extracted by Soxhlet in 250 mL of each solvent, then the obtained extracts were concentrated and free of solvent under reduced pressure, using a rotary evaporator, then, ethanol and methanol extracts were obtained. Water extracts were obtained in the same process as Soxhlet in 250 mL of distilled water. Then, the residue of water was eliminated through freeze-drying process. Briefly, the aqueous solution obtained by Soxhlet was frozen at −25 °C for about 1 day, and then submitted to a freeze-dryer (ilShin BioBase Co. Ltd, Made in Korea, ilShin Europe B.V. Ede, The Netherlands, Co. Ltd, Model No: TFD5503 Serial No: LC3000) for about 18 h. At the end of the process, extracts were obtained by drying water. All extracts were stored in airtight bottles in a refrigerator at 4 °C until further analysis.
Qualitative Tests for Preliminary Phytochemical Screening
Detection of Phenolics and Flavonoids
The ethanol and water extracts (10 mg) were completely dissolved in 10 mL of distilled water. The obtained stock solution was used for the phytochemical screening of polyphenols and flavonoids following the methodology of 8 with moderate modifications. To detect phenolic compounds, 500 µL of the diluted crude extracts, 1500 µL of the Folin-Ciocalteu reagent, and 1200 µL of 7.5% saturated aqueous sodium carbonate (Na2CO3) were mixed; the mixture was then thoroughly homogenized and incubated for 45 min at 40 °C. The appearance of a blue color indicates the presence of phenolics. To detect flavonoids compounds, 200 µL of each crude extract was mixed with 2000 µL of distilled water and subsequently with 150 µL of sodium nitrite solution (5% NaNO2); the mixture was incubated for 6 min at room temperature. Thereafter, 150 µL of aluminum trichloride solution (10% AlCl3) was added and then allowed to stand for 6 min, afterward, 2 mL of sodium hydroxide solution (4%NaOH) was added to the mixture, and then 200 µL of distilled water was added. Then, the mixture was thoroughly mixed and allowed to stand for 30 min at room temperature. The appearance of a golden or yellowish color indicates the presence of flavonoids.
Detection of Saponins
The method of 9 was adopted in testing for saponins in the A. indica. A quantity of 5 g of sample was boiled with 50 mL of distilled water and filtered to obtain aqueous extracts. To the 25 mL filtrate of aqueous extracts obtained, 2 mL of the aqueous extracts were added in a tube test and shaken by hand vigorously many times. A foam layer was obtained on the top of the test tube. After 5 min the foam was still stable. This foam layer indicated the presence of saponins.
Detection of Steroids
The method of 10 was adopted for testing steroids with moderate modifications. About 5 g of sample powder leaves of A. indica was weighed out and stirred with 50 mL of distilled water and kept in a balloon heater for 15 min of extraction. After that the solution was filtered and the aqueous extracts were obtained, 2 mL of this aqueous extract was versed in a tube test and 2 mL of anhydride acetic was added and allowed the solution to cool well in a refrigerator followed by the addition 1 mL of concentrated H2SO4 carefully. The upper layer in the test tube turned red and the sulfuric acid layer showed yellow with green fluorescence. It showed the presence of steroids.
Detection of Terpenoids
The method of 11 was adopted for testing terpenoids. 5 g of sample powder of A. indica leaf was weighed out mixed with 50 mL of ethanol and kept for 30 min for extraction. After that, the solution was filtered and the ethanol was evaporated in a water bath; 2 mL of ethanol extract was versed in a tube test and 2 mL of anhydride acetic was added, followed by the addition of 1 mL of concentrated H2SO4 solution carefully. The formation of a reddish-violet color shows the presence of terpenoids.
Detection of Alkaloids
This is based on the utilization of standard conventional protocols as reported by 7 and demonstrated by. 12 5 g of sample powder of A. indica leaf was weighed out mixed with 50 mL of ethanol and kept for 30 min for extraction. Then, the solution was filtrated and the ethanol was evaporated in a water bath, then, 3 mL of hydrochloride acid HCl was added to dissolve completely the solution. Then the solution was filtered. 1 mL of the extract solution was versed in a tube test and a few drops of the picric acid solution were added. A yellow color indicated the presence of alkaloids.
Detection of Tannins
This is also based on the utilization of standard conventional protocols as illustrated by 7 and demonstrated by. 12 5 g of sample powder was stirred with 50 mL of distilled water heated for 15 min in a balloon heater for aqueous extraction. Then, the solution was filtered and the aqueous extracts were obtained. After that, 3 mL of hydrochloride acid was added to dissolve completely the solution. Then the solution was filtered. 500 µL of the filtrated solution was versed in a tube test and a few drops of 10% ferric chloride solution were added. A blue-green color appeared, indicating the presence of tannins.
High-Performance Liquid Chromatography Coupled with Mass Spectrometry Detection Analysis (HPLC-MS)
Separations were performed using a Thermo Scientific™ Dionex™ UltiMate™ 3000 XRS LC system with a Thermo Scientific™ Hypersil™ Gold aQ, 100 × 2.1 mm ID, 1.9 µm particle HPLC Column. The flow rate was 200 µL/min using the following chromatographic gradient:
Mobile Phase: A: 80% Formic Acid 0.3% (pH 2.5) and 20% (v/v) of methanol B: Methanol + 0.1% Formic Acid. The gradient was programed as follows: 100:0 (solvent A: solvent B) for 10 min, from 100:0 to 80:20 in 3 min, remaining at 80:20 for 5 min, to 60:40 in 8 min, remaining at 60:40 for 3 min, to 100:0 in 1 min. The flow rate was 1.0 mL/min and the oven temperature were set at 30 °C.
Mass Spectrometer Method: Samples were analyzed using a TSQ Endura triple-quadrupole mass spectrometer Full Scan mode. Ionization was performed using the new Ion Max NG ion source operated in heated electrospray ionization mode H(ESI), with a vaporizer temperature of 300 °C and a capillary temperature of 350 °C. Sheath and auxiliary gas flows were 45 and 10 (arbitrary units) respectively with an ionization voltage of 3000 volts in both positive and negative ion modes. Collision gas pressure was set at 1.5 m Torr throughout the experiments.
Antibacterial Activity
Microorganisms Used
The leaf extracts of A. indica were evaluated for their in vitro antibacterial activity against various bacterial strains. This included Gram-positive bacteria such as Staphylococcus saprophyticus (SS), Methicillin-resistant Staphylococcus sp. (MRSS), and Staphylococcus aureus (SA) ATCC, as well as Gram-negative bacteria including Klebsiella pneumoniae (KP), Proteus mirabilis (PM), Acinetobacter baumannii (Ab), Escherichia coli (EC), Escherichia coli with extended-spectrum beta-lactamase (EC ESBL), and Salmonella sp. The bacterial strains selected for this study are commonly associated with various health issues, particularly headaches, fevers, skin infections, urinary tract infections, and foodborne and nosocomial infections. These strains were sourced from the Biology and Health Laboratory at the Faculty of Sciences in Tetouan. To prepare for the experiments, the bacteria were sub-cultured on nutrient agar (NA) plates and stored on nutrient agar slants at 4 °C until needed. A final inoculum concentration of 5 × 105 CFU/mL was used, following the guidelines set by the European Committee on Antimicrobial Susceptibility Testing (EUCAST). 13
Method of Diffusion
Antibacterial test was carried out using Mueller Hinton's (MH) method of diffusion. Leaf extracts were diluted in dimethyl sulfoxide (DMSO 10%) with a concentration of 80 mg/mL. After solidification, the cylindrical molds were removed generating at their site wells filled with 50 µL of the leaf extracts. Diffusion of different components of the leaf extracts in the agar medium was improved by a pre-incubation of Petri dishes at 4 °C for 30 min. The Petri dishes were then incubated at 37 °C for 24 h. The antibacterial test of the leaf extracts results in the appearance of an inhibitory zone around the wells.14,15
Determination of Minimum Inhibitory Concentration (MIC) by Microdilution Method
The minimum inhibitory concentration (MIC) was determined by the standardized method of microdilution in a liquid medium according to the recommendations of EUCAST. 16 The study was carried out in 96-well sterile micro-plates. The dilutions of the samples were distributed in the wells starting from the highest to the lowest concentration. 10 µL of the bacterial inoculum (5 × 105 CFU/mL) was added to each well according to the recommendations of EUCAST.13,16 Control wells did not contain leaf extracts. The microplates were incubated for 18 h at 37 °C. The MIC was determined after verification of the bacterial growth using the salt MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) as a redox indicator. After 2 h of incubation, the MIC (mg/mL) corresponded to the lowest concentration of antibacterial agents in leaf extracts which did not produce any change in MTT color (yellow to purple) (absence of growth) (Figure 2).16-18 Cefiderocol is considered the positive control (reference standard from 16 ).

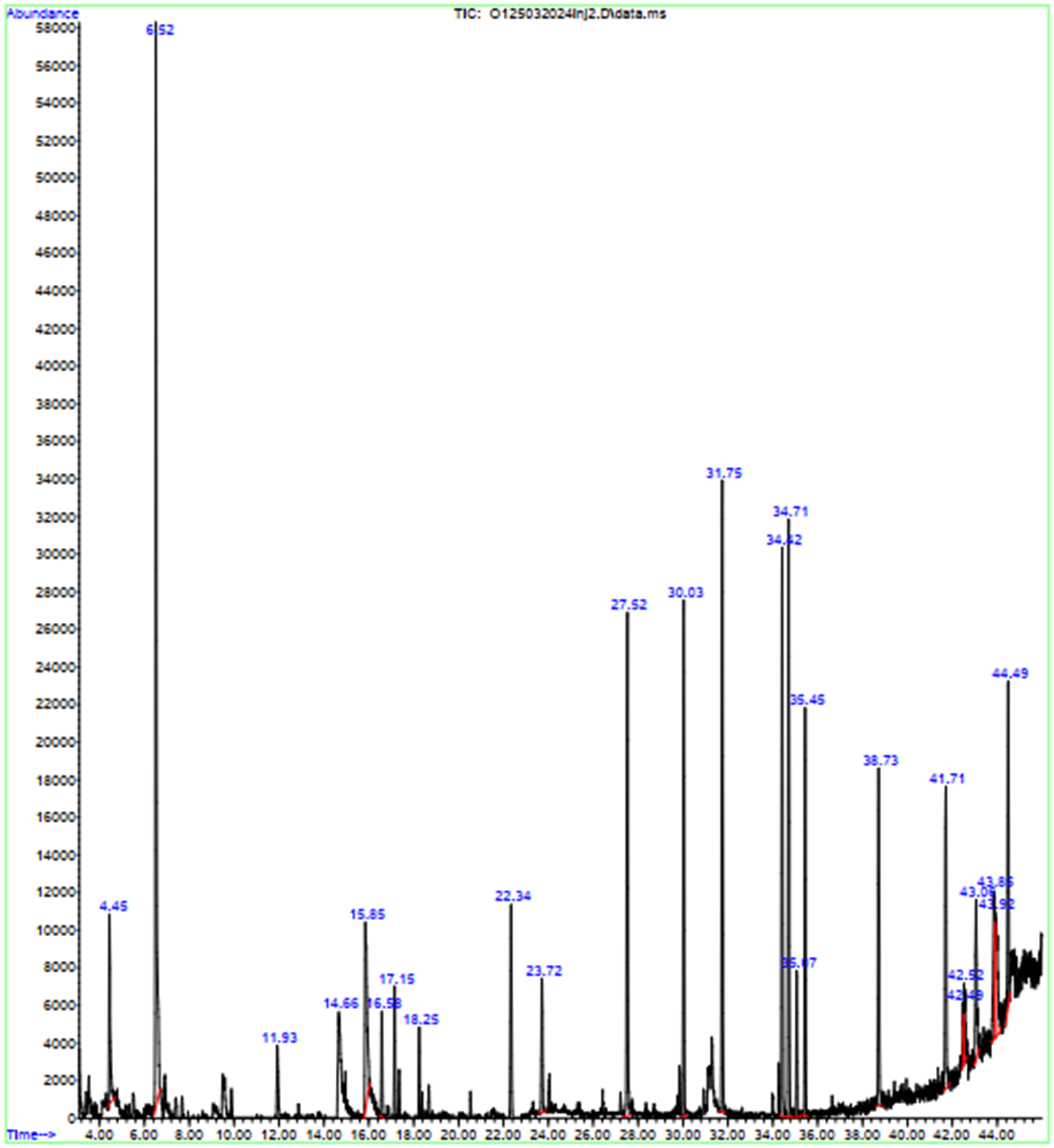
Chromatogram of compounds identified in the water leaf extract of A. indica using HPLC/MS analysis.
Determination of Minimum Bactericidal Concentration (MBC)
10 µL of each well that did not show turbidity or change in MTT color staining was sub-cultivated on the Mueller Hinton agar and incubated at 37 °C for 24 h to determine the minimum bactericidal concentration (MBC). The MBC in mg/mL refers to the lowest concentration of A. indica leaf extracts at which no bacterial overgrowth is observed. 18 This concentration is considered bactericidal, while any concentration that allows for overgrowth is classified as bacteriostatic. 19
Statistical Analysis
Data and graphs were analyzed using Microsoft Excel Worksheet software 2021, Version 2108 (Build 14332.20517). The results are presented as mean ± standard deviation (SD) from triplicate experiments (n = 3). Differences were considered significant when P < .05. The mean inhibition zone diameters (IZDs) of the leaf extracts against the various bacterial strains were compared using one-way ANOVA.
Results
Phytochemical Screening
The qualitative tests conducted for preliminary phytochemical screening revealed the presence of various compounds, including saponins, tannins, carbohydrates, steroids, and alkaloids, in the three leaf extracts (see Table 1). Specifically, saponins, terpenoids, and tannins were detected in the water extracts. In contrast, alkaloids and steroids were found in the ethanol extract. The methanol extract contained polyphenols, flavonoids, saponins, terpenoids, tannins, carbohydrates, alkaloids, and steroids (refer to Table 1).
Qualitative Tests for Phytochemical Screening of A. indica Leaf Extracts.

(−), absence of compound; (+), low compound content; (++), medium compound content; (+++), high compound content.
HPLC-MS Analysis
The A. indica leaf extracts demonstrated a variety of significant natural compounds, as detailed in Tables 2 and 3, when three different solvents were utilized in the extraction process. The HPLC-MS analysis of the ethanol extracts, illustrated in Figure 3, identified a major bioactive compound with strong antibacterial properties: diethyl phthalate, which accounted for 92.31% of the extract (see Table 2). Additionally, the ethanol extracts contained phenolic compounds such as eugenol (0.30%), and terpenoids with notable antioxidant properties, including phytol (1.68%), germacrene B, and vomifoliol. The extracts also included fatty acids such as palmitic acid methyl ester (0.63%), lauric acid methyl ester (0.48%), stearic acid methyl ester (0.21%), myristic acid methyl ester (0.10%), and indole (0.19%) (refer to Table 2).
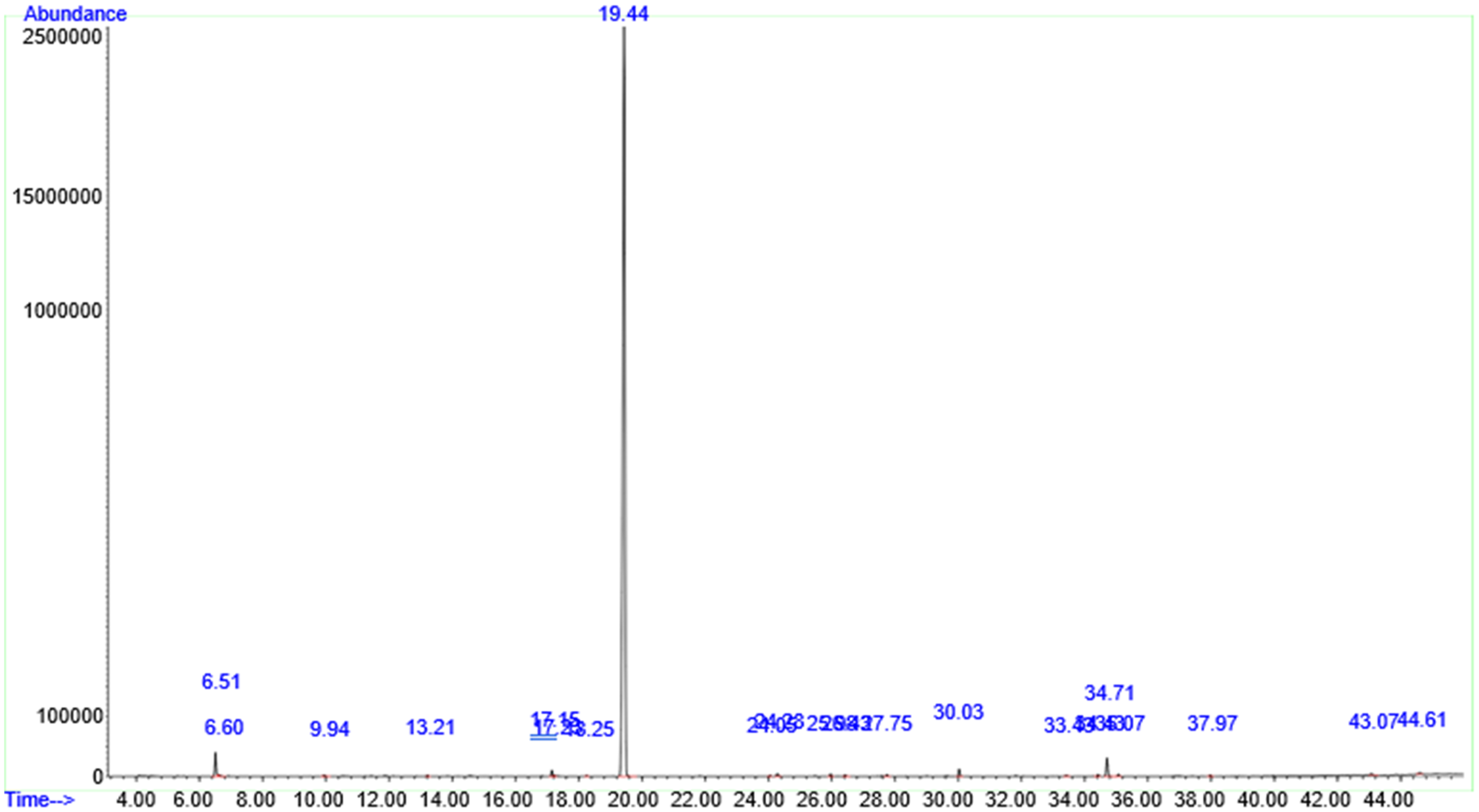
Chromatogram of compounds identified in the ethanol leaf extract of A. indica using HPLC/MS analysis.
Compounds Identified Through HPLC-MS Analysis of the Ethanol Extract from A. indica.

Compounds Identified Through HPLC-MS Analysis of the Water Extract from A. indica.

The HPLC analysis of water leaf extracts (Figure 2) revealed the presence of several phenolic compounds with significant antioxidant properties. These compounds include butylated hydroxytoluene (BHT, found in A. indica leaf as a natural antioxidant) at 28.12%, benzoic acid at 5.61%, 2-methoxy-4-vinylphenol at 3.45%, eugenol at 2.12%, and syringol (2,6-dimethoxyphenol, a derivative of syringic acid) at 1.58%. Other detectable compounds were vanillin at 1.17%, 1,2-benzenediol at 0.62%, and phenylacetic acid at 0.46%.
Additionally, the water leaf extracts contained bioactive molecules such as 2,3-dihydro-benzofuran (Table 3), which exhibit diverse biological activities, including antibacterial, anticancer, antiallergic, anti-inflammatory, and pesticide properties. Oleyl alcohol (cis-9-octadecen-1-ol) (Table 3) is also present, known for its emulsifying, emollient, and thickening properties in skin creams, lotions, and various cosmetic products.
The HPLC-MS chromatogram of the methanol leaf extracts displayed significant peaks (Figure 4) and confirmed the presence of effective compounds, as shown in Table 4. Major compounds detected include benzenamine N, N-dimethyl- (14.69%), phytol (7.75%), 1,1,1,5,5,5-hexamethyl-3-((trimethylsilyl)oxy) trisiloxane (7.52%), linolenic acid methyl ester (6.73%), palmitic acid methyl ester (hexadecanoic acid) (6.72%), and bistrimethylsilyl N-acetyl eicosasphinga-4,11-dienine (4.91%), among other important molecules (Table 4).
Chromatogram of compounds identified in the ethanol leaf extract of A. indica using HPLC/MS analysis.
Compounds Identified Through HPLC-MS Analysis of the Methanol Extract from A. indica.

Antibacterial Activity
The antibacterial properties of A. indica leaf extracts were examined against various Gram-positive and Gram-negative bacterial strains using the well-diffusion technique. This method allowed for the measurement of the IZDs. Additionally, the MIC were determined using the micro-dilution method, while the MBC were assessed with petri dishes containing Mueller-Hinton medium.
The results obtained by the well-diffusion method showed that the A. indica leaf extracts exhibit antibacterial activity on Gram + strains, while no antibacterial activity was detected on all Gram–strains tested.
The antibacterial activity varied significantly based on the solvent extract used. The IZDs indicated that all the Gram-positive strains, including SS, MRSS, and SA, were sensitive to the tested leaf extracts, though the degree of sensitivity varied (see Figure 4). The IZDs for the Gram-positive sensitive strains ranged from 10 ± 1 mm to 15 ± 1 mm. The largest inhibitory zone was observed against SA with the aqueous extract, measuring 15 mm, while the smallest was observed against MRSS with the ethanol extract at 10 mm. The methanol extract demonstrated greater activity against SS and MRSS, showing an inhibition zone of 14 mm, and moderate activity against MRSS with a zone of 12 mm. The water extract was most effective against SS, with an inhibition zone diameter (IZD) of 13 mm, and moderately effective against SARM at 11 mm (see Figure 5).

Inhibitory zone diameters (IZDs) (mm) of A. indica leaf extracts on bacteria strains.
The MIC determined by the micro-dilution method are presented in Figure 6. The MIC values ranged from 0.06 to 0.50 mg/mL, as shown in Table 5. Notably, the methanol extract against SA exhibited the lowest MIC value of 0.03 mg/mL. Interestingly, the extracts that demonstrated the largest inhibitory zones in the well-diffusion method (specifically the methanol extracts against SA) also had the lowest MIC values, as indicated in Table 5. Importantly, no inhibitory concentration was observed against any of the Gram-negative strains.

MIC values determined by the micro-dilution method.
MIC and MBC Values of A. indica Leaf Extracts.

MIC, Minimal inhibitory concentrations (MIC) (mg/mL); MBC, Minimal bactericidal concentrations (MBC) (mg/mL); Final bacterial density was around 5 × 105 CFU/mL; ND, Not determined.
In Figure 7, the MBC demonstrated a bacteriostatic effect against SS when using the methanol extract (Figure 7a). For MRSS, the water and methanol extracts also exhibited a bacteriostatic effect (Figure 7b). Regarding SA, the aqueous extract showed a bacteriostatic effect (Figure 7c), while both the methanol extract and ethanol extract had a bacteriostatic effect (Figure 7d). Notably, all MBC values were greater than 0.5 mg/mL (Table 5).

Minimal bactericidal concentrations (MBC) (mg/mL) of A. indica leaf extracts in the Mueller-Hinton agar: (a): SS Bacteriostatic with the methanol extract in (5). (b): MRSS Bacteriostatic with the water extract in (3), and the methanol extract in (4). (c): SA Bacteriostatic with the aqueous extract in (6). (d): SA Bacteriostatic with the methanol extract in (7), and the ethanol extract in (8).
Discussion
The qualitative phytochemical screening tests showed that compounds such as the polyphenols, flavonoids, saponins, terpenoids, tannins, steroids, and alkaloids were all present in the A. indica leaf extracts (ethanol, water and methanol). Viewing the previous works, we found in the same way that the biochemical screening test of crude neem leaf extracts carried out by 9 showed that alkaloids, steroids, flavonoids, tannins, saponins, and amino acids were present in all polarities of crude neem leaf extracts except anthraquinones and triterpenoids. Our results bear similarities to ones obtained by 20 who showed that saponins, steroids, and terpenoids are most present in the aqueous extract of A. indica, while tannins and glycoside are in moderate concentration, while alkaloids, flavonoids, phenol, and oxalic acid have low concentration. The bio-active compounds such as alkaloids, flavonoids, glycosides, and saponins for example exert multiple biological effects like anti-inflammatory, anti-allergic, antioxidant, anti-diabetic, anti-viral and anti-cancer activities, anti-leprosy activities, and antimicrobial activity. 20 These bio-active compounds screened in this study presented several advantages with few secondary effects and safety cures compared to synthetic drugs. The advantages are generally fewer side effects, better patient tolerance, relatively less expensive, and renewable. 20 Alkaloids were reported to possess an antispasmodic, antimalarial, analgesic, and diuretic activities; terpenoids are known for their antiviral, anthelmintic, antibacterial, anticancer, antimalarial, and anti-inflammatory properties; glycosides are reported for antifungal and antibacterial properties; phenols and flavonoids have an antioxidant, antiallergic, and antibacterial properties; and saponins have shown anti-inflammatory and antiviral activities.4,21,22 Flavonoids act as chemical messengers, physiological regulators, and cell cycle inhibitors. 20 Tannins are found in many plant species, where they serve as pesticides and help regulate plant growth. 23 In plants, saponins may serve as antiparasitic, insecticidal, antifeedant and protect against microbes and fungi. 24 In some studies, A. indica leaf can be used for the development of novel contraceptive strategies targeting the male gametes. 25 Nimbolide treatment weakens the antioxidant enzymes in rat sperm, specifically glutathione peroxidase, superoxide dismutase, and catalase. It also increases hydrogen peroxide (H2O2) production and lipid peroxidation (LPO) in a dose-dependent manner. This shows that nimbolide can harm the antioxidant defense system and the sperm's membrane, leading to oxidative stress in epididymal sperm. 25
During the HPLC-MS analysis of the ethanol leaf extracts (ELE) from A. indica, a major compound was identified: diethyl phthalate (1,2-benzenedicarboxylic acid, diethyl ester, also known as anozol), which comprised 92.31% of the extract. Diethyl phthalate is an organic compound known for its significant antimicrobial activity. 26 On the other hand, the HPLC-MS analysis of water leaf extracts (WLE) revealed the presence of various promising phenolic compounds. These include BHT (28.12%), identified in this study as a natural antioxidant, as well as benzoic acid (5.61%), 2-methoxy-4-vinylphenol (3.45%), eugenol (2.12%), syringol (2,6-dimethoxyphenol, a derivative of syringic acid) (1.58%), vanillin (1.17%), 1,2-benzenediol (0.62%), and phenylacetic acid (0.46%). These compounds are naturally occurring in A. indica. Notably, BHT is recognized as a commonly synthesized antioxidant used in food products but is rarely found in natural sources. 27 Nevertheless, its detection in this study indicates that it can also be a natural antioxidant in the Chadian A. indica. Additionally, BHT has been detected in the fruit of Litchi (Litchi chinensis Sonn.) through electrospray ionization mass spectrometry and Nuclear Magnetic Resonance (NMR) spectroscopy. 28
In our study, extracts from A. indica water leaves contained 5.61% benzoic acid, a type of phenolic acid known for its strong antioxidant properties. 29 This compound was also identified in another study, 30 where it was reported as the most abundant compound in pure A. indica oil, with a concentration of 12.6 ± 1.35 µg/mL as determined by HPLC-PDA analysis. Additionally, T-cinnamic acid was reported at a concentration of 1.9 ± 0.2 µg/mL. Furthermore, benzoic acid was detected in A. indica leaf extracts in a study on the chemical and biological profiles of Algerian A. indica leaf extract, with a concentration of 56.82 ± 0.04 µg/g of extract. 31 Vanillin (1.17%) detected in our study in the water leaf extracts was also identified by the authors 31 in the A. indica leaf from Algeria by LC–MS/MS, with a quantity of 123.89 ± 0.03 µg/g extract.
Vanillin (1.17%) detected in our study of water leaf extracts was also identified by authors in Algeria in A. indica leaf samples using LC-MS/MS, with a measured quantity of 123.89 ± 0.03 µg/g of extract. Vanillin is a phenolic aldehyde compound known for its high antioxidant activity, 32 antimicrobial properties, 33 and antigenotoxic effects. 34 The third major phenolic compound detected in our study was 2-methoxy-4-vinylphenol (3.45%), which has been reported to exhibit strong antioxidant activity and to serve as an antimicrobial preservative in food and beverages. 35 Additionally, other important minor phenolic compounds detected include eugenol, phenylacetic acid, 1,2-benzenediol, and syringol (2,6-dimethoxyphenol). The latter is a derivative of syringic acid, which is recognized as a promising phenolic compound with high antioxidant activity.36-39
In this study, the methanol leaf extract was found to contain several important compounds, including benzenamine N, N-dimethyl- (14.69%), phytol (7.75%), and 1,1,1,5,5,5-hexamethyl-3-((trimethylsilyl)oxy) trisiloxane (7.52%). Other notable components included linolenic acid methyl ester (6.73%), palmitic acid methyl ester (hexadecanoic acid) (6.72%), and bistrimethylsilyl N-acetyl-eicosasphinga-4,11-dienine (4.91%). One of the key compounds, N, N-dimethylaniline, is an aromatic amine commonly used as a solvent and as an intermediate in the production of dyes, Michler's ketone, and vanillin. 40 Linolenic acid (ALA) is an essential fatty acid that humans must obtain through their diet. It provides several health benefits, including protection for the cardiovascular system. 41 ALA is predominantly found in higher plants and algae, with particularly high concentrations in certain plant oils, such as those derived from chia seeds, walnuts, and flaxseeds (linseed). 42
Our results demonstrated that A. indica leaf extracts possess antibacterial activity against the studied Gram-positive bacteria, specifically SS and MRSS, which are known to cause skin and urinary infections. The well-diffusion method results indicated significant IZDs ranging from 10 ± 1.00 to 14 ± 1.00 (see Table 4), which is consistent with findings from other studies. The MIC of the leaf extracts against the bacteria, along with the corresponding inhibitory zones, revealed that the lowest MIC values ranged from 0.06 to 0.50 mg/mL. It is noteworthy that the extracts exhibiting the largest IZDs—specifically, the methanol extracts with SS—do not always correspond to the lowest MIC values, which are observed in the ethanol extracts with SS. This discrepancy can be attributed to several factors. The IZDs observed in the well-diffusion method primarily indicate the extract's potential for qualitatively inhibiting microbial growth. This method captures a wider range of compounds present in the extract, all of which may work together to contribute to the observed inhibition.43,44 Conversely, the MIC quantitatively determine the lowest concentration of an extract that effectively inhibits visible microbial growth after overnight incubation.45,46 This assessment takes into account the potency of specific bioactive components within the extract, which may not always correlate with the overall size of the inhibitory zone. Variability in compound solubility, diffusion rates, and the susceptibility of target microorganisms also contributes to this difference. In summary, while the inhibitory zones from the well-diffusion method indicate overall inhibitory potential, 44 the MIC provides insight into the concentration of specific bioactive compounds. 47
This study showed that leaf extracts of A. indica can inhibit bacterial strains that cause fever and urinary infections. The authors 48 reported that A. indica leaves are beneficial for enhancing the immune system, reducing the incidence of malarial fever, and treating neuromuscular pain as well as fungal infections, such as athlete's foot. However, we found that extracts from A. indica leaves showed no antibacterial activity against the Gram-negative strains KP and PM. This difference in results between Gram-positive and Gram-negative bacteria can be explained by the fact that Gram-negative bacteria are generally less susceptible to antibacterial agents than Gram-positive bacteria. 49 Structurally, the outer membrane of Gram-negative bacteria contains hydrophilic lipopolysaccharides (LPS) molecules, which act as a mechanical barrier to macromolecules and hydrophobic compounds, providing Gram-negative bacteria with higher tolerance toward hydrophobic antimicrobial compounds.49,50 There are other studies reported good antimicrobial activity of A. indica leaf extracts. Indeed, 51 investigated the antimicrobial activity using the disc diffusion method of neem leaves of alcoholic extracts against human pathogenic bacteria including E. coli, Staphylococcus aureus, Pseudomonas aeruginosa, Salmonella typhimurium, and Bacillus pumilus, where they found that the methanol and ethanol extract shows maximum inhibition on Bacillus pumilus, Pseudomonas aeruginosa, and Staphylococcus aureus in ascending order when compared with gentamycin 200 mg and gentamycin 10 mg. 51 Also, methanol extract of A. indica leaf demonstrated the highest antimicrobial activity in a study by 52 showing potent antibacterial effects against various strains of bacterial pathogens. The study reported an IZD of 24 mm for E. coli at a concentration of 1.5% and an IZD of 23 mm for Salmonella spp. at a concentration of 2.5%. These results were closely comparable to the antimicrobial activity of gentamicin used in the study of. 52 The authors 53 have shown that leaf extracts of A. indica, when using hexane, ethyl acetate, chloroform, butanol, methanol, and water, did not demonstrate significant antibacterial activity against the tested Gram-positive and Gram-negative bacteria. Specifically, the bacteria tested included Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa, and Proteus vulgaris, even at a maximum concentration of 2 mg/mL. The lack of activity observed against the Gram-positive strains in their study may be attributed to the low concentration of the crude leaf extracts used (2 mg/mL). The ethanol extract of neem leaves demonstrated antibacterial activity in vitro against both Staphylococcus aureus and MRSS, with the most significant zones of inhibition observed at a 100% concentration. 54 Researchers 55 studied neem leaf ethanolic extract (Neem EE) for its effects on Methicillin-resistant Staphylococcus aureus (MRSA) and Schistosoma mansoni worms. They found azadirachtin (AZA) in the extract at 0.147 ± 0.02 mg/L. Neem EE significantly reduced biofilm adherence at 62.5 mg/mL for sensitive Staphylococcus aureus and 125 mg/mL for two MRSA strains. As Neem EE concentration increased from 250 to 1000 mg/mL, one MRSA strain formed larger aggregates. In tests against Schistosoma worms, Neem EE caused 100% death of female worms at 50 mg/mL after 72 h and required 300 mg/mL for male worms to achieve the same result after 24 h. They concluded that neem leaf ethanolic extract effectively inhibits MRSA biofilm and is harmful to Schistosoma mansoni worms. 55 In another study by, 48 A. indica leaf contains sodium nimbate, cyclic trisulfide, and cyclic tetrasulfide, polysaccharides which have respectively antifungal and anti-inflammatory activity. Some researchers 56 studied the in vitro antiviral properties of A. indica and found that polysaccharides extracted from its leaves exhibited effective virucidal effects against poliovirus. Another study conducted by 57 examined the antiviral activity of A. indica leaf extract and discovered that it contains a variety of compounds, including flavonoids, triterpenoids, and their glycosides, which contribute to its antiviral action against group B coxsackieviruses. The authors 58 reported that a mixture of acetone and water (in a 1:1 volume ratio), which was fractionated using high-performance liquid chromatography, is a more effective solvent than water alone and exhibits significant anti-malarial activity. A. indica has been recognized as one of the most promising sources of compounds for insect control and has antimicrobial properties. 51 Quercetin and β-sitosterol, polyphenolic flavonoids, were purified from fresh A. indica leaves and are known to possess antibacterial and antifungal properties. 59 Additionally, the oil in the seeds contains valuable constituents, including gedunin and azadirachtin. 59
The findings of this current study represent the first report on the A. indica leaf from Chad. However, the study had limitations, particularly regarding the no antibacterial activity against Gram-negative pathogens using diffusion and microdilution methods; many other studies have also reported negative results. In perspective, combining the leaf extract with other bioactive compounds from various plant or animal sources, or mixing it with bee propolis, could create a synergistic effect that may effectively combat Gram-negative pathogens. The limitation also applies to the qualitative test for phytochemical screening, which remains preliminary. A quantitative method is essential to determine the exact quantities of each compound screened, allowing for better characterization of the chemical analysis. Another limitation involved the HPLC-MS analysis used to detect the chemical compounds in the extracts. This technique is not yet fully sufficient, and further refinement is needed. It would be beneficial to apply additional methods to separate the compounds into pure states using chromatographic or physicochemical techniques, followed by identification through spectroscopic methods, particularly NMR spectroscopy.
Conclusion
The qualitative screening test indicated that leaf extracts of A. indica contain various beneficial compounds, including polyphenols, flavonoids, saponins, steroids, terpenoids, tannins, and alkaloids. HPLC-MS analysis of ethanol, water, and methanol leaf extracts of A. indica identified several phenolic compounds and other substances with promising pharmacological effects. Notably, diethyl phthalate (1,2-benzenedicarboxylic acid, diethyl ester) was found in significant concentration at 92.31%. Additionally, BHT, identified for the first time as a natural antioxidant in A. indica leaf extract, made up 28.12%. Other identified compounds included benzoic acid (5.61%), 2-methoxy-4-vinylphenol (3.45%), and eugenol (2.12%). Leaf extracts demonstrated significant antibacterial activity against common Gram-positive bacterial pathogens, specifically SS, SA, and MRSS. The inhibition zones measured between 10 ± 1.00 mm and 15 ± 1.00 mm. The MIC ranged from 0.06 to 0.50 mg/mL, while the MBC were greater than 0.5 mg/mL. This study suggests that A. indica leaf extracts contain compounds with potential health benefits, particularly antibacterial properties. These properties support their traditional use by healers in the Republic of Chad for treating conditions such as headaches, fever, skin infections, and urinary tract infections. This research offers important insights regarding A. indica leaf extracts for the scientific community in Chad and internationally.
Footnotes
Acknowledgments
We would like to express our gratitude to the head of the Chemistry Department at the Faculty of Sciences for their technical support. We also extend our thanks to Professor El Hassan Sakar from the Laboratory of Biology, Ecology, and Health at the Faculty of Sciences in Tetouan, along with his collaborators, for their assistance with the HPLC-MS analysis. We would like to express our gratitude for the technical support provided by Professor Ahmed Ibn Mansour from the Laboratory of Applied Organic Chemistry in the Chemistry Department at the Faculty of Sciences in Tetouan, particularly regarding the HPLC-MS analysis results. Additionally, we extend our thanks to Professor Abdelfettah Maouni from the Superior Normal School of Martil (Ecole Normale Supérieure) and the Biology, Environment, and Sustainable Development Laboratory at Abdelmalek Essaâdi University for his valuable feedback and suggestions on the antibacterial methods. We would like to express our gratitude to the Toumaï University herbarium in N'Djamena, especially to Ms. Khadidja Brahim, as well as to the team from the Fédération Nationale des Associations des Praticiens de la Medicine au Tchad (FENAPMT) for their assistance in collecting and identifying the species we studied.
Authorship Contribution Statement
OBM: Investigation, Resources, Software, Formal Analysis, and Writing - original draft. OBM, ZM: Materials, Method, Analysis, Interpretation. IB, ZM: Visualization, Critical Review. YS, BBO: Supervision, Visualization, Validation.
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
