Abstract
D scandens (Derris scandens Benth.) and E scaber (Elephantopus scaber Linn.) contain flavonoids and phenolic acids, which have antitumor activity in various cancer cell lines. Oral cancer is among the most common cancers in Southeast Asia, and the survival rate remains low. Thus, this study screened 2 ethanolic plant extracts for cytotoxicity on the oral human squamous carcinoma cell line (HSC-2), and compared the mechanisms of action. Extracts of D scandens and E scaber showed cytotoxicity against HSC-2 cells in a dose-dependent manner. Observation of nuclear morphology by Hoechst 33342 staining revealed chromatin condensation. Apoptosis was confirmed by Annexin V-FITC staining and cell sorting (fluorescence-activated cell sorting) analysis. We demonstrated that cancer apoptosis was accompanied by changes in the expression of procaspase 3 and that D scandens-mediated apoptosis in HSC-2 cells was potentiated by protein kinase B (Akt) and B-cell lymphoma-2 (Bcl-2), while E scaber apoptosis was mediated by mitogen-activated protein kinase (MAPK) pathways, involving stress-activated protein kinases/jun amino-terminal kinase (SAPK/JNK) and p38-MAPK. Further investigation into targets for apoptosis induction by these plant extracts may have potential in oral cancer therapy.
Introduction
Oral cancer is among the 5 most common cancers in Thailand with an age-standardized rate incidence of 15.7 and 10.7 per 100 000 males and females, respectively. 1 This is mainly caused by betel and areca nut chewing, 2 tobacco use, 3 alcohol consumption, 4 and the human papillomavirus. 5 Treatment of oral cancer by conventional methods includes surgery, chemotherapy, and radiation; however, the survival rate has remained unimproved for many years. 6 Thus, the identification of novel, effective therapeutic agents targeting apoptosis continues to be an important success factor for cancer prevention. Phytochemicals in plants are a source of drug discovery due to the presence of bioactive compounds such as flavonoids and sesquiterpenes, as well as their precursors, many of which are associated with anticancer activity.7-9
Research in the ability of phytochemicals in non-toxic botanicals to induce cell apoptosis for non-invasive management of oral cancer is increasing. 10 Derris scandens Benth. (D scandens) and Elephantopus scaber Linn. (E scaber) have been used in traditional Thai medicine since the 13th century AD as active ingredients in longevity concoctions, and cancers are considered an age-related disease. 11 E scaber is mainly used in longevity concoctions arising from central Thailand, while D scandens is a more popular ingredient in traditional medicine from Northeast Thailand. Comparisons between the efficacy of these 2 extracts have not been conducted. Extracts of E scaber have been found to inhibit tumor necrosis factor alpha (TNF-α) and interleukin-1β (IL-1β) in lipopolysaccharide-stimulated monocytes, 12 while deoxyelephantopin in E scaber has been found to promote wound healing in rats, 13 and antitumor activity has been reported for the leaves of the plant against Dalton's ascitic lymphoma 14 in mice. 15 Meanwhile there is evidence that D scandens can enhance radiation therapy in human colon cancer cell line HT-29 cells, and a decrease in Erk1/2 activation may be involved. 16
Mitogen-activated protein kinases (MAPKs) regulate cell proliferation, differentiation and survival pathways, including apoptosis, 17 and along with the phosphatidylinositol-3-kinase/protein kinase B (PI3K/Akt) cascade, are highly involved in oncogenic pathways. 18 Thus, research into bioactive phytochemicals targeting key modulators of downstream MAPK/PI3K signaling are beneficial for cancer therapy. Studies have shown that activation of downstream constituents of the MAPK cascade are important mediators of apoptosis in various cancer cell lines.19-21 Another pathway that regulates apoptosis and cell survival involves the anti-apoptotic protein B-cell lymphoma 2 (Bcl-2) and its family member proteins such as Bax (a proapoptotic protein), which regulate the mitochondria-dependent apoptotic pathway and are often observed alongside caspase 3 activation.22-24 Interaction between Bcl-2 family member proteins and MAPK cascades is known to regulate apoptosis.25-27 Dysregulation of the PI3K/Akt pathway is also well-associated with cancer pathological processes, and Akt and Bcl-2 downregulation have been observed in phytochemical-induced cytotoxicity.28,29 We investigated the role of phytochemical-induced apoptosis in the human squamous carcinoma (HSC-2) cell line, and compared the mechanisms through which D scandens and E scaber exert antitumoral or proapoptotic effects, focusing on the modulation of components of the MAPK and/or PI3K/Akt pathways, which have not been reported in previous literature.
Results and Discussion
Plant Extract-Mediated HSC-2 Cytotoxicity is Dose-Dependent
Ethanolic extracts of plant species were tested for their cytotoxic effects on HSC-2 cell lines by MTT assay; the results are summarized in Figure 1. Cells treated with 3 mg/mL E scaber extract caused a ∼4-fold decrease in viability compared to cells treated with the control (24.5 ± 1.3 vs 100 ± 0.01, P < .01), and a ∼73% decrease in viability compared to cells treated with 0.375 and 0.75 mg/mL (24.5 ± 1.3 vs 92.1 ± 2.3, P < .05, 24.5 ± 1.3 vs 91.2 ± 10.1, P < .05). Treatment with 1.5 mg/mL E scaber extract only caused a 40% decrease in cell viability compared to the control (60 ± 2 vs 100 ± 0.01, P < .05). This dose-dependent pattern of cytotoxicity is similarly seen upon treatment with extract of D scandens at the same concentrations. Treatment with 3 mg/mL D scandens extract caused a ∼97% decreased cell viability compared to treatment with 0.375 and 0.75 mg/mL and control (2.6 ± 0.5 vs 92.4 ± 4.5, P < .05, 2.6 ± 0.5 vs 95.5 ± 12, P < .05, 2.6 ± 0.5 vs 100 ± 0.01, P < .01). Treatment with 1.5 mg/mL D scandens extract showed a ∼27% decrease in viability compared to the control (72.9 ± 3.7 vs 100 ± 0.01, P < .05).

HSC-2 cytotoxicity was measured using MTT assay. Cell viability in response to varying concentrations of extracts, D scandens and E scaber, are displayed as the percentage of viable cells after treatment compared to control. Data expressed as means ± SD; n = 3 *P < .05, **P < .01.
HSC-2 Cell Apoptosis by Extracts of E scaber and D scandens
To investigate the nature of E scaber and D scandens-induced cell death, a double staining method using FITC-labelled Annexin V and propidium iodide (PI) was used to detect apoptotic cells using fluorescence-activated cell sorting (FACS) analysis (Figure 2A). Apoptotic cells were recognized by Annexin V and PI-positive staining, while viable cells were negative for both Annexin V-FITC and PI. No difference in cytotoxicity was found in cells treated with 0.75 mg/mL of either extract of E scaber or D scandens compared to the control (data not shown). Treatment with 1.5 mg/mL E scaber extract for 24 h caused significant percentage of apoptosis in HSC-2 cells compared to control (35.7% ± 3.8 vs 10.4% ± 4.7, P < .05), however this was not observed after treatment with 1.5 mg/mL D scandens extract (Figure 2B). At 3 mg/mL, higher apoptotic events were significantly observed compared to control upon treatment with extracts of E scaber (52.3% ± 12.1 vs 10.4% ± 4.7, P < .01) and D scandens (69.2% ± 7.3 vs 12.4% ± 4.6, P < .01).

Induction of apoptosis after treatment with E scaber and D scandens. (A) FACS analysis showed percentage of apoptotic cells compared to control. (B) Graph representing the percentage of apoptotic cell death with Annexin V-FITC and PI staining. Data represented as mean ± SD. *P < .05, **P < .01. (C) DNA condensation was detected by Hoechst 33342 staining. Regions of condensed chromatin and apoptotic bodies appear as denser areas indicated by arrows. Scale bar: 60 μm (low magnification images). (D) Graph representing the percentage of apoptotic cells out of total number of cells. Data expressed as mean ± SD; n = 3; **P < .01.
Hoechst 33342 staining was conducted to detect the presence of chromatin condensation, nuclear fragmentation, and apoptotic bodies as these are characteristics of cellular apoptosis. 30 Cells in the control group with intact DNA had weaker fluorescence compared to brighter, visible fluorescence in cells treated with extracts of E scaber and/or D scandens due to chromatin condensation (Figure 2C). Treatment with 1.5 and 3 mg/mL E scaber extract for 24 h caused a ∼10-fold and a ∼50-fold increase in apoptotic bodies and chromatin condensation compared to the control, respectively (4.3 ± 0.6 vs 0.4 ± 1.3, P < .01; 21 ± 2.6 vs 0.4 ± 1.3, P < .01) (Figure 2D). Treatment with 1.5 and 3 mg/mL D scandens extract also caused significant increase in percentage apoptosis compared to the control (12.3 ± 0.6 vs 0.4 ± 1.3, P < .01; 31 ± 1 vs 0.4 ± 1.3, P < .01). The pattern of HSC-2 apoptosis observed with Annexin V-FITC and PI staining and Hoechst 33342 after treatment with 1.5 and 3 mg/mL E scaber and D scandens extracts were similar.
D scandens-Mediated HSC-2 Cytotoxicity is by Apoptosis Induction Involving Procaspase 3
Activation of the effector caspase 3 through cleavage at specific internal residues is important for initiating the apoptosis cascade,31,32 thus the detection of procaspase 3 cleavage by western blotting indicates apoptotic events. Here we show that treatment with E scaber for 24 h produced no changes in procaspase 3, however treatment with 1.5 and 3 mg/mL D scandens revealed significant activation of procaspase 3 (0.4 ± 0.06 vs 1 ± 0.07, P < .01; 0.5 ± 0.02 vs 1 ± 0.07; P < .01, respectively; Figure 3A). Densitometric analysis of protein bands revealed over 1.5-fold decrease in procaspase 3 after treatment with 3 mg/mL D scandens. Peak activation was observed at a concentration of 1.5 mg/mL, which caused approximately a 2.5-fold change in procaspase 3 expression (Figure 3B). Cleavage of caspase 3 by D scandens is concentration-dependent, whereas treatment with E scaber did not produce significant change in protein expression.

Caspase 3 activation in E scaber- and D scandens-induced HSC-2 apoptosis. (A) Detection of procaspase 3 proteins was used to determine cell apoptosis after exposure to 0 to 3 mg/mL E scaber and D scandens. (B) Data expressed relative to untreated cells (control). β-actin was used as loading control. Results are expressed as means ± SD; n = 3; **P < .01.
Extract-Induced Apoptosis Involves MAPK and Activation of Akt
MAPKs and PI3K/Akt pathways have roles in apoptosis and cell survival mechanisms.21,33 Thus, we performed gel electrophoresis experiments to observe fold changes and post-translational modifications of proteins involved in these cascades, namely Akt1, Bcl-2, p44/42 (Erk1/2), p54/46 (SAPK/JNK), and p38-MAPK, in response to treatment with 3 mg/mL E scaber and D scandens (Figure 4A). We found a steady decrease in Akt1 phosphorylation after 15 min of treatment with E scaber (Figure 4C). A similar pattern was observed after treatment with D scandens, where activation of Akt1 was seen after 15 min, followed by a steady decline in phosphorylated protein until 120 min.

Involvement of MAPK and PI3K proteins in D scandens and E scaber-stimulated HSC-2 apoptosis. (A) Gel electrophoresis bands were used to detect phosphorylated Akt1, Bcl-2, phosphorylated p44/42 (Erk1/2), phosphorylated SAPK/JNK, and phosphorylated p38-MAPK proteins to evaluate the change in expression of proteins involved in the MAPK and PI3K cascades. (B-F) Phosphorylated protein bands were normalized to β-actin, which was used as loading control. Results are expressed as means ± SD; n = 3; *P < .05, **P < .01.
We did not find significant fold change in the antiapoptotic protein, Bcl-2, after treatment with 3 mg/mL E scaber, however Bcl-2 activation was observed after 10 and 15 min of HSC-2 exposure to D scandens (2.2 ± 0.08 vs 1 ± 0.3; P < .01, and 1.9 ± 0.05 vs 1 ± 0.3; P < .01, respectively; Figure 4B). This activation was followed by a steady decrease in Bcl-2 proteins between 30 and 120 min. SAPK/JNK and p38-MAPK are activated after cell exposure to stress and mediate pro-apoptotic responses by controlling Bcl-2 family proteins, 25 while Erk1/2 regulate growth factor-stimulated signaling. 34 We found phosphorylation of Erk1/2 after exposing HSC-2 cells to E scaber for 60 min (3.6 ± 0.1 vs 1 ± 0.02; P < .01) which lasted until 120 min (Figure 4D).
After 30 min of treatment with 3 mg/mL E scaber, there was significant increase in SAPK/JNK phosphorylation (1.7 ± 0.2 vs 0; P < .05) and peak activation was observed after 60 min (3.7 ± 0.5 vs 0; P < .05; Figure 4E). In contrast, D scandens extract produced protein activation after 120 min of exposure (3.1 ± 0.1 vs 0; P < .01). p38-MAPK proteins have been shown to phosphorylate Bcl-2, facilitating apoptosis induction. 25 Here we show that p38-MAPK was activated by phosphorylation after treatment with 3 mg/mL E scaber after 30 to 120 min (5.28 ± 0.6 vs 1 ± 0.05; P < .01, 10.1 ± 0.7 vs 1 ± 0.05; P < .01, 11.6 ± 0.7 vs 1 ± 0.05; P < .01; Figure 4F).
E scaber-Induced Apoptosis is Inhibited by MAPK Inhibitors
To further investigate the relationship between extract-induced apoptosis in HSC-2 carcinoma and proteins involved in the MAPK and PI3K/Akt pathways, cells were pre-treated with inhibitors to Erk1/2 (U0126), PI3K (LY294002), p38MAPK (SB203580), and JNK1/2 (SP600125) for 1 h, followed by treatment with 3 mg/mL E scaber and D scandens for 24 h, before analysis by flow cytometry and Annexin V-FITC staining. We found apoptosis inhibition with pre-treatment of SB203580, SP600125, and LY294002 followed by 1.5 mg/mL E scaber (Figure 5). However, pre-treatment with U0126 did not inhibit extract-induced apoptosis (16.4 ± 0.2 and 21 ± 6.6; P < .05). Pre-treatment with various inhibitors followed by 1.5 mg/mL D scandens did not affect cytotoxicity and the percentages of apoptosis observed were similar to treatment with the extract alone (Figure 5B).
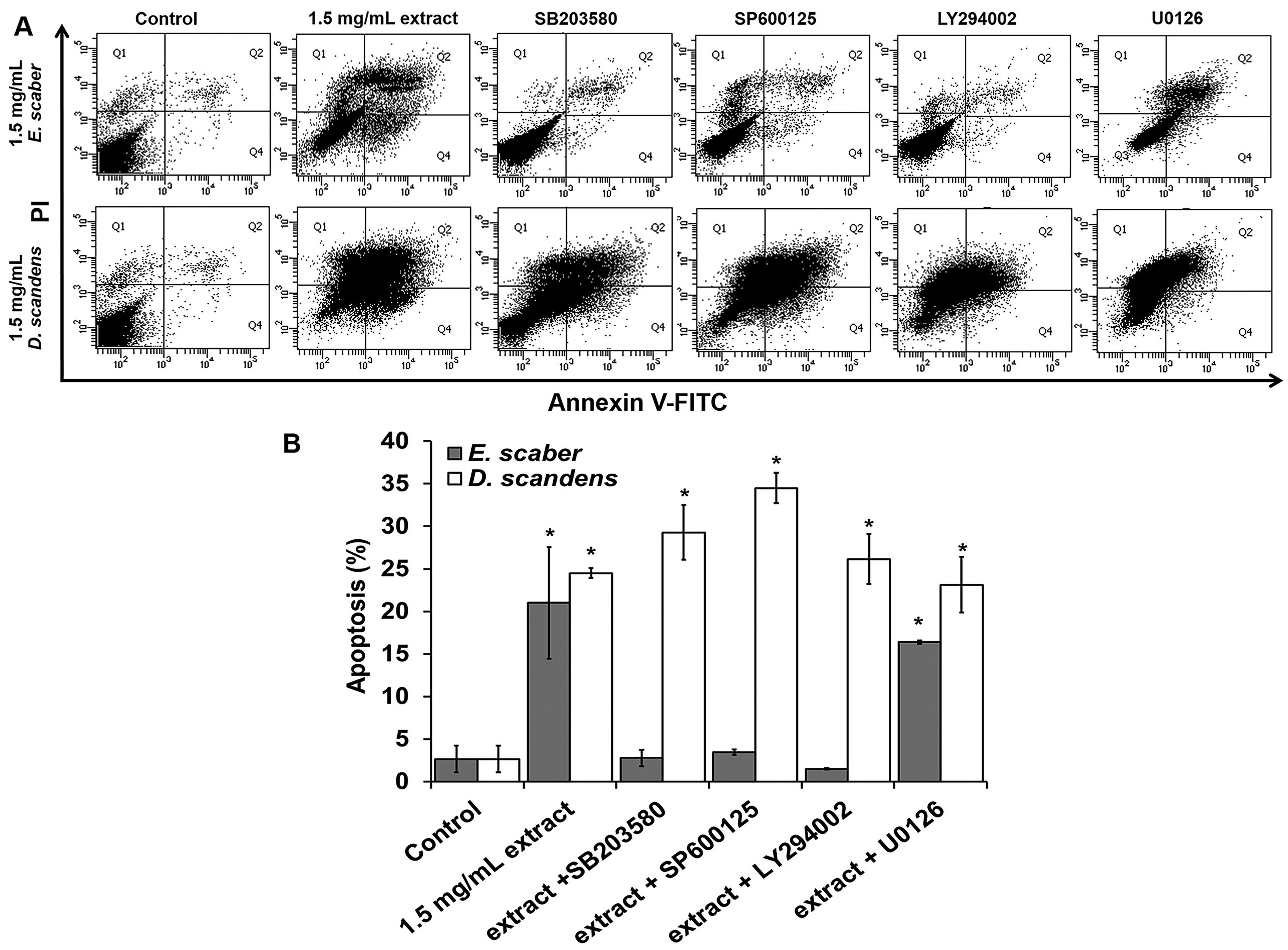
Inhibition of E scaber- and D scandens-induced apoptosis in HSC-2 by pre-treatment with inhibitors SB203580, SP600125, LY294002, and U0126. (A) FACS analysis showed percentage of apoptotic cells compared to control. (B) Graph representing the percentage of apoptotic cell death with Annexin V-FITC and PI staining. Data represented as mean ± SD. *P < .05.
Discussion
Natural products are increasingly being developed as a means of non-invasive anticancer therapy in conjunction with current conventional therapeutic methods such as chemotherapy and radiation. 10 Phytochemicals remain a valuable source for discovering new compounds which target downstream components of pathways regulating cancer initiation, progression, and proliferation. 10 The ability of bioactive compounds to induce apoptosis in cancer remains fundamental to the success of cancer treatment. Our study reports for the first time that crude ethanolic extracts of Derris scandens Benth. (D scandens) and Elephantopus scaber Linn. (E scaber) induce apoptosis in the human squamous carcinoma cell line 2 (HSC-2) in a dose-dependent manner. D scandens are an excellent source of flavonoids including genistein, luteolin, and scandinones, 35 while E scaber have a high concentration of sesquiterpene lactones such as deoxyelephantopin that display antitumor effects.36,37 Evidence in the literature suggests that scandinones are the main bioactive compounds in D scandens which affect various components of inflammation,35,38 and the herb has been implicated for the treatment of osteoarthritis and muscle aches and pains in Thailand. It is possible that the plant's cytotoxic effects originate from genistein and luteolin as both have been substantially implicated in apoptosis of various cancer types including of the breast and liver.39-41 The presence of chromatin condensation and apoptotic bodies indicate cell death by apoptosis, and our study revealed these occurrences upon treatment with D scandens, as well as E scaber, which was in line with our cytotoxicity results.
One major event in apoptosis is characterized by a feedback loop involving cytochrome c release, which induces caspase 9 activation, caspase 3 cleavage, leading to subsequent proteolysis of Bcl-2 and resulting in more cytochrome c release causing cleavage of various downstream caspases. 22 Bcl-2 is an antiapoptotic protein and it preserves mitochondrial membrane integrity by preventing Bax/Bak oligomerization, therefore preventing the release of apoptogenic molecules from the mitochondria. 23 Our results show that treatment with D scandens induces procaspase 3 and Bcl-2 downregulation. Because activated caspase 3 is known to cause cleavage of Bcl-2, converting it into a Bax-like fragment, 24 it is possible that caspase 3 cleaved from the procaspase form may facilitate Bcl-2 proteolysis in the cytosol, causing cleaved caspase 3 itself to be used up as a substrate in a reaction, and inducing a feedback loop causing apoptosis. 42 Treatment with E scaber did not produce a change in procaspase 3 nor caspase 8 (Supplemental Figure S1), despite apoptotic events observed through flow cytometry. However, western blotting revealed upregulation of phosphorylated SAPK/JNK prior to peak p38-MAPK activation. Studies in the literature have found that p38-MAPK activation can cause phosphorylation of Bcl-2 and suppression of caspase 3 cleavage.25-27 This may explain the lack of fold change observed in caspase 3 after cell exposure to E scaber. The increase in SAPK/JNK prior to peak p38-MAPK observed was similar to other studies investigating antitumor properties of nutritional components. 43
PI3K and Akt/Bcl-2 pathways play significant roles in regulating oncogenesis,17,18 and our study has shown that crude ethanolic extracts of D scandens and E scaber altered the signaling of these pathways and caused cell apoptosis. Akt is activated by PI3K and phosphorylates Bad at Ser136, resulting in caspase 3 inhibition through (a) the maintenance of Bcl-x function and (b) prevention of cytochrome c release from the mitochondria. 29 Therefore, reduced available Akt would cause increased apoptotic events. Our study found that the amount of available Akt isoform, Akt1, was significantly reduced after treatment with E scaber and D scandens in a dose-dependent manner, suggesting that Akt1 play a role in plant extract-induced cytotoxicity of HSC-2 cells. There is evidence in the literature that phytochemicals are able to regulate Akt directly. One such example is genistein, a bioactive component in D scandens, which was found to downregulate Akt and causes inactivation of NFκB 44 ; a key regulator of genes involved in cell activation and proliferation. 45 This direct interaction between D scandens and Akt may be independent of MAPK/PI3K pathways as pre-treatment with MAPK/PI3K inhibitors did not prevent extract-induced cell death. Because Akt is activated by PI3K to inhibit apoptosis, 29 our results suggest that D scandens causes cytotoxicity by direct Akt1 downregulation. Existing literature also demonstrates the targeting of Akt by a compound found in E scaber, deoxy-elephantopin, to induce apoptosis or autophagy in many cell types, including microglia, 46 SiHa (cervical carcinoma) cells, 47 and HCT116 (colorectal cancer) cells. 48
Our results show phosphorylation of Erk1/2 after treatment with E scaber. Erk1/2 activation via phosphorylation causes nuclear translocation of the molecule after it is dissociated from Mek1/2, and this translocation determines cell proliferation or apoptosis depending on the intensity and duration of pro- and anti-apoptotic signals.49,50 Akt phosphorylates and stabilizes astrocytic phosphoprotein (PEA-15), a protein which sequesters Erk1/2 into the nucleus, and causes cell proliferation. 51 Our results show decreased Akt, causing Erk1/2 to be retained in the cytosol and augmenting apoptosis, which is similarly reported in existing literature.49,51,52 Pre-treatment of the Erk1/2 inhibitor, U0126, did not directly inhibit E scaber-induced cytotoxicity, suggesting that Erk1/2 plays a supportive role downstream of Akt: once Akt1 is decreased, Erk1/2 is retained in the cytosol and prevents cell proliferation. In contrast, pre-treatment of inhibitors involved in the MAPK and PI3K/Akt pathways were able to inhibit cytotoxicity induced by E scaber, further suggesting their crucial involvement as main mediators of E scaber-induced HSC-2 apoptosis.
Our results suggest that D scandens-induced apoptosis is mediated by direct Akt activation, while Akt1 downregulation and p38-MAPK activation seem to mediate E scaber-induced HSC-2 apoptosis. We thereby propose a mechanism of action for cytotoxic effects of E scaber on human squamous carcinoma 2 (HSC-2) cells, firstly by decreasing Akt1, and secondly, by direct activation of SAPK/JNK, causing phosphorylation of p38-MAPK and the anti-apoptotic protein Bcl-2, which suppresses caspase 3 cleavage. Similar findings have been reported with other flavonoids such as curcumin activating p38-MAPK, Bcl-2, and Akt, as well as other signaling proteins involved. 28 Based on our findings, we propose a possible mechanism in which D scandens and E scaber cause HSC-2 apoptosis (Figure 6). The regulatory scope of apoptosis induction by phytochemicals is increasingly being recognized as a means for cancer treatment, thus further studies to elucidate the exact mechanism of action of these plant extracts on oral cancer cell lines are warranted.

Proposed mechanism of action of E scaber and D scandens in HSC-2 apoptosis. (A) E scaber causes Akt1 downregulation, leading to apoptosis. Our results also suggest another pathway of E scaber induced apoptosis via phosphorylation of SAPK/JNK, leading to p38-MAPK phosphorylation causing Bcl-2 phosphorylation and inhibition of caspase 3 cleavage. (B) D scandens directly downregulates Akt1, causing proteolysis of Bcl-2 leading to cytochrome c release. This causes cleavage of caspase 3, which subsequently cleaves Bcl-2, creating a positive feedback loop for HSC-2 apoptosis induction.
Materials and Methods
Reagents and Antibodies
3-(4, 5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) solution was obtained from Promega (Madison, WI, USA). Annexin V-fluorescein isothiocyanate (Annexin V-FITC) with PI kit was obtained from Miltenyi Biotec (Bergisch Gladbach, Germany). Antibodies anti-phospho-Erk1/2 (p44/42 MAPK) (rabbit mAb cat. no. 9910), anti-phospho-SAPK/JNK (p54/46 MAPK) (rabbit mAb cat. no. 9910), anti-phospho-p38 MAPK (rabbit mAb cat. no. 9910) were obtained from Cell Signaling Technology 53 ; rabbit monoclonal anti-phospho-alpha serine/threonine-protein kinase (Akt1) (phospho S473) (cat. no. ab81283) and rabbit polyclonal B-cell lymphoma 2 (Bcl-2) (cat. no. ab59348) were obtained from Abcam (Cambridge, UK); mouse monoclonal anti-β-actin (Santa Cruz Biotechnology, Inc., Dallas, USA), and secondary antibodies of horseradish peroxidase (HRP)-conjugated polyclonal goat anti-rabbit immunoglobulin 54 (cat. no. P0448) and HRP-conjugated polyclonal goat anti-mouse IgG (cat. no. P0447) were obtained from Dako (Glostrup, Denmark). Rabbit polyclonal anti-caspase 3 (p17 (H-60); cat. no. sc-98785) were obtained from Santa Cruz Biotechnology, Inc. (Dallas, USA). MAPK inhibitors used were SP600125 (cat. no ab120065), LY294002 (cat. no. ab120243), U0126 (cat. no. ab120241), SB203580 (cat. no. ab146589); all were obtained from Abcam (Cambridge, UK). Unless stated otherwise, all other reagents were purchased from Sigma-Aldrich Inc.
Cell Culture
Human squamous carcinoma (HSC-2) cell line was purchased from the National Institute of Biomedical Innovation, Health and Nutrition (Japanese Collection of Research Bioresources Cell Bank [JCRB]; Osaka, Japan) and cultured in 1X Eagle's minimum essential medium with Earle's salts, without L-glutamine and phenol red (Corning, USA), containing 10% fetal bovine serum (FBS) (Hyclone, USA) and 100UI/mL penicillin and streptomycin (Hyclone, UK). Cells were seeded at a density of 7 × 105 cells/cm2 before being harvested by trypsinization (Trypsin; Hyclone, USA) and counted under a microscope for subsequent experiments. Cell culture was kept at 37 °C in an incubator containing 5% CO2. Cells were treated with varying concentrations of either E scaber or D scandens extract for 24 h (at 0.75, 1.5, or 3 mg/ml), or pre-treated with inhibitors SB203580, SP600125, LY294002, and U0126 for 1 h before plant extracts were added.
Plant Material Preparation
D scandens stems were obtained from Khao Chakan Arboretum, Sra Kaeo, Prachin Buri, and leaves of E scaber were obtained from Sakunothayan Arboretum, Wang Thong, Phitsanulok Province, Thailand. Leaves of E scaber and fresh stems of D scandens were thoroughly washed with tap water, cut into small pieces, and dried for 15 h. Extraction was performed using standard procedures. Materials were reduced to coarse powder and macerated with ethyl alcohol for 48 h, and the filtrate was evaporated using a Büchi B-490 rotary evaporator (Büchi Labortechnik AG, Flawil, Switzerland) and lyophilized with a freeze dryer (Labconco Corp., Kansas City, MO, USA) to obtain dried extract using a vacuum evaporator. Powdered forms of plant samples were stored at room temperature. Before each experiment, dried powder was dissolved in ethanol to obtain stock solutions of 300 mg/mL and diluted to varying concentrations. All chemicals were of analytical grade purity. Extracts were prepared fresh prior to treatment and filtered twice before use, once through a70 μm cell strainer (Corning, USA) and once through a0.45 μm sterile- and endotoxin-free filter (Whatman, UK).
MTT Cytotoxicity Assay
MTT colorimetric assay was used to determine cell viability, as previously reported. 55 Cells were seeded at a density of 1 × 104 with fresh medium before treatment for a 24 h period with varying plant extracts prepared to concentrations of 0.375, 0.75, 1.5, and 3 mg/mL. MTT solution were added to each well for 2 h before dimethyl sulfoxide was added. Absorbance was measured at 570, 550, and 690 nm (blank).
FACS Analysis
Briefly, cells were trypsinized and washed with phosphate-buffered saline (Sigma Chemicals, USA), before washing with binding buffer, each time centrifuging for 10 min at 300×g. 10 μL Annexin V-FITC and 5 μL PI (1 μg/mL) per 1 × 106 cells were added according to the manufacturer's recommendations (cat. no. 130-092-052; Miltenyi Biotec, Germany) and mixed thoroughly before incubation. PI was added immediately before analysis with flow cytometry, as per previously reported. 56 Analysis was done by FACSDiva Version 6.1.3 (BD Biosciences, New Jersey, USA).
Nuclear Morphological Staining
Cells were seeded at 4 × 103 cells/chamber (Lab-Tek II Chamber Slide System, Sigma-Aldrich, USA) and were stained with 5 μg/mL Hoechst 33342 (cat. no. H3570; Thermo-Fisher Scientific, Waltham, MA, USA) for 30 min at 37 ˚C and fixed onto a microscope slide and examined under a fluorescence microscope as previously reported. 56 The number of cells exhibiting condensed nucleus was determined from random sampling of 3 × 102 cells per experiment.
Western Blot Analysis
HSC-2 cells were seeded at 7 × 105 cells per T75 flask (Corning, USA) and treated with 0.75, 1.5, and 3 mg/mL D scandens and E scaber for 24 h. Cells were also treated with a maximum dose of plant extracts (3 mg/mL) for 0, 5, 10, 15, 30, 60, and 120 min. Protein samples were lysed using mammalian protein extraction reagent (Thermo-Fisher Scientific, USA) supplemented with protease inhibitor cocktail (Sigma-Aldrich, USA). Protein concentration was determined using PierceTM BCA protein assay (Thermo-Fisher Scientific, USA) with bovine serum albumin as a standard. Western blotting was used to detect change in protein expression as per methods previously reported. 57 Protein was loaded into 10% SDS-polyacrylamide gels and electrophoretically transferred onto polyvinylidene difluoride membranes and blocked with 3% bovine serum albumin 1 h at room temperature and subsequently incubated at 4 ˚C overnight with primary antibodies against phospho-Erk1/2 (p44/42 MAPK) (1:1000), phospho-SAPK/JNK (p54/46 MAPK) (1:1000), phospho-p38MAPK (1:1000), phospho-Akt1 (1:1000), Bcl2 (1:1000), and caspase 3 (1:1000). The membranes were then washed with Tris-buffered saline with 0.1% Tween 20 and was incubated with secondary antibodies (HRP-conjugated goat anti-rabbit IgG or HRP-conjugated goat anti-mouse IgG; 1:1000) at room temperature for 1 h. Proteins were visualized using a chemiluminescence kit and x-ray films (Bio-Rad Laboratories., CA, USA). Protein band densitometric analysis was done by a Scion Image Software (Scion Corp., Frederick, MD, USA).
Statistical Analysis
All experiments were performed in triplicate. Results were expressed as mean ± SD. The differences between multiple groups were assessed using one-way analysis of variance 58 followed by Tukey-Kramer method, and differences between 2 groups were assessed using Student's t-test. The level of significance was set at P < .05. All analyses were performed using SPSS 20.0 (SPSS Institute, Cary, NC, USA).
Supplemental Material
sj-jpg-1-npx-10.1177_1934578X221107970 - Supplemental material for Induction of Human Oral Squamous Carcinoma Apoptosis by Derris scandens Benth and Elephantopus scaber Linn Extracts
Supplemental material, sj-jpg-1-npx-10.1177_1934578X221107970 for Induction of Human Oral Squamous Carcinoma Apoptosis by Derris scandens Benth and Elephantopus scaber Linn Extracts by Petchpailin Leenutaphong, Salunya Tancharoen, Thamthiwat Nararatwanchai, Chareerut Phruksaniyom, Ariya Sarikaphuti, Pakhawadee Palungwachira and Sirintip Chaichalotornkul in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
