Abstract
Diabetic nephropathy (DN) is the leading cause of uremia and clinical mortality, characterized by the progressive deterioration of kidney structure and function due to prolonged exposure to hyperglycemia. This condition often necessitates renal replacement therapy for patients with end-stage-renal-disease (ESRD). Consequently, early detection, diagnosis, and treatment of DN are crucial to mitigate disease progression, enhance patient outcomes, and maintain a good quality of life. Exploring relevant biomarkers for the early diagnosis of DN holds significant importance. In recent years, numerous researchers have identified various novel biomarkers associated with diabetic nephropathy, which are critical for predicting both the onset and progression of the disease. This article aims to provide a comprehensive overview of the biomarkers related to DN.
Introduce
The pathogenesis of multifactorial and multi-stage DN is intricate, primarily driven by hyperglycemia-induced inflammatory processes affecting both hemodynamic and metabolic pathways. Hemodynamic alterations elevate intraglomerular pressure and activate vasoactive hormone pathways, while metabolic changes result in mesangial cell expansion and structural modifications. These processes ultimately lead to inflammation, endothelial dysfunction, and fibrosis. In clinical practice, the estimation of glomerular filtration rate (eGFR) and albuminuria serve as common markers for the early diagnosis of DN; however, they also act as significant predictors of cardiovascular disease and mortality, with their prognostic relevance extending beyond DN alone. 1 A notable limitation of this approach is the increasing prevalence of the nonalbuminuric phenotype in DN, where the absence of albuminuria is becoming more common. 2 Furthermore, when albuminuria is present, the associated renal damage often reaches an advanced stage, resulting in a rapid renal function decline to ESRD. It is important to note that proteinuria does not consistently precede a decline in renal function, and relying solely on proteinuria detection is inadequate for monitoring the incidence and progression of DN, indicating early involvement of the tubulointerstitial compartment rather than the glomerulus. 3 Additionally, serum creatinine levels are influenced by factors such as muscle mass and dietary protein intake, rendering eGFR estimates unreliable as biomarkers for diagnosing and stratifying DN progression, particularly in patients with type 2 diabetes mellitus (T2DM) have low eGFR estimates using the CKD-EPI equation. 4 Moreover, despite treatment with current renin-angiotensin-aldosterone system (RAAS) blockers and sodium-glucose cotransporter-2 inhibitors (SGLT2is), there remains a significant residual risk of progressive renal function decline, highlighting urgent the need for targeted therapeutic interventions. 5 Current clinical diagnostic markers for DN exhibit several limitations, including the late onset of albuminuria during disease progression, the occasional absence of proteinuria in typical DN cases, and inaccuracies in eGFR. Consequently, there is an urgent need to identify alternative biomarkers that can be detected earlier or concurrently with microalbuminuria. Such advancements would facilitate earlier disease identification, enhance risk stratification, and potentially delay the progression of DN.
Over the past few years, significant progress has been made in understanding the pathophysiology of DN and the interpretation of associated biomarkers. The purpose of this review is to describe the current biomarkers of DN from multiple perspectives such as DN physiology and pathology, pathogenesis, and omics techniques, as shown in Figure 1.

The current biomarkers of DN from multiple perspectives.
Physiological and Pathological Biomarkers of DN
DN induces morphological and functional changes in the kidney, mainly affecting the glomeruli and tubulointerstitium. Analysis of the molecules that cause kidney lesions has the potential to predict the course of DN. As shown in Figure 2, the single biomarkers of DN in physiology and pathology are summarized.

Single biomarkers of DN in physiological and pathological.
Biomarkers of Renal Tubular Injury
Research indicates that tubular injury in diabetic patients can occur prior to glomerular injury, and the extent of tubulointerstitial damage may play a crucial role in the deterioration of renal function. 3 Consequently, tubular biomarkers hold significant importance for the early diagnosis of diabetic nephropathy.
Cystatin C
Cystatin C (CysC) is a low-molecular-weight protein consisting of 122 amino acids, synthesized and released into plasma by all nucleated cells at a constant rate. This small, positively charged protein is freely filtered through the glomerular membrane and is subsequently completely reabsorbed and degraded in the proximal tubule, without being secreted back into circulation. Consequently, CysC is typically absent in urine.6–8 As a member of the cysteine protein inhibitor family, CysC serves as the most abundant and potent endogenous inhibitor of both intracellular and extracellular enzyme activity, playing a significant role in various pathological processes, including cardiovascular disease and inflammation. 6 Recent studies have identified CysC as a valuable biomarker for tubular injury, aiding in the quantification and classification of different stages of DN. 9 Research conducted by Jeon 10 et al demonstrated that CysC levels increase with advancing stages of chronic kidney disease (CKD), from stage I to III, and correlate positively with the albumin/creatinine ratio, particularly during the transition from normal albuminuria to microalbuminuria. Furthermore, Xu 11 et al confirmed the association of elevated CysC levels with the progression of DN. Notably, serum CysC levels are independent of muscle mass and remain unaffected by age, presenting distinct clinical advantages over creatinine (Cr). Both serum CysC and Cr exhibit a hyperbolic relationship with GFR, allowing their inclusion in the same predictive equation for GFR. 8 CysC is particularly advantageous for patients experiencing muscle mass loss or degenerative muscle diseases, proving to be a more accurate biomarker. 9 Thus, utilizing serum CysC as a biomarker for the early diagnosis of acute kidney injury (AKI) can effectively reflect early changes in renal function and a decrease in estimated GFR.
KIM-1
KIM-1 is a 38.7-kDa type I transmembrane glycoprotein expressed on the parietal membrane of renal proximal tubular (PT) cells. It is shed from these cells during AKI and contributes to tubular epithelial hyperplasia. 12 Elevated concentrations of urinary KIM-1 (uKIM-1) are associated with further damage to the tubulointerstitium, leading to increased fibrosis and inflammation.13,14 Notably, estimates of uKIM-1 levels can occur prior to the elevation of serum creatinine, serving as predictors of AKI; thus, KIM-1 has been utilized as a biomarker in studies investigating drug nephrotoxicity. 14 Presently, KIM-1 is the most extensively studied biomarker for DN. 15 Clinical studies have demonstrated a gradual increase in uKIM-1 values among patients with T1DM and T2DM, particularly those exhibiting microalbuminuria.16,17 In a nested case-control study involving 380 participants and a prospective cohort study with 1156 participants, serum KIM-1 (sKIM-1) levels were independently associated with a heightened risk of decreased eGFR in patients with either early or advanced DN. 18 Similarly, a case cohort study of 894 individuals with DN and renal insufficiency demonstrated that higher sKIM-1 levels correlated with an increased risk of DN progression. 19 Additionally, KIM-1 may be linked to the risk of developing DN in children and adolescents diagnosed with T1DM; however, further prospective cohort studies are necessary to elucidate this relationship. 20 Moreover, fully printed photonic crystal microarrays can quantitatively detect uKIM-1 levels in less than 10 min. 21 Beyond its role in AKI, KIM-1 serves as a valuable tool for detecting CKD progression and guiding therapeutic interventions for patients at risk of CKD.
Neutrophil Gelatinase-Associated Lipocarrierin, NGAL
NGAL, a 25 kDa protein that belong to the lipocalin family, is synthesized by neutrophils and epithelial cells in various tissues, including the kidney. This protein is released into circulation and subsequently filtered through the glomerular membrane, with reabsorption occurring in the proximal tubule.9,22 NGAL has been extensively validated as a reliable biomarker for AKI, 23 and prospective analyses have demonstrated a strong association between urinary NGAL (uNGAL) levels and the histopathological severity of inflammatory kidney injury in patients with DN. 24 Significant elevations in serum NGAL (sNGAL) and uNGAL have been observed in individuals with normal to micro-massive albuminuria T2DM,20,25 indicating that tubular and glomerular injuries may occur in the early stages of DN, thereby enabling the detection of early nephropathy prior to the onset of proteinuria. A meta-analysis further supports the potential diagnostic value of uNGAL for DN. 26 He et al 27 conducted a meta-analysis involving 19 studies, suggesting that both sNGAL and uNGAL could assist in the classification of DN and possess diagnostic utility in normoalbuminuric nephropathy. In a study of 300 subjects with T2DM, both uNGAL and sNGAL were predictive of albuminuria and could serve as noninvasive tools for diagnosing, grading, and monitoring the progression of DN. 28 Furthermore, Duan et al 29 identified an optimal cut-off value for the uNGAL-to-creatinine ratio of 60.685 ng/mg, which was recognized as an independent risk factor for DN in patients with CKD and DM, exhibiting high specificity (90.5%). Additionally another study involving T2DM patients found that NGAL levels were positively correlated with renal function tests, including creatinine, blood urea nitrogen, and albuminuria. The sensitivity and specificity of uNGAL at a cut-off point of 565 ng/mL were reported to be 97.5% and 100%, respectively, underscoring the accuracy of both uNGAL and sNGAL as biomarkers for the early diagnosis of DN. 30 Recent reports regarding NGAL have revealed significant insights, indicating that asymptomatic SARS-CoV-2 infections can lead to elevated levels of uNGAL, comparable to those observed in symptomatic COVID-19 patients. This results suggests that NGAL is more closely associated with immune system activation than with renal function. 31
These findings imply that NGAL could serve as a non-invasive tool for diagnosing, staging, and monitoring the progression of DN, while also offering additional diagnostic value in patients with nephropathy characterized by similar levels of albuminuria. However, large-scale prospective studies are essential to validate the application of NGAL as a biomarker for routine clinical practice.
Hepatic Fatty Acid-Binding Protein, L-FABP
L-FABP is a 14-kDa intracellular protein predominantly synthesized in the cytoplasm of proximal tubule (PT) cells, where it plays a crucial role in the metabolism of long-chain fatty acids. This protein is essential for maintaining fatty acid homeostasis, as fatty acids within the cytoplasm bind to L-FABP. A loss of intraglomerular fatty acid-binding albumin can lead to excessive accumulation of intraglomerular fatty acids, which induces inflammatory responses and manifests as tubular lesions in CKD. The upregulation of the L-FABP gene accelerates fatty acid metabolism and mitigates inflammation, functioning as a protective response. 32 In a three-year follow-up study involving 48 patients with T1DM, uL-FABP emerged as an early marker of DN, correlating with disease progression over time, independent of albuminuria. 33 A cross-sectional study comparing patients with T2DM and nondiabetic individuals found that changes in uL-FABP levels during renal impairment preceded alterations in urinary albumin levels, suggesting that uL-FABP is a more reliable indicator than the albumin-to-creatinine ratio (ACR). 34 Zhong et al 35 reported significantly elevated L-FABP levels in elderly patients with T2DM, which were closely associated with the onset and progression of type 2 DN. Furthermore, a meta-analysis conducted by Zhang et al 36 indicated that uL-FABP could serve as a potential biomarker for detecting all stages of DN and for predicting the progression and severity of the condition in patients with both T1DM and T2DM. A 12-year follow-up study corroborated these findings, demonstrating a significant increase in uL-FABP, a 50% reduction in GFR, and a decrease in the incidence of cardiovascular disease among patients with T2DM. 37 The assessment of uL-FABP in individuals with T2DM appears to be a promising tubular marker for predicting the incidence of cardiovascular disease and renal impairment. However, a 14-year follow-up study by Panduru et al, which involved 2329 individuals with T1DM found that uL-FABP was an independent predictor of stroke and mortality in this population, but did not predict other cardiovascular endpoints. 38
Furthermore, Hirowatari 39 et al reported that uL-FABP levels were significantly reduced following the administration of sodium-glucose cotransporter 2 inhibitors, indicating that uL-FABP may serve as a marker for the therapeutic efficacy of DN treatment. Additionally, Mohsen et al proposed that the DPP4 inhibitor saxagliptin may reduce albuminuria by mitigating tubular injury, suggesting that uL-FABP could be a valuable marker for monitoring the efficacy of gliptins in the management of DN. 40 Importantly, advancements in clinical decision-making, such as the development of chemiluminescence rapid quantitative tests 41 and microchip immunoassays, 42 have facilitated the rapid, cost-effective, and high-sensitivity quantitative detection of biomarkers like uL-FABP, which is beneficial for precise and frequent monitoring of renal function.
Retinol-Binding-Protein-4, RBP4
RBP4 is a low-molecular-weight transporter that facilitates the transport of vitamin A (retinol) in the bloodstream. Elevated RBP4 levels have been associated with the onset of metabolic diseases, as RBP4 promotes inflammation and oxidative stress through the activation of immune cells and releasing pro-inflammatory cytokines. 43 Furthermore, RBP4 is regarded as a potential diagnostic marker for renal impairment due to its ability to detect tubular damage early and to sensitively reflect the extent of injury to the renal proximal convoluted tubules. 44
Evidence indicate that elevated RBP4 levels can be identified in the early stages of type 2 DN, with urinary RBP4 (uRBP4) and microalbumin levels significantly higher in the diabetic cohort compared to both the control group and those with nephropathy. 45 A retrospective study involving 1946 patients with T2DM, with or without DN, found that elevated uRBP4 was linked to an increased risk of DN in T2DM patients, as well as worsening albuminuria and declining renal function in those with DN. 46 Additionally, Javed reported a significant correlation between RBP4 levels and creatinine clearance. 47 A recent study evaluated the diagnostic utility of uRBP4 in patients with T2DM, reporting an area under the curve (AUC) of 0.74. 22 Additionally, a meta-analysis indicated that RBP4 demonstrates strong sensitivity and specificity in T2DM patients with DN, underscoring its promising diagnostic potential. 44 Xing et al 48 established that RBP4 levels are positively correlated with urinary albumin concentration, achieving a sensitivity of 85.7% and a specificity of 56.2%. This study, which included a cohort of 135 T2DM patients, proposed that RBP4 could serve as a biomarker for the early diagnosis of DN. Furthermore, a prospective study involving 438 T2DM patients found that RBP4 was positively correlated with the degree of renal impairment, effectively identifying DN in this population. 49
RBP4 also reflects the onset of diabetic retinopathy 50 and is regarded as a promising biomarker for predicting cardiovascular disease. Moreover, medicinal plants that target RBP4 may aid in the treatment of cardiometabolic diseases. 51 For instance, a study involving patients with prediabetes or early T2DM who were supplemented with mulberry leaves for 12 weeks revealed that the down-regulation of RBP4 is associated with improved insulin resistance. Collectively, these findings suggest that RBP4 is a valuable biomarker for both the prevention and treatment of early DN. 52 Collectively, it may serve as an effective marker for identifying the early onset of DN and for predicting the progressive stages of renal impairment in individuals with both T1DM and T2DM.
Biomarkers of Glomerular Injury
The glomeruli serve as the primary sites of damage in DN. This progressive DN is characterized by several pathological changes, including the loss of podocyte foot processes, thickening of the glomerular basement membrane, and expansion of mesangial cells within the glomeruli. Collectively, these alterations lead to a reduction in glomerular surface area and impaired glomerular filtration. 53 Table 1 summarizes the biomarkers associated with current glomerular injury.
Biomarkers of Glomerular Injury.

DN Pathogenesis Biomakers
Oxidative Stress Biomakers
Reactive oxygen species (ROS) are the primary contributors to oxidative stress, which is associated with disease progression. In the kidneys, ROS predominantly generated by mitochondria and the nicotinamide adenine dinucleotide phosphate oxidase (NOX) family. Additionally, oxidative stress arises from advanced glycation end-products (AGEs) induced by hyperglycemia during the later stages of non-enzymatic glycation of sugars and proteins. This hyperglycemia-induced oxidative stress leads to several detrimental outcomes, including DNA damage, lipid peroxidation, mitochondrial dysfunction, and the infiltration of inflammatory cells, ultimately culminating in renal cell damage. 14
8-OHdG
8-Hydroxy-2'-deoxyguanosine (8-OHdG) is a byproduct of DNA damage resulting from oxidative stress in living cells, particularly in the context of DN. It is excreted in urine through nuclease activity and can also be detected in plasma.13,68 Cohort studies involving patients with T1DM have indicated that elevated plasma levels of 8-OHdG are independently associated with an increased risk of kidney disease, implying its potential utility in monitoring the progression of DN. 69 Wakeel et al demonstrated that urinary 8-OHdG is a reliable marker of oxidative DNA damage in T1DM patients with diabetic retinopathy. 70 Furthermore, urinary excretion of 8-OHdG may serve as an independent predictor of disease progression and the onset of both microvascular and macrovascular complications. However, Serdar et al reported that urine 8-OHdG measurement did not effectively predict progressive DN when compared to uACR. 71 Consequently, further research is warranted to elucidate the clinical significance of these biomarkers and to explore the role of novel therapeutic agents in diabetes management.
Pentosidine
Pentosidine, a product of higher sugar oxidation, is formed through the covalent binding of amino groups to glucose. Machowska et al observed that plasma pentosidine levels correlate with low GFR, oxidative stress, and inflammation, serving as independent predictors of mortality in patients with CKD. 72 Furthermore, Perkins et al reported that patients with microalbuminuria exhibited approximately a two-fold increase in pentosidine excretion and an early decline in GFR compared to those with normal albuminuria. 73 In diabetic patients, elevated pentosidine levels have been identified as independent predictors of diabetic retinopathy, cardiovascular disease, and all-cause mortality. 74 Collectively, these findings suggest that measuring pentosidine levels in both urine and serum may aid in identify patients at risk for early GFR decline and could serve as a promising biomarker for both microvascular and macrovascular complications associated with diabetes.
Uricacid
Uricacid, a byproduct of purine metabolism, has been demonstrated to independently predict the progression of DN. Numerous clinical studies have concentrated on the relationship between uric acid levels and DN prognosis. In a cohort study involving 277 patients with T1DM, elevated serum uric acid levels were identified as predictors of persistent massive albuminuria. 75 Similarly, among a cohort of 422 patients with T2DM who had been diagnosed for at least 15 years, initial hyperuricemia emerged as an independent risk factor for DN progression. 76 A four-year follow-up study by Cosmo et al revealed a significant association between serum uric acid levels and albuminuria; furthermore, the cumulative incidence of GFR decline was notably higher in patients with hyperuricemia, which was identified as an independent risk factor for progression in T2DM patients. 77 Additionally, a significant correlation exists between urinary lactate levels and biomarkers indicative of renal tubular injury and epithelial stress, suggesting that elevated urinary lactate levels could serve as potential biomarkers for the risk of renal disease progression.
Collectively, this evidence indicates that serum uric acid may function as an independent predictor for the subsequent development of massive albuminuria in patients with both T1DM and T2DM.
Acylcarnitine
Acylcarnitine, an esterification product of carnitine, plays a crucial role in fatty acids oxidation and is significantly associated with an increased risk of T2DM. 78 Mirzoyan et al 79 demonstrated that in cases of hyperlipidemia, the development of diabetes correlates with a diminished capacity of the kidneys to oxidize fatty acids and amino acids. Sulaj et al 80 proposed that acylcarnitine has historically been an important diagnostic marker for congenital fatty acid oxidation disorders. Mu et al 81 found that short- and medium-chain acylcarnitines were less prevalent, while longer acylcarnitines were more abundant in the late stages of DN, typically observed in diabetes with normal albuminuria and microalbuminuria. The increase in long-chain acylcarnitines is believed to compensate for the adaptive oxidation of fatty acids in the early stages of DN, whereas the reduction of long-chain acylcarnitines in advanced DN stages is attributed to incomplete fatty acid oxidation. Consequently, acylcarnitine may serve as a predictive marker for the risk of DN. Hassan et al 82 emphasized the importance of monitoring plasma free amino acid and acylcarnitine profiles children with T1DM, particularly those testing positive for JAK2 (V617F), to facilitate the early diagnosis of DN. Li and Xuerui et al 83 conducted an analysis of clinical data from 1032 participants with T2DM, measuring levels of 25 acylcarnitine metabolites in fasting plasma via mass spectrometry. Their findings indicated that certain plasma acylcarnitine metabolites were elevated in T2DM patients with DN, and incorporating acylcarnitine into traditional risk models enhanced the predictive value for DN. Esmati et al 84 employed flow injection tandem mass spectrometry (FI-MS/MS) to simultaneously quantify amino acids and carnitine/acylcarnitines in patients with T2DM, meeting analytical performance requirements within a brief timeframe. The concurrent detection of amino acid and acylcarnitine profiles serves as prospective tandem biomarkers for DN, offering valuable insights for predicting the risk of DN at the time of initial diagnosis. While acylcarnitines have demonstrated a strong predictive capacity for DN, further clinical data and scientific research are necessary to enhance the acylcarnitine risk model.
Acrolein
Acrolein, an α, β-unsaturated aldehyde, is endogenously produced during lipid peroxidation. It forms highly reactive conjugates with proteins, leading to alterations in protein function. Renal fibrosis is a hallmark of DN, and it has been demonstrated that acrolein modification at Cys358 in PKM2 results in its inactivation, which is implicated in the pathogenesis of DN. 85 Currently, there are very few reports on acrolein involving DN biomarkers.
Inflammatory Biomarkers
Monocyte Chemoattractant Protein-1, MCP-1
MCP-1, also known as C-C motif chemokine ligand 2 (CCL2), is a pro-inflammatory cytokine synthesized by monocytes, cortical tubular epithelial cells, and podocytes. It is responsible for mediating the migration of monocytes and macrophages to tissues via the nuclear factor-κB pathway. MCP-1 has been implicated in various renal pathologies, including inflammation, glomerular injury, tubular atrophy, and fibrosis. 86 Notably, excessive renal inflammation can lead to a rapid decline in renal function, and elevated levels of MCP-1 have emerged as a promising biomarker for predicting progressive decline in non-DN renal function. 87
In a median follow-up period ranging from 4.2 to 7.1 years, Menez et al found that 266 patients (30.8%) experienced a composite CKD outcome following cardiac surgery. The suggesting indicates that uMCP-1 is independently associated with CKD post-surgery, and suggests its potential as a noninvasive indicator of tubular injury. 88 Similarly, Fufaa et al found a significant correlation between uMCP-1 concentrations and changes in renal interstitial volume, as well as disease progression in normotensive patients with albuminuria and T1DM. 89 In a cohort of 75 patients with T2DM exhibiting varying degrees of DN, uMCP-1 levels were observed to increase with albuminuria, demonstrating a sensitivity of 92% and a specificity of 100% for early diagnosis. 90 Additionally, in a prospective study involving 83 high-risk patients with T2DM and DN, Satirapoj et al showed that uMCP-1 was positively associated with early declines in DN GFR, independent of conventional risk factors. 91 A case-cohort study of 894 participants with T1DM and T2DM reported that high levels of MCP-1 were also associated with an increased risk of DN progression. 19
Overall, MCP-1 proves to be a valuable biomarker for detecting the incidence or progression of CKD in patients undergoing cardiac surgery as well as for stratifying long-term CKD risk in other recognized clinical settings for kidney injury. 87 These observations suggest that MCP-1 as a promising inflammatory marker for diagnosing early progressive renal dysfunction and for assessing long-term CKD risk.
Tumor Necrosis Factor -α, TNF-α
TNF-α has been identified as a notable inflammatory marker that predicts the progression of DN. 92 A meta-analysis by Qiao et al 93 demonstrated a significant elevation in TNF-α levels among patients with T1DM compared to healthy controls. Furthermore, Sharad et al established a robust correlation between microalbuminuria and the activity of soluble serum TNF-α receptors and the IL-6 pathway in T1DM patients, indicating the critical role of TNF-α in disease progression. 94 Umapathy et al reported that a gradual increase in normal-micro-massive albuminuria in T2DM is associated with the rs1800629 polymorphism in the TNF-α gene. 95 Additionally, Khaloo et al found that elevated TNF-α levels were linked to diabetic retinopathy in T2DM, achieving an area under the curve (AUC) of 0.84. 96 Consequently, the assessment of TNF-α emerges as a promising biomarker for predicting cardiovascular disease and progressive renal function impairment.
The TNF-α receptor is a type 1 transmembrane protein distinguished by cysteine-rich motifs present in glomeruli and tubular cells. It comprises two types: TNF-α receptor 1 (TNFR1, 55 kDa) and TNF-α receptor 2 (TNFR2, 75 kDa). TNF-α binds to these receptors and is also detected in its soluble form within the systemic circulation. 13 Studies have indicated that soluble TNF-α receptor 1 (sTNFR1) 97 and soluble TNF-α receptor 2 (sTNFR2) 19 sever as significant predictors of DN progression in patients with T2DM, with sTNFR1 emerging as the most robust prognostic biomarker. 98 Paykov et al reported that serum levels of TNF receptors were independently associated with decreased renal function in T2DM and ESRD. 99 In a nested case-control and prospective cohort study, elevated plasma levels of TNFR-1 and TNFR-2 were linked to a heightened risk of DN progression, independent of age and other laboratory parameters. 18 Moreover, circulating levels of sTNFR have been shown to independently predict progressive renal function decline and a ten-year risk of ESKD in cohorts of patients with T1DM and T2DM. 100 In contrast, Martin et al reported that sTNFR1 predicts of short- to medium-term mortality; but not of progressive renal function decline in an elderly cohort of T2DM patients with CKD. They also noted that the parallel assessment of uACR could provide complementary prognostic information. 101 Furthermore, sTNFR1 and sTNFR2 are emerging as promising biomarkers for forecasting progressive impairment of renal function.
YKL-40
YKL-40, also known as chitinase 3-like-1 (CHI3L1), is a 40-kDa glycoprotein secreted by various cell types that protects renal tubular cells from apoptosis. 102 AKI is a prevalent syndrome among critically ill patients, and elevated levels of YKL-40 are associated with poorer clinical outcomes. Biomarkers are essential for in the early recognition and management of AKI, potentially enhancing patient outcomes. 103 Urinary YKL-40 levels correlate with the severity and mortality of AKI during hospitalization, and its elevation is considered a promising urinary biomarker for AKI diagnosis. It demonstrates comparable diagnostic efficacy in identifying patients with AKI at or above KDIGO stage 2 within 24 h; however, further studies are warranted to validate its clinical applicability.103,104 A single-center, prospective cohort study involving 249 AKI patients indicated that urinary YKL-40 levels were associated with disease progression and/or mortality, thereby enhancing clinically determined risk reclassification. 105 Additionally, Conroy et al identified YKL-40 as an independent risk factor for mortality in AKI related to malaria pathogenesis, highlighting its potential as a biomarker for AKI. 106 Elevated serum YKL-40 concentrations have also been documented in patients with T2DM. 107 In a case-cohort study involving 894 T2DM participants, higher YKL-40 levels were linked to an increased risk of DN progression. 19 The integration of YKL-40 with other AKI biomarkers, such as NGAL, may facilitate a more comprehensive assessment of progression risk. 108 However, additional research is required to establish the universal applicability of YKL-40 and its relationship with long-term AKI outcomes.
EGF
Epidermal growth factor (EGF) is a 6-kDa mediator that regulates and stimulates the proliferation, migration, and differentiation of mesenchymal cells, particularly within epithelial tissues. It is expressed predominantly in the distal tubules and plays a crucial role in the proliferation and survival of renal tubular cells, typically being excreted in the urine. Recent data suggest that urinary EGF serves as a reliable noninvasive indicator of functionally normal tubular volume and the status of tubular repair following AKI. 109 Norvik et al demonstrated that lower levels of urinary EGF (uEGF) were associated with a rapid decline in GFR and an increased risk of CKD events in the general population, based on a median follow-up of a large sample size (n = 5883) over an average of six years. These findings align with observations in CKD and high-risk populations, indicating that uEGF can be utilized as a broadly applicable tubular-specific biomarker for assessing clinical risk. 110 Additionally, the uEGF/MCP-1 ratio is independently linked to rapid renal progression in patients with DN. 91 Lower urinary EGF levels correlate with increased interstitial fibrosis and tubular atrophy, while higher EGF levels are associated with improved treatment responses and remission in various glomerular diseases. 111 Therefore, the therapeutic use of exogenous EGF may not be an appropriate strategy for long-term treatment.
FGF-23
Fibroblast growth factor 23 (FGF-23), a 28 kDa hormone, is crucial for regulating renal phosphate excretion and maintaining mineral balance. 112 Phosphate homeostasis is compromised when the GFR declines. 113 An observational study involving 60 patients with stages Ⅲ-Ⅴ CKD found that elevated phosphate levels correlated with increased FGF-23/Klotho ratios, suggesting that these ratios may serve as risk factors for evaluating CKD patients and as protective indicators for treatment as well as prognosis. 113 Additionally, a separate study of 60 CKD patients undergoing hemodialysis revealed significant associations between FGF-23 and levels of creatinine, urea, phosphorus, and calcium. 112 A systematic review and meta-analysis conducted by Castillo et al indicated that aerobic physical exercise can decrease FGF-23 levels and slow the progression of CKD. 114 Furthermore, several studies have elucidated the mechanisms underlying the upregulation of FGF-23 in AKI, establishing that quantitative changes in FGF-23 may serve as a reliable biomarker for predicting adverse outcomes and prognosis in AKI patients.115,116 FGF-23 is a potential marker for DN, but there are currently few research reports on DN.
Other Inflammatory Factor Biomarkers
Recent studies have updated our understanding of inflammatory factor biomarkers in DN. For instance, Hostet et al 102 demonstrated that elevated urinary levels of C-C motif chemokine ligand 14(CCL-14) are associated with renal impairment, identifying it as the most reliable predictor of AKI severity and persistence. Furthermore, the expression levels of renal interleukin-17 receptor E (IL-17RE) have been correlated with albuminuria and the severity of DN. 117 Interleukin-6 (IL-6) is recognized for its dual role in inflammation and glycemic control, making it a promising biomarker and therapeutic target for both DN and T2DM. 118 Additionally, decreased serum levels of IL-18 have been linked to the development of DN in the Pakistani population. 119 The overexpression of Wilms’ tumor 1(WT1) in podocytes has been shown to induce podocyte injury associated with DN. 120 Moreover, angiotensin-converting enzyme-2 (ACE-2) and connective tissue growth factor (CTGF) have emerged as early and reliable predictors of disease progression in DN. 121 Lysine lactylation and acyl-CoA synthetase family member 2 (ACSF2), which are implicated in the pathogenesis of metabolic and inflammatory diseases, may also contribute to the progression of DN. 122 Notably, a reduction in MCPIP1 has been identified as an independent risk factor for renal progression in patients with DN. Additionally, centrally administered dapagliflozin has been reported to exert anti-inflammatory effects by upregulating MCPIP1 levels in microglia and altering lipid metabolism in kidneys affected by DN, 123 These findings suggest that MCPIP1 levels may serve as a valuable biomarker for assessing the efficacy of dapagliflozin in the treatment of DN.
Future Trends in Biomarkers of DN
In the future, DN's research will develop from the direction of multi-biomarker combination, universal application of omics technology, and personalized treatment.
Multi-Biomarkers Combination
DM is a heterogeneous disease characterized by various pathophysiological mechanisms, which complicates the early diagnosis of DN due to the absence of a singular diagnostic index. The integration of multiple biomarkers significantly enhances both the early diagnosis rate and the predictive capacity for DN. By considering factors such as inflammation, oxidative stress, renal function, metabolites, and genetic markers, a more comprehensive assessment of the disease state can be achieved, thereby improving the sensitivity and specificity of the diagnosis. This multifaceted evaluation strategy is anticipated to yield better clinical outcomes. The diagnostic efficacy of individual biomarkers is not exclusive to DN; it can also be affected by other factors, including the course of infection, which may influence molecules such as IL-18, NGAL, and calprotectin, without necessarily indicating AKI. 124 Consequently, it is crucial to consider the effects of comorbidities on these biomarkers, necessitating a reassessment of the cut-off points for diagnosis, follow-up, and prognosis. In a cohort study involving individuals with T2DM, ShiCaifeng et al 125 identified and integrated seven candidate urine metabolic markers (isovaleric acid, isobutyric acid, leucine, S-adenosylhomocysteine, propionic acid, oxodioic acid, and propionylcarnitine) to construct a diagnostic model. This combined marker model demonstrated significantly superior performance compared to individual metabolic markers, indicating that the use of urine metabolic markers may represent a novel strategy for diagnosing DN. Similarly, Trifonova et al 126 conducted an analysis of plasma samples from 80 patients with T1DM and DN, revealing that a combination of 15 compounds—including amino acids and their derivatives, monosaccharides, organic acids, and uremic toxins—exhibited high diagnostic performance for DN, achieving an early diagnosis accuracy of 81% and a late diagnosis accuracy of 99%. Research has shown that a combination of clinical factors—including age, the presence of proliferative diabetic retinopathy, duration of T2DM, and hemoglobin A1c levels—along with four specific urine metabolites (alanine, choline, N-phenylacetylglycine, and triglycine) is highly effective in predicting patients with T2DM who have biopsy-confirmed DN (AUC = 1). 127 The combined SERPINA4/UCR and gGT1/UCR indexes have been identified as potential biomarkers for DN following adjustment for urine creatinine levles. 128 Zhao Xinxin 129 reported that the simultaneous detection of IL-18, CysC, RBP4, and NGAL yields a higher diagnostic accuracy and sensitivity for DN, thereby facilitating the early detection of diabetic kidney injury. Gonzalez et al 130 proposed a model that incorporates age, proteinuria, EGF, dickkopf-related protein 3 (DKK-3), and MCP-1 to evaluate interstitial fibrosis in patients with DN; their findings indicated that this model effectively assesses the progression of interstitial fibrosis in individuals with normal renal function. Additionally, the combination of sTNFR1, neutrophil gelatinase-associated lipocalin, C-reactive protein, and complement component 3a with cleaved C-terminal arginine (C3a-desArg) enhances the prediction of CKD progression and mortality. 98 In patients with T2DM, urine mitochondrial DNA (mtDNA) levels are elevated during the early stages of kidney injury and positively correlate with the severity of the injury. Notably, urine leukocyte and glucose levels do not interfere with urine mtDNA levels, which are positively associated with the development of early DN. Other clinical indicators, when combined with urine mtDNA, may assist in the diagnosis of early DN. 131
Numerous researchers have conducted co-diagnostic cohort analyses to identify common physiopathological markers in DN, as summarized in Table 2. For instance, the combined assessment of sRBP4 and CysC has revealed that elevated levels of both markers in DN patients are positively correlated with the duration of DM. This finding suggests that NGAL and RBP4 may serve as valuable biomarkers for the early diagnosis of DN, with the combined detection method demonstrating superior diagnostic efficacy. 131 Furthermore, this approach shows significant potential for identifying DN in its early stages and aids in evaluating the severity of kidney disease, thereby providing essential insights for clinical treatment planning.
Combined Biomarkers Based on a Retrospective Analysis Study.
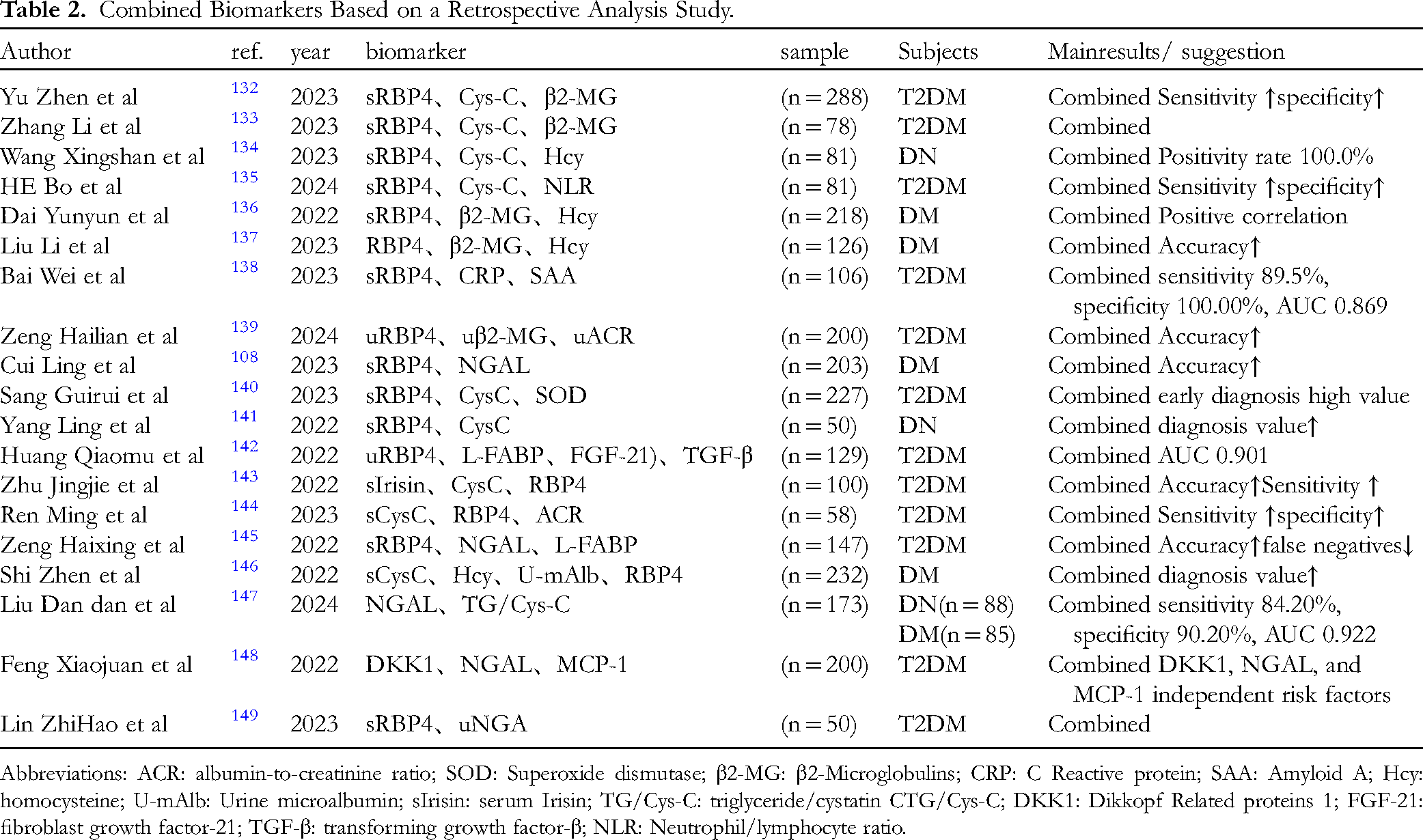
Abbreviations: ACR: albumin-to-creatinine ratio; SOD: Superoxide dismutase; β2-MG: β2-Microglobulins; CRP: C Reactive protein; SAA: Amyloid A; Hcy: homocysteine; U-mAlb: Urine microalbumin; sIrisin: serum Irisin; TG/Cys-C: triglyceride/cystatin CTG/Cys-C; DKK1: Dikkopf Related proteins 1; FGF-21: fibroblast growth factor-21; TGF-β: transforming growth factor-β; NLR: Neutrophil/lymphocyte ratio.
Future research should focus on investigating a panel of biomarkers that exhibit low intercorrelation. Furthermore, the majority of existing studies have been constrained by small sample sizes; therefore, larger population studies are necessary to validate these biomarkers.
Omics Platform-Based Approach
The absence of albuminuria in the non-albuminuria phenotype of DN represents a significant limitation of the current study. Omics testing encompasses a range of analytical methods that integrate multiple molecular measurements, allowing for the capture of a comprehensive spectrum of peptides or metabolites within a short timeframe. This approach enables the quantification of various biomolecules, including RNA, lipids, amino acids, proteins, and metabolites, in a biological sample. The resulting data is interpreted through computational models, yielding a wealth of unbiased and clinically relevant information that can be employed for diagnosis, predicting disease progression, and evaluating treatment responses. Consequently, the omics platform-based approach has emerged as a powerful tool for biomarker discovery in recent years. 150
Genomic Biomarkers
Advancements in genomic technology have led researchers to increasingly investigate gene-related biomarkers. Specific gene polymorphisms have been identified as potential indicators of susceptibility to DN. By utilizing genetic testing, healthcare providers can assess a patient's risk, thereby facilitating them to implementation targeted preventive strategies.
Sarwaretal 151 identified the genes Cyp2d22, Slc1a4, and Ddah1 as indicators of the dysregulation of renal function-related pathways and DN. Genetic predisposition may contribute to the complications associated with T2DM. Wong 152 demonstrated that the genes GABRR1 and ELMO1 are associated with DN, which leads to glomerular damage. Additionally, the duplication of the FRMD3, CARS, and MYO16/IRS2 genes has been linked to DN. The elevated expression of ICAM-1, along with the C667 T polymorphism in the THBS2, NGAL, PIP, and TRAF6 genes, has been shown to increase susceptibility to DN in patients with T2DM. Furthermore, polymorphisms in cytokine genes are considered critical for to diabetes mellitus and DN. Nikolovetal 153 found strong correlations between serum creatinine levels, creatinine clearance, and the albumin-to-creatinine ratio with the C677 T polymorphism of the MTHFR gene and the 4G/5G polymorphism of the PAI-1 gene, suggesting a novel approach to enhancing early diagnostic methods for DN in individuals with type 2 diabetes. Dabhietal 154 analyzed 648 participants from West India using PCR-RFLP and found that the IL-6-634C/G and IL-10-592C/A polymorphisms are associated with DN in this population. Future large-scale studies should consider racial differences to gain a clearer understanding of the associations between genetic factors and the risks of diabetes and its complications.
Yan et al 155 identified two DN clusters associated with pyroptosis through bioinformatics analysis. They classified DN cases into two subtypes, primarily linked to inflammation, immune response activation, and cellular metabolism, and predicted 65 potential DN therapies targeting key genes, which may provide valuable references for DN treatment. A critical factor in DN is immune cell infiltration, where immune-related genes significantly contribute to inflammation and the recruitment of immune cells. Wang et al 156 analyzed DN samples using bioinformatics technology and identified hub genes—LYZ, CCL5, ALB, IGF1, CXCL2, NR4A2, and RBP4—associated with immune infiltration in DN, which may serve as potential genetic biomarkers for the disease. An Ming et al 157 concluded that DUSP1 and FOSB demonstrate strong diagnostic efficacy in DN and are associated with glomerular immune cell infiltration, positioning them as potential new markers and therapeutic targets for DN diagnosis. Additionally, studies have shown 158 that reducing the expression of DNA damage response 1 (REDD1) and enhancing Nrf2 signaling through modulation of the AKT/GSK-3β pathway can prevent hyperglycemia-induced apoptosis, oxidative stress, and inflammatory damage in mouse podocytes, underscoring the potential role of REDD1-mediated podocyte damage in the development of DN. Renal fibrosis is a common terminal pathway in nearly all advanced chronic kidney diseases. In their study, Fu et al utilized the NanoString nCounter® Human Fibrosis V2 Panel to analyze kidney biopsy specimens from subjects, identifying peroxisome proliferator-activated receptor α (PPARA) fibrosis-related genes that may serve as diagnostic markers for DN. 159 Some recent omics biomarkers are presented in the accompanying Table 3.
Some Recent Omics Biomarkers.

Transcriptomics Biomarkers
Transcriptome profiling provides insights into gene expression under specific conditions, enabling the inference of functions for previously uncharacterized genes and elucidating the mechanisms of action for particular regulatory genes. This molecular tagging, derived from gene expression profiles, facilitates not only the identification of the phenotypic characteristics of cells but also serves as a crucial tool for disease diagnosis. Sequencing technology is the predominant method for uncovering these distinctions. For instance, Wu and Yanjun explored the protective mechanisms of P. capitata against DN, identifiing the involvement of mRNA transcripts encoding ENTPD5, EPHX1, ARVCF, and TREH, alongside six long non-coding RNAs (TCONS_00024194, TCONS_00085825, TCONS_00006937, TCONS_00070981, TCONS_00074099, and TCONS_00097913), as well as two circular RNAs (novel_circ_0001514 and novel_circ_0017507) in this protective response. 179
MicroRNA (miRNA) is a small, non-coding RNA that regulates gene expression through epigenetic mechanisms. It primarily exerts its effects by being transported via exosomes and microvesicles, which subsequently repress target mRNA. Urinary exosomes, ranging from 40 to 100 nm in size, are minute vesicles secreted by various cell types. The exosomes can carry an array of proteins, nucleic acids, and other bioactive substances that are indicative of renal failure and structural damage. Cao et al observed a positive correlation between TGF-β stimuli and miRNA expression in albuminuria, noting its strong diagnostic efficiency. 180 Currently, several drugs targeting the pathogenic signaling of DN, primarily through protein-coding genes, are currently under development. Furthermore, the early detection of DN necessitates the use of miRNA panels to enhance the understanding of the pathogenic signaling associated with this condition. 181
Long non-coding RNAs (lncRNAs), defined as transcripts exceeding 200 nucleotides in length and lacking protein-coding potential, have emerged as crucial regulators of gene expression through various mechanisms. This recognition paves the way for novel therapeutic interventions and the development of biomarker, thereby advancing precision medicine in nephrology. 182 The AMP-response-element-binding-protein has been shown to mediate podocyte damage in DN by targeting the lncRNA DLX6-AS1. Consequently, the AMP-response-element-binding-protein may represent an effective and promising therapeutic target for the treatment of DN. 183
The evidence supporting the early diagnosis and risk stratification of microRNAs (miRNAs) in the clinical context of DN remains inconclusive, necessitating larger cohort studies to validate these preliminary findings. Urinary exosomal miRNAs hold significant clinical relevance as non-invasive early indicators of renal impairment. Importantly, exosomes function as carriers that facilitate cell-to-cell communication and are implicated in the pathogenesis of DN. 184 Furthermore, the proteins contained within exosomes are crucial for the diagnosis and management of DN. For instance, the identification of miRNAs secreted by urinary exosomes from DN patients indicates that altered ferroptosis-related proteins may serve as novel candidate biomarkers for early disease progression and/or therapeutic response. 185 Additionally, both monomers and compounds can influence multiple signaling pathways, including TGF-β1, potentially delaying the progression of DN lesions through modifications in exosome expression. 186 Moreover, exosomes present a promising avenue for drug delivery systems and are poised to become a significant focus in the research of traditional Chinese medicine for the diagnosis and treatment of DN.
Proteomics Biomarkers
Proteomics offers a less invasive and more feasible approach, enabling the simultaneous quantification of multiple protein markers in biological samples. 187 Urine samples address the limitations inherent in blood samples, such as low dosage, significant heterogeneity, and high hydrolytic activity. Furthermore, urine can be collected in substantial volumes and is non-invasive. Consequently, the identification of diagnostic and prognostic biomarkers for kidney disease through urine proteomics is expected to attract growing interest.
(Ⅰ) CKD273
The capillary electrophoresis-mass spectrometry-based urine peptide classifier CKD273 is the most extensively studied and validated proteomic classification, providing robust evidence for its utility in the management of DN. 188 This mass spectrometry technique synthesizes data from 273 urine peptides to generate a composite score, which is highly effective in predicting the onset of de novo albuminuria. Furthermore, a recent meta-analysis has demonstrated that CKD273 is a cost-effective biomarker for forecasting renal function decline in patients with normoalbuminuric diabetes and DN. 170
In a study involving 1014 patients with T1DM and T2DM, all of whom had a baseline eGFR of ≥70 mL/min/1.73 m² and nonalbuminuria, CKD273 was found to effectively identify progression of eGFR to levels below 60 mL/min/1.73 m². Notably, this progression was not associated with albuminuria, age, blood pressure, or baseline eGFR. 171 In a prospective cohort study involving 1775 patients with nonalbuminuria T2DM, with a median follow-up period of 2.5 years, high-risk patients identified by CKD273 were more likely to develop microalbuminuria. Moreover, CKD273 was demonstrated to predict the development of DN independently of HbA1c levels, systolic blood pressure, baseline albuminuria, and eGFR. 172 A further prospective clinical trial involving 52 living-donor kidney transplant recipients, monitored over eight years, assessed the significant role of CKD273 in the prognostic risk stratification of long-term kidney transplant function. 173 Additionally, CKD273 is frequently utilized to investigate markers of treatment response. In a 24-week exploratory trial of linagliptin in patients with type 2 diabetes and nephropathy, stratification based on the CKD273 classifier indicated that linagliptin may have the potential to slow the progressive decline in renal function among patients with high-risk CKD, correlating significantly with declines in clinical renal parameters and eGFR. 174
CKD273 has the capability to predict the onset of albuminuria independently of other renal biomarkers commonly used to forecast the development or progression of DN. However, the application of proteomics for the early diagnosis of DN is constrained by the absence of robust and well-validated diagnostic criteria, regardless of the sample type utilized for proteomic analysis. Therefore, further research is necessary to thoroughly evaluate the clinical utility of proteomic biomarkers in the management of DN before their widespread implementation can be warranted.
(2) Other Therapeutic Markers
Only a limited number of trials utilized biomarkers other than albuminuria or eGFR for participant enrollment and risk stratification, and even fewer studies assessed the effects of treatments using novel biomarkers. 189 The current standard of care for DN involves the blockade of the renin-angiotensin system through the use of ACE inhibitors or ARBs. However, these medications are effective for treating DN primarily at an early stage; otherwise, monotherapy can only serve to prevent or slow the progression of the disease. The incorporation of additional medications, such as dipeptidyl peptidase-4 inhibitors, pentoxifylline, statins, and vasodilator β-blockers, may enhance renal function in patients with preserved DN. 190 Biomarkers indicating the response to pentoxifylline have been identified, and Sakashita suggests that TNF could serve as a biomarker for monitoring the response to pentoxifylline therapy in patients with CKD. 175 So far, there are few biomarkers for treatment response of DN.
S100A8 and S100A9
S100A8 and S100A9 are proteins that play crucial roles in immune and inflammatory responses. The study conducted by Du et al reported a significant upregulation of S100A8 and S100A9 expression in renal epithelial cells within diabetic kidneys. Furthermore, the application of the small molecule inhibitor AB38b to suppress the aberrant expression of S100A8 and S100A9 may represent a novel therapeutic approach for addressing renal interstitial fibrosis induced by DN. 176
ACSL1
The expression of ACSL1 in exfoliated urinary cells and urine supernatant from patients with DN was significantly elevated and closely correlated with renal function. Furthermore, ACSL1 expression was markedly increased in the kidney tissues of fibrotic db/db mice. Knockdown of ACSL1 in HK-2 cells demonstrated a reversal of renal fibrosis induced by high glucose levels. 177 Therefore, reducing ACSL1 expression may represent a novel therapeutic strategy for treating DN-induced renal fibrosis.
DsbA-L
DsbA-L interacts with catalase in peroxisomes to mitigate renal tubular oxidative damage in DN. It regulates catalase activity through binding thereby reducing stress and apoptosis induced by high glucose (HG, 30 mM) and palmitic acid (PA, 250 μM). Notably, the expression of DsbA-L in the renal tubules of DN patients was significantly decreased and positively correlated with peroxisome function. These findings suggest that DsbA-L may serve as a therapeutic marker for alleviating tubular injury in DN. 178
Metabolomics Biomarkers
Metabolomics involves the measurement of low-molecular-weight intermediates and biochemical processes in biological fluids with high sensitivity and specificity, enabling the detection of a wide range of metabolites. This approach is often regarded as a superior platform for assessing a patient's molecular phenotype when compared to biological information derived from the genome, transcriptome, and proteome. By facilitating early diagnosis and improved management of patients with DN, metabolomics may contribute to a reduction in the progression to ESRD. 191 Further metabolomic investigations by Guo et al have demonstrated that α-ketoglutarate (AKG) serves as a crucial intermediate in multiple metabolic pathways. Moreover, dysregulation of AKG levels is associated with kidney disease. 192 Recent metabolomic markers are presented in the accompanying Table 3.
Sharma et al highlight that metabolomics uncovers critical pathways involved in the progression of DN, with biomarkers such as glycosylated albumin and free fatty acids offering valuable insights into the complexities of DN, thereby informing potential treatment strategies. Metabolomics focuses on the detection of small molecule metabolites, which facilitates the identification of disease-specific patterns that support personalized care. 193 However, interpreting metabolomics results can be challenging due to various confounding factors, including lifestyle, medication, and nutritional status. Ongoing research into metabolomics and multi-omics approaches is essential for identifying new therapeutic targets and anticipating clinical interventions in response to adverse disease outcomes, based on individual molecular profiles.
Novel Biomarkers
Microbial Markers
Kumaresan emphasizes that microbial populations coexist in symbiosis with the host to maintain homeostasis and regulate the immune response. Dysbiosis of the microbiota can result in diseases, including kidney disease and systemic dysfunction, underscoring the significance of salivary microbes in conditions such as diabetic nephropathy, chronic kidney disease, membranous nephropathy, and IgA nephropathy. Consequently, salivary microbial markers as noninvasive tools for the detection of diverse kidney diseases. 194 What's more, a multitude of studies have clarified the roles of gut microbiota and the gut-liver axis in the pathophysiology of diabetes. Notably, respiration facilitated by hepatic mitochondria is linked to systemic alterations in oxidative stress, insulin resistance, and lipid metabolism. 195 Moreover, Crudele et al 196 emphasized that the gut microbiota serves as a new prognostic biomarker which may have therapeutic implications for diabetes. In a related study, Mokhtari et al 197 found that adolescents suffering from type 1 diabetes and nephropathy displayed slight changes in the taxonomic makeup of their gut microbiome, with significant characteristics such as Faecalibacterium and Streptococcus.
Potential Drug Candidates and new Methods
Recent research has identified gentiopicroside (GPS), primarily composed of iridoid glycosides, as a promising candidate for the prevention and treatment of T2DM. 198 Zhang et al 199 have highlighted that active components of traditional Chinese medicine, such as saponins, may mitigate cardiovascular diseases by modulating the ferroptosis regulatory mechanism characterized by iron-dependent lipid peroxidation. This finding offers a novel direction and a scientific foundation for utilizing traditional Chinese medicine in the prevention and management of diabetes and its associated complications.
Identify new uses for existing drug therapies and enable rapid, low-cost drug repurposing. Drug-gene associations were discovered by leveraging publicly accessible databases that include the most comprehensive genome-wide relationships with T2DM. These results were later confirmed using data collected from electronic health records and Mendelian randomization techniques. The findings suggest that angiotensin-converting enzyme inhibitors and calcium channel blockers are strong candidates for reducing blood sugar levels while also lowering the likelihood of cardiovascular disease. 200 Additionally, Mendelian randomization has shown that metformin positively influences body mass index (BMI) and blood pressure in individuals without diabetes. 201 This novel method provides important insights and a solid basis for exploring the repurposing of existing drugs for DN or for discovering biomarkers for drug monitoring.
Conclusion
DN research is continually advancing, and the identification of new biomarkers offers valuable tools for early diagnosis and disease monitoring. While it would be any potential challenges or limitations in using these novel biomarkers in clinical practice. This article presents several novel biomarkers associated with DN identified in recent years. In terms of content, this article comprehensively discusses biomarkers from the multi-dimensional parallel intersection perspectives of physiology, pathology, mechanism, and new technologies. From a structural point of view, this paper proposes an asymptotic relationship between points, lines, and surfaces from single to multiple to biomarker spectra. Although traditional indicators such as albuminuria and eGFR remain essential in clinical practice, emerging studies on inflammatory markers, oxidative stress markers, combinatorial markers, and omics markers have demonstrated their potential in the management of DN. Future research should prioritize the integration of multiple biomarkers and personalized treatment approaches to enhance patients’ quality of life and extend survival. Additionally, these biomarkers should be considered as enrollment criteria or surrogate endpoints in randomized clinical trials. To strengthen the evidence base, larger study cohorts are necessary, including kidney biopsy samples alongside urine and plasma or serum samples from the same patients. To ensure the generation of comparable and reproducible data, a consensus protocol for sample collection, processing, and analysis must be established among collaborating researchers. As our understanding of the pathophysiology of DN deepens, an increasing number of novel biomarkers are expected to emerge. Therefore, it is imperative to incorporate these new findings and biomarkers into the foundational elements of future clinical trials aimed at improving the prognosis of patients with DN. it would be interesting to explore future directions or ongoing research in the field of DN biomarkers.
Footnotes
Acknowledgments
This research was funded by the National Natural Science Foundation of China (no.81473427), Xinglin Talent Program of Chengdu University of Traditional Chinese Medicine(QJJJ2023017), Scientific Research Project of Sichuan Provincial Administration of Traditional Chinese Medicine(2024MS046), Sichuan Provincial Science and Technology Program(2021YJ0112).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, Xinglin Talent Program of Chengdu University of Traditional Chinese Medicine, Sichuan Provincial Science and Technology Program, Scientific Research Project of Sichuan Provincial Administration of Traditional Chinese Medicine, (grant number 81473427, QJJJ2023017, 2021YJ0112, 2024MS046).
